Abstract
In the context of an ageing population, the burden of disease and medicine use is also expected to increase. As such, medicine safety and preventing avoidable medicine-related harm are major public health concerns, requiring further research. Potentially suboptimal medicine regimens is an umbrella term that captures a range of indicators that may increase the risk of medicine-related harm, including polypharmacy, underprescribing and high-risk prescribing, such as prescribing potentially inappropriate medicines. This narrative review aims to provide a background and broad overview of the patterns and implications of potentially suboptimal medicine regimens among older adults. Original research published between 1990 and 2021 was searched for in MEDLINE, using key search terms including polypharmacy, inappropriate prescribing, potentially inappropriate medication lists, medication errors, drug interactions and drug prescriptions, along with manual checking of reference lists. The review summarizes the prevalence, risk factors and clinical outcomes of polypharmacy, underprescribing and potentially inappropriate medicines. A synthesis of the evidence regarding the longitudinal patterns of polypharmacy is also provided. With an overview of the existing literature, we highlight a number of key gaps in the literature. Directions for future research may include a longitudinal investigation into the risk factors and outcomes of extended polypharmacy, research focusing on the patterns and implications of underprescribing and studies that evaluate the applicability of tools measuring potentially inappropriate medicines to study settings.
Plain Language Summary
Medicine use in older age is common. Older adults with more than one chronic condition are likely to use multiple medicines to manage their health. However, there are times when taking multiple medicines may be unsafe and the number of medicines, or the combination of medicines used, may increase the risk of poor health outcomes. The term medicine regimens is used to describe all the medicines an individual takes. There are several ways to measure when a medicine regimen may be inappropriate and, therefore, potentially harmful. Much research has been published looking into potentially inappropriate medicine regimens. To bring together the current research, this review provides a background on the different measures of potentially inappropriate medicine regimens. It also summarizes how many people may experience potentially inappropriate medicine regimens, the impact it is having on their health and who may be at greater risk. In doing so, we found a number of gaps in the existing evidence, indicating that our understanding of potentially inappropriate medicine regimens is incomplete. This review highlights gaps in knowledge that can be addressed by future research. With an improved understanding of potentially inappropriate medicine regimens, we may be able to better identify those at greater risk to prevent or minimize the impact of poorer health outcomes related to unsafe medicine use.
Keywords
Introduction
Medicine safety and medicine-related harm are major public health concerns. Older adults are particularly vulnerable to adverse events associated with pharmaceutical intervention. Potentially suboptimal medicine regimens is an umbrella term that encompasses a range of tools and indicators for measuring medicine use that may increase the risk of harm in older adults. Medicines safety has become a policy priority in many countries since the World Health Organization (WHO) launched its Medication Without Harm initiative in 2017, which aimed to reduce medicine-related harm by 50% within 5 years. 1 While there is a substantial body of evidence investigating potentially suboptimal medicine regimens internationally, how risk is defined and measured appears to vary across the literature. This narrative review aims to provide an overview of the current evidence of potentially suboptimal medicine regimens, namely, polypharmacy, underprescribing and potentially inappropriate medicines (PIMs). The objective is to highlight key gaps in the literature to inform targets for future research.
Methods
This is a narrative literature review that aims to synthesize the current research on the prevalence and risk factors of potentially suboptimal medicine regimens, longitudinal patterns of polypharmacy and outcomes of polypharmacy, PIMs and underprescribing. We searched MEDLINE (1990–2021) and using manual checking of reference lists for original research. Databases were searched using combinations of free text and Medical Subject Headings (MeSH) terms for polypharmacy, inappropriate prescribing, medication errors, drug prescriptions, age, adults, risk factors, drug interactions, potentially inappropriate medication lists and trends (see Box 1 for a more detailed sample of search terms). This review focused on general populations, excluding populations experiencing specific disease states. Data on population, indicators of suboptimal regimens, prevalence, associations and clinical outcomes were extracted and tabulated for synthesis.
Examples of key search terms.

Ageing – epidemiology
Globally, projections suggest the number of older adults, aged 65 years and above, will exceed 1.5 billion by 2050. 2 Multimorbidity is estimated to affect approximately 65% of adults aged 65–84 years and up to 82% among those aged 85 years or older. 3 To manage these morbidities, medicines are one of the most common treatments in health care. However, older adults are a heterogeneous group, encompassing those who are robust and in good health, as well as those with significant frailty and high burden of disease. 4 These factors can make optimal prescribing complex. Ageing is associated with a range of physiological changes, including decreased kidney and liver function and loss of total muscle mass, 5 which can affect the absorption, distribution, metabolism and excretion of medicines, and may increase the risk of adverse events and medicine-related harm.6,7 Consequently, older adults are regularly excluded from drug trials and dosing is often extrapolated from younger, healthier populations, which may not be appropriate in older age. 8 To further confuse the situation, prescribing guidelines often focus on single diseases and do not consider potentially harmful medicine combinations or disease contradictions associated with having two or more chronic conditions, which is common in older age. 8 With increasing burden of disease and complexity of medicine regimens, medicine-related harm is increasingly being understood as a new risk factor for disease in older adults.9,10
Medicine-related harm
Medicine-related harm is often recognized as a preventable cause of harm, which may occur with an error in the provision or management of therapy. 11 Medicine-related harm has been associated with poor health outcomes, including increased falls and fractures, confusion, loss of appetite, functional decline, hospitalization and mortality. 4 The WHO has projected that medicine errors cost US$42 billion per annum. 1 Estimates suggest that 1 in 10 patients admitted to hospital will have experienced an adverse medicine event, either leading to or during their hospitalization. 12 A recent systematic review indicated that more than half of adverse events could have been prevented with safer prescribing practices. 13 Inability to consider the total impact of age-related changes, burden of disease and the number and type of medicines used by older adults may put them at greater risk of adverse events and medicine-related harm. 14 A such, careful and regular review is essential for identifying risk and preventing avoidable harm. To support this effort, there is an expanding body of research describing and applying a range of tools and indicators for identifying potentially suboptimal medicine regimens among older adults both in the community and higher risk settings.
Potentially suboptimal medicine regimens
Potentially suboptimal medicine regimens is a term that considered an individual’s entire regimen and captures a range of indicators that aim to identify medicine use that may increase risk of medicine-related harm. The concept measures the intensity of multiple medicine use, which is called polypharmacy. It also considers the quality of medicine regimens by identifying specific medicines used, or not used, to determine their potential appropriateness. These indicators include underprescribing, and high-risk prescribing, which encompasses the use of PIMs, specific medicines with anticholinergic or sedative properties, which are a subset of PIMs, and prescribing cascades. Table 1 provides a brief outline of the key indicators of potentially suboptimal medicine regimens.
Indicators of potential suboptimal medicine regimens.

