Abstract
Objective:
Between 2012 and 2017, the U.S. Food and Drug Administration (FDA) approved 10 antidiabetic indicated therapies. Due to the limited literature on voluntarily reported safety outcomes for recently approved antidiabetic drugs, this study investigated adverse drug reactions (ADRs) reported in the FDA Adverse Event Reporting System (FAERS).
Research Design and Methods:
A disproportionality analysis of spontaneously reported ADRs was conducted. FAERS reports from January 1, 2012 to March 31, 2022 were compiled, allowing a 5-year buffer following drug approval in 2017. Reporting odds ratios were calculated for the top 10 ADRs, comparing new diabetic agents to the other approved drugs in their therapeutic class.
Results:
127,525 reports were identified for newly approved antidiabetic medications listed as the primary suspect (PS). For sodium-glucose co-transporter-2 (SGLT-2) inhibitors, the odds of blood glucose increased, nausea, and dizziness being reported was greater for empagliflozin. Dapagliflozin was associated with greater reports of weight decreased. Canagliflozin was found to have a disproportionally higher number of reports for diabetic ketoacidosis, toe amputation, acute kidney injury, fungal infections, and osteomyelitis. Assessing glucagon-like peptide-1 (GLP-1) receptor agonists, dulaglutide and semaglutide were associated with greater reports of gastrointestinal adverse drug reactions. Exenatide was disproportionally associated with injection site reactions and pancreatic carcinoma reports.
Conclusion:
Pharmacovigilance studies utilizing a large publicly available dataset allow an essential opportunity to evaluate the safety profile of antidiabetic drugs utilized in clinical practice. Additional research is needed to evaluate these reported safety concerns for recently approved antidiabetic medications to determine causality.
Plain language summary
Keywords
Introduction
The 2020 Centers for Disease Control and Prevention National Diabetes Statistics Report estimates that 34.2 million people in the United States population has diabetes. 1 Each year, an estimated 1.5 million Americans over 18 years of age are diagnosed with diabetes, still approximately 7.3 million cases are undiagnosed. Diabetes and related complications are the seventh leading cause of death in the United States representing significant mortality. In 2017, the economic burden of diabetes totaled $327 billion in direct and indirect costs. Current data shows the economic impact and prevalence of diabetes will continue to rise with a predicted 60.6 million adults diagnosed with diabetes in the United States by the year 2060.2,3
Type 2 diabetes accounts for 90–95% of all diabetes cases in the United States. 4 This unique patient population faces a significant medication burden in order to manage type 2 diabetes and its comorbid conditions. At time of the diagnosis, type 2 diabetes patients receive medications for an average of five unique indications. Post diagnosis, the average number of disease states patients receive medications for increases to almost seven. 5 With the use of many concomitant drugs, potential risk of adverse drug reactions (ADRs) increases. 6 One study found that ADR related hospital admission increased five-fold in those treated with more than three drugs, and nine-fold in those treated with more than 10. 7 A complete understanding of the safety profile of all drugs used to treat type 2 diabetes patients is essential for effective and safe care.
Between 2012 and 2017, the U.S. Food and Drug Administration (FDA) approved 18 new agents for the management and treatment of type 2 diabetes. These approvals consisted of novel medication classes, expansion of existing classes and combination products. Prior to market availability, the largest scale safety evaluations for new drugs occurs in Phase III clinical trials, with usual treatment populations limited to a few hundred to a few thousand subjects for only the prespecified length of the study. This is an insufficient sample size and duration-of-use to identify rare ADRs or comprehend common events. 8 Through exclusion and inclusion criteria, subjects in Phase III trials also fail to reflect the diversity of the general population (e.g., demographics, comorbidities). Additionally, pre-FDA approval trials do not compare the safety profiles of the unique drugs within medication classes. To overcome these gaps in knowledge, postmarking surveillance is commonly used to identify safety signals and trends. 8
One post-marketing surveillance tool available to analyze the reporting of drugs and adverse events is the FDA Adverse Event Reporting System (FAERS). 9 FAERS is a public database and reporting platform available for use by healthcare professionals, consumers, and manufacturers. Its goal is to improve public health through compiling and analyzing safety reports. FAERS primarily serves the U.S. population, however, anyone may report suspected ADRs in FAERS with corresponding patient, medication, and ADR information. This centralized data source was used to extract a large cache of ADR reports related to the numerous antidiabetic agents approved in a 5-year timeframe. The purpose of this study was to investigate ADRs reported for recently approved antidiabetic medications in FAERS.
