Abstract
Introduction:
Drug-induced QTc-prolongation is a well-known adverse drug reaction (ADR), however there is limited knowledge of other drug-induced arrhythmias.
Purpose:
The objective of this study is to determine the drugs reported to be associated with arrhythmias other than QTc-prolongation using the FAERS database, possibly identifying potential drug causes that have not been reported previously.
Methods:
FAERS reports from 2004 quarter 1 through 2019 quarter 1 were combined to create a dataset of approximately 11.6 million reports. Search terms for arrhythmias of interest were selected from the Standardized MedDRA Queries (SMQ) Version 12.0. Frequency of the cardiac arrhythmias were determined for atrial fibrillation, atrioventricular block, bradyarrhythmia, bundle branch block, supraventricular tachycardia, and ventricular fibrillation and linked to the reported causal medications. Reports were further categorized by prior evidence associations using package inserts and established drug databases. A reporting odds ratio (ROR) and confidence interval (CI) were calculated for the ADRs for each drug and each of the 6 cardiac arrhythmias.
Results:
Of the 11.6 million reports in the FAERS database, 68,989 were specific to cardiac arrhythmias of interest. There were 61 identified medication-reported arrhythmia pairs for the 6 arrhythmia groups with 33 found to have an unknown reported association. Rosiglitazone was the most frequently medication reported across all arrhythmias [ROR 6.02 (CI: 5.82-6.22)]. Other medications with significant findings included: rofecoxib, digoxin, alendronate, lenalidomide, dronedarone, zoledronic acid, adalimumab, dabigatran, and interferon beta-1b.
Conclusion:
Upon retrospective analysis of the FAERS database, the majority of drug-associated arrhythmias reported were unknown suggesting new potential drug causes. Cardiac arrhythmias other than QTc prolongation are a new area of focus for pharmacovigilance and medication safety. Consideration of future studies should be given to using the FAERS database as a timely pharmacovigilance tool to identify unknown adverse events of medications.
Keywords
Introduction
Cardiac arrhythmias can result in all-cause mortality, sudden cardiac death, decreased quality of life, thromboembolism, and increased health costs. 1 It is estimated that there is approximately 1% prevalence of atrial fibrillation in the U.S. adult population. 2 While atrial fibrillation and other arrhythmias may be treated with medications, treatment may escalate to ablations. 2 -4 Ablations may be cost-effective at eliminating arrhythmias, 3,4 but healthcare professionals may be able to limit the need for intensifying therapies by minimizing medications that could induce arrhythmias.
Medications causing QTc-prolongation are used in both inpatient and outpatient settings and have been thoroughly studied. 1,5 -7 While drug-induced QTc-prolongation is recognized due to the potential for Torsades de Pointes (TdP), 7 literature for many other drug-induced arrhythmias is limited to case reports 8 -12 outside of drug-induced arrhythmia reviews with atrial fibrillation 13 and bradyarrhythmias. 14 -16 A newly published review of drug-induced arrhythmias did add further to the literature by including a section on atrioventricular nodal reentrant tachycardia. 17 Antineoplastic agents are a class of medications with mounting evidence of bradyarrhythmias that may further exacerbate cardiotoxicities already caused by atrioventricular nodal blocking agents. 14 A recent review of oncology literature has linked multiple chemotherapeutic agents to various cardiac arrhythmias ranging from sinus bradycardia to sudden cardiac death. 15 Although the most common drug-induced arrhythmias are thought to be related to QTc-prolongation, atrial fibrillation, and bradyarrhythmias, there are a few medications thought to be associated with development of atrioventricular block. Atrioventricular block can also be caused by medications, such as beta-blockers and digoxin, which can result in the placement of a permanent pacemaker even after the offending medication is discontinued. 18
The Food and Drug Administration Adverse Event Reporting System (FAERS) is a post-marketing surveillance database used to analyze the reported association between drugs and adverse events. FAERS includes voluntarily reported data from both healthcare providers and the public that is submitted through MedWatch and manufacturer information, making FAERS the largest adverse event report database in the United States. 19 The FAERS database provides an opportunity to supplement published case reports and case series by quickly identifying ADRs on a larger scale and improving pharmacovigilance. 20 On average it takes the FDA 12 years to identify ADRs that yield safety warnings or withdrawal of a medication from the market. Evaluations of large databases, such as FAERS, could be used to provide earlier safety signals for ADRs that may have been overlooked in pre-approval studies. 21 Therefore, the objective of this study was to highlight other drug-associated arrhythmias by determining their frequency of reports from a large-scale database to identify new potential drug adverse effects that have not been reported previously.
