Abstract
Adverse drug reactions (ADRs) are common in older adults, with falls, orthostatic hypotension, delirium, renal failure, gastrointestinal and intracranial bleeding being amongst the most common clinical manifestations. ADR risk increases with age-related changes in pharmacokinetics and pharmacodynamics, increasing burden of comorbidity, polypharmacy, inappropriate prescribing and suboptimal monitoring of drugs. ADRs are a preventable cause of harm to patients and an unnecessary waste of healthcare resources. Several ADR risk tools exist but none has sufficient predictive value for clinical practice. Good clinical practice for detecting and predicting ADRs in vulnerable patients includes detailed documentation and regular review of prescribed and over-the-counter medications through standardized medication reconciliation. New medications should be prescribed cautiously with clear therapeutic goals and recognition of the impact a drug can have on multiple organ systems. Prescribers should regularly review medication efficacy and be vigilant for ADRs and their contributory risk factors. Deprescribing should occur at an individual level when drugs are no longer efficacious or beneficial or when safer alternatives exist. Inappropriate prescribing and unnecessary polypharmacy should be minimized. Comprehensive geriatric assessment and the use of explicit prescribing criteria can be useful in this regard.
Introduction
The prevalence of adverse drug reactions (ADRs) increases with age, with twice as many patients aged 65 years and older being hospitalized because of ADR-related problems than their younger counterparts [Beijer de Blaey, 2002]. Pirmohamed and colleagues, in a prospective study of 18,820 patients attending two general hospitals in the United Kingdom, found that ADRs contributed to 6.5% of admissions and 4% of bed capacity over a 6-month period, with most ADRs occurring in older patients [Pirmohamed et al. 2004]. Kongkaew and colleagues identified ADRs in 10% of hospitalized older adults and 6.3% of younger adults [Kongkaew et al. 2008]. Cooper reported that 67.4% of 332 nursing home residents experienced at least one ADR over a 4-year period, many having repeated ADRs related to antipsychotic drugs, nonsteroidal anti-inflammatory drugs (NSAIDs) and insulin [Cooper, 1996]. Recent data indicate that 60% of nursing home residents continue to experience ADRs, thus emphasizing ADRs as a pervasive problem in this vulnerable older cohort [Dilles et al. 2013].
More than 50% of ADR-related hospital admissions are preventable [Chan et al. 2001], most being attributable to diuretics, NSAIDs, antiplatelet, anticoagulant and antidiabetic drugs [Howard et al. 2007; Budnitz et al. 2011]. ADR-related hospitalizations have a median stay of 8 days [Pirmohamed et al. 2004]. In-hospital incident ADRs cause a 9% increase in length of stay and a 20% increase in cost of care encompassing bed consumption, laboratory and treatment costs [Khan, 2013]. An example of additional expenditure is the use of blood products which are needed in up to 20% of all ADR-related hospitalizations to treat gastrointestinal bleeding. Preventing such ADRs could reduce demand for blood products and could also impact on mortality [Rottenkolber et al. 2012]. A prospective study of 1225 ADR-related hospital admissions reported that 20 of 28 deaths were due to gastrointestinal or intracranial bleeding [Pirmohamed et al. 2004]. Overall ADR-related mortality in hospitalized patients ranges from 0.14% to 4.7%, death being more likely in those over 55 years and the greatest risk being in those aged 75 years and older [Pirmohamed et al. 2004; Lazarou et al. 1998; Budnitz et al. 2011].
