Abstract
Implantable infusion pumps might improve the convenience and safety of intravenous treprostinil for pulmonary arterial hypertension. The LENUS Pro® pump (approved in Europe) has a fixed flow rate. Based on 126 pumps and 2853 refills, we retrospectively analyzed the actual flow rate from 09/2010 to 09/2018. A relevant flow rate variance is evident after three years; therefore, flow rate monitoring and dose adjustment are mandatory.
Use of parenteral treprostinil for the treatment of pulmonary arterial hypertension is limited due to the inconvenient route of administration and risk of catheter-associated infections or local side-effects.1,2 Fully implantable pumps may help to address these issues. The LENUS Pro® implantable pump (Tricumed Medizintechnik GmbH, Kiel, Germany) is approved in Europe for intravenous treprostinil administration and has a fixed flow rate.3–7 Treprostinil solution is injected into a drug reservoir within the pump via a silicone septum, and a gas-driven titanium bellows generates a constant flow from the reservoir regulated by a chip capillary. 7 In routine clinical practice, percutaneous refills are performed under aseptic conditions using specifically designed refill needles either at the referral center or by a specialized healthcare service in the patient’s home, based on local availability. 5 Intervals between percutaneous refills are usually 14–28 days depending on the flow rate and pump size (20 or 40 mL). The dose is adjusted at each refill based on the size of the reservoir, the flow rate, and the concentration of the treprostinil solution (≤10 mg/mL). However, the variance of the fixed flow rate during long-term follow-up has not yet been analyzed.
We conducted a retrospective database analysis of all pump refills documented by a specialized healthcare service (pro-samed, Berlin, Germany) supervised by expert centers between September 2010 and September 2018. The specialized healthcare service measures the volume of residual drug (in mL) using a specifically designed collection syringe after puncture of the filling silicone septum at every refill. By combining this information with the intervals between refills, the actual flow rate in each interval was calculated and expressed as a percentage change from the expected fixed flow rate (which was provided by the distributor of the pump (OMT GmbH, Frittlingen, Germany) at the time of implantation). Furthermore, the absolute amount of treprostinil administered in mg/day (adjusted to the actual flow rate) was recorded. Due to differences in the intervals between percutaneous refills, the percentage change from the expected flow rate and the absolute amount of treprostinil administered per day were averaged over three-month intervals for each patient. In addition, the relative flow rate deviation between each individual refill was calculated in % and classified into the following categories: below −10%, −10% to +10%, and above +10%.
Data collection and analyses were approved and the need to obtain written informed consent from each patient was waived by the ethics committee of the Faculty of Medicine at the University of Giessen (Approval No. 241/16). Data are reported as mean ± standard error of the mean or as median (interquartile range).
During the study period, 315 pumps were implanted in Germany (personal communication from OMT GmbH), of which 126 (40%) were systematically followed and refilled by the specialized healthcare service with supervision from pulmonary hypertension expert centers (median follow-up: 12 (6 to 24) months). The median distributor-specified fixed flow rate was 1.3 (1.2 to 1.3) mL/day, and the median initial rate of treprostinil administration (during the first three months after implantation) was 1.5 (1.1 to 2.7) mg/day. Twenty-one patients (16.7%) had a 20 mL pump and 69 patients (54.8%) had a 40 mL pump (missing data, n = 36 (28.6%)). The actual flow rate was lower than the distributor-specified fixed flow rate during the first three months after implantation (variance of the fixed flow rate: mean, −6.5 ± 4.9%; median, −6.7 (−9.2 to −3.8)%) and steadily increased thereafter: the mean variance of the fixed flow rate was −1.2 ± 7.3% (median, −1.9 (−6.5 to 1.7)%) at >12–15 months, 8.7 ± 11.1% (median, 7.5 (1.9 to 15.5)%) at >24–27 months, and 16.1 ± 10.7% (median, 16.9 (10.5 to 21.4)%) at >36–39 months. Of note, mean flow rate variances of up to 42.6 ± 4.2% were observed beyond 39 months after implantation, but these results were significantly biased by the low number of patients with available data during those time intervals (Fig. 1a). A similar pattern of flow rate increase was observed in subgroups with low versus high distributor-specified fixed flow rates (<1.3 mL/day and ≥1.3 mL/day, respectively; Fig. 1b), low versus high initial rates of treprostinil administration (<1.5 mg/day and ≥1.5 mg/day, respectively, at >0–3 months; Fig. 1c) and 20 mL versus 40 mL pumps (Fig. 1d). Based on 2853 refills, the relative flow rate deviation between each individual refill was between −10% and +10% in the vast majority of cases (94.5%). The deviation was below −10% in 2.7% of refills and above +10% in 2.9% of refills (Fig. 1e). Three cases (0.1%) had a relative flow rate deviation of more than 40% from the previous refill (one case of −40% and two cases of +40%).
