Abstract
Objective
To undertake a network meta-analysis to compare the relative efficacy of a dual peroxisome proliferator-activated receptor (PPAR)α and PPARγ agonist, glucagon-like peptide-1 receptor agonists (GLP-1RAs) and metformin in patients with non-alcoholic fatty liver disease (NAFLD).
Methods
Electronic databases, including Embase®, PubMed® and The Cochrane Library, were searched systematically for eligible studies from inception to 20 July 2022. Randomized controlled trials (RCTs) that investigated aspartate aminotransferase, alanine aminotransferase (ALT) and triglyceride levels were considered for inclusion. Data were extracted using a standardized data collection table. A network meta-analysis was performed. Relative risk and 95% confidence interval were calculated for continuous data and
Results
A total of 22 RCTs involving 1698 patients were eligible for inclusion in the analysis. Both direct analysis and indirect analysis showed that saroglitazar was significantly superior to GLP-1RAs in improving ALT levels. Metformin improved ALT levels, but the effect was not as good as saroglitazar.
Conclusion
Saroglizatar was the most effective drug for improving NAFLD.
INPLASY registration number: INPLASY202340066
Introduction
The prevalence of non-alcoholic fatty liver disease (NAFLD) is gradually increasing worldwide. 1 Non-alcoholic steatohepatitis (NASH) is the severe stage of NAFLD and may further progress to cirrhosis and hepatocellular carcinoma. 2 Currently, the management of NAFLD mainly focuses on lifestyle-related interventions, including healthy diets, caloric restriction and increased aerobic exercise. 3 However, the morbidity and mortality caused by NAFLD are still high in modern society. 4 Recently, several medications for NAFLD have been developed. 5
Hepatic lipid accumulation is one of the pathogenetic causes of NAFLD. 6 Peroxisome proliferator-activated receptor (PPAR) α and PPARγ are nuclear receptors that play key roles in modulating hepatic lipid metabolism. 7 Saroglitazar, a novel dual PPARα and PPARγ agonist, can improve alanine aminotransferase (ALT), aspartate aminotransferase (AST) and triglyceride (TG) levels in NAFLD patients. 8 Due to its efficacy, saroglitazar is now available in India as the first pharmacotherapy for NASH. 9 Glucagon-like peptide-1 (GLP-1) is a pleiotropic hormone secreted by the gut with broad pharmacological potential in lowering blood glucose levels. 10 GLP-1 receptor agonists (GLP-1RAs) have been widely applied in treating type 2 diabetes mellitus (T2DM). 11 Interestingly, several studies have shown that GLP-1RAs also have a potential therapeutic effect on preventing the development of NAFLD. 5 GLP-1Ras, including liraglutide and semaglutide, have a good effect on reducing ALT and AST levels in patients with NAFLD. 12 Metformin has been a classic glucose-lowering drug for patients with T2DM over the past 60 years. 13 More importantly, its ability to combat the disease process involved in the development of NAFLD has been identified in recent years. 13 Treatment with metformin for 72 weeks can improve ALT levels in patients with NAFLD. 14
Data directly comparing the efficacy of various drugs on NAFLD are lacking, which may limit their clinical application. This current study undertook a network meta-analysis to compare the relative efficacy of dual PPARα and PPARγ agonists, GLP-1RAs and metformin in patients with NAFLD.
Materials and methods
Search strategy
This network meta-analysis was conducted in accordance with the PRISMA guidelines. 15 Electronic databases, including Embase®, PubMed® and The Cochrane Library, were searched from inception to 20 July 2022 using the following keywords: (“liraglutide”[Mesh] OR “semaglutide” OR “dulaglutide” OR “saroglitazar” OR “metformin”) AND (“Non-alcoholic Fatty Liver Disease”[Mesh] OR “NAFLD” OR “Nonalcoholic Fatty Liver Disease”. The search was limited to human studies published in the English language. In error, this study was not prospectively registered, but it has been now registered retrospectively at INPLASY: registration number INPLASY202340066.
Eligibility criteria
Two reviewers (Z.Y.Z and Q.Y.) independently screened records according to the title/abstract and then screened the full text of the relevant records according to prespecified screening criteria. Disagreements during this process were resolved by consensus and a third reviewer (W.H.W.). The trials included in this network meta-analysis were selected based on the following criteria: (i) they were phase II, III or IV randomized controlled trials (RCTs); (ii) they included adults and adolescents patients with NAFLD, which was confirmed by biopsy or other testing; (iii) they compared dual PPARα and PPARγ agonist (saroglitazar) or GLP-1RAs (including semaglutide, liraglutide and dulaglutide) or metformin with placebo or each other; (iv) they had a follow-up duration of at least 4 months; (v) they reported the primary outcome of AST and ALT levels and/or the secondary outcome of serum TG levels.