ADRs can be difficult to recognize in older people as they often present with nonspecific symptoms, for example falls, fatigue, cognitive decline or constipation, all of which have several aetiologies. Falls, for example, may be secondary to chronic conditions such as osteoarthritis, visual impairment, myopathy or peripheral neuropathy. Numerous medications also independently increase the risk of falls, for example sedative hypnotics [Mets et al. 2010; Shuto et al. 2010], antihypertensives [Verhaeverbeke and Mets, 1997; Aronow, 2009] and antiarrhythmics [Ham et al. 2014]. Thus it can be difficult to precisely conclude whether or not a fall is attributable to an ADR in a multimorbid older patient who is prescribed such a drug. Nonetheless, potential ADRs should be part of every differential diagnosis in older patients. Failure to recognize an ADR may result in a prescribing cascade whereby a new drug is prescribed to treat the adverse effect of the culprit drug, thus exposing the patient to continuing risk of ADR from the culprit drug and additional risk from the newly prescribed drug [Rochon and Gurwitz, 1997]. An example of such practice is the prescription of an anticholinergic drug (e.g. biperiden) to treat the extrapyramidal effects of an antipsychotic drug (e.g. risperidone). The anticholinergic drug increases the risk of cognitive decline, orthostatic hypotension, blurred vision, constipation and urinary retention, which may adversely precipitate falls, delirium, functional and cognitive decline.
ADRs should be defined, recorded and their causality identified in order to minimize the risk of ADR recurrence and optimize outcomes for older patients. However, it can be difficult to reliably define and classify ADRs in everyday clinical practice using existing tools, particularly with regard to causality and preventability in patients with multiple contributory factors. In this narrative review article we discuss commonly used terminology, including definition, classification, causality and avoidability of ADRs. We describe the risk factors for ADRs in older adults and highlight the challenges in identifying those at greatest risk. We examine existing ADR risk prediction tools and other approaches to minimizing ADR risk, including medication reconciliation (MR), comprehensive geriatric assessment and pharmacovigilance. Finally, we describe a practical clinical approach to identifying and reducing ADR risk in older patients.
ADRs: terminology
Definition and classification
An ADR is defined as ‘an appreciably harmful or unpleasant reaction resulting from an intervention related to the intentional use of a medicinal product, which predicts hazard from future administration and warrants prevention or specific treatment, or alteration of the dosage regimen or withdrawal of the product’ [Edwards and Aronson, 2000, p. 1255]. ADRs were traditionally classified as being either ‘type A’ (typically dose related, predictable and accounting for 80% of all ADRs) or ‘type B’ (typically nondose related, unpredictable or idiosyncratic) [Rawlins and Thompson, 1977]. However, this simplistic classification has evolved to include six definitive categories, presented with examples in Table 1 [Edwards and Aronson, 2000]. An adverse drug event (ADE)is defined as ‘any physical or mental harm resulting from medication use be it misuse, under-dosing or overdosing’ [Nebeker et al. 2004, pp. 797–798]. Unfortunately, the terms ADR and ADE are often used incorrectly and interchangeably, thus leading to difficulties with reporting, interpretation and comparison of epidemiological and interventional studies. Standardized definition and robust classification of ADRs is essential to correctly identify and measure their frequency, predictor variables and related outcomes.
Examples of ADRs using Edwards and Aronson classification system [Edwards and Aronson, 2000].

ADR, adverse drug reaction.
Causality, severity and avoidability
Several standardized methods of assessing ADR causality exist, the advantages and disadvantages of each being summarized in Table 2 [Hire et al. 2013]. None is universally accepted or used in everyday clinical practice. No method is specifically validated for use in older patients with multiple comorbidities and multiple medications. The World Health Organization–Uppsala Monitoring Center (WHO-UMC) criteria [WHO] (Table 3) and the Naranjo criteria [Naranjo et al.1981] are frequently cited in the medical literature. Some of the variables required by the Naranjo criteria are difficult to apply to older patients with suspected ADR, for example it may be unethical to rechallenge if there is a high index of suspicion of ADR and risk of recurrence or harm is high. It is unethical to give placebo drugs in most jurisdictions. In addition, the Naranjo criteria do not allow for drug–drug interactions as a cause of ADR. Often, many of the required variables cannot be completed, thus making it unlikely for any older patient to score higher than ‘possible or probable’ in this causality system, thereby limiting its utility. ADRs are often subjectively classified as being mild, moderate, severe and lethal. Hartwig and colleagues robustly categorize ADR severity into seven groups according to clinical consequence, including resultant harm and intensity of medical intervention required (see Table 4) [Hartwig et al. 1992].
Commonly cited ADR causality assessment tools.
