Abstract
Current dogma is that pathological hypertrophy of the right ventricle is a direct consequence of pulmonary vascular remodeling. However, progression of right ventricle dysfunction is not always lung-dependent. Increased afterload caused by pulmonary vascular remodeling initiates the right ventricle hypertrophy, but determinants leading to adaptive or maladaptive hypertrophy and failure remain unknown. Ischemia in a hypertrophic right ventricle may directly contribute to right heart failure. Rapidly enlarging cardiomyocytes switch from aerobic to anaerobic energy generation resulting in cell growth under relatively hypoxic conditions. Cardiac muscle reacts to an increased afterload by over-activation of the sympathetic system and uncoupling and downregulation of β-adrenergic receptors. Recent studies suggest that β blocker therapy in PH is safe, well tolerated, and preserves right ventricle function and cardiac output by reducing right ventricular glycolysis. Fibrosis, an evolutionary conserved process in host defense and wound healing, is dysregulated in maladaptive cardiac tissue contributing directly to right ventricle failure. Despite several mechanisms having been suggested in right heart disease, the causes of maladaptive cardiac remodeling remain unknown and require further research.
Introduction
Pulmonary hypertension (PH) is a heterogeneous group of diseases characterized by right ventricle hypertrophy and pulmonary vascular remodeling. 1 Failure of the right heart is the major cause of death in PH patients.2,3 Current dogma is that enlargement of the right ventricle is a consequence of pulmonary vascular remodeling. This is supported by the observation that lung transplantation in patients with pulmonary arterial hypertension (PAH) and pulmonary endarterectomy in patients with chronic thromboembolic pulmonary hypertension (CTEPH) reverses the right ventricle hypertrophy.4–7 However, progression of right ventricle dysfunction is not always lung-dependent. For example, subsets of patients on vasodilator therapy continue to develop worsening of right ventricle function despite improved pulmonary artery hemodynamics.8–11 In a small cohort study, improved right ventricle ejection volume, but not decreased pulmonary vascular resistance, predicted survival rates in patients on vasodilator drugs. 12 Although there is consensus that increased afterload caused by pulmonary vascular remodeling initiates the right ventricle hypertrophy, determinants leading to adaptive remodeling versus right ventricle failure (maladaptive hypertrophy) remain unknown. Here we provide a synopsis of mechanisms that are linked to maladaptive right ventricle hypertrophy in patients and preclinical models.
Right ventricular ischemia
Ischemia in a hypertrophic right ventricle may directly contribute to right heart failure. Several lines of evidence support that blood supply to the right ventricle in PH is impaired. Right ventricles of rats in hypoxia/Sugen and monocrotaline models have capillary rarefaction.13–17 In patients, chest pain, reduced technetium 99m sestamibi uptake to evaluate myocardial perfusion, 18 and increased retention of 18F-fluorodeoxyglucose (FDG) in the right ventricle19,20 indicate metabolic reprogramming and potentially the presence of ischemia. However, histological studies also show a reduced microvessel density in the right ventricle of patients with PAH associated to scleroderma. 15 The precise causes for right ventricle ischemia remains unknown, but several mechanisms have been suggested in the literature, including impaired angiogenesis to meet the increasing demands of a hypertrophic myocardium,13,16,17,21–23 endothelial dysfunction,13,24 or dropped coronary perfusion pressure. 25 Loss of capillary density may mark the transition from adaptive to maladaptive right ventricle hypertrophy. In the monocrotaline model, adaptive right ventricle hypertrophy was associated with preserved capillary density. 22 Coagulopathy may be another mechanism causing right heart ischemia. 11 In mice xenografted with bone marrow CD133+ hematopoietic stem cells isolated from PAH patients, ischemia and infraction in hypertrophic right ventricles were observed without changes in the right ventricle capillary density. 26 It is noteworthy that ischemia has not been directly assessed and stereologic approaches of right ventricle angiogenesis depicts a somewhat different concept. In a small cohort, patients with PH showed compensatory angiogenesis in the right ventricle which may be helpful to sustain right ventricle function. 27 In a mouse model of chronic hypoxia-induced PH, early adoptive right ventricle angiogenesis was observed. 28 Therapeutic strategies to potentially target right ventricle ischemia will thus depend on further research identifying the precise causes and whether or not ischemia occurs in the right heart in PH.