PIMs, potentially inappropriate medicines.
The appropriateness of an individual’s medicine regimen is often highly contextual. A range of quality indicators have been developed, including both implicit and explicit measures. Factors including overall health and life expectancy, current diagnoses, which may include multiple comorbidities, previously unsuccessful treatments or intolerances, as well as the patient care goals and values must all be considered to investigate true regimen appropriateness.17,20 Implicit measures, or qualitative assessments, are judgement-based and capture these contextual factors. Trained health care professionals provide an individualized assessment of a patient’s medicine regimen, which is often informed by a patient interview and/or review of full medical history. While this level of detail is achievable in clinical settings, access to these data is less common in research, particularly in large epidemiological studies. Therefore, research often relies on explicit tools to assess the quality of medicine regimens. These tools are more objective and criteria-based measures, designed to minimize the need for clinical judgement. These features make the application of explicit tools more accessible to a wider range of users and may be appropriate for measuring regimen quality across populations. However, with the ease of application, assumptions must be made on a population level; therefore, explicit measures may only provide an estimate of
Polypharmacy (overprescribing)
Polypharmacy, polytherapy or the use of multiple medicines is one of the broadest measures of potentially suboptimal medicine regimens. The intensity of medicine use may be estimated using the number of medicines to indicate where potential overprescribing may be occurring. There is no universally agreed-upon definition for polypharmacy; it may be captured according to a continuous measure of the number of medicines or be defined by a cut point of a specific number of medicines. 21 A recent systematic review investigating polypharmacy definitions found substantial heterogeneity in the cut points applied across the literature, ranging from the use of ⩾2 concurrent medicines up to ⩾21 medicines. 15 Variability has also been observed in the criteria for measuring polypharmacy, including numeric only measures, for example, ⩾10 medicines; numeric measures with conditions, for example, ⩾6 medicines taken in the previous 7 days; or qualitative measures, for example, more medicines than clinically necessary. 15 While qualitative measures may provide the best estimate of whether overprescribing may be occurring, access to quality data is often a limitation. Therefore, the quantitative, ⩾5 medicine cut point is the most common measure of polypharmacy in published research,15,21 and appears to be the most appropriate cut point for identifying those at possible risk of harm.22,23 Hyperpolypharmacy or excessive polypharmacy is generally considered to be ⩾10 medicines.
Applying measures of polypharmacy
Point prevalence
Polypharmacy is a commonly applied indicator of potentially suboptimal medicine regimens. Most studies focus on older cohorts, aged 65 years or older, some studies also include middle-aged adults, while the addition of younger cohorts appears less common among general populations (Table 2). Applying a ⩾5 medicine cut point, the prevalence of polypharmacy reported in the literature ranged from 7.0% up to 83.0%;24,25 however, there was substantial heterogeneity across the literature in terms of study population, age group, methods for counting medicines and geographic locations (Table 2). The inclusion of younger cohorts generally appears to lower the prevalence of polypharmacy across the literature; however, similar estimates were observed between an older sample of community-dwelling men (35.9%) 26 and primary care outpatients aged 20 years or older (39.2%), 27 which suggests in some cases the study context may be just as important a determinant as age (Table 2). Hyperpolypharmacy (⩾10 medicines) was less commonly measured and ranged from 2.0% to 23.8%.28,29
Summary of studies reporting polypharmacy prevalence estimates. a

CAMs, complementary and alternative medicines; ICU, intensive care unit; OTC, over the counter; PRN, as required.
The table sorted according to polypharmacy measures.
Polypharmacy definitions also included more nuanced methods of counting, beyond whether medicines were doctor-prescribed or self-prescribed. While some definitions were non-specific, others counted medicines according to the number of active ingredients or focused on specific administration routes, such as only counting medicines with systemic action (Table 2). Polypharmacy was also defined according to administration frequency, medicines taken regularly or PRN (as required), or used within specific time frames, such as co-prescription, which describes the number of medicines dispensed at one time, the total medicines or mean medicines used daily, weekly, monthly or in the previous year (Table 2). However, because study setting, age group and methodology vary substantially between studies, it is unclear the effect definitions have on overall prevalence measures and whether results can be compared. Furthermore, it should also be acknowledged that quantitative measures of polypharmacy, alone, are unlikely to distinguish between appropriate and inappropriate polypharmacy. Appropriate polypharmacy is possible within the context of multiple comorbidities, where the prescription of several medicines is following the best evidence; while inappropriate polypharmacy occurs when regimens include unnecessary treatments or potentially harmful medicine combinations. 69
Risk factors for polypharmacy
Identifying the risk factors associated with polypharmacy may highlight who may be disproportionately affected by potential overprescribing. Of the studies investigated, increasing age appears to be a consistent predictor of polypharmacy (Table 3), although there is some evidence risk may decrease slightly among the very old (aged 90 or older).32,70 The relationship between sex and polypharmacy was mixed; however, being female was a commonly identified risk factor (Table 3). Indicators of poorer health were variably defined across the literature and may include the Charlson comorbidity index, 71 crude number of chronic diseases,32,41,43,60,62,70 binary indicators of specific chronic conditions28,30,33,50,67,68 or comorbidity (present/absent),51,55 frailty 28 or poor self-perceived health.30,33,67 Poorer health appears to be a strong predictor of polypharmacy and hyperpolypharmacy across a range of age categories and study populations (Table 3), though it remains unclear whether polypharmacy is causing poorer health or polypharmacy is required due to poor health.
Direction of association between polypharmacy and commonly reported risk factors. a