Methods
This was a retrospective pharmacovigilance disproportionality analysis using the FAERS database. FAERS reports from 2004 quarter 1 through 2019 quarter 1 were combined to create a dataset of around 11.6 million reports. Because the FAERs database is publicly available, IRB approval was not needed for this study. Search terms for arrhythmias of interest were selected from the Standardized MedDRA Queries (SMQ) Version 20.0 using their arrhythmia hierarchy. The highest-level descriptor based on SMQ was “cardiac arrhythmias.” The next level included “cardiac arrythmia terms,” “arrhythmia related investigations, signs and symptoms,” and “congenital and neonatal arrhythmias.” “Arrhythmia related investigations, signs and symptoms,” and “congenital and neonatal arrhythmias” were excluded to prevent the influence of examinations/tests and congenital arrhythmias influencing the results. Under “cardiac arrythmia terms,” there were 3 categories, “bradyarrhythmias,” “cardiac arrythmia terms, nonspecific,” and “tacharrhythmias.” The selected terms included arrhythmias listed under these 3 categories. Torsades de Pointes was listed in its own category and was not collected with the selected search terms. From the 3 selected terms (“bradyarrhythmias,” “cardiac arrhythmias non-specific,” and “tachyarrhythmias”), lower level arrhythmia descriptors resulted, such as “atrial fibrillation,” “sinoatrial block,” and “ventricular tachycardia.” The pool was further narrowed using the lower level descriptors after discussion with cardiology clinicians to include 6 groups of arrhythmias: atrial fibrillation, atrioventricular block, bradyarrhythmia, bundle branch block, supraventricular tachycardia, and ventricular fibrillation. Atrial fibrillation included “atrial fibrillation” and “atrial flutter” due to the potential difficulty in distinguishing between the 2 arrhythmias and because they both have a similar treatment strategy. 22 Bradyarrhythmia included reports with “bradyarrhythmia” and “sinus bradycardia.” “Bundle branch block,” “bundle branch block left,” “bundle branch block right,” and “bundle branch block bilateral” was included in the bundle branch block group. Supraventricular tachycardia included “arrhythmia supraventricular,” “atrial tachycardia,” and “Wolff-Parkinson-White syndrome” and excluded “atrial fibrillation” and “atrial flutter.” “Atrioventricular block” and “ventricular fibrillation” were the only low-level descriptors used for each of those arrhythmia groups.
Counts of the identified adverse drug reactions (ADRs) were determined and linked to the reported causal medication. The top 10 reported drugs in each of the arrhythmia groups were identified. Reports without a medication name were omitted from analysis. Medications names were standardized to generic names to account for misspellings, and varying ways that medications were entered into the FAERS database. Combination medications with 2 or more drug components were considered unique medications. For example, if metformin/glipizide combination medication was reported, it was considered its own medication and only evaluated in the combination drug group. As such, metformin/glipizide combination medication was not included with metformin or glipizide due to in inability to differentiate which active drug contributed to the ADR report.
Medications and reported frequency of the cardiac arrhythmias were determined and classified into pairs. Medications reported with cardiac arrhythmia events were cross-referenced using Micromedex®, Up-to-Date®, and package inserts to classify the cardiac arrhythmia adverse event into prior evidence association categories. Reported causal medications were categorized as: 1) known proarrhythmogenic; 2) potential proarrhythmogenic and 3) unknown (i.e., newly identified in this study) proarrhythmogenic. The medication was considered to be a known proarrhythmogenic if the event was listed in all 3 of the cross references. Similarly, the medication was considered to potentially proarrhythmogenic if the event was listed in 1-2 of the cross references. If the ADR was not listed in any of the cross references, it was considered as an unknown proarrhythmogenic.