Right ventricle metabolism
During postnatal life, mitochondrial fatty acid oxidation becomes the predominant source of energy, in addition to glycolysis in lesser amount.29,30 Several lines of evidence indicate that mitochondrial metabolism is suppressed during right ventricular hypertrophy. The fast enlarging cardiomyocytes switch from aerobic to anaerobic ATP generation similar to cancer cell growth (Warburg effect), allowing rapid cell growth under hypoxic conditions.31,32 Glucose uptake is increased in hypertrophic right ventricles, as demonstrated by FDG-PET cardiac imaging,19,20,30,33 while Iodine-123-labeled 15-(p-iodophenyl)-3-(R,S)-methylpentadecanoic acid (BMIPP) SPECT showed impaired fatty acid uptake in the failing hypertrophied RV. 34 Increased expression of glycolysis-related genes or glycolysis enzymatic activities were observed in right ventricles in the hypoxia/Sugen and monocrotaline rat models.15,35 Oxidative energy generation is lessened in hypertrophic right ventricles by several mechanisms. Pyruvate dehydrogenase in the mitochondria that links cytosolic glycolysis to the Krebs cycle by conversion of glycolysis end-product pyruvate into acetyl CoA, may be inhibited by upregulated pyruvate dehydrogenase kinase as illustrated in a case report. 36 A key question is: what are the alternations in mitochondrial bioenergetics that mark the transition from adaptive to maladaptive right ventricle hypertrophy? In the monocrotaline rat model, the maladaptive right ventricle hypertrophy was associated with decreased pyruvate dehydrogenase kinase levels and decrease glucose uptake and an increase in mitochondrial reactive oxygen species production. 37 In a case report, long-term PAH survival was also correlated with lower expression of aerobic glycolysis regulators compared to a patient with decompensated right heart failure. 24 Glutaminolysis, the conversion of glutamine into α-ketoglutarate to fuel the Krebs Cycle, another maladaptive metabolic pathway allowing rapid cell growth, may also play a role in pathological right ventricle hypertrophy. 38 Although the Warburg effect and glutaminolysis may provide benefit to hypertrophic cardiomyocytes to maintain normal function during initial stages of PH, it is insufficient in progressive right ventricle hypertrophy, supported by the observation that fatty acid oxidation is maintained or even increased in left ventricle hypertrophy, but reduced in left ventricle failure. 39 Thus, therapies targeted to restore normal mitochondrial bioenergetics may be beneficial to PH patients.
Right ventricular β-adrenergic receptor function
β-adrenergic receptors have critical roles in pathogenesis of several cardiac diseases such as chronic left heart failure, coronary artery by-pass grafting, and ischemic heart disease. 40 Heart tissue expresses both β1- and β2-adrenergic receptors, which mediate myocardial contractility and heart rate. Both right and left ventricles react to an increased afterload by overactivation of the sympathetic system and uncoupling and downregulation of β-adrenergic receptors.30,41–43 In the rat monocrotaline model, β1-adrenergic receptors were downregulated in a failing hypertrophied right ventricle, but not in a hypertrophied right ventricle that maintained normal function. 44 Several animal studies demonstrated that β-adrenergic receptor blockade may rescue receptor function and have improved outcomes. In the hypoxia/Sugen and/or monocrotaline rat models, β-blocker therapy improved right heart function26,45 but did not affect right ventricle afterload.21,46,47 β-blocker treatment restored β-adrenergic signaling 47 and reduced right ventricle fibrosis,21,46,47 by downregulating TGFβ 1 expression and blocking pro-fibrotic signaling, 46 and decreased capillary rarefaction, by upregulating vascular endothelial cell growth factor (VEGF). 21 Expression of apoptotic markers in the right ventricle were also lessened in rodents treated with β-blocker. 46 However, progression of right ventricle hypertrophy and failure induced by pulmonary trunk banding in rats was not improved by β-blocker therapy. 48 Despite major favorable effects of β-blocker therapy in these preclinical studies, clinical use of β-blockers for the treatment of PH is not without risks. Indeed, β-blocker therapy was initially contraindicated in PH due to concern of possible mortality. 49 However, several non-randomized small clinical cohorts showed that β-blocker therapy may be safe in the treatment of PAH patients and provided clinical benefits.26,49–56 A recent six-month double-blind, placebo-controlled, and randomized clinical trial demonstrated that treatment of patients with a non-selective β-blocker/vasodilator carvedilol is well tolerated and safe. 57 β-blocker therapy in this study preserved right ventricle function and cardiac output. Mechanistically, patients treated with carvedilol, but not placebo, had reduced right ventricular glycolysis and increased β-adrenergic receptor levels. 57 Overall, this promising clinical study warrants a larger clinical trial to further evaluate β-blocker therapy in PAH.