Data extraction and quality assessment
Data from the included literature were extracted into a standardized table in Microsoft Excel 2022 (Microsoft, Redmond, WA, USA) and included the following: (i) study characteristics (author names, year of publication, research duration); (ii) participants characteristics (age, sex, body mass index [BMI], diabetes history); (iii) treatment characteristics (sample size, intervention time of each group); (iv) outcome assessments (changes in AST, ALT and TG levels from baseline in each treatment group).
The Cochrane Risk of Bias Tool was used to evaluate the risk of bias of each study, including the random generation sequence, concealment of assignments, patient and investigator blinding, outcome assessment blinding, incomplete outcome data, selective results reporting or other sources of bias. 16 Risk of bias was classified as low, high or unclear.
Statistical analyses
The comparison of different interventions was made by calculating alterations from baseline to the end of intervention of the primary/secondary outcomes. According to the data provided by the original literature, the changes of the mean ± SD were calculated. When median, range and size of a sample were used instead of the mean ± SD, a previously published method was used for the estimation.
17
A random-effects model was applied to estimate 95% confidence interval (CI) in the direct meta-analysis. Direct comparisons were performed using RevMan software (version 5.3; Cochrane Collaboration, Oxford, UK).
Due to the limited sample size, indirect comparisons were used to explore the difference in efficacy between two regimens. In order to evaluate the consistency of direct comparison and indirect comparison, a node splitting method was used to estimate the effect of indirect comparisons. A
Results
The initial database search identified 486 articles. Of these, 22 studies were selected for this meta-analysis (Figure 1).18–39 The characteristics of the included studies are summarized in Table 1.18–39 A total of 1698 patients with NAFLD were included in the 22 studies, with a mean age of 44.3 years and a median follow-up time of 12–96 weeks. Among the included studies, one study explored the efficacy of the dual PPARα and PPARγ agonist saroglitazar, 20 while seven studies assessed the effects of GLP-1RAs.18,19,21–23,38,39 The remaining 14 studies evaluated the efficacy of metformin.24–37 Twenty studies used placebo as the control;18–37 and two studies compared the effect between liraglutide and metformin.38,39 According to the Cochrane Collaboration’s tool for assessing risk of bias, all of these studies were at a low-to-moderate risk of bias (see supplementary materials, Supplementary Figure 1).

Flow diagram of eligible studies showing the number of citations identified, retrieved and included in the final network meta-analysis to compare the relative efficacy of dual peroxisome proliferator-activated receptor (PPAR)α and PPARγ agonists, glucagon-like peptide 1 receptor agonists and metformin in patients with non-alcoholic fatty liver disease (NAFLD).
Baseline characteristics of the 22 studies included in the final network meta-analysis to compare the relative efficacy of dual peroxisome proliferator-activated receptor (PPAR)α and PPARγ agonists, glucagon-like peptide-1 receptor agonists and metformin in patients with non-alcoholic fatty liver disease (NAFLD).
