Abstract
Pulmonary arterial hypertension (PAH) is a progressive disease, which can be potentially fatal. The management of a complex disease like PAH requires a multidisciplinary approach from a team consisting of physicians, nurses, social workers, and pharmacists. Adherence to PAH-specific therapy is one of the key factors in the management of this disease. Poor adherence to treatment is a common problem in PAH as it is in many chronic diseases. Management of medication interruptions is a challenge in patients with PAH that can lead to negative consequences. However, for most PAH-specific drugs, there are no clear guidelines on how to manage temporary or abrupt medication discontinuations. In this review, we summarized the available literature and provide suggestions on how to manage interruptions of PAH-specific therapies.
Keywords
Introduction
Pulmonary arterial hypertension (PAH) is a rare, rapidly progressive, debilitating, and potentially fatal pulmonary vascular disease. 1 The pathobiology of PAH includes pulmonary vasoconstriction, vascular remodeling, endothelial dysfunction, smooth muscle proliferation, and in situ thrombosis. These changes increase the pulmonary vascular resistance that eventually leads to right heart failure and death.
Several pathways are involved in the pathogenesis of PAH, including the nitric oxide (NO), endothelin (ET), and prostacyclin (PGI2) pathways. Besides calcium channel blockers (CCBs), current Food and Drug Administration (FDA) approved therapies for the treatment of PAH act on these pathways. Nitric oxide activates soluble guanylate cyclase (sGC) in the vascular smooth muscle cells to form cyclic guanosine monophosphate, which causes pulmonary vasodilation. The phosphodiesterase-5 (PDE5) inhibitors and the sGC stimulator riociguat act on this pathway. Endothelin-1 exerts its effects on the smooth muscle cells via the ET-A and ET-B receptors to cause vasoconstriction and cell proliferation. The endothelin receptor antagonists (ERAs) act on this pathway. Prostacyclin acts on the IP receptor, leading to production of cyclic adenosine monophosphate and subsequent vasodilation with antiproliferative effects. Drugs targeting this pathway include the PGI2 analogues and the PGI2 IP receptor agonist, selexipag. 2,3 None of the currently available PAH therapies are curative or act directly on pulmonary vascular remodeling. In recent years, improved understanding of the pathogenesis of PAH has led to the development of new therapeutic targets, but at this time, more research is needed to support the use of these investigational agents in PAH. 4 -9
The REVEAL registry reported a 5- and 7-year survival rate for patients with PAH of 57% and 49%, respectively. 10,11 Given this high mortality, early diagnosis and adequate treatment are of paramount importance. 12,13 Studies showed that PAH-specific therapies improve functional class, 6-minute walk distance (6MWD) and pulmonary hemodynamics as well as prolong time to clinical worsening. 14 Pulmonary arterial hypertension–specific therapies also reduce PAH-related hospitalizations and, based on meta-analyses, may improve survival. 15,16 Combination therapy further reduces clinical worsening and improves exercise capacity, functional class, and hemodynamic status compared to monotherapy. 15,17 -21
The recently released proceedings of the Sixth World Symposium on pulmonary hypertension (PH) emphasized the need to use dual combination therapy as the initial treatment of patients with PAH, except in selected circumstances. 12 Furthermore, in patients who have a persistent moderate or high mortality risk, triple combination therapy is advised. Despite these recommendations, patients frequently interrupt or discontinue therapies due to a myriad of reasons. 22 However, there are limited data on the effect of these treatment interruptions or discontinuations. In addition, there is scarce information on how PAH-specific medications need to be reintroduced after an interruption.