Right ventricular fibrosis
Fibrosis is conserved during evolution as a highly regulated and critical process in host defense and wound healing, but results in uncontrolled disease. 58 In cardiac tissue, fibrosis is triggered by early inflammation in response to pressure overload. 59 Several magnetic resonance imaging (MRI)-delayed gadolinium enhancement studies showed presence of interstitial or local myocardial fibrosis in PH patients.60–62 Not all animal models with PH develop right ventricle fibrosis, including models of compensated right ventricle hypertrophy such as wild-type (WT) rodents under chronic hypoxia and pulmonary artery banding.13,35 In contrast, substantial right ventricle fibrosis is observed in models of decompensated right ventricle failure including Sugen/hypoxia, pulmonary artery banding combined with Cu2+-depleted diet or high dose monocrotaline treatment,13,35,63 and caveolin-1-deficient mice.64–67 Collagen synthesis kinetics in rabbits with increased right ventricle afterload caused by pulmonary artery banding showed a rapid increase in right ventricle collagen synthesis followed by degradation, 68 supporting the notion that controlled fibrosis may be part of a reparative process in compensated right ventricle hypertrophic response. 69 Growth factors and cytokines involved in cardiac fibrosis have been elegantly reviewed in a previous report. 69 Myofibroblasts are key cells in fibrosis and have several origins including differentiation of tissue resident fibroblasts, bone marrow hematopoietic stem cell-derived, or endothelial-derived via endothelial to mesenchymal transition.70–73 A recent report demonstrated that bone marrow-hematopoietic stem cell-derived fibroblasts are mobilized and recruited into the left ventricle where they differentiate into myofibroblast in response to transverse aortic constriction induced pressure overload in mice and substantially contribute to cardiac fibrosis. 74 In line with these findings, another report showed that right ventricle fibrosis in caveolin-1-deficient mice is prevented by transplantation of WT bone marrow independent of pulmonary vascular disease. 64 Whether caveolin-1 affects bone marrow hematopoietic stem cell-derived fibroblast progenitors needs to be further investigated. Fibrocytes, a subset of circulating hematopoietic cells with established role remodeling of the pulmonary artery wall in animal models of PAH75,76 may also be involved in cardiac fibrosis. It is unclear at what point during right ventricle hypertrophy fibrosis transits from a reparative process into excessive extracellular matrix deposition and pathological remodeling of the myocardium. The reversal of right ventricle fibrosis by β-blocker therapy21,46,47 and associated beneficial effects indicate that fibrosis may be therapeutic target in PH.
Conclusions
Several adaptive mechanisms have been identified in the right ventricle in PAH, some of which may be detrimental and others beneficial to sustain right heart function. Blocking all adaptation will be harmful, because the exact mechanisms of right ventricle failure in PAH remain unidentified. Inflammation and angiogenesis induced by an increased right ventricle afterload may result in regenerative fibrosis resulting in compensated right ventricle hypertrophy with sustained mitochondria and β-adrenergic receptor function. Impaired angiogenesis, coagulopathy, reduced coronary perfusion, or other unknown factors may derail the reparative process resulting in ischemia and pathological fibrosis. Glutaminolysis, increased glycolysis, and dysfunctional β-adrenergic receptor may further impair right ventricle function resulting in decompensated hypertrophy and failure. Further research is required to identify the determinants of adaptive and maladaptive right ventricle hypertrophy (Fig. 1). Heterogeneity likely exists in mechanisms initiating right heart disease. Discovery of root cause(s) of right ventricle failure will aid in the development of cardiac targeting therapies.