Flow rate variance of the fully implantable LENUS Pro® pump for the delivery of intravenous treprostinil. Mean flow rate variance of (a) all pumps, (b) pumps with low and high distributor-specified flow rates, (c) pumps with low and high initial treprostinil infusion rates, and (d) 20 mL and 40 mL pumps, and (e) the relative flow rate deviation between each individual refill (2853 refills in total). n = number of patients with ≥1 visit during the corresponding three-month interval. Dashed horizontal lines show the upper and lower limits of technical deviation as specified by the distributor (±7.5%). Error bars show standard error of the mean. *Low and high distributor-specified flow rates were defined as <1.3 mL/day and ≥1.3 mL/day, respectively. †Low and high initial treprostinil infusion rates were defined as <1.5 mg/day and ≥1.5 mg/day, respectively, at 0–3 months.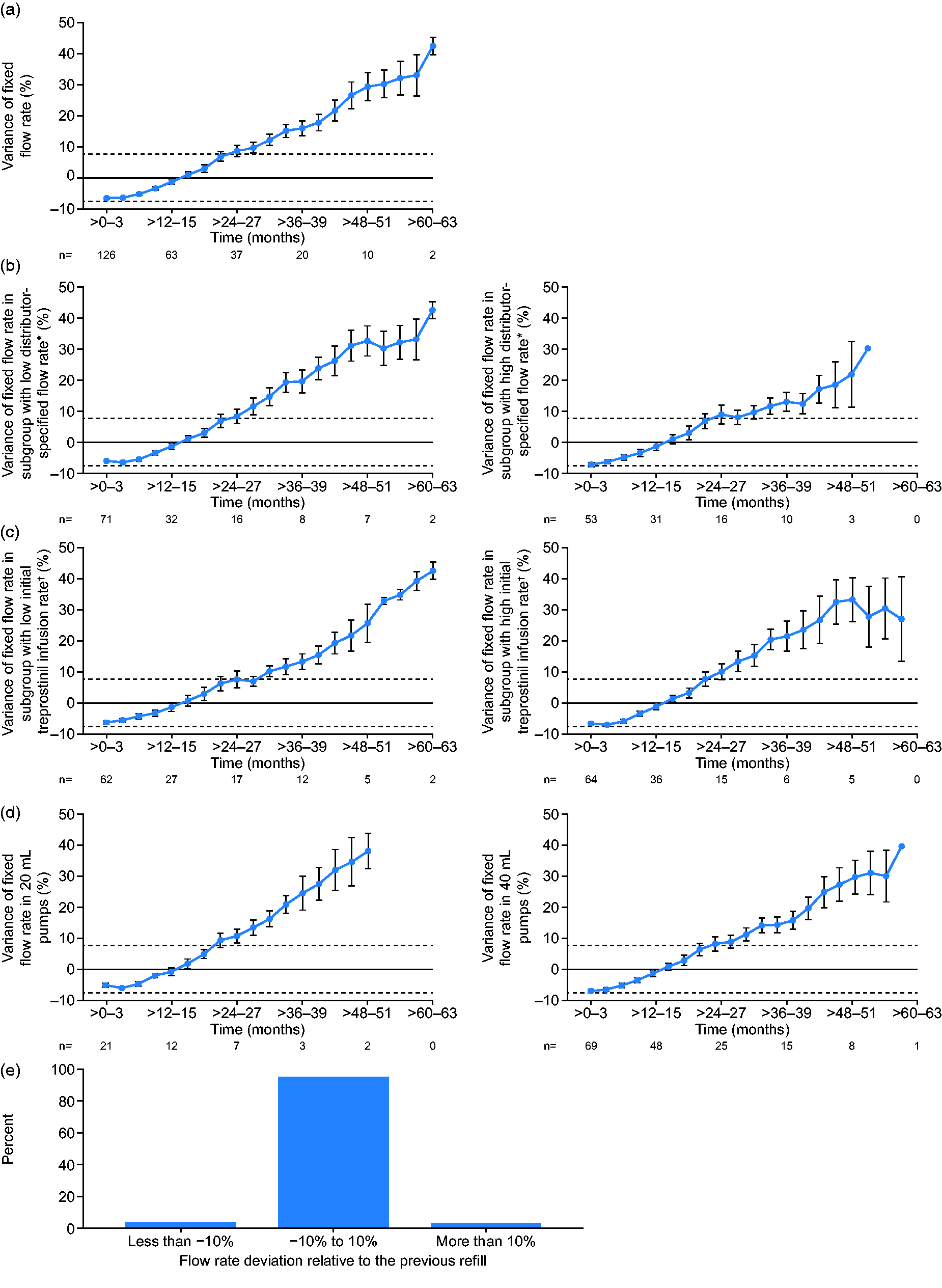
In the present study, we performed the first detailed evaluation of the flow rate variance of the fully implantable LENUS Pro® pump for treprostinil infusion. Our data suggest the following: (1) flow rate variance is time dependent and an increase of the fixed flow rate above +7.5% (which is set as the upper limit of technical deviation by the distributor, based on product information (April 2018) provided by OMT GmbH) is evident after three years; (2) surprisingly, within the first three months after implantation, the mean actual flow rate is lower than specified by the distributer, although it does not exceed the lower limit of technical deviation (−7.5%); (3) regardless of the initial absolute dose of treprostinil, the initial distributor-specified fixed flow rate, or the pump size, we observed a steady flow rate variance over time, and (4) the individual flow rate deviation from the previous refill was mostly within the range of the technical deviation.
