Abstract
Common variable immunodeficiency is a chronic illness plagued with recurrent infections and the potential to develop autoimmune disease. These patients may manifest a spectrum of complications ranging from hematologic malignancy to chronic parenchymal lung disease. Regular immunoglobulin replacement therapy improves immunologic debility but does not mitigate other features of this disease. Here, we discuss a complication of common variable immunodeficiency not previously characterized in the literature. We present two cases of advanced pulmonary vascular disease associated with common variable immunodeficiency treated with pulmonary vasodilators.
Introduction
Common Variable Immunodeficiency (CVID) is a severe primary immunodeficiency caused by antibody production failure and characterized by recurrent bacterial infections. 1 Prior registries have found no association between CVID and pulmonary arterial hypertension (PAH), although patients with CVID and severe hypoxic lung disease may develop pulmonary hypertension (PH). 2 The literature on CVID-associated PH is limited to case reports attributing pulmonary vascular disease to destructive lung disease or development of secondary amyloidosis due to chronic, ongoing inflammation; a proportion develop autoimmunity as well.3,4 Here, we present two cases of pre-capillary PH with pathologic changes in pulmonary arteries and veins of intimal hyperplasia, with near-occlusive arterial remodeling. Both patients had clinical improvement with PAH-specific therapies. These findings are consistent with a pulmonary vasculopathy and remodeling, out of proportion to the severity of Granulomatous and Lymphocytic Interstitial Lung Disease (GL-ILD) in these two CVID patients.
Case presentations
Case 1
A 42-year-old man with Stage V chronic kidney disease and CVID with hyperimmunoglobulin M was evaluated for PH during a hospital admission for acute right heart failure. He was diagnosed with CVID at 19 years old and was treated with intravenous immunoglobulin (IVIg) since age 38. An echocardiogram showed a severely dilated right ventricle (RV), RV hypertrophy, severe RV systolic dysfunction with an estimated RV systolic pressure (RVSP) of 73 mmHg. His brain natriuretic peptide (BNP) was 1789 pg/mL. Right heart catheterization (RHC) revealed RA pressure of 14 mmHg, mean PA pressure of 54 mmHg, pulmonary capillary wedge pressure (PCWP) of 8 mmHg with a Fick of cardiac output of 3.05 L/min/m2 and pulmonary vascular resistance (PVR) of 15 Woods Units. Chest computed tomography (CT) showed enlarged pulmonary arteries, ground glass opacities in the bilateral lower lobes, no findings of chronic thromboembolic disease (Fig. 1a). Pulmonary function testing (PFT) revealed mild restriction and severely reduced diffusion capacity (forced expiratory volume 2.6 L, 71% predicted; forced vital capacity 3.5 L, 76%; FEV1/FVC 75%; total lung capacity 4.8 L, 78% predicted; diffusing capacity for carbon monoxide 48% predicted). He started sildenafil and ambrisentan but developed worsening RV failure and renal failure prompting admission for dialysis and inhaled epoprostenol. He transitioned to IV epoprostenol therapy with improvement to mild RV systolic dysfunction and BNP normalized to 58 pg/nL.