CAMs, complementary and alternative medicines; NA, no association; NL, non-linear association; OTCs, over the counters; PRN, as required; ↑, positive association; ↓, negative association.
The table sorted according to sample age.
Born between 1956 and 1965.
Born between 1946 and 1955.
Born before 1946.
Several studies also investigated the relationship between education and polypharmacy, with a growing body of evidence to suggest lower education may be associated with the use of more medicines (Table 3). Of interest, studies finding no association or the inverse relationship tended to apply a definition that included over the counter (OTC) and complementary or alternative medicines (CAMs), in addition to prescription medicines.55,73 This suggests the predictors of prescription, OTC and CAMs use may differ according to education level. The relationship between polypharmacy and indicators of social disadvantage seems to be less frequently investigated (Table 3). As with measures of poor health, social disadvantage was also defined according to a range of measures, including area-level indicators of relative social advantage and disadvantage,32,72 household income43,55,62 and employment status.55,73 The emerging evidence appears to have been observed among relatively younger cohorts, compared with other risk factors, with inconclusive results (Table 3). Two studies reported models that did not adjust for indicators of poorer health,72,73 as a likely confounder in the social disadvantage–polypharmacy relationship.74,75 A single study found greater social disadvantage was protective against polypharmacy in Brazil, 43 though it remains unclear whether the decreased risk may be driven by barriers to accessing health care and subsequent potential underprescribing.
From a broader perspective, the direction of the relationships predicting polypharmacy appears relatively stable among studies investigating associations among older cohorts (Table 3). However, the conflicting results observed for sex,41,60 education 73 and social disadvantage 43 seem to be occurring in samples that include younger and middle-aged adults, which may suggest the predictors of polypharmacy differ across age groups (Table 3). Two studies conducted age sub-analyses in Australian and South Korean populations. In South Korea, the study found no change in direction of associations between paediatric and adolescent participants (aged <20 years) and adults (aged ⩾20 years), although the strengths of relationships did vary between the age groups. 62 The Australian study stratified age groups into young baby boomers [aged 43–52: estimated from the year of data collection (2008) and birth year defined as 1956–1965], baby boomers [aged 53–61: estimated from the year of data collection (2008) and birth year defined as 1946–1955] and older adults [aged ⩾62: estimated from the year of data collection (2008) and birth year defined as born before 1946]. The study found both significant and non-significant associations for sex and education across the three age strata. 73 Despite substantial evidence to support a relationship with increasing age, there is limited research investigating how age interacts with other potential predictors of polypharmacy.
Longitudinal patterns of polypharmacy
Having investigated risk factors cross-sectionally, this section of the review focuses on studies measuring polypharmacy over more than one timepoint. Several studies have investigated the ecological trends in polypharmacy over time, using a repeat cross-sectional study design. Findings indicate the prevalence of polypharmacy and hyperpolypharmacy have increased over the last one to two decades.9,72,76–78 Studies have detected a near doubling of those using ⩾5 prescription medicines (8.2–15%;
Changes in prevalence over time
Of the studies investigating polypharmacy over time, following the same cohort, definitions varied. Studies applied binary cut points ranging from ⩾2 to ⩾10 medicines and continuous measures indicating the mean number of medicines (Table 4). Over the study durations, which ranged from 3 to 12 years, both the prevalence of polypharmacy and mean number of medicines use increased (Table 4). While one Swiss population-based study found a significant increase in polypharmacy over 5.5 years among those aged 35–75, 80 studies largely focus on older populations aged ⩾65 years (Table 4). The underlying reason for the growth in the number of medicines used by older adults is likely multifactorial. Proposed explanations have included the availability of new medicines, changes in prescribing recommendations, increased focus on preventive therapies and clinical guidelines for single disease states.37,54,72 The optimal management of some common chronic conditions may result in the prescription of multiple medicines. Anecdotally, research investigating the development of polypharmacy in younger cohorts tends to focus on specific disease contexts, for example, among patients with HIV, 81 cerebral palsy 82 or mental illness, 83 where the use of multiple medicines may be expected. Studies tracking the development of polypharmacy among the general population before reaching older age appear less common. With findings indicating that medicines use appears to be increasing both in the community and over time, cross-sectional research may be insufficient in identifying who may be at risk of polypharmacy in the future.
Change in polypharmacy prevalence over time. a