Reports of flow rate variance with the LENUS Pro® pump have been published previously.3,5,8 The constant flow rate is regulated by a glass capillary, and it was speculated that chemical substances within the intravenous treprostinil sodium formulation slowly cause alterations within the glass capillary over a long period of time; 3 this is consistent with the results of the current study in which relevant flow rate variances only occurred in the third year after pump implantation. In the recently published long-term safety study of the LENUS Pro® pump, variances of the non-adjustable flow rate were classified as pump defects. 5 However, clinically relevant events (defined as serious adverse events leading to hospitalization or pump exchange) caused by extreme flow rate variance were reported for only two of 129 pumps during long-term follow-up: in one case, the variance of flow rate resulted in cardiogenic shock requiring pump replacement, and in the other case, the patient developed cardiac decompensation but recovered without requiring pump replacement (instead, the treprostinil dosage was adjusted to the increased flow rate). 5 Those rare (0.1% in the present study) but extreme individual deviations from the previous refill might be able to trigger a clinically relevant adverse event. In contrast, the potential of the steady, slow flow rate variance over time to cause a clinically relevant event (taking into account previous publications3,5,9) seems low if routine monitoring of the actual flow rate and compensatory adjustment of the treprostinil dosage are performed on a monthly basis at an expert pulmonary hypertension center.
Limitations of the current study include its uncontrolled and retrospective design without direct assessment of causality of the flow rate variance. The study is not an analysis of a medical device registry (which is not available for this pump), nor is it industry-sponsored. To ensure consistent measurement and complete documentation of all refills during follow-up, our study only included pumps that were systematically refilled by a specialized healthcare service with supervision by our centers. Patients who died directly after implantation or refused the specialized healthcare service were not included. Although the study included only a subset (40%) of all pumps implanted in Germany during the study period, we believe that any resulting bias is likely to be small as we suspect a systematic flow rate variance.