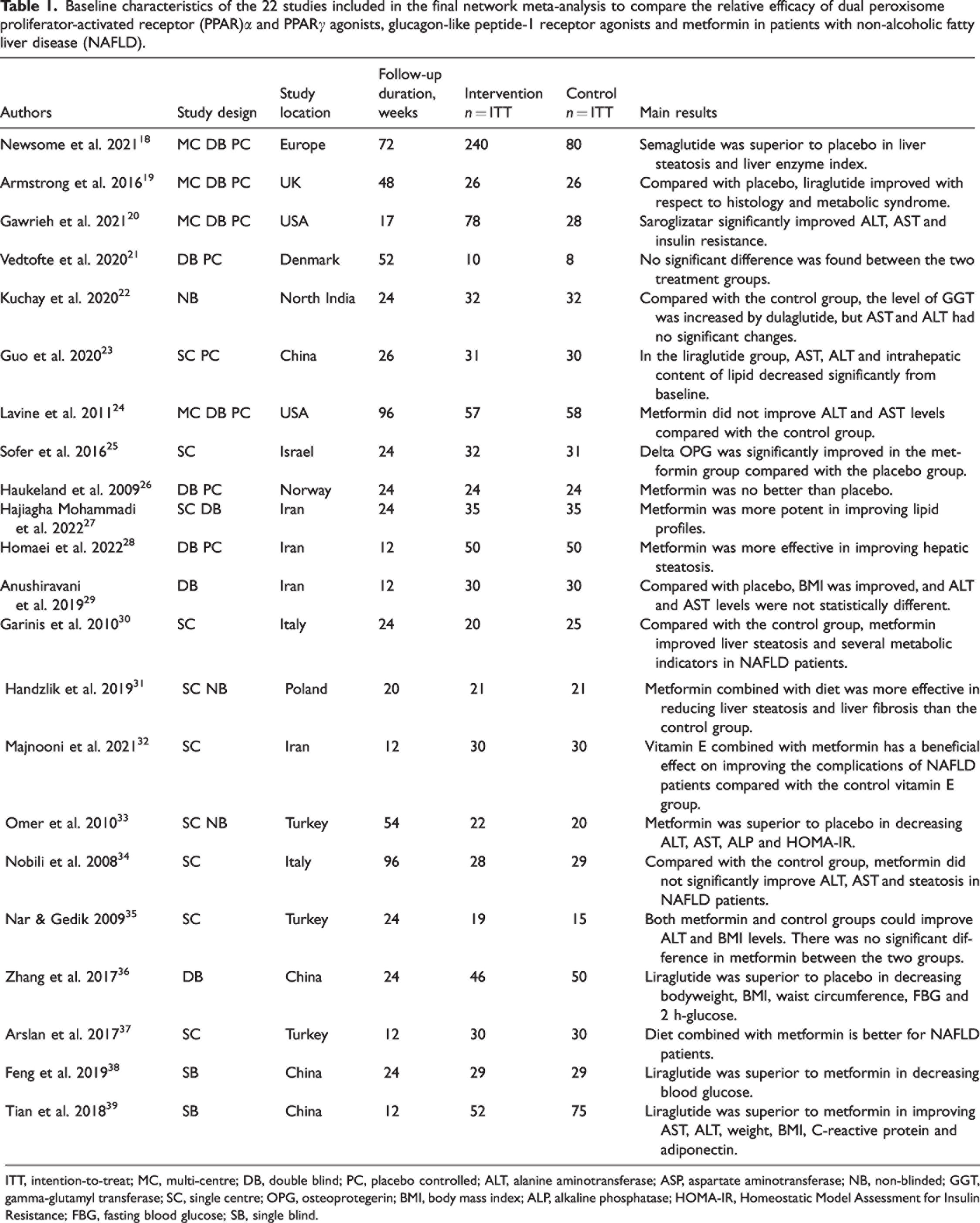
ITT, intention-to-treat; MC, multi-centre; DB, double blind; PC, placebo controlled; ALT, alanine aminotransferase; ASP, aspartate aminotransferase; NB, non-blinded; GGT, gamma-glutamyl transferase; SC, single centre; OPG, osteoprotegerin; BMI, body mass index; ALP, alkaline phosphatase; HOMA-IR, Homeostatic Model Assessment for Insulin Resistance; FBG, fasting blood glucose; SB, single blind.
With regard to the primary outcome of lowering circulating ALT levels, the direct meta-analysis included 21 RCTs.18–36,38,39 Compared with placebo, saroglitazar was associated with an improvement in ALT levels in patients with NAFLD (95% CI: −1.66, −0.74) (Figure 2a). GLP-1RAs such as semaglutide, liraglutide and dulaglutide or metformin did not significantly improve ALT levels in NAFLD patients (Figure 2a).

Direct meta-analysis (a), network graphs (b) and network meta-analysis (c) for the primary outcome of alanine aminotransferase levels. In the reticulation diagram (b), the size of the dots represents the number of patients with relevant interventions and the thickness of the wires represents the number of included studies.
With regard to the primary outcome of lowering circulating ALT levels, the indirect meta-analysis demonstrated the network comparisons of available treatments in assessing the effects in improving circulating ALT levels (Figure 2b). Consistent with the direct meta-analysis, saroglitazar significantly improved ALT levels in NAFLD patients when compared with placebo, metformin and liraglutide (
With regard to the primary outcome of lowering circulating AST levels, the direct meta-analysis demonstrated that when compared with placebo, saroglitazar were associated with a decrease in AST levels (95% CI: −1.46, −0.55) (Figure 3a). Several of the GLP-1RAs, including semaglutide, liraglutide and dulaglutide, did not show significant improvements in AST levels in patients with NAFLD compared with placebo. The effect of metformin on improving AST levels in NAFLD patients was not obvious. No significant differences in reducing circulating AST levels were observed between the other interventions.