Data from other diseases showed that treatment interruption ranges from 19.3% to 94%. 22 -25 A report published by the World Health Organization (WHO) found that only 50% of patients in developed countries with chronic conditions adhere to their prescribed medication regimens. 26 A meta-analysis of 569 studies of nonpsychiatric medication adherence by DiMatteo revealed an average nonadherence rate of 24.8%, with the lowest adherence rate in patients with diabetes, pulmonary, or sleep disorders. 27 In patients with heart failure, medication adherence rates vary widely from 2% and 93%, based on differences in definition and tools used to measure medication adherence. 28 Importantly, low adherence is associated with an increase in heart failure exacerbations, hospitalizations, and mortality. 29 Similarly, in patients with systemic arterial hypertension, the rate of nonadherence is high at 50% to 80%. 26
Limited information exists on the adherence to PH medications. A study by Grady et al published in 2018 measured PH-targeted therapy adherence using a Morisky Medication Adherence Scale 8 (MMAS-8) questionnaire. 30 The authors noted a 47.9% rate of high adherence (MMAS score < 6), 40.3% rate of moderate adherence (MMAS scores 6-7), and 11.8% rate of low adherence (MMAS score of 8). Interestingly, the factors associated with poor adherence included a higher dosing frequency, greater length of time on PAH-specific therapy, combination therapy compared to monotherapy, and lack of pill organizer use. 30 Likewise, Studer et al showed that adherence was higher among patients on combination therapy versus monotherapy (90.2% vs 46.6%). 31 A more recent study reported a high adherence rate to sildenafil and tadalafil of 94% in patients with PAH, while a study by Waxman et al reported a suboptimal adherence rate of 46.8% for the same drugs. 23,32
Pulmonary arterial hypertension imparts a significant cost burden on the health-care system. 25,33 -36 Sikirica et al found that post-initiation of index therapy with PAH-specific drugs, the medical costs decreased, but this was offset by the pharmacy costs that increased. 25 Berger et al showed similar findings with sildenafil as the index therapy drug. 37 Burger et al evaluated the cost associated with use of PGI2s and found that there was an increase in the overall health-care costs as well as PAH-related health-care costs after initiating this therapy. This increase was mainly driven by the pharmacy cost of the medications. 36 Although we know that medication nonadherence in other chronic diseases leads to increased health-care costs, the economic impact of medication nonadherence in PAH has not been directly studied. 38 The white paper by express scripts estimated that higher adherence in patients with PAH led to lower medical expenses for patients (USD$13 000 per patient per year). 39
One study describes clinical worsening after withdrawal/discontinuation of PAH-specific medications. In this study, sildenafil withdrawal in patients (n = 9) with PAH with WHO functional classes 2 to 4 resulted in worsening functional class and drop in 6MWD in the majority of patients. 40 A few reports describe rebound PH with life-threatening hemodynamic deterioration after abrupt withdrawal of NO. 41 -43 In fact, lambs treated with inhaled NO (40 ppm) for 24 hours had a doubling in ET-1. 44 Abrupt discontinuation of inhaled NO in these animals resulted in a 78% increase in pulmonary vascular resistance, a response that was prevented by treatment with an ET-A receptor antagonist. 44
Medication Adherence Hurdles
It is important to appropriately counsel patients about the importance of strict adherence to their prescribed medication regimen. Medication adherence is traditionally defined as the extent to which an individual follows recommendations from a health-care provider with respect to timing, dosage, frequency, and duration of a particular therapy. 45,46 Beyond the safety concerns of using PAH-specific medications in patients who are nonadherent, the efficacy of the intended regimen becomes uncertain, directly affecting decisions regarding the dose, number, and type of treatments. In fact, poor adherence directly impacts patient outcomes, increases health-care costs, and complicates the disease management. 47 -49
Medication nonadherence is a multifaceted health-care problem that includes factors related to the patients, health-care providers, disease, and the drug (Table 1). Nonadherence can occur at different stages from obtaining/filling the prescription to taking the recommended dosage and observing drug titration. 45 Medication nonadherence can be intentional or unintentional.
Factors Impacting Patient Adherence to Medications.a

a Table modified from References 22 and 40.
Intentional nonadherence refers to a deliberate and rational process on the part of the patient. It is frequently motivated by patients’ beliefs about their disease, prognosis, treatment, as well as their personal or others’ experiences with prior or current treatments. For instance, patients may become skeptical after reading the drug’s package insert or hearing about unpleasant side effects from physicians or other people. To overcome this type of nonadherence, health-care personnel need to investigate the reasons for not following the indicated treatment and provide the necessary education/support.
Unintentional nonadherence refers to unplanned occurrences, mainly driven by a lack of capacity or resources to comply with a certain medication regimen. It may be the result of forgetfulness, inattentiveness, lack of health literacy, or adequate insurance coverage. 45,46 This type of nonadherence requires patient education, simplification of the regimen, setting reminders, and/or finding support to encourage patients to take their medication.