Statistics
Reporting odds ratios (ROR) were used to assess the association between the reported causal drug and one of the 6 arrhythmias (atrial fibrillation, atrioventricular block, bradyarrhythmia, bundle branch block, supraventricular tachycardia, and ventricular fibrillation). ROR was chosen because it is a validated, common disproportionality analysis method to determine a potential association between a medication and ADR. 20,21,23 -26 The ROR was determined using the formula: ROR = (n11 × n00)/(n10 × n01), where n11 = arrhythmia reports for drug of interests, n01 = arrhythmia reports not including the drug of interest; n10 = reports not including arrhythmia for the drug of interest; n00 = reports including neither arrhythmia nor the drug of interest. 17,19 Using this formula resulted in the ratios of the odds of the reported event of interest A for the drug B to the odds of event of interest A in the absence of drug B. 19 ROR > 1 was considered to have greater odds of being proarrhythmogenic than other ADRs. Ninety-five percent confidence intervals (CIs) were calculated for each ROR using the formula: CI = e Ln (ROR) ±1.96 √ [(1/n11)+(1/n10)+(1/n00)+(1/n01)]. 19,23 Statistical analysis was completed using IBM SPSS version 25 (Armonk, N.Y.) and Stata (College Station, TX). 27,28
Results
Of the 11.6 million entries in the FAERS database, 76,049 entries were listed with an arrhythmia specific to atrial fibrillation, atrioventricular block, bradyarrhythmia, bundle branch block, supraventricular tachycardia, and ventricular fibrillation. The groups were then narrowed to individual reports for atrial fibrillation, atrioventricular block, bradyarrhythmia, bundle branch block, supraventricular tachycardia and ventricular fibrillation (Figure 1).

Study flow. aAtrial fibrillation included atrial fibrillation and atrial flutter; bbradyarrhythmia included bradyarrhythmia and sinus bradycardia; cbundle branch block included bundle branch block left, bundle branch block right, bundle branch block bilateral, and bundle branch block group; dsupraventricular tachycardia included arrhythmia supraventricular, atrial tachycardia, and Wolff-Parkinson-White syndrome and excluded atrial fibrillation and atrial flutter.
Reports were most commonly entered by a healthcare professional with 39.0% from physicians, 5.2% from pharmacists, 0.1% from nurses, and 20.1% from other health care professionals. Consumers did contribute 20.1% of entries and lawyers 3.4%. Arrhythmia reports resulted in serious outcomes such as death (5.4%), hospitalization (26.4%), life-threatening events (3.9%), disability (1.5%), requiring intervention to prevent impairment (0.6%), other serious events not defined (20.9%) and the remaining reports did not provide an outcome.
The medications most frequently reported for each arrhythmia are listed in Table 1. Of these 61 medication-arrhythmia pairs, 33 (54%) were found to have an unknown reported association. Reported potential prior evidence association pairs were found with only 12/61 (19.7%). Sixteen of the 61 pairs (26.2%) were deemed to have a known reported association with a cardiac arrhythmia (Figure 2). For example, rosiglitazone demonstrates 3,856 reports across the 6 arrhythmia groups of interest with unknown proarrhythmic associations for atrial fibrillation, atrioventricular block, bundle branch block, supraventricular tachycardia, and ventricular fibrillation. Rosiglitazone was not among the top 10 medications for bradyarrhythmias.
Top 10 Medications for Each of the 6 Arrhythmia Groups.

a N = number of reports for that arrhythmia.
b n = number of reports for that medication and arrhythmia.

Medications and reported proarrhythmic association for 6 arrhythmia groups. This figure represents the 84 identified medication-reported arrhythmia pairs for the 6 arrhythmia groups (atrial fibrillation, atrioventricular block, bradyarrhythmia, bundle branch block, supraventricular tachycardia, and ventricular fibrillation) after using cross references to categorize the reported pairs.