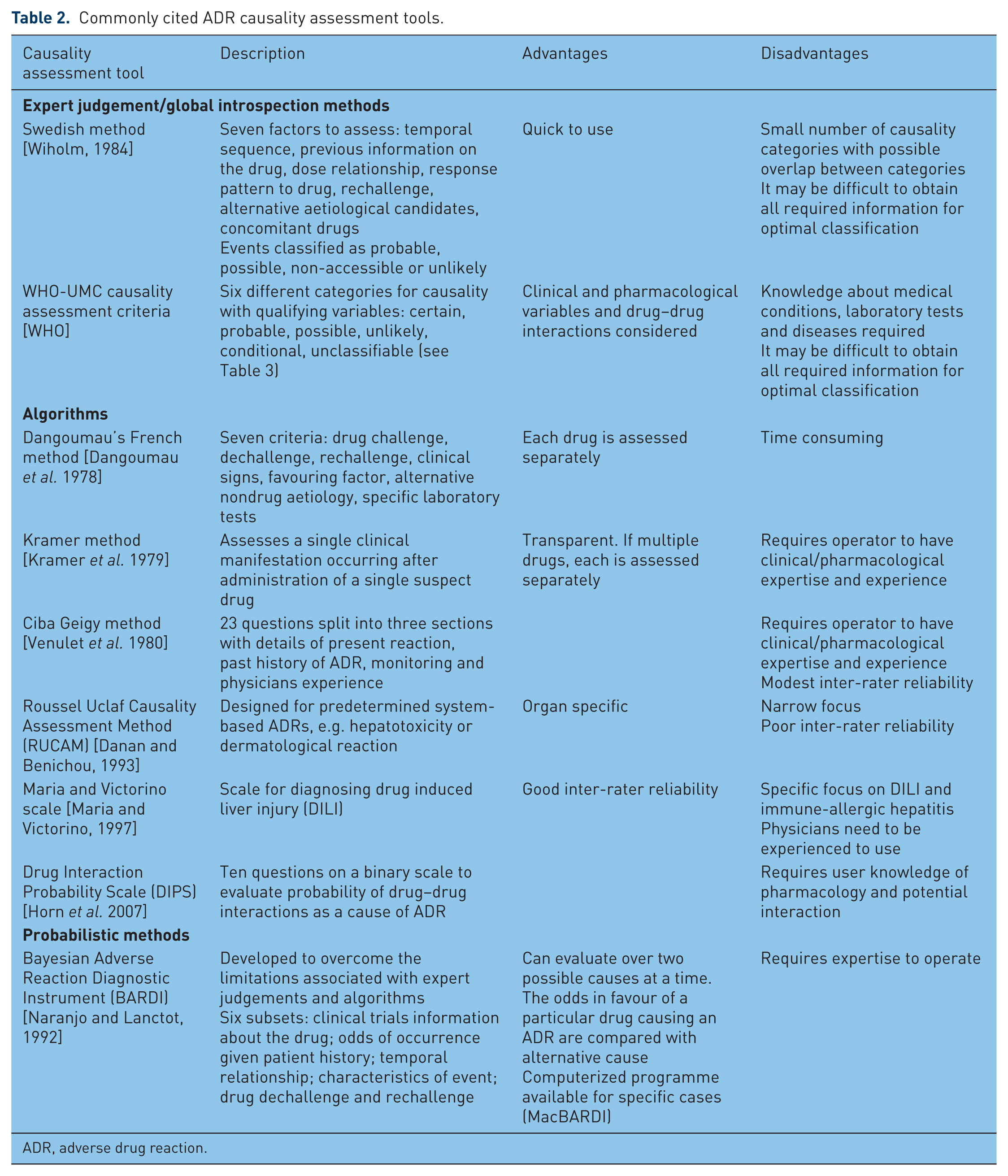
ADR, adverse drug reaction.
The WHO-UMC causality criteria [WHO].

UMC, Uppsala Monitoring Center; WHO, World Health Organization.
