Abstract
Background:
The aim of this study was to evaluate efficacy and safety of poly-de-prescribing (PDP) based on the Garfinkel method in older people with polypharmacy.
Methods:
A longitudinal, prospective, nonrandomized study in Israel was carried out between 2009 and 2016. Comprehensive geriatric assessments were performed at home in people age ⩾66 years consuming ⩾6 prescription drugs. Exclusion criteria were life expectancy <6 months and a seeming unwillingness to cooperate (poor compliance). PDP of ⩾3 prescription drugs was recommended. Follow up was at ⩾3 years. Between April 2015 and April 2016 Likert scale questionnaires were filled by all participants/families to evaluate overall satisfaction and clinical outcomes. The outcome measures were change in functional, mental and cognitive status, sleep quality, appetite, continence; major complication, hospitalizations, mortality, and family doctor’s cooperation.
Results:
Poly-de-prescribing of ⩾3 drugs was eventually achieved by 122 participants (PDP group); ⩽2 drugs stopped by 55 ‘nonresponders’ (NR group). The average age was 83.4 ± 5.3 in the PDP group, and 80.8 ± 6.3 in the NR group (
Conclusions:
This self-selected sample longitudinal research strongly suggests that the negative, usually invisible effects of polypharmacy are reversible. PDP is well tolerated and associated with improved clinical outcomes, in comparison with outcomes of older people who adhere to all clinical guidelines and take all medications conventionally. Future double-blind studies will probably prove beneficial economic outcomes as well.
Keywords
Introduction
The huge increase in lifespan is accompanied by unprecedented growth of subpopulations of Very Old, those with multiple COmorbidities, Dementia, Frailty (and disability) and Limited life- EXpectancy; the author has suggested to address these ‘at risk’ subpopulations using the acronym VOCODFLEX.1,2 Multiple morbidities lead to an increased number of drugs prescribed (polypharmacy) thus increasing the risk of adverse drug events (ADEs). De-prescribing is the process of intentionally stopping a medication or reducing its dose to improve the person’s health or reduce the risk of ADEs.
A major predictor for inappropriate medication use is polypharmacy3–7; therefore, both iatrogenic problems are addressed here together as ‘the epidemic’. Major causes of this epidemic are: (1) the increased number of doctors/specialists and clinical guidelines; (2) the lack of evidence-based medicine (EBM) and knowledge regarding drug–disease–patient interactions in polymedicated VOCODFLEX; (3) barriers/fears of medical doctors to de-prescribe.
This research is based on several prespecified hypotheses: in most older people particularly VOCODFLEX, the sum total of negative effects of ‘the epidemic’ outweighs the sum of all beneficial effects of all specific medications; the best way of treating polypharmacy is poly-de-prescribing (PDP), defined by the author as simultaneous discontinuation of as many nonlife-saving drugs as possible; as most medications are not ‘life-saving’, a monitored therapeutic trial of PDP is well tolerated; patient/family approval, and doctor’s cooperation, are crucial for successful PDP.
The purpose of this research is to examine an approach to alleviating ‘the epidemic’ and achieving better quality of life (QoL) for older people. The results shown are the follow up to two studies; PDP was associated with a significant reduction in both mortality and referrals to hospitals, in nursing home patients, 8 and with improved clinical outcomes and QoL in community-dwelling older people. 9 The present research evaluates the long-term efficacy and safety of the Garfinkel method of PDP, as judged by a variety of clinical outcomes, hospitalizations and mortality rates.
Method
Study setting and design
This is a longitudinal, prospective, nonrandomized study of PDP in older people. Since 2009, several hundred older patients have been referred for a second opinion to the author either by their family doctor/general practitioner (GP), the family or as a self-referral. Comprehensive geriatric assessments were performed at the patient’s home and PDP recommended. Only elderly patients who consumed at least six medications (not including vitamins, minerals, food additives, over-the-counter and topical preparations) were included. Exclusion criteria were age younger than 66; life expectancy shorter than 6 months and communication problems of patient/family expected to interfere with follow up.
Main principles of the Garfinkel method
In older people, each drug requires rethinking and re-evaluation; adhering to all clinical guidelines to achieve ‘traditional target numbers’ [e.g. blood pressure (BP), serum glucose, lipid concentrations] is not the main goal, but rather to achieve the best QoL possible as perceived by the patient/family; and discuss with the patient/family the known EBM pros and cons for each drug and achieve their consent to PDP (stopping as many drugs as possible, simultaneously). When several drugs are prescribed for the same indication, PDP is staggered, stopping one drug at a time with a detailed plan. For example, discontinue one anti-hypertensive compound per week until the optimal BP of 150/90 is achieved.10–14 Clinical judgement and an individually tailored strategy based on patient disease characteristics determine which drug to stop first, and when needed, prescribe drugs for new diagnoses or symptoms. A detailed consultation with references is sent to the family doctor/GP, with recommendations for drug and nondrug therapy. Availability for further ‘over the phone consultations’ is crucial.