Direct meta-analysis (a), network graphs (b) and network meta-analysis (c) for the primary outcome of aspartate aminotransferase. In the reticulation diagram (b), the size of the dots represents the number of patients with relevant interventions and the thickness of the wires represents the number of included studies.
With regard to the primary outcome of lowering circulating AST levels, the indirect meta-analysis demonstrated the network comparisons of available treatments in assessing the effects of affecting circulating AST levels (Figure 3b). Saroglitazar was significantly superior to placebo in improving circulating AST levels (
With regard to the secondary outcome of lowering circulating TG levels, the direct meta-analysis demonstrated that saroglitazar (95% CI: −1.27, −0.35) was associated with a reduction in TG levels in NAFLD patients compared with placebo (Figure 4a). Compared with placebo, liraglutide and dulaglutide could also improve the circulating levels of TG. Semaglutide did not reduce TG levels when compared with placebo. Liraglutide did not significantly improve TG levels when compared with metformin. In a controlled comparison with placebo, metformin was not superior to placebo in reducing TG levels.

Direct meta-analysis (a), network graphs (b) and network meta-analysis (c) for the secondary outcome of triglycerides. In the reticulation diagram (b), the size of the dots represents the number of patients with relevant interventions and the thickness of the wires represents the number of included studies.
With regard to the secondary outcome of lowering circulating TG levels, the indirect meta-analysis demonstrated the network comparisons of available interventions evaluating the improvement of TG level (Figure 4b). In indirect comparisons, saroglitazar and liraglutide were consistent with the direct comparisons in improving TG levels compared with placebo. Semaglutide and dulaglutide did not significantly reduce TG levels compared with placebo (Figure 4c). Saroglitazar and liraglutide ranked first and second in terms of improving TG based on the SUCRA ranking system (see supplementary materials, Supplementary Figure 2C). The intervention of liraglutide was significantly better than metformin in lowering TG levels in the indirect comparison, which was inconsistent with the result of the direct comparison.
Discussion
This current network meta-analysis comprehensively evaluated the relative efficacy of different drugs, including GLP-1RAs, saroglitazar and metformin, in improving the development of NAFLD. The results indicated that the effects of saroglitazar on decreasing circulating ALT and AST levels were better than that of GLP-1RAs. However, there was no significant difference between saroglitazar and GLP-1RAs in improving TG levels. Metformin could also decrease circulating AST levels, but the effect was not as obvious as that of saroglizatar. Meanwhile, saroglitazar was more effective than GLP-1RAs and metformin in improving serum ALT and TG in NAFLD patients.
At present, there is increasing evidence that PPAR agonists and glucose-lowering drugs such as metformin and GLP-1RAs have potential value in the treatment of NAFLD. 40 However, there are few direct comparative studies on the effects of these drugs on NAFLD. Although the efficacy between PPAR agonists and GLP-1RAs has been discussed previously, the authors only analysed the mechanisms of these drugs. 36 A meta-analysis of the relevant clinical data was not performed. 41 The main purpose of this current network meta-analysis was to directly compare the effects of these drugs on the progression of NAFLD, especially with regard to liver enzymes and TG levels.
The development of NAFLD is a dynamic process, ranging from steatosis to fibrosis. 42 During this process, the levels of liver enzymes such as ALT and AST are closely associated with the severity of NAFLD. 43 If there are large amounts of enzyme in the cytoplasm of hepatocytes, ALT and AST can usually be detected in the serum of healthy people at a relatively low level. Once hepatocyte injury and apoptosis occur, the serum ALT and AST levels significantly increase. 44 Therefore, alterations of the ALT and AST levels are considered to be important indicators of NAFLD improvement. Serum ALT is independently correlated with liver TG content, therefore it is more suitable as a predictor of NAFLD than AST. 45 In Asian NAFLD patients, ALT levels are closely related to intrahepatic TG accumulation, steatosis, inflammation and fibrosis. 46
During the progression of NAFLD, in addition to the hepatic inflammatory response, there is also lipid accumulation in the liver.
42
Increased
It has been reported that PPAR agonists have an important role in regulating several biological processes associated with NAFLD. 47 PPARα/γ ameliorates liver inflammation by reducing inflammatory cytokines and chemokines. 48 PPARα is mainly expressed in the liver and can promote the processes of fatty acid oxidation, ketogenesis, lipid transport and gluconeogenesis. 7 PPARγ is predominantly present in the liver and adipose tissue, and its activation increases insulin sensitivity. 49 Both oxidative stress and inflammation are involved in the process of NAFLD. 50 Saroglizatar is a novel PPAR agonist with predominantly PPARα and moderate PPARγ agonist activity. 47 The current results demonstrated that saroglizatar was the most effective drug in improving ALT and AST levels in NAFLD patients. In addition, saroglizatar was also superior to other drugs in decreasing TG levels.