ROR for the top 10 medications appearing across the 61 pairs of medications with reported proarrhythmic association were calculated (Table 2). All 10 medications had a statistically significant ROR. Rosiglitazone resulted the most frequently across all arrhythmias with a ROR of 6.02 (5.82-6.22). Of note adalimumab’s ROR of 0.37 (0.35-0.40) indicates lower odds of being reported with these 6 arrhythmias as compared to other ADRs.
Reported Odds Ratios for Top 10 Reported Medications in 6 Arrhythmia Groups.
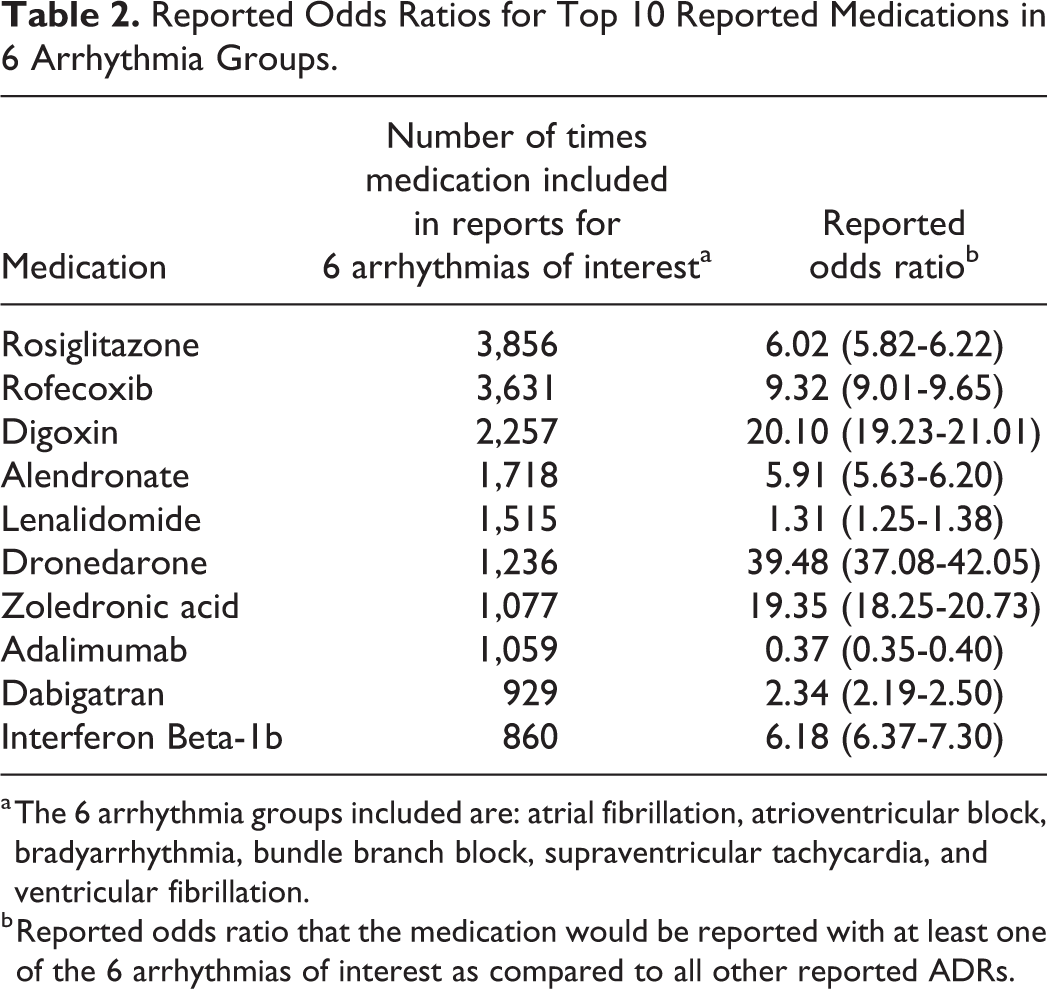
a The 6 arrhythmia groups included are: atrial fibrillation, atrioventricular block, bradyarrhythmia, bundle branch block, supraventricular tachycardia, and ventricular fibrillation.
b Reported odds ratio that the medication would be reported with at least one of the 6 arrhythmias of interest as compared to all other reported ADRs.
