Abstract
The Breelib™ nebulizer was designed to reduce iloprost inhalation times for patients with pulmonary arterial hypertension (PAH). In 30 patients with PAH, rapid inhalation of iloprost 2.5 µg using Breelib™ caused significant improvements in invasively measured afterload and cardiac index but not echocardiographic right ventricular strain during 30 min post-inhalation.
To the Editor:
Parameters in PAH patients before and after inhalation of a single dose of iloprost 2.5 µg via the Breelib nebulizer. Data are shown as mean ± standard deviation or median [interquartile range].
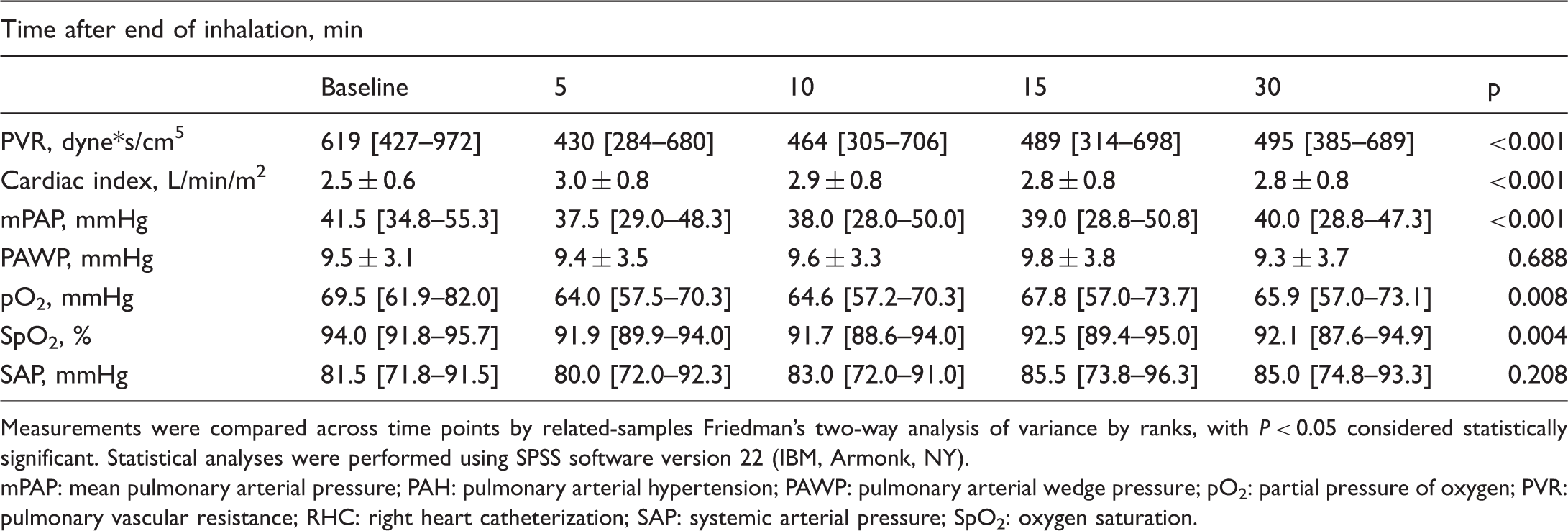
Measurements were compared across time points by related-samples Friedman’s two-way analysis of variance by ranks, with P < 0.05 considered statistically significant. Statistical analyses were performed using SPSS software version 22 (IBM, Armonk, NY).
mPAP: mean pulmonary arterial pressure; PAH: pulmonary arterial hypertension; PAWP: pulmonary arterial wedge pressure; pO2: partial pressure of oxygen; PVR: pulmonary vascular resistance; RHC: right heart catheterization; SAP: systemic arterial pressure; SpO2: oxygen saturation.
Our study shows a substantial decrease in afterload (PVR) after rapid inhalation of iloprost 2.5 µg via the Breelib™ nebulizer in PAH patients. The observed concomitant increase in cardiac index was previously attributed to indirect positive inotropic effects (due to systemic vasodilation and activation of the arterial baroreflex) in PAH. 8 Without causing a clinical relevant adverse event blood oxygenation (also observed in previous studies) was worsening after inhalation.3,9 Therefore, use of the Breelib™ nebulizer in patients with respiratory insufficiency and concomitant high long-term oxygen therapy might not be feasible. Surprisingly, we saw no acute effect of rapid iloprost inhalation on RV strain, although RV strain was recently described as mirroring afterload and RV diastolic function. 10 Therefore, the acute effects of inhaled iloprost or other pulmonary vasoactive drugs on RV function, which have been previously been proposed,11,12 merit further investigation. The clinical need to shorten iloprost inhalation times is unquestionable. Our study supports this concept as rapid inhalation of iloprost 2.5 µg via the Breelib™ nebulizer resulted in a substantial improvement from baseline in PVR and cardiac index.
Footnotes
Acknowledgments
Editorial assistance during the preparation of this article was provided by Dr Claire Mulligan (Beacon Medical Communications Ltd, Brighton, UK).
Authors’ contribution
Richter had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. He contributed as the principal investigator for this study to project oversight, organization, data collection, and writing of the manuscript. Wan contributed to data collection, critical reviewing of the manuscript, and final approval of the version to be published. Ghofrani contributed to the conceptual study design, data collection, writing and critical reviewing of the manuscript, and final approval of the version to be published. Seeger contributed to the conceptual study design, data collection, writing and critical reviewing of the manuscript, and final approval of the version to be published. Gall contributed to the conceptual study design, data collection, statistical analysis, writing and critical reviewing of the manuscript, and final approval of the version to be published. Rieth contributed to the conceptual study design, data collection, writing and critical reviewing of the manuscript, and final approval of the version to be published. Tello contributed to the conceptual study design, data collection, writing and critical reviewing of the manuscript, and final approval of the version to be published.
Conflict of interest
The author(s) declare the following conflicts of interest: Richter has received support from United Therapeutics and Bayer; speaker fees from Actelion, Bayer, MSD and OMT; and consultancy fees from Bayer. Wan has nothing to disclose. Ghofrani has received consultancy fees from Bayer, Actelion, Pfizer, Merck, GSK, and Novartis; fees for participation in advisory boards from Bayer, Pfizer, GSK, Actelion, and Takeda; lecture fees from Bayer HealthCare, GSK, Actelion, and Encysive/Pfizer; industry-sponsored grants from Bayer HealthCare, Aires, Encysive/Pfizer, and Novartis; and sponsored grants from the German Research Foundation, Excellence Cluster Cardiopulmonary Research, and the German Ministry for Education and Research. Seeger has received speaker/consultancy fees from Pfizer and Bayer Pharma AG. Gall has received fees from Actelion, AstraZeneca, Bayer, BMS, GSK, Janssen-Cilag, Lilly, MSD, Novartis, OMT, Pfizer, and United Therapeutics. Rieth has received a research grant from Pfizer and speaker fees from Servier, St. Jude Medical, Cardiokinetix, and Actelion. Tello has received speaking fees from Actelion and Bayer.
Ethical approval
The study was approved by the ethics committee of the Faculty of Medicine at the University of Giessen (approval number 111/16).
Funding
The study was funded by Bayer Vital GmbH, Leverkusen, Germany. Editorial support was funded by the University of Giessen.
Guarantor
MJR.
*These authors contributed equally to this work.