Glucagon-like peptide-1, secreted by the gastrointestinal tract, has drawn considerable attention due to its glucose-lowering effects. For example, GLP-1 can regulate insulin secretion through GLP-1R in pancreatic β cells; 51 and it can also reduce blood glucose by inhibiting the secretion of glucagon in islet α cells. 52 GLP-1 can also reduce food intake by enhancing satiety through central mechanisms in the hypothalamus and brainstem. 53 In addition, the effects of GLP-1RA on improving hepatic lipotoxicity have been demonstrated in animal and human studies.54,55 However, based on this current network meta-analysis, the effect of GLP-1RAs in improving ALT and AST levels was not as obvious as that of saroglizatar. Liraglutide and dulaglutide were also effective in improving TG levels.
Metformin, a classic anti-diabetic drug, has an anti-inflammatory effect in the liver.56,57 In NAFLD patients, metformin increases hepatic β-oxidation and reduces gluconeogenesis. 58 In addition, metformin can also reduce caloric intake by inhibiting appetite, thereby reducing body weight and total body fat and visceral fat content. 59 In this current network meta-analysis, metformin could reduce serum ALT levels in NAFLD patients, although its effect is not superior to that of saroglizatar.
The current network meta-analysis had several limitations. First, the gold standard for NAFLD testing is liver biopsy, 60 which is difficult to achieve in clinical studies. The current analysis was based on the evaluation of the liver histology from a biochemical perspective. Secondly, due to the relatively small number of publications, the results of indirect and direct comparisons were not completely consistent. The inhibitory effects of GLP-1RAs, saroglizatar and metformin on ALT, AST and TG levels in the indirect comparison were supported by low-quality evidence. Therefore, larger randomized trials are needed to further verify these results.
In conclusion, GLP-1RAs and other drugs such as metformin and saroglizatar have some effects on improving the process of NAFLD. However, this network meta-analysis suggests that saroglizatar is the best one among these drugs in preventing the development of NAFLD. This network meta-analysis provides a focus for the future direction of the treatment of NAFLD and lays a foundation for subsequent drug research and development.
Supplemental Material
sj-jpg-1-imr-10.1177_03000605231177191 - Supplemental material for PPAR-alpha/gamma agonists, glucagon-like peptide-1 receptor agonists and metformin for non-alcoholic fatty liver disease: A network meta-analysis
Supplemental material, sj-jpg-1-imr-10.1177_03000605231177191 for PPAR-alpha/gamma agonists, glucagon-like peptide-1 receptor agonists and metformin for non-alcoholic fatty liver disease: A network meta-analysis by Zhuo-Ya Zhang, Qi Yan, Wen-Hao Wu, Yuan Zhao, Hua Zhang and Jin Li in Journal of International Medical Research
Supplemental Material
sj-jpg-2-imr-10.1177_03000605231177191 - Supplemental material for PPAR-alpha/gamma agonists, glucagon-like peptide-1 receptor agonists and metformin for non-alcoholic fatty liver disease: A network meta-analysis
Supplemental material, sj-jpg-2-imr-10.1177_03000605231177191 for PPAR-alpha/gamma agonists, glucagon-like peptide-1 receptor agonists and metformin for non-alcoholic fatty liver disease: A network meta-analysis by Zhuo-Ya Zhang, Qi Yan, Wen-Hao Wu, Yuan Zhao, Hua Zhang and Jin Li in Journal of International Medical Research
Footnotes
Acknowledgement
The authors thank Dr Huijie Zhang at Nanfang Hospital at Southern Medical University for their kind guidance and discussion.
Author contributions
Zhuo-Ya Zhang designed the research, wrote the manuscript and collected the data; Qi Yan conducted the research and data collection; Wen-Hao Wu analysed the data; Yuan Zhao and Hua Zhang modified the manuscript. Jin Li had primary responsibility for the final content. All authors read and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This work was supported by grants from the National Natural Science Foundation of China (no. 81970725, No. 82270915), Fok Ying Tong Education Foundation (no. 171031) and Shanxi Preferential Funding Projects for Scientific and Technological Activities of Returns (2019).
Supplementart materials
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