Methods
Study design
A disproportionality analysis was completed to examine voluntary ADR reporting trends for recently FDA approved antidiabetic agents. This study utilized de-identified public data and did not require institutional review board approval.
Medication selection
To identify medications approved by the FDA from 2012 to 2017, FDA.gov and Drugs.com were searched.10,11 In addition to the aforementioned sources, UptoDate® and Mircomedex® were utilized to determine each medication’s approval year, generic and brand name, mechanism of action, indication, and international name(s).12,13 From this list, medications indicated for the treatment of type 2 diabetes were identified. A 5-year approval period between 2012 and 2017 was chosen to allow at least a 5-year buffer between the approval of the most recently marketed antidiabetic drugs included in this study and the reports submitted for these newly marketed drugs. This meant reports for the year 2022 were included in this study. Insulin products were excluded due to newly approved products largely being reformulations of existing drugs, in addition to narrow the focus of reported ADRs to the type two diabetes patient population. Additionally, newly approved combination products were excluded due to low ADR reports after database standardization. The 10 antidiabetic agents approved by the FDA in the 5-year period include five glucagon-like peptide-1 (GLP-1) agonists, four sodium-glucose co-transporter-2 (SGLT2) inhibitors, and one dipeptidyl peptidase-4 (DPP-4) inhibitor. Since there were no comparators that met the inclusion criteria for DPP-4 inhibitors, only SGLT2 inhibitors and GLP-1 agonists were included in the analysis. The stepwise process of identifying medications of interest for analysis is displayed in Figure 1.

Stepwise process of identifying antidiabetic medications approved between 2012 and 2017.
Standardization
FAERS data was downloaded from public FDA website. Using the process described by Banda et al. 14 drugs, indications, reactions and outcomes were standardized to Observational Health Data Science and Informatics (OHDSI) Common Data Model V5 Concepts. The mapping of FAERS drug names into RxNorm standard code ingredients and multi-ingredients was completed using the OHDSI Usagi tool, the National Library of Medicine’s Metamap program,15,16 and manual review. FAERS reported adverse events were mapped to Medical Dictionary for Regulatory Activities (MedDRA) by the FDA.
Data collection
FAERS reports from 2012 quarter 1 through 2022 quarter 2 containing the nine anti-diabetic medications of interest were compiled for analysis. The analysis was limited to reports where one of the nine anti-diabetic medications of interest was indicated as the primary suspect (PS) medication for causing the ADR. ADRs were reported out at the MedDRA Preferred Term level. Clinically similar ADRs were collapsed to the respective higher MedDRA level code (Higher Level Term) to be included in the statistical analysis. ADRs condensed to the Higher Level Term (HLT) group included diabetic ketoacidosis and injection site. Reports were further narrowed to a single entry to exclude duplicates of the primary identification number and case numbers. All ADRs listed in each unique report were included in the analysis. Duplicates for primary identification number and case number were removed. Additional data collected were patient age, sex, reporter country, and reporter profession.
Statistical analysis
Patient demographics were analyzed using descriptive statistics. Means and standard deviations were reported for continuous variables and frequencies were reported for categorical variables. New drugs were grouped by mechanism of action in the following categories: GLP-1 receptor agonist and SGLT-2 inhibitor. For each drug, the frequency of all ADRs was evaluated from the time of drug approval for a pre-specified duration so each medication within a drug class would be evaluated for the same time period post-approval (Supplemental Table 1). The 10 most frequent events for each drug within each grouping were selected. Reporting odds ratios (RORs) were calculated for the top 10 ADRs for each individual drug and compared to all other drugs in the same therapeutic drug class. ROR is a measure of reporting disproportionality commonly used for signal detection in pharmacovigilance studies.17,18 The ROR was calculated based on the formula:
Results
A total of 127,525 reports of interest were identified as having an antidiabetic drug of interest listed as the PS within the database for the designated analysis times. Demographics for patients are listed in Table 1, including reporter professions. Patients were more frequently female (47.8%), and between the ages of 18 and 69 (41.6%). Consumers were the most common reporters with 57.5% of all reports for antidiabetic agents, physicians were the next most frequent with 23.0% of reports. The top 10 ADRs for each newly approved anti-diabetic medication can be found in Supplemental Table 2.