Baseline data
The initial baseline visit included a detailed list of diagnoses, prescription and nonprescription medications consumed (topical preparations and drugs taken occasionally were not included). Functional status was determined using a 5-point scale: 1 = independent, 2 = frail (as defined by Fried and colleagues 15 ); 3 = mild disability [needs help in 1–2 activities of daily living (ADLs)]; 4 = disability (needs help with at least 3 ADLs); 5 = severe disability/bed ridden. Cognitive status was assessed using the Folstein mini-mental test (MMSE). Depression was assessed using the short Geriatric Depression Scale (GDS). In patients with severe dementia GDS was not performed.
Intervention
The intervention used, concurrent de-prescribing of multiple medications, is based on the Garfinkel algorithm (Figure 1) and the already mentioned principles of the method; it combines ethics, EBM and clinical judgement while giving the highest priority to patient/family preferences.
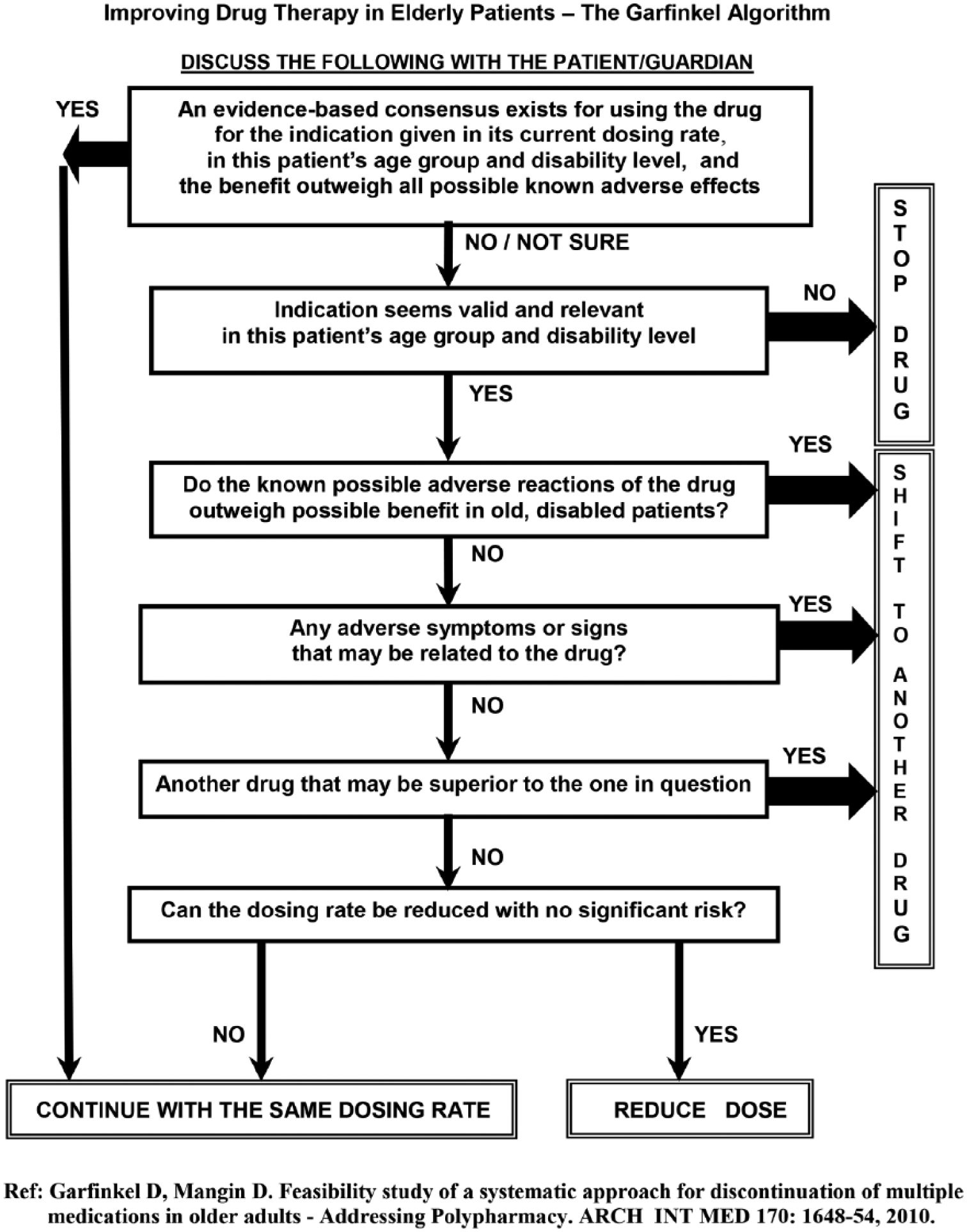
The Garfinkel algorithm.