CAMs, complementary and alternative medicines; CI, confidence interval; OTC, over the counter; PRN, as required; SD, standard deviation.
The table sorted according to polypharmacy measures.
Associations with changes in polypharmacy
Studies investigating associations with changes in polypharmacy used a range of study designs and polypharmacy measures to analyse longitudinal data. However, the outcome was most defined according to the number of medicines or polypharmacy status at baseline and follow-up (Table 5). Less frequently applied methods included incidence of polypharmacy, 70 exposed to polypharmacy for ⩾80% of the study period (chronic exposure), 90 or a multinomial analysis investigating differences in associations between polypharmacy initiation, reduction or maintenance according to exposure baseline and follow-up. 80 For most studies, the time to follow-up ranged from 3 to 5.5 years (Table 5), except for one study that assessed the long-term predictors of medicine used over 11.7 years. 89 However, with data on only 160 older adults and a substantial number of predictor variables, this study was likely underpowered.
Associations with change in polypharmacy. a

β, coefficient; CI, confidence interval; HR, hazard ratio; NA, no association; OR, odds ratio; OTC, over the counter.
The table sorted according to age group.
Time in study was used.
Increasing age was associated with a greater number of medicines, polypharmacy incidence and probability of high exposure time at follow-up (Table 5). One analysis measured time in the study, which is likely to act as a function of age, with similar findings. 85 A reduction in polypharmacy was also associated with increasing age when compared with those with no polypharmacy at baseline or follow-up. 80 The number of medicines used at baseline appears to be a consistent predictor of higher medicine use and polypharmacy at follow-up (Table 5). Evidence also suggests that greater morbidity may increase the likelihood of polypharmacy in the future, particularly among those with diagnosed coronary heart disease, heart failure and diabetes, as well as positive correlations between polypharmacy and total number of comorbidities (Table 5). However, there is limited research investigating the relationship between indicators of socioeconomic status and changes in exposure to medicines or polypharmacy over time (Table 5). Of the three studies considering factors such as education, source of income or whether individuals were living as a couple or alone,70,80,89 education appears to be the only factor showing any significant association (Table 5).
While longitudinal studies may provide insight into the mechanism driving polypharmacy, there is emerging evidence highlighting that polypharmacy is not a time-stable exposure, rather it may be transient or consistent over time and within-person trajectories may vary.86,91,92
Of the research investigating longitudinal associations with polypharmacy, only two studies considered polypharmacy as a time-variant exposure.80,90 Abolhassani
Clinical implications of polypharmacy
There is a substantial body of research investigating the clinical implications of polypharmacy, including several reviews that have synthesized the existing research.93–96 Of note, the association between polypharmacy and drug–drug or drug–disease interactions and adverse events is generally accepted across the literature.97,98 However, research is heterogeneous, often focusing on more sensitive indicators of high-risk prescribing, such as specific drug–drug or drug–disease interactions, rather than considering the broader total number of medicines use. This section provides a brief overview of studies reporting longitudinal outcomes of polypharmacy (Table 6).
Outcomes of polypharmacy. a

CI, confidence interval; HR, hazard ratio; ICU, intensive care unit; NA, no association; OR, odds ratio.
The table sorted according to study duration.
Borderline significant.
Lower score indicates reduced capacity or function.
Lower score indicates a greater degree of malnourishment.
There is some evidence to suggest those experiencing polypharmacy (binary ⩾5 medicines) may have a greater probability of mortality, with research also demonstrating a significant dose–response relationship with the number of medicines used (continuous measure; Table 6). While two studies showed no association between polypharmacy and mortality, the null findings were observed among relatively younger cohorts.46,103 The number of medicines used also appears to be a significant predictor of re-hospitalization post-discharge, with each additional medicine contributing to a 3–11% increase in risk.46,101 When investigating the relationship between physical function and polypharmacy, capacity was measured using a range of tools and tests, with mixed findings (Table 6). Of interest, using participants with no polypharmacy at baseline or follow-up as the reference group, a UK study found extended exposure may be linked to a significant reduction in sit-to-stand and walking speed, balance and grip strength. 102 However, associations between current or previous exposure to polypharmacy were less consistent across the same indicators of physical function. 102 There is some evidence that polypharmacy may be associated with a decline in cognitive function; however, findings were only significant when a ⩾10 medicine cut point was applied, 87 among those exposed to ⩾5 medicines at more than one follow-up. 102 The findings from an Australian study also suggest that the greater number of medicines used may increase the risk of experiencing a cardiovascular event in the following 4.5 years. 26 While a study in Finland found older adults with hyperpolypharmacy may experience a 38% decline in their nutritional state over a 3-year follow-up, however, no association was observed among those using 5–9 medicines. 87
Gaps in the literature
Future polypharmacy research may address several gaps in the literature, including an investigation into the impact of different polypharmacy definitions on polypharmacy prevalence estimates and how the predictors of polypharmacy may vary across the age groups. Studies applying quantitative definitions may also consider qualitative indicators of polypharmacy or broader measures of overall prescribing quality, including exposure to PIMs and potential underprescribing. This distinction may enable an enhanced ability to distinguish between instances of appropriate and inappropriate polypharmacy While cross-sectional research may provide insight into who may be at greater risk of potentially suboptimal medicine regimens at a single timepoint, this design is unable to address the temporal nature of relationships. Current research investigating longitudinal associations with polypharmacy appears to focus on older adults; therefore, future work should include adult and middle-aged populations, with the potential to identify characteristics present in younger age that may predict polypharmacy in older age. Research investigating the transitions between states of polypharmacy exposure and non-exposure is also needed; however, data collection at each timepoint should capture medicine use over a set period to distinguish between chronic and potential transient polypharmacy exposure. Finally, future work should investigate associations between within-person trajectories in medicine use, identifying those who may be at greater risk of more rapid increases in medicine use, further exploring how medicine use in younger age may influence trajectories of medicine use in older age. The implications of these gaps in the literature suggest that polypharmacy research may not be developed enough for clinical application at this time as appropriate cut points remain uncertain.
Omitted medicines (underprescribing)
Underprescribing, prescribing omissions or omitted medicines occurs when an individual is not prescribing a potentially beneficial medicine, indicated for the treatment or prevention of a disease or condition. 37 Paradoxically, polypharmacy has been identified as a risk factor for underprescribing.37,104,105 In the context of an already complex medicine regimen, clinicians may hesitate to prescribe preventive therapies or contribute to the overall medicine burden and choose to prioritize the management of current conditions. 38 In some instances, particularly in end-stage care, the rationale for underprescribing is valid; however, avoiding essential pharmacotherapy can also pose a risk to patients’ safety and may reduce quality of life.8,20
Tools have been developed to assist clinicians and researchers to evaluate when potential underprescribing may be occurring. One of the most common tools is the Screening Tool to Alert doctors to the Right Treatment (START), an explicit list of criteria that considers common instances of potential underprescribing, where no contraindications exist and where life expectancy and functional status justify the prescription. 17 While implicit tools, such as the Assessment of Underutilization (AOU) tool, will likely provide a more accurate estimate of potential underprescribing, the AOU requires a detailed medical history, a complete list of current medicines and the clinical judgement of a trained health care professional, which are not always available in population-based research. 17
Applying measures of underprescribing
Point prevalence
Studies measuring underprescribing were less commonly reported in the literature than polypharmacy. Most studies were sampled from a patient population, with only four studies reporting prevalence estimates for community-dwelling or population-based samples (Table 7). This pattern may reflect the challenges associated with collecting complete data on current medicines and diagnoses, both of which are required to determine when a potentially beneficial medicine may have been omitted. Prevalence estimates ranged from 12% to 64.2%;50,104 however, study settings varied substantially between studies (Table 7).
Summary of studies reporting prevalence estimates for underprescribing. a