Patient and reporter demographics in adverse events reports of newly approved anti-diabetic medications.

ADR reports will not add up consistently to the total amount of reports due to FAERS not requiring all fields to be filled out for reports. Data are number, n, and (%) of reports in FAERS unless otherwise specified.
ADR, adverse drug reaction; FAERS, FDA Adverse Event Reporting System; GLP-1, glucagon-like peptide-1; SGLT-2, sodium-glucose co-transporter-2.
SGLT-2 inhibitors
SGLT-2 inhibitors identified for analysis in the FAERS database were canagliflozin (n = 25,957), dapagliflozin (n = 17,715), and empagliflozin (n = 28,266). ADRs for each drug were assessed for the 32 quarters following each drug approval date, respectively. ADRs occurring frequently for each of these medications included diabetic ketoacidosis, fungal infection, weight decreased, and blood glucose increased (Table 2). The odds of reporting blood glucose increased (1.19; 1.11–1.27), dizziness (1.28; 1.18–1.38), and nausea (1.32; 1.22–1.42) was disproportionally greater for empagliflozin compared to other medications within the drug class. Dapagliflozin was associated with greater reports of weight decreased, although this was not significant (1.074; 0.996–1.16). Canagliflozin had several ADRs present in its top 10 most frequently reported events unique to the medication such as toe amputation, osteomyelitis, and acute kidney injury. Notably, canagliflozin was found to have disproportionally greater RORs for the previously listed ADRs (acute kidney injury 3.42; 3.18–3.71; osteomyelitis 42.2; 34.2–26.5; toe amputation 22.7; 19.4–26.5), as well as fungal infection (1.20; 1.13–1.29) and diabetic ketoacidosis (1.12; 1.08–1.17). Refer to the Supplemental Tables 3 and 4 for analysis by age and sex. A summary of reports by quarter for SGLT2 inhibitors with new indication approval dates and warning dates is presented in Figure 2.
Reporting odds ratios for adverse drug reactions of SGLT-2 inhibitors approved between 2012 and 2017.

Reporting odds ratios are a comparison of each drug to all other drugs in the same therapeutic class for each ADR.
Lower 95% confidence interval > 1.0.
ADR, adverse drug reaction; ROR, reporting odds ratio; SGLT-2, sodium-glucose co-transporter-2.

Timeline of reports by quarter for SGLT2 inhibitors with new indication approval dates and warning dates.
GLP-1 receptor agonists
GLP-1 receptor agonists identified for analysis in the FAERS database were exenatide (n = 16,159), albiglutide (n = 5011), dulaglutide (n = 20,931), and semaglutide (n = 14,066). ADRs for each drug were assessed for the 19 quarters following each drug approval date, respectively. Frequent ADRs identified in each GLP-1 receptor agonists included nausea, vomiting, diarrhea, abdominal pain, and injection site reactions shown in Table 3. In our analysis, dulaglutide and semaglutide were associated with greater reports of gastrointestinal related ADRs including nausea (D: 1.61, 1.54–1.86; S: 1.24, 1.18–1.30), diarrhea (D: 1.61, 1.51–1.72; S: 1.44, 1.34–1.54), and vomiting (D: 1.40, 1.31–1.50; S: 1.50, 1.40–1.60). Nausea and vomiting were frequently reported more often for females, while diarrohea was similar between the sexes (see Supplemental Table 5). Reporting frequencies for nausea, vomiting, and diarrhea were not different between the compared age groups (see Supplemental Table 6). The odds of reporting blood glucose increased was disproportionally greater for dulaglutide compared to the other GLP-1 receptor agonist medications (1.68; 1.59–1.78). Exenatide was found to have disproportionally higher number of reports for injection site reactions (2.34; 2.24–2.45), decreased weight (1.81; 1.70–1.93), and pancreatic carcinoma (15.5; 12.8–18.8). Semaglutide additionally had a disproportionately greater ROR for constipation compared to other medications within the drug class (2.35; 1.35–1.59). Refer to the Supplemental Tables 5 and 6 for analysis by age and sex. A summary of reports by quarter for GLP1 receptor agonists with new indication approval dates and warning dates is presented in Figure 3.
Reporting odds ratios for adverse drug reactions of GLP-1 receptor agonists approved between 2012 and 2017.