Outcomes
The researcher (author) was available by phone to all participants. Furthermore, all patients/families were contacted by phone at least once a year and their comments recorded. Apart from this routine follow up, between April 2015 and April 2016, all participants who had been followed for at least 3 years were interviewed by phone by the author and asked to respond to a questionnaire that had been sent to them in advance (Appendix 1). This last follow up was considered the study endpoint. Participants were required to assess the change between baseline and last follow-up visits regarding several clinical outcomes using a 5-point Likert scale (1 = much improved, 2 = improved, 3 = no change, 4 = worsened, 5 = much worse). The parameters for evaluation were overall satisfaction, functional, mental and cognitive status, night sleep quality, daytime sleepiness, appetite, and incontinence. The scores given were subjective, based on the patient’s perception, or primary caregiver’s impression when patients had severe dementia or disability, or chose not to fill it themselves. Patients/families were asked to report on all types of medications taken, new diagnoses, complications and hospitalizations since the baseline visit; all were confirmed by medical documents. Major complications were classified as: significant vascular events (acute coronary event, cerebral stroke, pulmonary emboli, major arterial occlusion, deep vein thrombosis), new significant arrhythmia, pulmonary edema, major bleeding, fractures, severe infection (e.g. pneumonia, sepsis). Other factors assessed were the GP response to the recommended PDP, the time between de-prescribing and improvement and the length of time this improvement persisted. Mortality was assessed, the date of death and QoL in the last period before death were recorded by the caregiver/family.
Group affiliation was determined by the absolute number of drugs consumed at the last follow-up visit: the PDP group in whom ⩾3 prescription drugs had been discontinued and the nonresponder group (NR) in whom either ⩽2 drugs were stopped, or more drugs were prescribed. When ⩾3 drugs had been discontinued but new drugs added so that eventually, the total number was not reduced by 3 or more, the participant was classified among the NR group. Vitamins, minerals, food additives, over-the-counter or topical preparations, even when discontinued, were not counted in among de-prescribed drugs.
The follow up including the questionnaires were performed by the researcher (author) himself in all participants.
Ethics
The study protocol was approved several times since 2009 by the Ethics Committee of the Shoham Geriatric Medical Center, Pardes-Hana, and lastly by the Ethics Committee of Wolfson Medical Center, Holon, Israel.
Statistical methods
Descriptive statistics are given as mean ± standard deviation (SD) or medians with interquartile range (IQR) for continuous variables and frequency distribution for categorical variables. Comparison between the two study groups with regard to demographic and baseline clinical factors were performed using Student’s
Survival was calculated from the time of baseline visit to time of death or, for those still living, to the date of last follow up, but not later than 3 years. Survival estimates were calculated according to Kaplan–Meier method and are demonstrated by the Kaplan–Meier curves. The follow up was limited to 3 years because the number of patients followed for longer period was small. These curves were compared by the log-rank test. All statistical analyses were performed using SAS® 9.4 Companion for UNIX Environments, Sixth Edition. All
Results
Between April 2009 and January 2013, comprehensive geriatric assessment was performed by the author in 248 patients. A total of 55 patients were excluded: 2 were younger than 66 years, 17 consumed ⩽6 medications, 24 had an estimated life expectancy shorter than 6 months (e.g. metastatic cancer) and 12 because of communication problems. PDP was recommended for all other 193 patients. Following data analysis, 16 were lost to follow up or declined to participate in follow up. Eventually, 122 patients were included in the PDP group and 55 in the NR group. Most questionnaires were filled in by primary caregivers (spouse/offspring), 80% in the NR group, and 85% in PDP group; the others were filled by the patients themselves.
The mean age was 83.4 ± 5.3 years in the PDP group and 80.8 ± 6.3 years in the NR group (
Table 1 presents the list of chronic diseases, geriatric syndromes and common symptoms based on the medical records. The prevalence of most diseases was comparable in the PDP and NR groups. Diabetes mellitus was more prevalent in the NR group (
Prevalence of chronic diseases, geriatric syndromes and common symptoms a in NR and PDP groups.