Determining the avoidability of an ADR should theoretically minimize occurrence and prevent reoccurrence. The Hallas criteria categorize ADR avoidability into four groups: definitely avoidable, possibly avoidable, unavoidable and unclassifiable [Hallas et al. 1990]. ADRs that are definitely or possibly avoidable are usually those in which organ dysfunction, homeostatic dysregulation, age-related changes in pharmacokinetics and pharmacodynamics and known drug–drug interactions predictably and adversely influence drug handling and response.
Risk factors for ADRs in older patients
Clinicians must be vigilant for recognized risk factors for ADRs in older patients in order to minimize their occurrence. These risk factors are briefly summarized below.
Age-related changes in pharmacokinetics and pharmacodynamics
Age-related physiological changes affect drug pharmacokinetics (absorption, distribution, metabolism and excretion) and pharmacodynamics (the study of effects of a drug on the body) [Mangoni and Jackson, 2004]. There is a reduction in lean muscle mass and water content with a relative increase in the proportion of total body fat. These changes influence the volume of distribution of many drugs (e.g. benzodiazepines, antipsychotics and opioids) which can, in turn, increase the risk of toxicity or ADR if doses are unadjusted. Benzodiazepines are lipophilic and generally have a larger volume of distribution in older patients compared with younger patients. Consequently, their elimination half life is prolonged, which can result in prolonged effect, drug accumulation, and greater potential for adverse effects including drowsiness and falls. Conversely, the volume of distribution for hydrophilic drugs (e.g. lithium) is reduced; this may result in toxicity if drug doses are not adjusted, particularly if there is concomitant renal impairment which may impede drug excretion.
Many drugs are protein bound when circulating in plasma (e.g. phenytoin and ibuprofen) and exert their effects through their free or unbound fractions. Lower serum albumin levels can therefore result in higher free or active drug fractions with greater potential for adverse effects. Serum albumin levels are often reduced by chronic illness, malnutrition and dietary changes, which can be intentional or unintentional in older patients, for example disease or drug-related anorexia, reduced oral intake because of poorly fitting dentures or restricted food consistencies in patients with dysphagia. Liver mass and perfusion decline by as much as 40% with age [Woodhouse and Wynne, 1988], thereby impacting on drugs with a high hepatic extraction ratio, for example diltiazem, opiates and warfarin. Such drugs will have reduced first pass metabolism with consequent increase in systemic bioavailabilty and greater potential for higher serum drug levels and enhanced drug effect in older patients.
Prescribers must be aware of potential interactions between drugs that are metabolized by the hepatic cytochrome p450 enzyme system [Doan et al. 2013; Dolder et al. 2010]]. This is particularly relevant to older patients receiving multiple drugs, for example coprescription of haloperidol and amitriptyline. In this instance, haloperidol can impede the metabolism of amitriptyline through inhibition of cytochrome P450 2D6, thereby increasing the potential for anticholinergic side effects and orthostatic hypotension, which may contribute to falls and injury. Lists of cytochrome P450 enzyme inhibitors and inducers are readily available in most pharmacological texts. Glomerular filtration rate (GFR) declines with ageing because of reductions in renal size, perfusion and nephron function, which can be potentiated by diseases such as hypertension, diabetes and heart failure [Gottdiener et al. 2000]. Calculation of GFR is recommended for renally eliminated drugs, for example dabigatran or metformin, particularly in the context of acute illness when an acute deterioration in renal function may occur and medications may need to be adjusted or stopped accordingly in order to minimize the risk of an ADR.
Older patients are often more sensitive to the effects of medications than their younger counterparts because of altered pharmacodynamic responses. Common examples include excessive sedation and confusion with morphine, increased anticoagulant effect with warfarin, greater sensitivity to central nervous system effects of anticholinergic drugs and increased confusion with neuroleptics when used at ‘standard’ treatment doses. Such pharmacodynamic responses are generally predictable and can be minimized by starting at the lowest possible dose and titrating to response.