Discussion
Less is known about drug-associated proarrhythmic events in general, compared to those specifically related to QTc-prolongation. 8 Understanding these rarer events is important for future research, pharmacovigilance and patient care. This study identified 16 drug-arrhythmia pairs known for their proarrhythmic events. Still, known proarrhythmic agents are limited in familiar drug references like Micromedex® and Up-to-Date® 20 because we identified 45 potential or previously unknown drug-arrhythmia pairs. Identifying 73.8% of drug-arrhythmia pairs as potential or previously unknown may be expected because of notoriety bias since these 45 drugs are not a target for surveillance with the lack of data or mentioning in familiar databases. Also, this substantial number of unknown drug associations is likely due to much of the arrhythmia literature and databases focusing on QTc-prolongation and TdP with the exception of a recent review of drug-induced arrhythmias. 1,5 -7,17 A recent review from the American Heart Association only included information on 5 of the top 10 medications reported across the 6 arrythmias in our study: rofecoxib (under a class category and not specifically mentioned), digoxin, alendronate, dronedarone, and zoledronic acid. 17
The results of this retrospective pharmacovigilance disproportionality analysis display the significance of post-marking surveillance for arrhythmias. A prime example from our results is rosiglitazone. Rosiglitazone carries a known cardiovascular risk resulting in a black box warning with for heart failure and myocardial ischemia. 29,30 With rosiglitazone’s recognized cardiac adverse effect profile, it is not surprising that rosiglitazone would be the most reported medication in our study. However, it is unanticipated that it would appear in 5 of the 6 arrhythmia groups with each ADR being a new potential association. To our knowledge, there is no existing evidence for rosiglitazone correlating with atrioventricular block, bundle branch blocks, or supraventricular tachycardias. Interestingly, there is evidence opposing our atrial fibrillation findings for rosiglitazone. Thiazolidinediones (rosiglitazone and pioglitazone) are documented as having potential protective effects in preventing atrial fibrillation due to their pleiotropic effects. 31,32 It should be noted that pioglitazone, not rosiglitazone, could be the medication that confers protection for atrial fibrillation. 32 Nonetheless, Leonard et al examined outcomes of rosiglitazone versus pioglitazone in sudden cardiac death, which included ventricular arrhythmias. 33 Their findings indicated no difference in association between the 2 thiazolidinediones of sudden cardiac death or ventricular arrythmias. 33 Despite these findings, the FAERS database may yield key safety information for rosiglitazone and other potential medication associations found in our study.
Rofecoxib was the second most reported drug in the 6 arrhythmia groups. Similar to rosiglitazone, rofecoxib has an established adverse cardiac prolife, but limited studies examined the proarrhythmic adverse effects. 34 Rofecoxib is a COX-2 inhibitor, introduced into the market in 1999. It was used for the treatment of osteoarthritis pain. 34 In 2004, rofecoxib was voluntarily removed from the market by its manufacturer primarily driven by post-marketing studies finding an increased risk for myocardial infarction and stroke. 34 -36 Rofecoxib was only available for 9 months of our study period before being withdrawn from the market. Rofecoxib’s proarrhythmic effect was unknown prior to 2004 and may have contributed to increased stroke risk if secondary to atrial fibrillation. It is possible that monitoring pharmacovigilance databases such as FAERS may provide timely and insightful information to healthcare providers earlier than other approaches.
It is of interest that we found a lower reporting rate for chemotherapeutic associated arrhythmias compared to the literature considering the familiarity with chemotherapeutic related adverse cardiovascular side effect profiles supported by substantial published data. 37 -44 Only lenalidomide was in the top 10 most frequently reported drugs in the 6 arrhythmia groups of interests. Of note, there were 2 other immunomodulator medications that are not used as chemotherapy, adalimumab and interferon beta-1b, in the top 10. Lenalidomide was among the top drugs reported in atrial fibrillation, supraventricular tachycardia, and atrioventricular block. Of these 3, lenalidomide had a known association with atrial fibrillation and supraventricular tachycardia as these events where found in clinical trials. 45,46 On the other hand, lenalidomide’s association with atrioventricular block was found to be a new potential association. While we could not find any studies or case reports linking lenalidomide to atrioventricular block, there is literature to support thalidomide being identified as causing atrioventricular block, 17,47,48 which may have some significance as lenalidomide is structurally similar to thalidomide. 49 Our findings display a gap in lenalidomide literature for ADRs being reported, thus illustrating the use of FAERs as a tool for pharmacovigilance and medication safety.