RACF, residential aged care facility.
The table sorted according to study population/setting.
Risk factors for underprescribing
Evidence investigating the risk factors for underprescribing is limited and appears inconclusive (Table 8). While there is some evidence to support an association between older age and polypharmacy, a greater number of studies reported no significant relationship with these risk factors (Table 8). Education and income do not appear to be associated with underprescribing (Table 8), except for one study, which found a non-linear relationship with educational attainment among primary care patients in Kuwait. 52
Direction of association between potentially underprescribing and commonly reported risk factors. a

NA, no association; NL, non-linear association; ↑, positive association; ↓, negative association.
The table sorted according to study setting.
Clinical implications of underprescribing
Research investigating clinical outcomes of underprescribing is also limited (Table 9). Evidence suggests that underprescribing may be linked to increased risk of cardiovascular events in a sample of older Australian men. 26 While the odds of all-cause hospitalization were greater among Māori New Zealanders with underprescribing, however, no association was observed in the non-Māori study sample. 106 No association between underprescribing and mortality was observed in either study (Table 9).
Outcomes of underprescribing – associations with hospitalization and emergency department visits.
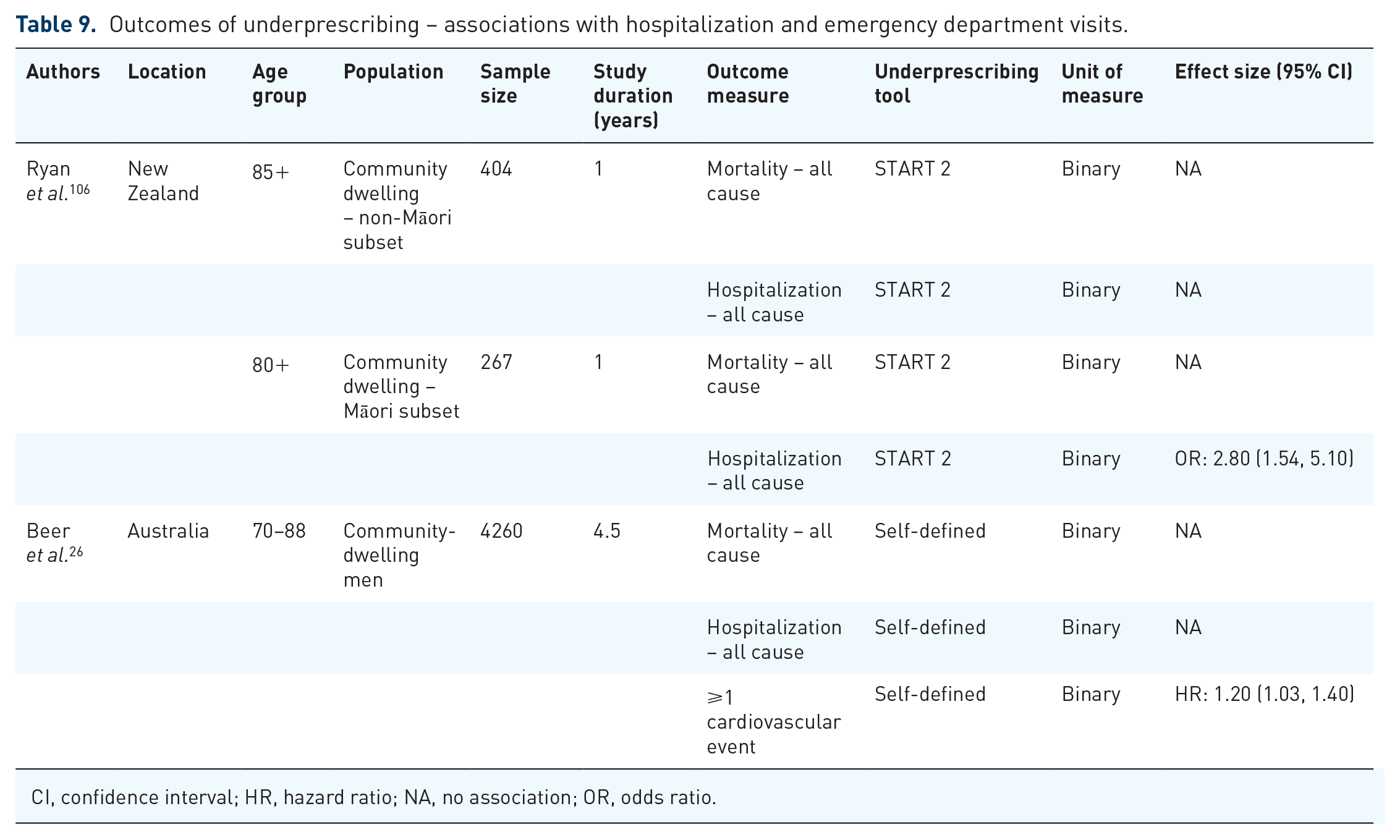
CI, confidence interval; HR, hazard ratio; NA, no association; OR, odds ratio.
Gaps in the literature
There is a paucity of research investigating the risk factors and clinical outcomes of potential underprescribing. It remains unclear whether there is a relationship between underprescribing and social disadvantage, and whether it is possible to distinguish between underprescribing resulting from the receipt of potentially suboptimal care or a lack of access to health care more generally. It is also challenging to interpret underprescribing at a population level and whether instances may be inappropriate or conscious, in the context of shared decision making. Likewise, in clinical practice, clear documentation regarding the reasons not to prescribe would be beneficial across transitions of care.
High-risk prescribing: PIMs
Indicators captured under the banner of high-risk prescribing evaluate the medicines used by older adults to provide a more sensitive assessment of potential inappropriateness. PIMs are medicines that are known to be potentially harmful when used by older adults, where the potential risk outweighs the anticipated benefit, particularly when safer or more effective alternatives for the same condition are available.110,111 To assist clinicians, pharmacists and researchers to evaluate the potential appropriateness of a regimen, a range of tools have been developed to monitor, prevent and minimize the use of PIMs in older populations. As with underprescribing, screening tools may be implicit (judgement-based) or explicit (criterion-based) by design. Implicit tools, (e.g. Medication Appropriate Index (MAI)) can be applied to any medicine and score their appropriateness according to a set of questions to evaluate factors such as indication, effectiveness, potential for interactions and duration. 112 This patient-level assessment is an effective quality assessment and may be applied to any regimen, in any setting or population. 112