Mechanisms of right heart disease in pulmonary hypertension.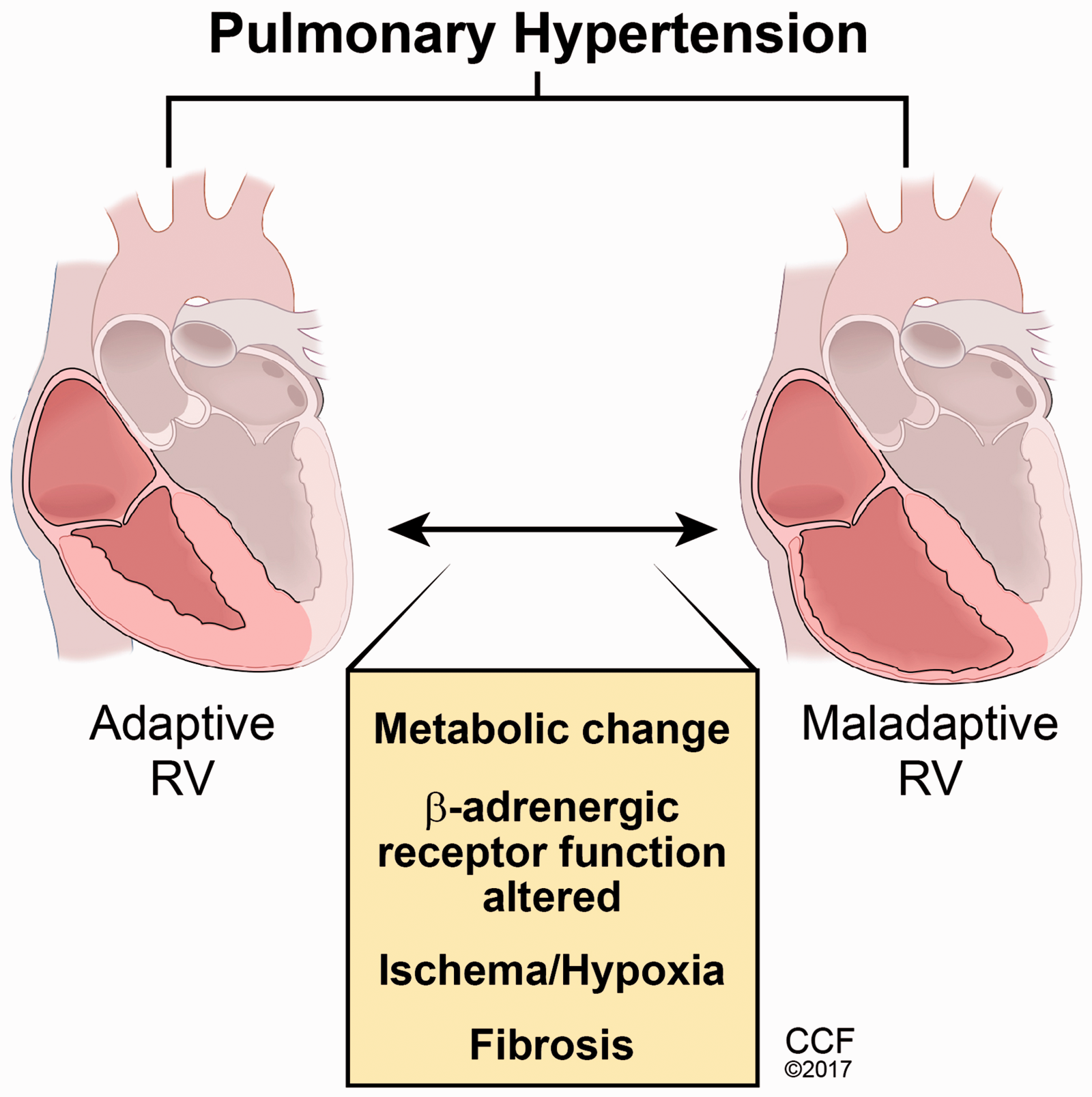
Footnotes
Acknowledgments
Illustration by David Schumick, BS, CMI. Reprinted with the permission of the Cleveland Clinic Center for Medical Art & Photography © 2017. All rights reserved.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
Grant support: HL125177, HL123767, HL115008, HL60917.
2017 Grover Conference Series
This review article is part of the 2017 Grover Conference Series. The American Thoracic Society and the conference organizing committee gratefully acknowledge the educational grants provided for the support of this conference by Actelion Pharmaceuticals US, Inc., Gilead Sciences, Inc., and United Therapeutics Corporation. Additionally, the American Thoracic Society is grateful for the support of the Grover Conference by the American Heart Association, the Cardiovascular Medical Research and Education Fund, and the National Institutes of Health.