Factors Affecting Medication Adherence
Forgetfulness is reportedly the most common reason for medication nonadherence. 46 Certain medications that are administered 3 or more times a day, such as sildenafil, riociguat, oral or inhaled treprostinil, and inhaled iloprost, can pose challenges to patients who often forget to take the mid-day dose due to work or other daytime chores. An important factor is the patient’s limited understanding of his or her disease and the poor prognosis it carries when inadequately treated.
There is a growing number of generic medications (sildenafil, tadalafil, ambrisentan, intravenous treprostinil, etc). Transition to a generic medication may be challenging since it may result in losing financial assistance or specialty pharmacy services. Copays and copay assistance programs may vary significantly depending on the medication and type of insurance (eg, commercial insurance vs Medicare, Medicaid, or other federal- or state-funded health-care program).
Gaps or delays in insurance coverage are relatively common. The paperwork required for new prescriptions, refills, and prior authorizations can lead to delays in starting medications or create unwanted interruptions. Changes in health insurance may affect the coverage of certain PAH medications, for instance, a particular insurer may cover the use of one but not all commercially available PDE5 inhibitors or ERAs. Drug delivery by specialty pharmacies may be affected if assistance programs are not renewed on time or the patient no longer meets the programs’ criteria to be eligible for assistance. There are digital platforms (eg, ZappRx) that offer solutions to streamline the process of collecting information (enrollment forms, digital patient consent) and monitoring the status of a prescription (prior authorization and prescription status tracking), with the goal of reducing the time from prescription to reaching the patient.
Oral medications are easier to take; but depending on the specific medication, there are important factors to consider. For instance, women of childbearing potential require enrollment in the Risk Evaluation and Mitigation Strategy Program before receiving treatment with ERAs (bosentan, ambrisentan, and macitentan) and sGC stimulator, riociguat. This drug safety program is required by the FDA to reduce the risk of fetal harm of certain medications. 50 As part of this program, women of childbearing potential must have a negative pregnancy test prior to the initiation of therapy, monthly during treatment and within 1 month of stopping it. In addition, given the potential for hepatotoxicity, bosentan requires monthly serum transaminase and bilirubin determinations. 51 In contrast, ambrisentan and macitentan do not require routine liver testing, and these determinations are performed when clinically indicated. Furthermore, due to the potential to induce anemia, a hemoglobin level at baseline, 1 month, and periodically thereafter is generally obtained when treating patients with ERAs. 52
Inhaled PAH-specific therapies have their own challenges since the delivery systems require additional time for assembly and administering the drug. Furthermore, these drug-specific inhalation systems need to be adequately maintained and transported when traveling. Fortunately, dry-powder inhalations systems are currently being tested (LIQ861, Liquida Technologies, NCT03399604). 53
Parenteral PAH therapies require proper maintenance of the intravenous or subcutaneous access, careful handling of the pump and tubing, and proficiency to reconstitute the medication. Fortunately, when patients achieve a stable dose of intravenous PGI2, they may consider the delivery of prefilled cassettes that reduce the time spent in preparing the medication.
Side effects have an impact on how well patients adhere to a particular medication regimen. For instance, side effects that are unexpected or poorly tolerated can lead to medication discontinuation, particularly when they compound on disease-related symptoms. 54 Even though the side-effect profile is broadly shared by all the medications in a specific treatment pathway, the type and frequency of side effects may vary for each medication. The PDE5 inhibitors can cause headache, dyspepsia, nausea, visual disturbances, nasal congestion, sinusitis, myalgia, back pain, and so on. 55,56 Endothelin receptor antagonists can be responsible for peripheral edema, headache, dyspepsia, oligospermia, anemia, nasal congestion, nasopharyngitis, and so on. 57 -59 The sGC stimulator riociguat can induce hypotension, headache, dizziness, dyspepsia, vomiting, diarrhea, anemia, and so on. 60 The side effects of PGI2 analogues depend in part on the route of administration, but in general are tachycardia, hypotension, flushing, skin rash, headache, diarrhea, nausea, vomiting, limb, join and jaw pain, and so on. 61 -66 The PGI2 IP receptor agonist, selexipag, has a similar side-effect profile as the PGI2 analogues, in addition to anemia. 67
To improve PAH medication adherence, it is critical that patients actively participate in their treatment. Patients need to understand the overall reasons and goals of treatment as well as some essential aspects of the pharmacokinetic and pharmacodynamics of the drug so that they know the rationale behind the dosing schedule, titration, and occurrence of side effects. It is important that patients have good communication with the PH team, in order to receive comprehensive care that can identify, prevent, and mitigate any barriers to treatment adherence.