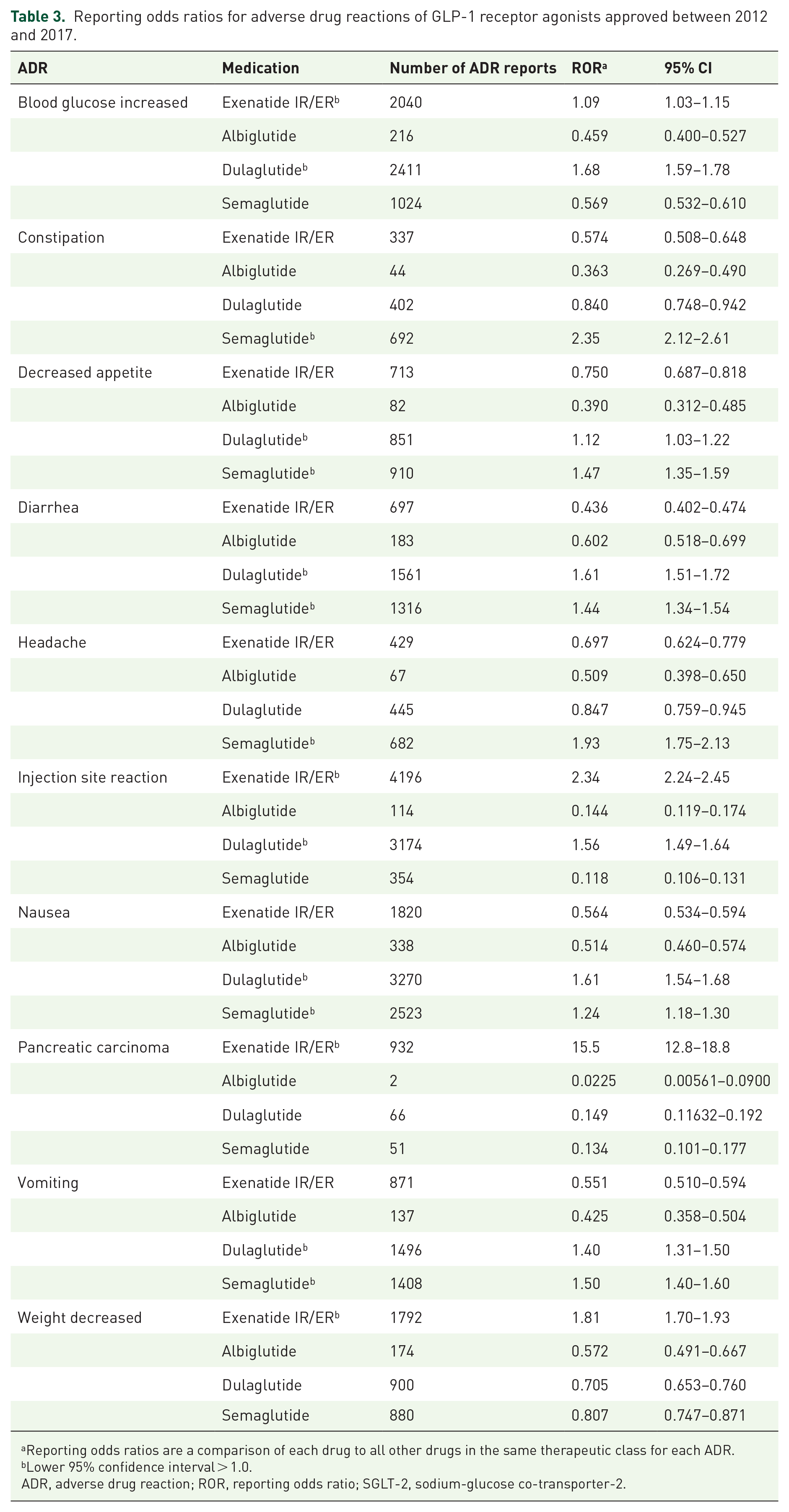
Reporting odds ratios are a comparison of each drug to all other drugs in the same therapeutic class for each ADR.
Lower 95% confidence interval > 1.0.
ADR, adverse drug reaction; ROR, reporting odds ratio; SGLT-2, sodium-glucose co-transporter-2.