Diagnostic images. Case 1: A chest CT (a) at the time of pulmonary arterial hypertension diagnosis showing ground-glass opacities in the bilateral lower lobes. (b) Hematoxylin and Eosin staining of lung biopsy specimen showing organizing pneumonia on a background pattern of non-specific interstitial pneumonia (NSIP), cellular pattern, and pneumocyte hyperplasia. (c) Elastin stain of lung biopsy specimens demonstrating a cross-sectioned pulmonary artery with reduplication of the elastin lamina, intimal hyperplasia, and luminal narrowing. (d) Elastin stain of lung biopsy specimen showing a cross-sectional view of a pulmonary vein with luminal obliteration. Case 2: A chest CT (e) at the time of PAH diagnosis showed inter- and intralobar septal thickening with bilateral bronchiectatic changes as well as ground glass opacities and nodular densities. (f) Hematoxylin and Eosin staining of lung biopsy specimen showing non-specific interstitial pneumonia, fibrosing pattern. (g) Elastin staining of lung biopsy specimen demonstrating pulmonary veins with intimal proliferation and significant luminal narrowing. (h) Elastin stain of a pulmonary artery showing duplication of the elastic lamina and non-occlusive intimal proliferative.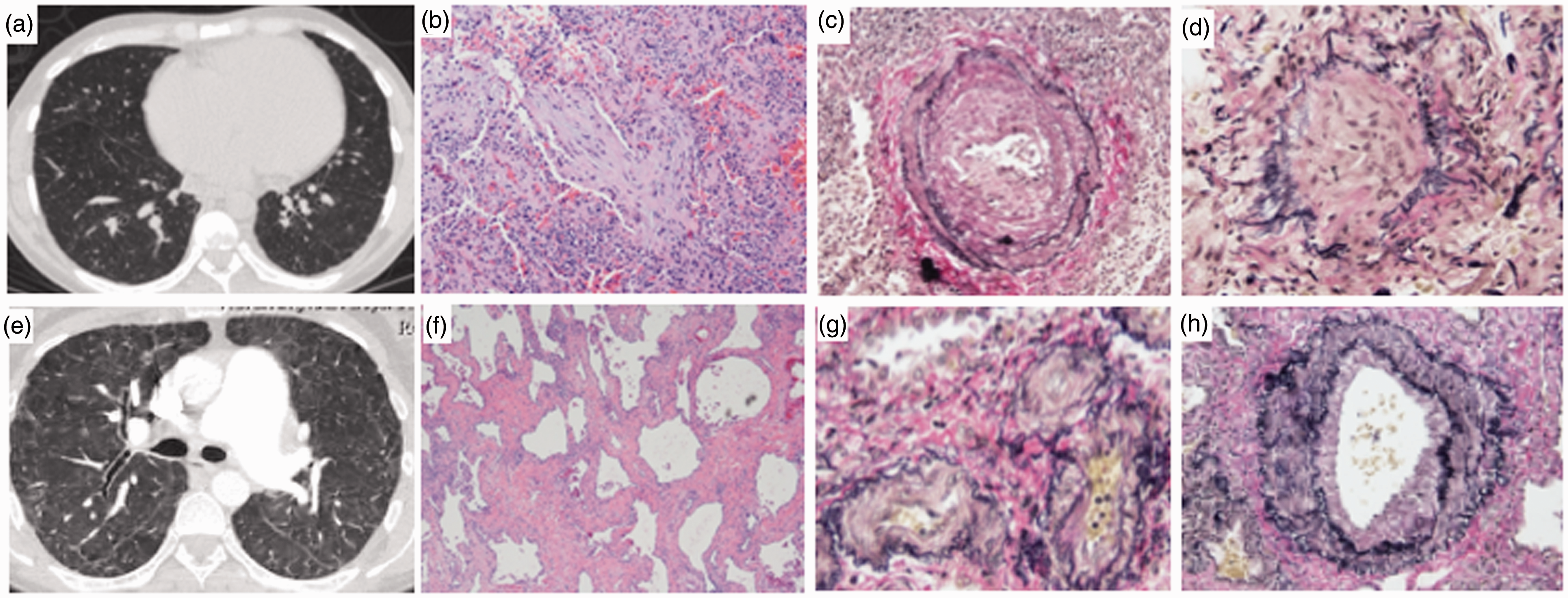
Two years later, he developed worsening dyspnea, cough, and hypoxemia, with a chest CT showed multifocal pulmonary infiltrates. Spirometry was stable with worsening diffusion capacity (30% predicted). His echocardiogram showed RVSP 50 mmHg and mildly depressed RV systolic function. Lung biopsy revealed a complex pattern of follicular bronchiolitis/hyperplastic bronchus-associated lymphoid tissue, nonspecific interstitial pneumonitis (NSIP)-like, and lymphoid interstitial pneumonitis (LIP)-like areas, scattered histiocyte aggregates consistent with poorly formed granulomas, and occasional foci of bronchiolitis obliterans-organizing pneumonia (Fig. 1b). Significant fibrosis was also present. Elastin stains demonstrated advanced hypertensive remodeling of both arteries and veins, with intimal hyperplasia (frequently occlusive) and reduplication of the elastic laminae; arterial changes were equivalent to Heath-Edwards grade 3 (no plexiform lesions were present) (Fig. 1c and d). There were no findings of amyloid deposition on pathologic specimens. He was diagnosed with GL-ILD and started on corticosteroids with rapid improvement in hypoxemia and resolution of radiographic abnormalities. He continues on IV epoprostenol for PAH.