Polypharmacy and inappropriate prescribing
Older patients are the major consumers of medications in most developed nations [Wynne and Blagburn, 2010; National Center for Health Statistics, 2013]. They have a higher prevalence of chronic disease and comorbidity [Salive, 2013] often requiring more medication to multiple illnesses, thus increasing ADR risk through drug–drug and drug–disease interactions. Poly-pharmacy, commonly defined as the use of multiple drugs, or more than are clinically indicated, is strongly predictive of ADRs [Hajjar et al. 2003; Viktil et al. 2007]. Goldberg and colleagues reported that patients taking two concurrent medications have a 13% risk of an adverse drug interaction, rising to 38% for four medications and 82% for seven or more medications prescribed simultaneously [Goldberg et al. 1996]. The highest rate of polypharmacy occurs in nursing home residents [Maher et al. 2014], so it is not surprising that ADRs are more prevalent in this group [Hajjar et al. 2007].
Inappropriate prescribing (IP) (the use of medications that pose more risk than benefit when safer alternatives exist) is highly prevalent in older patients. Approximately 50% of older adults take one or more medications that are not medically necessary [Maher et al. 2014]. IP has been identified in 12–40% of nursing home residents [Willcox et al. 1994] and 14–23% of community-dwelling older people [Ennis and Reichard, 1997]. The association between IP and ADRs is well established, Lindley and colleagues showing that 50% of ADRs in older adults are due to inappropriate prescribing [Lindley et al. 1992]. A significant association has also been found between IP, ADEs, acute hospitalization, death [Klarin et al. 2005; Lau et al. 2005] and higher healthcare costs [Chiatti et al. 2012].
Explicit criteria aim to identify IP in older adults, the most frequently cited being Beers criteria [Beers et al. 1991; Beers 1997; Fick et al. 2003; AGS, 2012] and STOPP/START criteria (Screening Tool of Older Persons Prescriptions/Screening Tool to Alert doctors to the Right Treatment) [Gallagher and O’Mahony, 2008; O’Mahony et al. 2015]. Such criteria comprise lists of drugs that should be avoided or used with caution in older adults, either independent of clinical condition, considering clinical condition or considering coprescribed medications. Potentially inappropriate medications listed in STOPP criteria, unlike those listed in Beers criteria, are significantly associated with avoidable ADEs in older people that cause or contribute to urgent hospitalization [odds ratio 1.8; 95% confidence interval (CI) 1.5–2.3] when adjusted for age, sex, burden of comorbidity, dementia, baseline functional status and number of medications [Hamilton et al. 2011]. A recent randomized controlled study showed that application of STOPP/START criteria to patients aged 65 years and older within 72 h of hospital admission results in an 11% absolute risk reduction in in-hospital ADEs [O’Connor, 2013]. These criteria have the potential to reduce IP and related ADEs but more randomized controlled studies in various clinical settings are required. In addition, explicit criteria can be time consuming to apply in everyday clinical practice and need regular updating in line with emerging literature.
Sex
Women have a 1.5 to 1.7 fold increased risk of developing an ADR compared with men [Rademaker, 2001]. This can be attributed to gender differences in immunological and hormonal physiology which influence pharmacodynamic and pharmacokinetic response, particularly in relation to cardiac and psychotropic medications [Soldin et al. 2011].
Genetic predisposition
Genetic factors are thought to play a role in serious ADRs that have been traditionally classified as idiosyncratic, for example drug-induced liver injury, statin-induced myotoxicity and drug-induced long QT syndrome [Wilke et al. 2007]. Genotyping at an individual level has the potential to optimize drug therapy thereby reducing ADRs [Meyer 2000]. This technology can be used to predict ADR risk in patients needing phenytoin, a widely used antiepileptic agent with a narrow therapeutic index and large variability in patient response. Such variability is partly due to variation in expression of the gene encoding the cytochrome P450 2C9 enzyme that metabolizes phenytoin. Recently published guidelines about the interpretation of genotyping are useful for drug dosing and could potentially reduce the occurrence of severe ADRs, such as Stevens Johnson syndrome [Caudle et al. 2014].