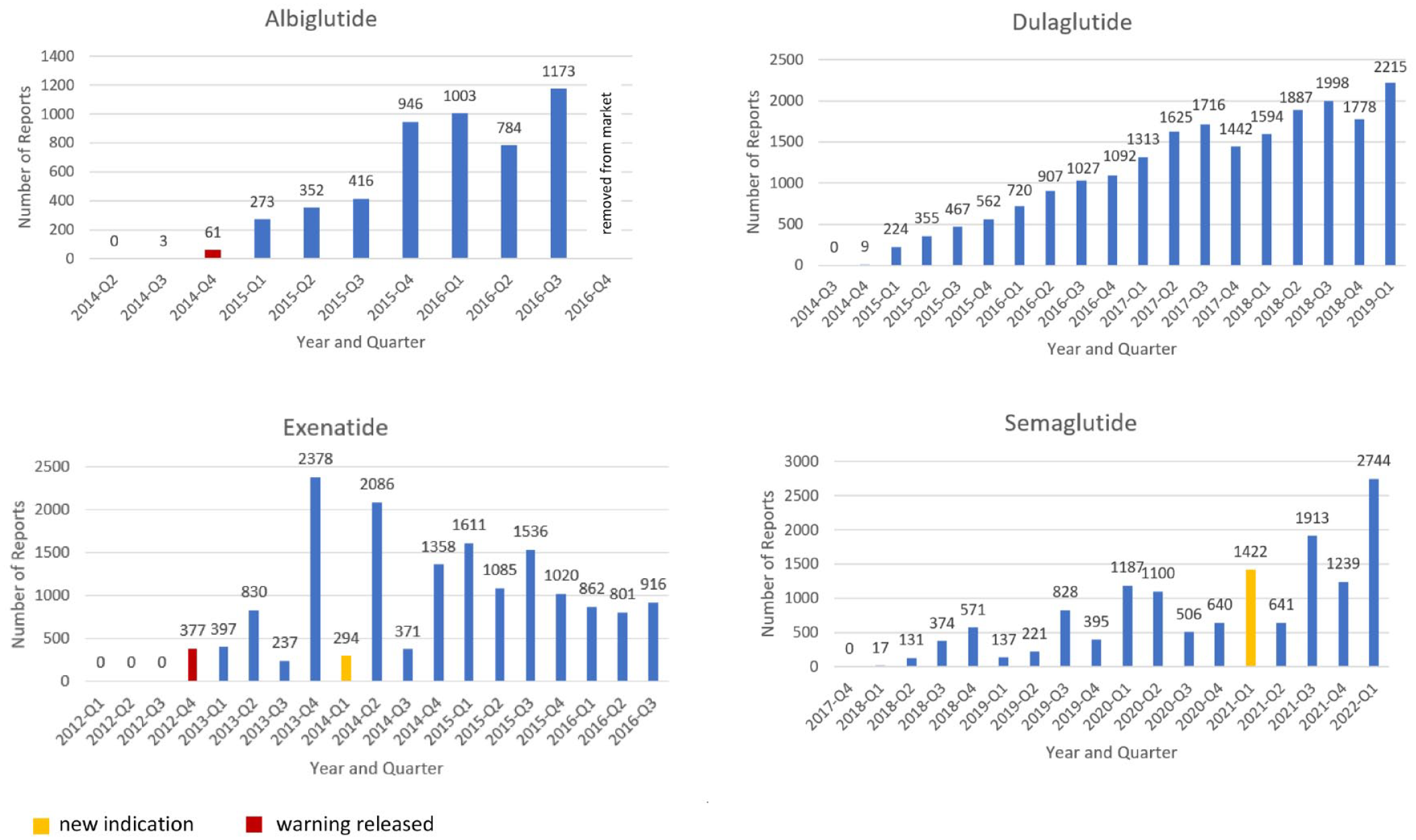
Timeline of reports by quarter for GLP1 receptor agonists with new indication approval dates and warning dates.
Discussion
This pharmacovigilance study provides unique insight into voluntarily reported ADRs for newer antidiabetic agents and an understanding of events reported for different agents within the same drug class. We offer a summary of reports that are supportive of ADRs known to occur with the use of SGLT-2 inhibitors such as diabetic ketoacidosis, urinary tract infection (UTI), and foot/toe amputation.21–23 Due to the nature of the FAERS database and this study being an analysis of voluntarily reported data, factors such as prescribing patterns, utilization rates, clinician opinion and product labeling are important considerations. These factors likely play a role in the number of reports in the FAERS database linked to each medication. Studies have shown canagliflozin and empagliflozin are more commonly initiated than dapagliflozin. 24 This is reflective of the number of ADRs identified in this study (Supplemental Table 1). Additionally, in 2016, the SGLT2 products canagliflozin and empagliflozin both had changes to their product labeling. Canagliflozin received a boxed warning for amputation, and empagliflozin added an indication for reduction in cardiovascular events and death. 24 Following these changes empagliflozin became more commonly prescribed than canagliflozin. 24 Canagliflozin’s label warning may have increased the number of ADRs associated with lower limb infection and amputation reported to the FAERs database as the linkage became widely known. However, we only see a slight spike in reporting after the safety announcement on May 5th, 2017 so this is unlikely.