Flow rate variance was also reported for another fully implantable treprostinil pump system (SynchroMed® II). Bourge et al. observed a decreasing flow rate with subsequent adjustment of the dose, and noted that this was at least partly attributable to “back pressure” from the special catheter 10 used in their study.11,12
In conclusion, during long-term application of intravenous treprostinil via the fully implantable LENUS Pro® pump, the actual flow rate differs from the distributor-specified fixed flow rate, starting below the expected rate in early months and increasing slowly but steadily above the expected rate with longer term use. Taking into account previous publications,3,5,9 the results of this study show that the variance has the potential to cause clinically relevant events. In rare cases with extreme deviations even life-threatening events can occur. Such extreme deviations require direct referral of the patient to the managing expert center. Frequent assessment of the flow rate and compensatory adjustments of the treprostinil dose are therefore considered mandatory and should be performed in expert centers. Fully implantable treprostinil pump systems need further technical advances.
Footnotes
Acknowledgments
Editorial assistance was provided by Dr Claire Mulligan (Beacon Medical Communications Ltd, Brighton, UK).
Conflict of interest
Richter has received support from Bayer Pharma AG, and speaker fees from Actelion and Bayer. Satenik Harutyunova has received speaker fees from Actelion and OMT and travel/accommodation/meeting expenses from Actelion, OMT, MSD, Bayer, GSK, United Therapeutics. Gall has received fees from Actelion, AstraZeneca, Bayer, GSK, Janssen-Cilag, Lilly, Novartis, OMT, Pfizer, and United Therapeutics. Gerhardt has received personal fees for lectures from Actelion, Bayer, GSK and United Therapeutics, and his institution has received research grants from Actelion, Bayer, Novartis, and United Therapeutics. Grünig has received fees for lectures and/or consultations from Actelion, Bayer, GSK, MSD, United Therapeutics, and Pfizer. Halank reports personal fees for lectures and consultations and travel/accommodation/meeting expenses from Actelion, AOP Orphan/OMT, AstraZeneca, Bayer, BERLIN CHEMIE, Boehringer, GILEAD, GSK, MSD, Novartis, and Pfizer, outside the submitted work. Lange has received speaker fees, honoraria for consultations, and research funding from Actelion, AOP Orphan Pharmaceuticals, Bayer, GSK, MSD, Pfizer, and United Therapeutics. Neurohr has received honoraria for lectures and/or served on advisory boards for Bayer, GSK, Actelion, and OMT. Nickolaus reports no conflicts of interest. Olsson has received personal fees from Pfizer, Actelion, Bayer, GSK, and United Therapeutics, outside the submitted work. Opitz’s institution has received speaker fees and honoraria for consultations from Actelion, Bayer, GSK, and Pfizer. Seyfarth has received fees for consulting and/or lectures and conference sponsorship from Actelion, Bayer, and GSK. Ghofrani has received consultancy fees from Bayer, Actelion, Pfizer, Merck, GSK, and Novartis; fees for participation in advisory boards from Bayer, Pfizer, GSK, Actelion, and Takeda; lecture fees from Bayer HealthCare, GSK, Actelion, and Encysive/Pfizer; industry-sponsored grants from Bayer HealthCare, Aires, Encysive/Pfizer, and Novartis; and sponsored grants from the German Research Foundation, Excellence Cluster Cardiopulmonary Research, and the German Ministry for Education and Research. Ewert has received speaker fees and fees for participation in advisory boards from United Therapeutics, OMT, Pfizer, GlaxoSmithKline (GSK), Actelion, Novartis, Bayer HealthCare, and Encysive/Pfizer; grants from Actelion and Boehringer Ingelheim; and publication support and industry-sponsored grants from OMT. All other authors report no conflicts of interest.
Ethical approval
Data collection and analyses were approved and the need to obtain written informed consent from each patient was waived by the ethics committee of the Faculty of Medicine at the University of Giessen (Approval No. 241/16).
Funding
Editorial assistance was funded by the University of Giessen.