ADR prediction tools
A recent systematic review identified and assessed the quality of validated ADR risk-prediction tools for use in adults over 65 years of age [Stevenson et al. 2014]. Four tools were identified from the literature; three related to ADRs [Onder et al. 2010; Tangiisuran 2009; McElnay et al. 1997] and one related to ADEs [Trivalle et al. 2011]. The three ADR prediction tools included dedicated development and validation phases but none addressed clinical impact or implementation, thereby limiting universal applicability. Stevenson and colleagues concluded that all of the ADR prediction tools had weaknesses with regard to definition and handling of predictor variables, and that all of the ADR risk prediction models had poor to modest performance [Stevenson et al. 2014].
The GerontoNet ADR risk prediction tool was developed on the basis of a large Italian database in which 383 of 5936 patients had an ADR (6.5%) [Onder et al. 2010]. The tool incorporates the following predictor variables: at least four comorbid conditions, heart failure, liver disease, number of drugs, history of ADR and presence of renal failure. When applied prospectively in a validation study of 483 patients, it correctly predicted ADRs in 71% of cases (95% CI 0.68–0.73) [Onder et al. 2010]. In a separate study, O’Connor and colleagues prospectively applied the GerontoNet risk score in an Irish hospital setting but found it incorrectly classified 38% of patients as being at low risk of developing an ADR [O’Connor et al. 2012]. ADR prediction models should ideally be tested on representative samples of a target population to determine clinical impact before widespread implementation.
A large number of variables contribute to ADR occurrence in older patients, thus rendering it difficult to develop a robust ADR prediction tool that is applicable to a heterogeneous population, that is a single tool is unlikely to correctly predict every ADR in every older patient. Therefore, ADR risk prediction strategies should focus either on commonly encountered harmful ADRs in all older patients (e.g. cognitive deterioration, orthostatic hypotension, falls, gastrointestinal or intracranial bleeding) or ADRs in older patients with a particular illness or clinical characteristic (e.g. renal impairment or dementia). Sharif-Askari and colleagues devised a tool to predict ADRs in hospitalized patients with chronic kidney disease (CKD) [Sharif-Askari et al. 2014]. This prospective, observational study evaluated patients with CKD stages 3–5, admitted to a renal unit. Baseline data were collected on all patients, the outcome being an ADR. A favourable C statistic of 0.84 was reported, that is ADRs were correctly predicted 84% of the time [Sharif-Askari et al. 2014]. Similar ADR risk prediction tools could be developed for patients with conditions such as dementia, delirium, orthostatic hypotension and falls.
Other strategies to minimize ADR risk in older adults
MR is a process of obtaining a patient’s medication list and accurately clarifying what the patient is taking, including over-the-counter (OTC) and herbal medicinal products. The Structured History-taking of Medication (SHiM) tool can be used for this purpose [Drenth-van Maanen et al. 2011]. It can identify discrepancies in all aspects of medication use in older patients e.g. differences between prescribed dose and dispensed dose, inappropriate formulations, intolerances, poor adherence and use of clinically relevant nonprescribed drugs, all of which can contribute to ADRs [Prins et al. 2013]. Medication review of hospitalized patients by a pharmacist or doctor can reduce future emergency contacts in the short term [Christensen and Lundh, 2013]. It can also identify and resolve unintentional discrepancies at care transition points, for example hospital discharge to nursing home [Kwan et al. 2013]. MR is resource intensive but its greatest benefit is likely to be in those at greatest risk of ADR, that is older patients on multiple medications.
Comprehensive Geriatric Assessment (CGA) uses a multidisciplinary approach to determine medical, psychological and functional capabilities of a frail older person. It aims to develop an individualized plan for a patient’s treatment and follow up. CGA can improve the quality of prescribing in older adults, Saltvedt and colleagues reporting a lower prevalence of drug–drug interactions (28% in patients with CGA intervention
Pharmacovigilance pertains to the detection, assessment, understanding and prevention of adverse effects or any other drug-related problem [Morin et al. 2015] with the aim of enhancing medication safety and patient care. E-pharmacovigilance (where a computer links a patient’s medications with their laboratory results) can inform prescribers of potential ADRs in the setting of abnormal laboratory values [Neubert et al. 2013]. This represents an important step towards a systematic drug safety process but requires clinician engagement, intensive resources and regular updates. Electronic prescribing and computerized alerts can provide guidance to a prescriber about dosage errors, transcription errors, potential drug interactions and drug monitoring. A recent systematic review concluded that electronic prescribing can reduce medical errors and ADEs, though there was wide variation in study design, quality and results [Ammenwerth et al. 2008].