Approaching a Temporary Medication Discontinuation?
As a general rule, PAH-specific medications should not be suddenly stopped. In the event that a PAH-specific medication needs to be discontinued, a downtitration in a monitored environment is usually recommended since hemodynamic decompensation might occur. Abrupt discontinuation of PAH-specific therapies poses unique challenges since there are limited data to guide decisions on how to restart and titrate medications in this particular situation. It remains unclear what dose would be safe and effective to restart PAH-specific therapies at after a brief discontinuation and how quickly to uptitrate. It is uncertain whether the uptitration to the previously achieved dose should be done following usual recommendations or in an accelerated manner based on tolerance.
Pulmonary arterial hypertension–specific medications are usually titrated up after initiation, except for macitentan, which is given at 1 dose (10 mg/d) and PDE5 inhibitors, which are started at a certain dose (sildenafil at 20 mg every 8 hours and tadalafil at 40 mg/d). In certain conditions in which tolerance may be challenging, PDE5 inhibitors are started at lower doses of 10 mg every 8 hours and 20 mg/d, respectively. Before reinitiation, consideration should be given to other factors that may impact titration, such as renal or hepatic dysfunction, hemodynamic instability, and intolerable side effects.
The drug insert provides recommendations in the case of missing a dose of medication. There is no published evidence on how to approach lapses in the medication treatment of more than 1 dose. We use the half-life of the medication as a guide. In the absence of interactions with other medications, renal or hepatic dysfunction, PAH-specific therapies are eliminated from the system after 4 to 5 half-lives of the last time of use. If < 4 to 5 half-lives have elapsed since the last dose, we start the medication at the currently prescribed dosage. If the time between doses is > 4 to 5 half-lives, then we would start treatment at a lower dose with rapid uptitration based on tolerance. During this process, it is essential to closely supervise the reintroduction of the PAH-specific treatment, particularly paying attention to signs or symptoms of PAH decompensation or side effects.
Phosphodiesterase Type-5 Inhibitors and sGC Stimulator
Pharmacokinetic and pharmacodynamic characteristics of PDE5 inhibitors are indicated in Table 2. Keogh et al showed worsening of the functional class and 6MWD after withdrawal of sildenafil. 40 Sudden cessation of sildenafil can cause rapid clinical worsening, acute right heart failure, and decrease in 6MWD. 55 However, no reports exist on the effect of sudden cessation of PDE5 inhibitors on pulmonary hemodynamics. There are no reports on potential consequences of abrupt riociguat interruptions.
Pharmacokinetic and Pharmacodynamics Properties of PDE5 Inhibitors and Riociguat.