Our analysis also identified a greater odds of reporting UTI and fungal infection for canagliflozin compared to dapagliflozin and empagliflozin (see Table 2). Additionally, these ADRs were reported more frequently in women than in men. The finding that UTIs and fungal infections are common ADRs associated with SGLT2′s is well-known and supported by evidence. However, the literature is unclear regarding the risk in men versus women and the most highly associated SGLT-2 inhibitor. One study found that women who used SGLT-2 inhibitors were at a 3.4-fold increased risk of fungal genital infection, and men at a 2.8-fold increased risk. 25 In general, while fungal genital infections and UTIs are more common in women, evidence suggests that the risk becomes similarly increased in men. Geerlings et al. 26 also suggested that the mechanism of action for SGLT-2 inhibitors may lead to an over-reporting of UTIs and fungal infections due to increased glucosuria being a known risk factor for genital infections. This may have generally inflated the RORs for these infections for SGLT-2 inhibitors, especially for women. Interestingly, a meta-analysis performed by Puckrin et al. 27 conflicts with our finding that canagliflozin has a greater odds of UTI reporting compared to other SGLT-2 inhibitors. The investigators found that dapagliflozin 10 mg was the only SGLT2 associated with significantly increased risk for UTI when compared to placebo (RR 1.33, 95% CI 0.93–1.61). In our study, dapagliflozin had a ROR of 1.00 which can be interpreted as the odds of UTI reporting for dapagliflozin was similar to others in the drug class. However, our results are supported by other reports that suggest genital infections are not limited to just dapagliflozin, but also includes other members of the drug class. 28
Each of the four analyzed GLP-1 receptor agonists included in this study are once weekly injections. This represents a strength in this study given the majority of existing research and review articles compare weekly and daily injectable GLP-1 receptor agonists.29,30 The treatment-associated gastrointestinal ADRs of nausea, diarrohea, and vomiting are recognized side effects of GLP-1 receptor agonists. 30 In our analysis, dulaglutide and semaglutide had greater odds of reporting compared to exenatide and albiglutide for each of these ADRs. Our findings agree with existing literature that exenatide and albiglutide may be associated with lower rates of gastrointestinal side effects. 30 Additionally, nausea and vomiting was reported more frequently for female subjects compared to males. Among clinicians the safety of GLP-1 receptor agonists has also been a concern for those with pancreatic risk factors. 31 In our analysis, the ADR pancreatic carcinoma was more likely to be reported for exenatide relative to the other GLP-1 receptor agonists. However, it is important to note, the number of reports for the other medications within the drug class were fairly low which could result in a falsely elevated signal. The occurrence of pancreatic carcinoma not supported by past studies examining the pancreatic safety of exenatide, which have failed to find a significant difference in the incidence of pancreatic cancer with exenatide use.31,32
In the literature, no difference was observed in weight loss between GLP-1 receptor agonists.33,34 Interestingly, our analysis found a greater odds of decreased weight reporting for exenatide relative to the other GLP-1 receptor agonists (1.81; 1.70–1.93). This suggests exenatide use may result in a greater weight loss compared to albiglutide, dulaglutide, and semaglutide. The lowest odds of reporting weight loss was seen for albiglutide (0.572; 0.491–0.667), which is supported by the limited head-to-head GLP-1 receptor agonist trials that exist. 33
Injection site reactions reports were disproportionally higher for exenatide compared to the other GLP-1 receptor agonists. This finding is supported by safety meta-analyses and in comparisons of their respective phase 3 trials, however no reason for the marked increase in injection site reactions has been put forth in the literature.35,36 Interestingly, exenatide ER pen injectors kits come with a 23G needle while semaglutide, dulaglutide, and albiglutide come with 29G needles or greater which provides a potential explanation for this finding.