While the FAERS database can provide helpful information regarding pharmacovigilance, some reports should be examined closely. For example, digoxin, an antiarrhythmic used to treat atrial fibrillation, was the third most frequently reported medication for atrial fibrillation and had a significant ROR. However, the high frequency of digoxin-atrial fibrillation reports could be due to lack of efficacy of digoxin instead of a causal effect between digoxin and atrial fibrillation. Although the FAERS data are useful for identifying associations between drugs and adverse events, it cannot be used to determine causal relationships between drugs and adverse events.
Large databases, such as FAERS, may promote investigation of events that may be overlooked possibly delaying research into important safety concerns. 21 This is displayed by our findings showing that none of the top 10 medications in the bundle branch block had known associations, yet bundle branch block had the second most reports (n = 6,792) of our 6 arrhythmia groups. Lack of drug-induced bundle branch block literature could be due to bundle branch blocks looking like or leading to other arrhythmias such as ventricular tachycardia and third-degree AV block. 50 Our findings highlight the need for further study of medication-induced bundle branch block.
Limitations
Arrhythmias can only be thought of as possibly associated events and not causal due to the spontaneous reporting nature of the data in this study. It is important to remember that these types of studies suggest association and not causation. The FAERS database uses voluntary reporting, and third parties are unable to adjudicate causality for the reported ADRs. Despite an inability to confirm causality, the severity assessment seems valid since 64.4% of reports were completed by healthcare professionals who have the appropriate expertise for this assessment. Still, some reports (20.1%) were entered by consumers who are unlikely to be trained for this type of assessment. Our results suggest that arrhythmias should be interpreted as serious events requiring medical attention based on the reported severity assessment.
It is difficult to assess drug-drug interactions that may play a role in a reported ADR and could go undetected with assessments in FAERS. 51 One study discussed evaluating drug interactions using pharmacovigilance data but this requires a post-hoc analysis using a chemical biology data. 52 Drug-drug interactions were not evaluated in our study.
There are other limitations to this study as well. Using a Standardized MedDRA Queries (SMQ) meant that some reports entered via a free text option were not included in the standardized query and therefore not included in this analysis. Additionally, the ability to identify potential and unknown reactions is limited by the adverse reactions included based on the cross-references’ regular review of the literature. Finally, it should be noted that these results could induce the notoriety effect (prior exposure to known drug-associated ADR) and future detection bias of arrhythmia ADRs. 53 Consumer reporting may be influenced by media and other societal norms. 54 Nevertheless, the FAERs database offers a unique opportunity to consider rare events (non-QT prolonging arrhythmias) that may otherwise not be investigated and should be considered hypothesis generating to stimulate further investigation.
Conclusion
The majority of drug-associated arrhythmias in the FAERS database were found to be unknown, thus non-QTc prolongation cardiac arrhythmias are a new area of focus for arrhythmia ADR pharmacovigilance and medication safety. Drugs associated with arrhythmias are not limited to one class of medications, and thus all medications classes should be evaluated for potential association with cardiac arrhythmias. This is a hypothesis generating study supporting the need for future research to confirm associations between drugs and arrythmias that have not been previously reported.
Footnotes
Author Contributions
Sandra L. Kane-Gill, Lindsay N. Moreland-Head, James C. Coons, and Amy L. Seybert conceived and designed the study; Matthew P. Gray and Lindsay N. Moreland-Head were involved in data acquisition; Matthew P. Gray and Lindsay N. Moreland-Head analyzed the data; Sandra L. Kane-Gill and Lindsay N. Moreland-Head prepared the manuscript; Matthew P. Gray, James C. Coons, and Amy L. Seybert reviewed the manuscript; Sandra L. Kane-Gill, Lindsay N. Moreland-Head, James C. Coons, and Amy L. Seybert, Matt P. Gray approved the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