PIMs can also be measured using explicit tools, which are generally developed through literature review, expert opinion and consensus panels of health care professionals. The tools can range from simple lists of medicines and medicine classes that should be avoided in older adults, to more complex lists that may also consider dosage, duration, other medicines, current diagnoses and functional state to assess regimens.113–115 PIMs tools may also vary in their target population, some developed for community-dwelling older adults, while others focus on specific settings or disease states.113–115 With minimal clinical judgement required, PIMs tools are often appropriate for application to a range of users and data, including routinely collected administrative data. 8 There has been a proliferation of PIMs tools over the past decade, developed internationally to capture country-specific approved medicines, local treatment practices and specific therapeutic, and prescribing guidelines.113,114 Two of the most common explicit tools are the US-developed Beers criteria, which is updated approximately every 3 years, 116 and the European consensus Screening Tool of Older Persons’ Prescriptions (STOPP), which is currently in its second iteration.116,117 While PIMs tools may be considered appropriate for application in the country of origin, their refection of national formularies is often recognized as a limitation to translation to other contexts.113,115
Applying measures of PIMs
Point prevalence
The prevalence of PIMs appears to be a widely reported statistic. Estimates range from 10.3% to 90.6%;25,44 however, the tools used to measure prevalence varied across study populations (Table 10). It was common for authors to acknowledge the differences between the tools by investigating more than one PIMs tool in the same study sample.36,44,52,64,65,100,103,108,118–125 Two studies were of particular interest, applying an implicit tool, the MAI, as a more sensitive indicator of inappropriate prescribing, alongside a selection of explicit tools to investigate their ability to accurately diagnose PIMs within a defined population.52,125 With the MAI as the reference tool, Table 11 provides a summary of the explicit tools’ accuracy according to sensitivity and specificity statistics. Across both studies, the explicit tools used do not appear to discriminate between those with and without PIMs well (Table 11). While the STOPP version 2 appears relatively consistent across both studies, with moderate sensitivity and good specificity (Table 11), variability in the overall results does highlight the potential for imprecision between the tools and the likelihood for error in identifying PIMs according to explicit criteria.
Summary of studies reporting PIMs prevalence estimates. a
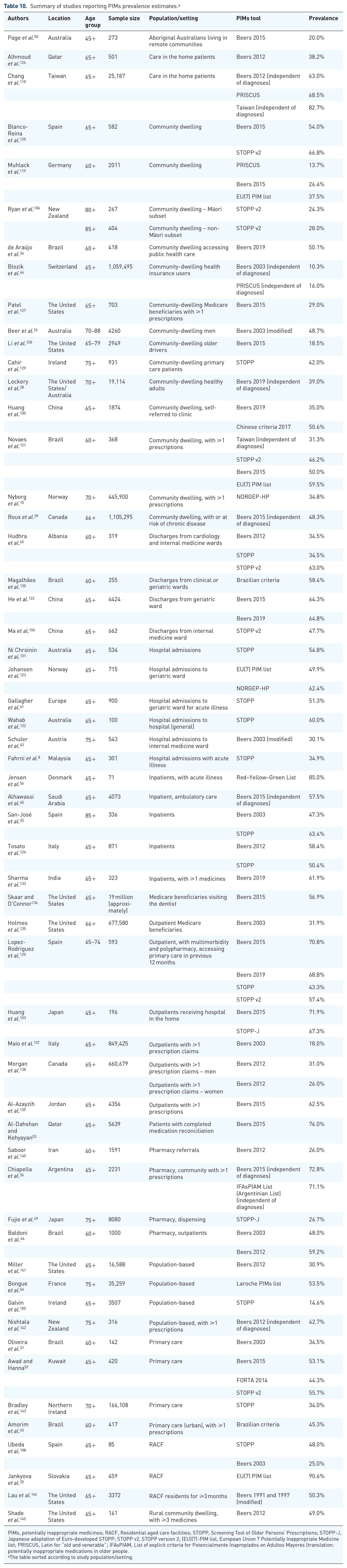
PIMs, potentially inappropriate medicines; RACF, Residential aged care facilities; STOPP, Screening Tool of Older Persons’ Prescriptions; STOPP-J, Japanese adaptation of Euro-developed STOPP; STOPP v2, STOPP version 2; (EU)(7)-PIM list, European Union 7 Potentially Inappropriate Medicine list; PRISCUS, Latin for “old and venerable”; IFAsPIAM, List of explicit criteria for Potencialmente Inapropiados en Adultos Mayores (translation: potentially inappropriate medications in older people.
The table sorted according to study population/setting.
Diagnostic test accuracy of explicit tools, using an implicit tool as the reference standard.