Abbreviations: CrCl, creatinine clearance; CYP, cytochrome; PAH, pulmonary arterial hypertension; PDE, phosphodiesterase type; qd, daily; TID, 3 times a day;
If a patient misses a dose of sildenafil, the drug should be taken immediately, unless there is less than 4 hours to the next regular administration time. It is advised that no more than 1 dose of sildenafil should be taken at any time. Similarly, tadalafil should be taken as soon as the patient remembers the lapse, but without taking more than a dose per day. Although no recommendations are provided for patients who missed ≥ 1 dose of PDE5 inhibitor, we use the time from the last dose as a guide. If < 4 to 5 half-lives (16-20 hours for sildenafil or 60-75 hours for tadalafil) have passed from the last PDE5 inhibitor dose, we resume the drug the previously tolerated dose; otherwise, we would restart sildenafil and tadalafil at ≤ 20 mg every 8 hours and 40 mg/d, respectively. If the patient was receiving higher doses of sildenafil, we would start at 20 mg every 8 hours and rapidly titrate based on tolerance. If a patient misses a dose of riociguat, he or she should take the next dose at the regular time interval. If patients received no riociguat for ≥ 3 days, the drug is usually started at 1 mg 3 times a day with titration as tolerated (consider risk/benefits of uptitrating riociguat at an accelerated pace). 60
Endothelin Receptor Antagonists
Pharmacokinetic and pharmacodynamic characteristics of ERA inhibitors are presented in Table 3. There is limited experience with abrupt discontinuation of ERAs in patients with PAH. If a patient misses a dose of bosentan, it should be taken as soon as possible, unless there is less than 4 hours to the next dose. Similarly, for ambrisentan and macitentan, the missed dose should be taken as soon as possible and the next dose the following day at the regular time. Patients should not take > 1 dose at the same time to make up for a missed dose. 57 -59 If ERAs are stopped abruptly and < 4 to 5 half-lives (20-25 hours for bosentan and 60-75 hours for ambrisentan) have passed, we resume the drug at the previously tolerated dose. If > 4 to 5 half-lives have elapsed since discontinuation of the drug, then we assess risk/benefits for initiating bosentan and ambrisentan at full dose. Macitentan has a unique dose, so it is restarted at 10 mg/d. In cases of associated liver dysfunction, consideration should be given to stopping the drug completely and switching to another pulmonary vasodilator, or lowering the dose of the ERA with careful uptitration and monitoring of liver function tests.
Pharmacokinetic and Pharmacodynamics Properties of Endothelin Receptor Antagonists (ERAs).

Abbreviations: BID, twice daily; CYP, cytochrome; qd, daily;
Prostacyclin Analogues and Prostacyclin IP Receptor Agonist
Pharmacokinetic and pharmacodynamic characteristics of PGI2 analogues and the PGI2 IP receptor agonist selexipag are presented in Table 4. There is limited experience with abrupt discontinuation of these medications in patients with PAH. Patients who had interruptions of > 3 days of selexipag in the GRIPHON study were retitrated up from 200 µg twice daily dosing. None of these patients experienced any acute clinical or hemodynamic deterioration during this time. 24 Given that parenteral PGI2 analogues are the most potent medications for PAH, abrupt discontinuations can have catastrophic consequences, including the rapid development of acute right ventricular failure. 68 -70 Drug inserts recommend avoiding abrupt withdrawal or sudden large reductions in dosing. No specific recommendations are provided in case of sudden cessation of the parenteral medications. 61,63 If the patient is chronically treated with a parenteral PGI2 analogue, we would consider risk/benefits of restarting the drug at the previously tolerated dose. Retitrating the drug from the lowest dose may be detrimental to the patient. In certain cases, we would start the parenteral PGI2 at a dose 10% lower and titrate as tolerated.
Pharmacokinetic and Pharmacodynamics Properties of Prostacyclin Analogues.