Limitations
Utilizing the FAERS database for pharmacovigilance research comes with several inherent limitations due to the public nature of the data. Given the FAERS database is a voluntary reporting system, reporter bias is likely present based on a reports individual experiences with medications. Additionally, this is seen given the large number of reports for those 18–69 years old compared to 70 years and older. It is possible the reporting system is easier to use for the adult population compared to the elderly, which could explain the difference. Data quality is a general limitation in terms of duplicative reports and incomplete ADR entries. In order to combat this limitation, we excluded duplicate reports and missing ADR data.
Furthermore, it is important to be aware that the existence of a report does not establish causation, and that the information in the database entries has not been verified which is why we emphasize that our data is hypothesis generating and not conclusive causation. The reports that document an ADR outcome of a patient does not explicitly mean that the suspect medication was the cause of the outcome, although we did attempt to mitigate this by only including reports indicating a drug of interest as the PS drug. 37 Of note, in the analysis of exenatide, the extended-release formulation was not able to be differentiated from the immediate release formulation due to the standardization process of multi-ingredient medications. Additionally, the intention of the analysis was to include the second quarter of 2022, however, only the first quarter was able to be obtained. As such, there is one less quarter analyzed for semaglutide (GLP-1 receptor agonist) and empagliflozin (SGLT-2 inhibitor) compared to the other medications within the respective drug classes, which may result in conservative ROR estimates for these two medications.
Using the FAERS database makes it impossible to quantify the true risk of a specific ADR since not every ADR is reported in the database and therefore there is no denominator for determining incidence rates which is why ROR were used to assess reporting risk instead of true hazard risk. However, RORs may potentially be skewed by the number of reports available where medications with a large number of reports will generate safety profiles that are more representative compared to a small number of reports because of the paucity of information available. Therefore, comparison between two medications with differing numbers of reports can generate bias. Medications with less than 600 reports were excluded to attempt to negate this, in addition to comparing medications within a drug class for the same amount of time after each respective approval date so the timeframe would be compared equally and likely generate similar numbers of reports. Moreover, differences in market approval times and therapeutic patterns in other countries may affect the total number of reports. However, ADRs reported outside of the United States were still included to provide a comprehensive scope of ADR reports of newer approved antidiabetic medications. Lastly, our data are limited by the Weber effect, peak in ADR reporting of a drug at the end of their second year of regulatory approval, followed by a continuous decline in reports. 38 This is demonstrated in Figure 2 with canagliflozin reports.
Conclusion
Due to the limited head-to-head trials comparing the safety antidiabetic medications, especially within classes of medications, this study provides a better understanding of ADRs voluntarily reported by healthcare professionals, consumers, and manufacturers. Real world experiences with medications were accounted for with reports from healthcare professionals, consumers, and manufacturers. Pharmacovigilance studies based on spontaneous reporting make it possible to identify essential signals for the analysis of the safety profile of drugs in clinical practice settings. Further defining ADR risk for medications within drug classes is important to improve clinician’s ability to make patient-specific decisions when choosing medications, counseling patients on adverse events, and building informed monitoring plans. The study we identified the SGLT-2 inhibitors with disproportionate rates of reporting important ADRs such as toe amputation, UTIs, and osteomyelitis. Similarly, the GLP-1 receptor agonists linked to gastrointestinal events, injections site reactions, and pancreas related side effects were identified through disproportional reporting. Increasingly, post-marketing surveillance systems and data mining techniques with measures of disproportionality are utilized to detect new or previously known ADRs. Future studies should consider safety signals related to newly approved combination products involving metformin. This study supports the need for further investigation of data from FDA pre-approval studies, and a need for phase IV randomized controlled trials or well-designed quasi-experimental studies with safety focused endpoints.
Supplemental Material
sj-docx-1-taw-10.1177_20420986231181334 – Supplemental material for Assessing adverse drug reaction reports for antidiabetic medications approved by the food and drug administration between 2012 and 2017: a pharmacovigilance study
Supplemental material, sj-docx-1-taw-10.1177_20420986231181334 for Assessing adverse drug reaction reports for antidiabetic medications approved by the food and drug administration between 2012 and 2017: a pharmacovigilance study by Britney A. Stottlemyer, Michael C. McDermott, Mackenzie R. Minogue, Matthew P. Gray, Richard D. Boyce and Sandra L. Kane-Gill in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