MAI: Medication Appropriate Index; STOPP, Screening Tool of Older Persons’ Prescriptions; STOPP v2, STOPP version 2.
Sensitivity/specificity interpretation: 91–100% – Excellent, 81–90% – Good, 71–80% – Moderate, 61–70% – Fair, 51–60% – Poor, <50% – Very poor.
Risk factors for PIMs
There also appears to be a substantial body of research investigating associations with PIMs use. The association between age and PIMs appears inconsistently across the literature (Table 12). Of interest, two studies reported conflicting associations between age and PIMs using two different tools in the same population.100,118 Both studies, conducted in China and Taiwan, found the risk of PIMs use decreased with age when PIMs were identified according to locally developed tools (the Chinese criteria 2017 and the Taiwan criteria, respectively); inversely, when PIMs were measured using US-developed Beers criteria, there was a positive association with increasing age.100,118 These findings further highlight potential variability in the applicability of tools in different study settings. The female sex was often associated with an increased risk of PIMs use; however, no association with sex was also just as common (Table 12). One study presented age sub-analyses looking at younger–older adults (65–74 years) and older adults (⩾75 years) and found being female was associated with an increased risk of PIMs use among the younger–old, while no association was observed among older adults. 146 These variable results suggest the risk factors of PIMs use may change with age and that sub-analyses may be an important consideration.
Direction of association between PIMs and commonly reported risk factors. a

NA, no association; NL, non-linear association; PIMs, potentially inappropriate medicines; STOPP, Screening Tool of Older Persons’ Prescriptions; STOPP v2, STOPP version 2; ↑, positive association; ↓, negative association.
~ stroke = NA; diabetes = ↑.
The table sorted according to study population.
The relationship between poorer health and the use of PIMs also appears mixed (Table 12). Studies that applied a modified version of a PIMs list, for example, excluding any criteria where diagnoses were required to assess potential appropriateness, tended to find an association between increased risk of PIMs use and poorer health.28,39,134,137,149 Alternatively, those that appeared to apply the full criteria, as published, were more likely to find no association with poorer health (Table 12). This suggests that accounting for diagnoses when identifying PIMs may, in part, be controlling for potential confounding by indication. Compared with indicators of poorer health, polypharmacy appears to be a more reliable predictor of PIMs use (Table 12). While the debate continues around the distinction between appropriate and inappropriate polypharmacy, this well-established link between polypharmacy and PIMs suggests even considered polypharmacy may contain specific drug–drug or drug–disease interactions that are suboptimal. Of interest, only a handful of studies that focused on measuring associations with PIMs provided a detailed definition of polypharmacy;28,45,52,64,66 however, it remains unclear whether associations vary between doctor-prescribed and self-prescribed medicines. The relationship between education and PIMs use appears to have been less extensively researched, relative to the polypharmacy literature (Table 12). Studies reporting an association between lower education appear to have been measured among community-dwelling or population-based samples,66,71,134,141 while those who found no association tended to be observed among patient populations.52,64,109
Evidence of an association between indicators of social disadvantage and PIMs use is mixed (Table 12). However, the relationship with social disadvantage is likely to be highly contextual and variability in the tools used to measure PIMs and the social, economic and political settings in which these findings were observed may have influenced the inconsistent results.
Clinical implications of PIMs use
One of the major limitations of published PIMs tools is that they have been developed
Mortality
There is some evidence to suggest older adults using one or more PIMs have an increased probability of mortality (Table 13). However, it would appear studies investigating a longer survival time (⩾5 years) were more likely to find an association, compared with those with a shorter study duration (Table 13). Of interest, studies applying locally developed PIMs tools appear more likely to have significant associations with mortality, relative to their internationally imported counterparts. For example, participants exposed to medicines listed on the Finnish-developed Med75+ criteria for 1, 3 and 6 months in Finland experienced an increased probability of mortality. 154 Similarly, adults with a disability receiving hospital in the home in Japan who used ⩾1 PIMs, defined according to the STOPP-J, the Japanese adaptation of Euro-developed STOPP, also saw a positive association with mortality. Yet when the US-developed Beers 2015 criteria were applied to the same Japanese sample, no association was observed. 103 This suggests that when applied within the intended geographic location, PIMs tools may be more precise in detecting clinically significant PIMs. Furthermore, looking back at the prevalence of PIMs, the same Japanese study 103 reported a higher PIMs prevalence for the Beers 2015 criteria (71.9%) compared with the STOPP-J (67.3%) (Table 10). This may highlight an issue of discrimination where utilization alone may be a misleading indicator of risk.
Outcomes of PIMs – associations with mortality. a