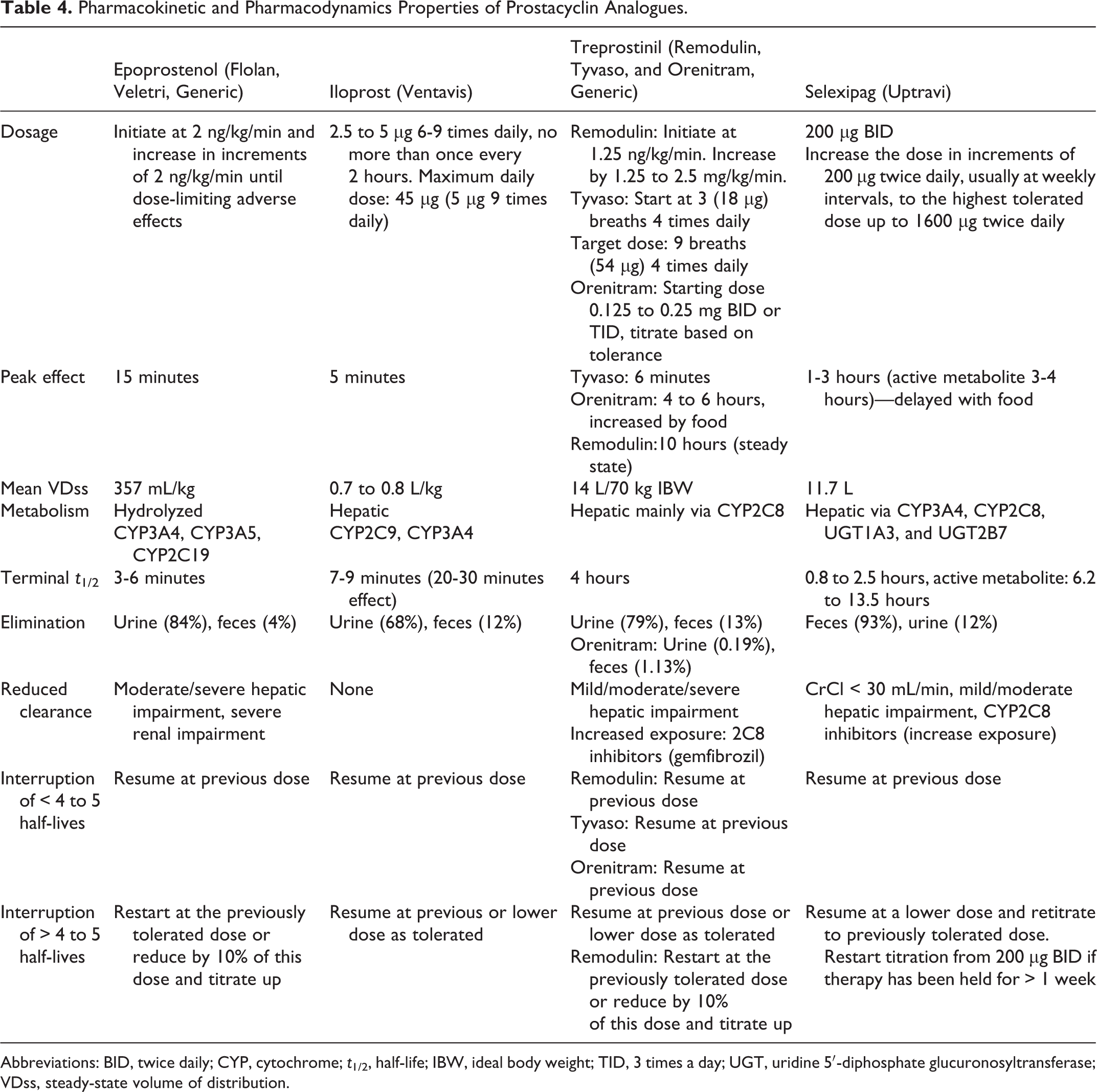
Abbreviations: BID, twice daily; CYP, cytochrome;
In the event of missing a dose of inhaled treprostinil or iloprost, oral treprostinil, or selexipag, patients should take the drug as soon as possible, except when it overlaps with the next scheduled dose. If > 4 to 5 half-lives elapsed between doses, risk/benefits of resuming at the same or lower dose should be considered. If selexipag is interrupted for ≥ 4 to 5 half-lives (up to 65 hours or ∼3 days for active metabolite), the medication should be started at a lower dose and then titrated. 67
Calcium Channel Blockers
Pharmacokinetic and pharmacodynamic characteristics of CCBs are presented in Table 5. Calcium channel blockers are used for patients with PAH (mostly idiopathic PAH) who show a positive response to a vasodilator challenge (usually inhaled NO) during right heart catheterization. 71 Commonly used CCBs for PAH are diltiazem, nifedipine, and amlodipine. Sudden withdrawal of CCBs in patients with angina has been reported to cause coronary ischemia. 72,73 In the case of PAH, sudden cessation of CCBs can cause clinical deterioration. The 2015 European Society of Cardiology/European Respiratory Society guidelines recommend adding a pulmonary vasodilator prior to stopping CCB therapy in patients who are nonresponders to this treatment, in order to prevent rebound PAH. 74 No specific recommendations are made in the drug inserts regarding treatment discontinuations in patients with PAH. In case of abrupt discontinuation of CCBs, the next dose should be taken as soon as possible. If more than 4 to 5 half-lives have elapsed since discontinuation, the medication should be started at a slightly lower dose and gradually titrated upward. If the patient experiences clinical signs of worsening PAH, another pulmonary vasodilator with a shorter half-life such as sildenafil can be added while the CCB is being titrated.