Case 2
A 41-year-old woman with CVID diagnosed at age 27, on chronic IVIg, was admitted for dyspnea and exertional hypoxia. Six years prior to this presentation, she was found to have bronchiectasis, septal thickening, ground glass opacities, and nodular densities on chest CT (Fig. 1e). Lung biopsy revealed follicular bronchiolitis with occasional LIP-like areas, variable bronchiolectasis, a few histiocyte aggregates consistent with poorly formed granulomas, and foci of NSIP-like pattern, both cellular and fibrotic (Fig. 1f). Elastin stains showed mild to moderate arterial hypertensive remodeling, with occasional non-occlusive intimal proliferative lesions and variable reduplication of the elastic laminae, equivalent to Heath-Edwards early grade 2; small veins had frequent mild intimal proliferation (Fig. 1g and h). No evidence of amyloid involvement was seen on pathology. Echocardiogram showed normal RVSP and RV function. She was diagnosed with GL-ILD and treated with corticosteroids with clinical improvement. Spirometry improved to mild-moderate obstruction (FEV1 1.98 L, 64% predicted) and moderately reduced diffusion capacity (50% predicted) and was stable for several years. At the current presentation, she had a new dilated, hypertrophied RV with moderately depressed RV systolic function and estimated RVSP of 87 mmHg on echocardiogram with a BNP of 894 pg/nL. Her PFTs showed similar degree of obstruction with severely reduced diffusion capacity (DLCO 7.1 mL/mmHg/min, 29% predicted). Chest CT showed a dilated PA but was otherwise unchanged compared to prior CT scans. An RHC revealed RA pressure of 7 mmHg, mean PA pressure of 58 mmHg, PCWP of 9 mmHg with a Fick of cardiac output of 4.02 L/min/m2 and PVR of 9.2 Woods Units. Based on advanced pre-capillary PAH with stable, mild parenchymal lung disease, she was offered PAH-directed therapies. She declined parenteral prostacyclin therapy and is currently on tadalafil and ambrisentan, with improvement in clinical symptoms and normalization of BNP (39 pg/nL).
Discussion
Here, we present two patients with CVID with pre-capillary PH by invasive hemodynamics and treated with pulmonary vasodilators. Our first case had no evidence of parenchymal lung disease at the time of PH diagnosis, whereas the second case had stable chronic lung disease with a new diagnosis of PH and RV failure. Examination of lung biopsy specimens showed marked pulmonary vascular remodeling involving both the pulmonary arterial and venous system. These pulmonary vascular findings have not yet been reported in this patient population and demonstrate a degree of pulmonary vascular remodeling disproportionate to degree of lung disease.
Echocardiographic evidence of PH and RV dilation has been reported in primary antibody deficiencies. 2 A small study found 20% of CVID patients had RV dilation and 45% had echocardiographic evidence of PH, the severity of which correlated with time to CVID diagnosis. Most of these patients had severe lung disease marked by obstruction on pulmonary function tests, hypoxia, and imaging abnormalities. This cohort had one patient with relatively minor pulmonary parenchymal findings and significant PH, like our patients. Cor pulmonale is a frequent cause of death in these patients; however, little data exist about the development of pulmonary vascular disease in this population. 5
Both of our patients were diagnosed with GL-ILD on biopsy. This variety of interstitial lung disease (ILD) is characterized by a non-infectious reaction with nodular peribronchial inflammation and non-necrotizing granulomas.6,7 These granulomas are seen along bronchovascular structures; however, little is published about associated pathology in the pulmonary vasculature of this condition. While both of our patients had parenchymal lung disease, the degree of pre-capillary PH with both venous and arterial vascular remodeling was out of proportion to the severity of ILD at the time of PAH diagnosis and is a finding not previously reported in CVID. Both of our patients had a beneficial response to PAH specific therapies with normalization of BNP and improvement of RV systolic function, this would not be expected with PH primarily related to chronic lung disease, as these therapies do not modify that pathology. The vascular lesions found in these patients indicate a discrete pulmonary vascular disease associated with CVID and warrants PAH specific therapies.
Lastly, both of our patients had been treated with long-term intravenous IVIg therapy, raising the question of whether the therapy, instead of the disease, could be implicated in development of PH. Administration of IVIg was problematic for both patients due to infusion volume, prompting transition to more frequent subcutaneous delivery. It is important to consider this variable in patients with RV dysfunction even if related to PH related to chronic lung disease. The literature is devoid of IVIg therapy-associated PH.
These cases demonstrate the importance of recognizing this vascular complication of CVID, especially in the setting of contemporary prolonged survival due to immunoglobulin and antibiotic therapy. In our experience, these patients have done well with PAH specific therapies.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
Dr. Hemnes is funded by NIH/NHLBI grant P01HL108800-10.