CI, confidence interval; HR, hazard ratio; NA, no association; PIMs, potentially inappropriate medicines; STOPP-J, Japanese adaptation of Euro-developed STOPP; STOPP v2, STOPP version 2.
The table sorted according to study duration.
Hospitalization
The use of PIMs has been linked to an increased risk of hospitalization, re-hospitalization and emergency department visits (Table 14). A novel study method was used in Germany among a population-based sample of older adults, where a case control–type design grouped exposed individuals, who used medicines on the German-developed PRISCUS list, and unexposed individuals, who used medicines that were considered to be the safer alternative to PIM on the PRISCUS list.
155
The study found that compared with those taking a safer alternative, those using ⩾1 PRISCUS PIMs were 38% more likely to be hospitalized in the proceeding 6 months.
155
In Japan, Huang
Outcomes of PIMs – associations with hospitalization and emergency department visits. a

CI, confidence interval; HR, hazard ratio; NA, no association; OR, odds ratio; PIMs, potentially inappropriate medicines.
The table sorted according to study duration.
Borderline significant.
Falls, fractures, physical function and frailty
There is also some evidence linking the use of PIMs with an increased risk of falls and fractures (Table 15). Both studies considered degrees of exposure, defined according to the number of months with a PIM or whether PIMs use was classified as regular or occasional use.154,158 When considering specific subclasses within a PIMs tool, it would appear some medicines are more likely to be associated with falls, 158 which suggests applying a complete list, in its entirety, may be a blunt tool for assessing some outcomes. The evidence to support an association between PIMs use and a decline in physical function or incidence of frailty, however, is less compelling. Of the three studies investigating these outcomes, each looked at more than one PIMs list to investigate associations, often with mixed findings (Table 16). Of note, a study from Germany found no association between the locally developed PRISCUS list and 6-year incidence of frailty; however, an increased probability was observed when PIMs were measured using the Beers 2015 criteria from the United States. 159 This appears to go against the trend observed in the Japanese study reporting associations with mortality and hospitalization using a locally developed tool. 103 A possible explanation is that the PRISCUS tool was published in 2010 and may no longer reflect the current challenges associated with inappropriate prescribing in Germany, and while the Beers 2015 criteria are not native to the study population, a more recently updated tool may be the sharper instrument for detecting clinically significant PIMs.
Outcomes of PIMs – associations with falls and fractures.

CI: confidence interval; HR, hazard ratio; NA, no association; OR, odds ratio; PIMs, potentially inappropriate medicines.
Subset of a combined list using the Beers 1991 criteria and the Laroche PIMs list.
Borderline significant.
Outcomes of PIMs – associations with physical function and frailty. a

CI, confidence interval; HR, hazard ratio; NA, no association; OR, odds ratio; PIMs, potentially inappropriate medicines; STOPP, Screening Tool of Older Persons’ Prescriptions; STOPP-J, Japanese adaptation of Euro-developed STOPP; STOPP v2, STOPP version 2.
The table sorted according to study duration.
Other clinically significant outcomes
In addition to the outcome discussed above, associations with quality of life and risk of cardiovascular events have also been considered (Table 17). There is some evidence that the use of two PIMs may be associated with a decrease in quality of life, according to the EuroQoL 5-Dimension (EQ-5D) utility, among a sample of community-dwelling older adults. 129 However, there is limited research investigating this outcome. One study investigated cardiovascular events as an outcome in Australia, finding no association. 26
Outcomes of potential suboptimal medicine regimens – other clinically significant outcomes.

β, coefficient; CI, confidence interval; NA, no association; PIMs, potentially inappropriate medicines; QoL, quality of life; STOPP, Screening Tool of Older Persons’ Prescriptions.
Gaps in the literature
There is evidence to indicate that PIMs are likely associated with poorer outcomes, which validates the tools beyond the expert opinion or consensus in which they were developed. However, it was not uncommon for studies to report conflicting results within the same study population when different PIMs tools were applied. While this suggests not all tools are equal in any given study setting, there is limited research available and it was not possible to compare the outcomes associated with specific tools across different study contexts. Research investigating the patterns and implications of PIMs use among the population must consider the applicability of the explicit tool(s) to the study setting. The most well-known PIMs tools may not be the most appropriate for all clinical contexts, and tool selection should be mindful of the intended purpose of the tool as well as the country in which it was developed. 115
Conclusion
There is a need for further research that distinguishes between transient and chronic exposure to polypharmacy and longitudinal studies that determine the trajectories of polypharmacy through adulthood into older age to better identify people at greatest risk. Research investigating underprescribing is limited and future research is warranted; however, it may be important to also consider indicators of health care utilization to better differentiate between instances of potential suboptimal prescribing and confounding by SES. Within PIMs research, substantial heterogeneity in tools, study contexts and populations of interest make it challenging to synthesize the evidence. It remains unclear how well PIMs tools developed internationally transfer to local settings, and thus the validity of many studies remains uncertain when applied internationally. As such, an evaluation of the applicability of tool(s) to specific contexts should be considered before the patterns and implications of PIMs are investigated. Addressing these gaps in the existing literature would contribute to the growing body of research on potentially suboptimal medicine regimens and build knowledge that may reduce the risk of medicine-related harm among older adults.

Footnotes
Acknowledgements
The authors have no acknowledgements; all contributors are listed as co-authors.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Prof. Christopher Etherton-Beer is an Editorial Board member of
Availability of data and materials
Not applicable.