Pharmacokinetic and Pharmacodynamics Properties of Calcium Channel Blockers.
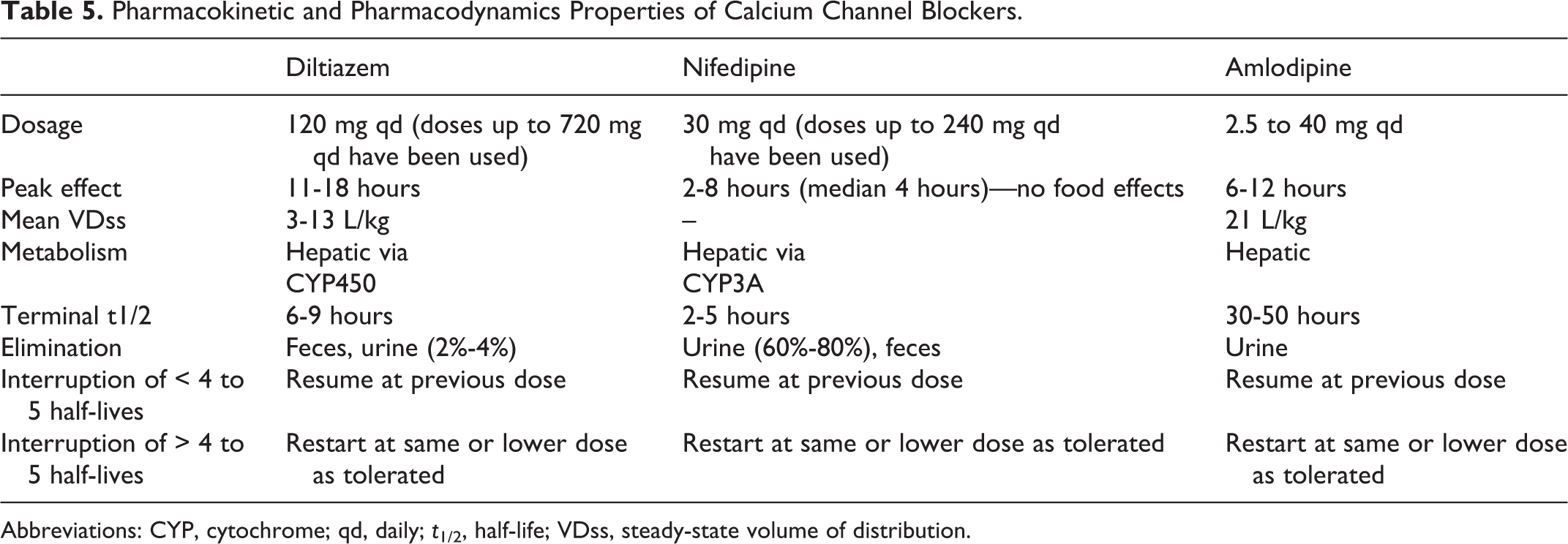
Abbreviations: CYP, cytochrome; qd, daily;
Medication Interruption in Patients Without Oral Access or With Feeding Tubes
In certain situations, patients are given orders for nothing by mouth with the resultant inability to swallow the PAH therapies. Examples of these conditions include acute encephalopathy, bowel obstruction, ileus, gastrointestinal perforation, dysphagia, intractable vomiting, or immediate postoperative periods after abdominal surgeries. In these circumstances, oral medications need to be temporarily discontinued. If the expected duration of interruption is prolonged, patients may need to be transitioned to inhaled or parenteral PAH medications.