A practical approach to assessing and preventing ADRs in everyday clinical practice
Diagnosing and managing ADRs in older patients requires physician awareness of physiology and pharmacology of aging. Physicians should have a high index of suspicion for ADRs in all older patients, particularly those who develop new symptoms after starting or dose adjusting a medication, or in those presenting with cognitive or functional decline. Similarly, ADRs should be considered as a cause of new symptoms in older patients with renal impairment, falls, orthostatic hypotension, heart failure, delirium, polypharmacy and previous history of ADR [Chan et al. 2001]. Sufficient time should be allotted for clinical assessment, detailed medication review (including prescribed and over-the-counter drugs) and supporting collateral history where necessary. Medication lists should be assessed in the context of a patient’s list of comorbid illnesses. Therapeutic duplication should be avoided, unnecessary medications discontinued and dosing frequency of remaining drugs should be optimized. Consideration should be given to medications that were prescribed at a younger age that may not have been adjusted for age-related pharmacodynamic sensitivity or for changes in renal or hepatic function.
Clear therapeutic goals should be established when starting a new medication and a date for review of efficacy and potential adverse reactions should be set. Medications which have not resulted in therapeutic benefit should not be used indefinitely, for example cognitive enhancing drugs for dementia. The use of antipsychotics, antihistamines, benzodiazepines, anticholinergic and hypnotic drugs should be limited to reduce the risk of falls. Drugs with a narrow therapeutic index should be avoided when possible. Prescribing appropriateness criteria such as STOPP/START and Beers criteria can be useful when selecting and reviewing medications in the context of a patient with multimorbidity, though these are not intended to replace clinical judgement.
In general, drugs should be started at the lowest possible dose and slowly uptitrated according to response. Only one new medication should be commenced at any one time. Clear concise information should be given to older patients and their carers about new medications, dose alterations, monitoring requirements and potential ADR-related symptoms, including the time frame in which they are most likely to occur. Stringent application of generic practice guidelines may not be appropriate for frail older patients with limited life expectancy. Indeed, quality of life outcomes and patient preference should take precedence over routine implementation of such guidelines. Similarly, clinicians should avoid treating every symptom with a medication, particularly because the symptom in question may be an adverse effect of another coprescribed medication. In some cases, it is appropriate to avoid prescribing new medications altogether, particularly when the intended clinical outcome is unlikely to be realized.
Conclusion
ADRs are a common clinical problem in older adults and contribute significantly to morbidity and mortality. They are often unrecognized in complex older patients because of competing contributory diagnoses and poor awareness amongst treating physicians. Furthermore, the terminology of ADRs can be confusing and existing causality and severity criteria can be prohibitively difficult to apply in everyday practice. A number of ADR risk prediction tools have been published, but none are universally accepted and none are used routinely in clinical practice. Robust ADR risk prediction tools should be aspired to. However, given the heterogeneity of the older population, research efforts should focus on either predictor variables for specific patient groups (e.g. those at risk of falls or those with cognitive impairment) or predictor variables for specific ADRs, for example bleeding, hypotension or hypoglycaemia.
Detecting and predicting ADRs in older patients is based on monitoring and regular review of prescribed and over-the-counter medicines. ADRs are a common cause of cognitive or functional decline, falls, gastrointestinal bleeding, heart failure and orthostatic hypotension. Clinicians should consider potential ADRs as part of every differential diagnosis. New medicines should be prescribed with a clear therapeutic goal. Medicines which are ineffective or no longer indicated should be deprescribed. Standardized tools, for example ‘appropriateness criteria’ or ‘risk prediction tools’ are useful adjuncts but do not replace sound clinical judgement underpinned by training in geriatric pharmacotherapy.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflict of interest in preparing this article.