In mechanically ventilated patients, enteral access is obtained with a naso- or orogastric tube or nasoenteric feeding tube. In these cases, medications need to be crushed for administration. Sildenafil is also available as an oral suspension and can be used through feeding tubes. 55 No recommendation is provided by the FDA regarding crushing sildenafil tablets. 55 Tadalafil tablets should not be crushed or split as this can result in getting a higher or lower dose than required. 56 A study by Pettit et al demonstrated the stability of an extratemporaneously prepared tadalafil suspension for 91 days. 75 This could be used for patients who are unable to swallow tablets. Riociguat tablets can be crushed and mixed with water or soft foods like applesauce. 60 Endothelin receptor antagonists should not be split, crushed, or chewed based on FDA recommendations. Bosentan, however, does come in a formulation for pediatric patients that can be dispersed in water to make a suspension, which can then be used through enteral feeding tubes. Given the teratogenic potential, nurses and pharmacists should be educated on taking adequate precautions like wearing gloves and masks while handling ERAs and riociguat. 58,76,77
Administration of Inhaled Therapies in Patients on Mechanical Ventilation
Inhaled therapies for PAH include inhaled treprostinil (Tyvaso) and iloprost (Ventavis). Tyvaso is to be administered only via a specialized Tyvaso inhalation system and Ventavis via the I-neb Adaptive Aerosol Delivery (AAD) System or the Prodose AAD System. 64 No recommendations have been provided by the FDA for using these medications in ventilated patients; but some physicians used them in mechanically ventilated patients via different adaptors. 78 -81 These methods need further investigation, to assure adequate drug delivery. More commonly, patients on Tyvaso or Ventavis who require mechanical ventilation are temporarily transitioned to a continuous inhalation of epoprostenol (Flolan, Veletri) or NO. 76,82
Management of PAH-Specific Therapy in Patients Under Palliative or Hospice Care
Pulmonary arterial hypertension is a debilitating disease with a significant symptom burden that impairs the patients’ health-related quality of life (HRQoL). 83 -85 The PAH-specific medications can elicit side effects like headaches, jaw pain, nausea, diarrhea, leg edema, and so on that can substantially affect patients’ HRQoL. Parenteral therapy can impair quality of life by causing local site pain and catheter-related complications such as catheter malfunction and infection. As the disease progresses, patients can develop acute on chronic right ventricular failure despite PAH-specific treatment. 1 The main focus of palliative care is symptom control and improving quality of life. Palliative care evaluation is recommended at earlier rather than later stages of the disease. 86
Patients with PAH can be continued on parenteral therapies when referred to palliative care. Withdrawal of parenteral PAH therapy, especially in patients who are on high doses, may lead to acute clinical deterioration and right heart failure. However, some patients may want to stop this treatment, given side effects or other reasons. There are no formal guidelines to guide clinicians in titrating these medications off; but some recommendations for PGI2 withdrawal have been outlined by the Care of Pulmonary hypertension patients at End-of-life Collaborative (Table 6). 87 If continued, parenteral PAH therapies can be administered at home with palliative care under the guidance of a PH physician. 86 Other noninvasive interventions include patient education, counseling, information about financial assistance programs, information about insurance coverage, support groups, and offering spiritual support. 86
Summary of COPE Collaborative Recommendations for Prostacyclin Discontinuation During End-of-Life Care.a

Abbreviation: COPE, Care of Pulmonary hypertension patients at End-of-life.
a Table modified from study by Bartlett and Landzaat (2013). 87
Conclusions
Temporary treatment discontinuation of PAH-specific therapies is common. Limited data exist on how to approach temporal interruptions of PAH-specific therapies. Patient engagement can be improved by improved communication between health-care team and patients, patient education, development of care plans, and a multidisciplinary approach. Patients with abrupt medication discontinuation should be monitored closely for signs of clinical deterioration. Both PDE5 inhibitors and ERAs may be resumed at the previously tolerated doses if interrupted for less than 4 to 5 half-lives. It is important to develop a system to report cases of temporary discontinuations of PAH treatments, to be able to provide a more structured guidance when these occur.
Footnotes
Author Contributions
Shraddha Narechania, MD, and Adriano R. Tonelli, MD, MSc, participated in the conception, design, writing, and critical revision of the manuscript for important intellectual content and final approval of the manuscript submitted. Heather Torbic, PharmD, participated in the writing and critical revision of the manuscript for important intellectual content and final approval of the manuscript submitted. Dr Tonelli is the guarantor of the paper, taking responsibility for the integrity of the work from inception to published article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