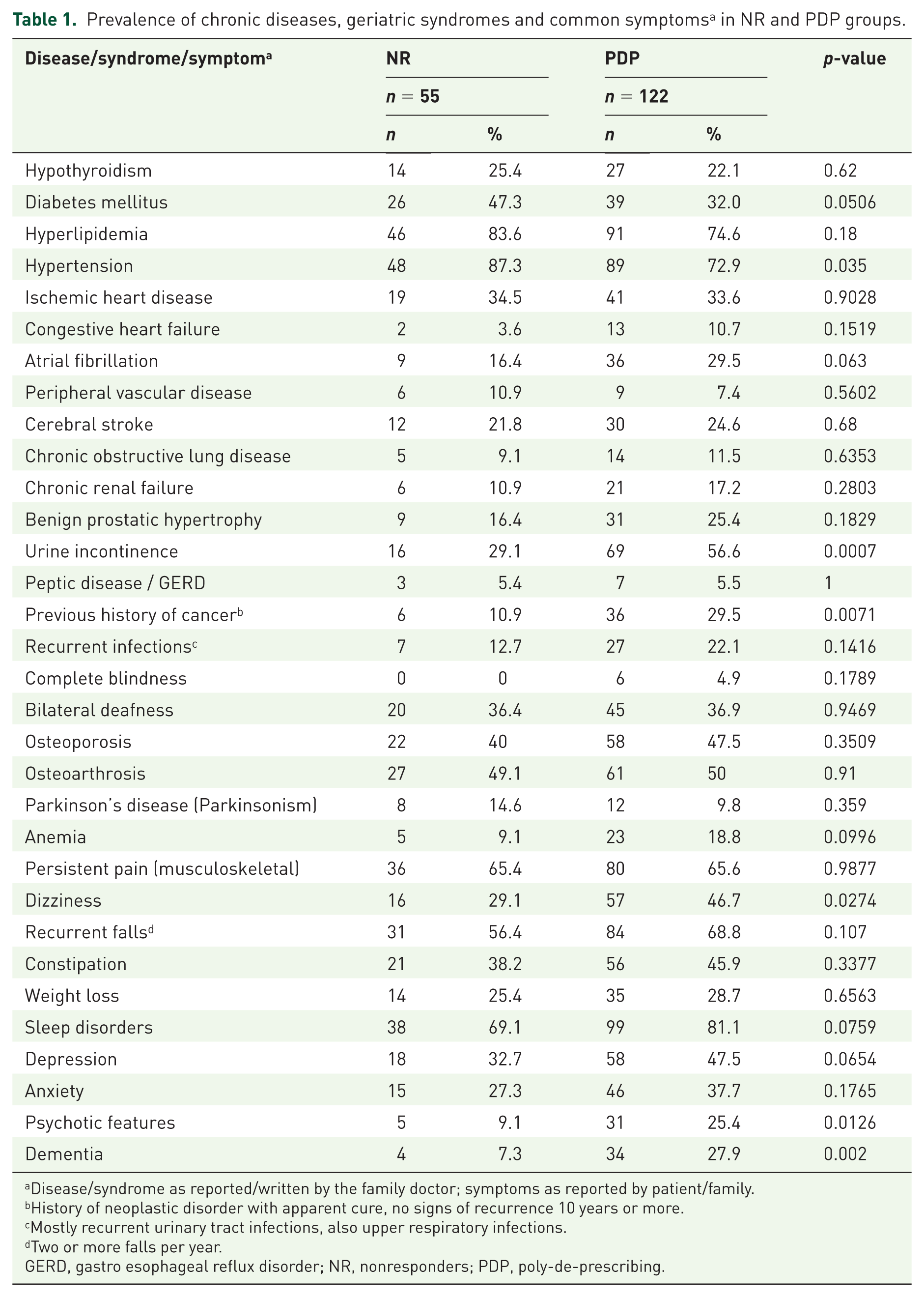
Disease/syndrome as reported/written by the family doctor; symptoms as reported by patient/family.
History of neoplastic disorder with apparent cure, no signs of recurrence 10 years or more.
Mostly recurrent urinary tract infections, also upper respiratory infections.
Two or more falls per year.
GERD, gastro esophageal reflux disorder; NR, nonresponders; PDP, poly-de-prescribing.
The main barrier to de-prescribing was the GP. In 48 out of 55 (87%) NRs, the GP (sometimes specialists) refused to discontinue medications. In the 122 patients of the PDP group, GPs adopted the PDP plan completely, mostly or partially in 86, 20 and 5 patients, respectively (91%). In six cases (3.3%) the patient/family decided to adopt the PDP recommendations despite their doctor’s refusal to comply. A univariate comparison with regard to all background variables mentioned above showed that the PDP participants were significantly older (83.4 ± 5.3 in PDP
Table 2 represents the most prevalent drugs taken by all 177 participants, the number of those recommended to de-prescribe and the actual number that was eventually discontinued. The last columns represent drugs that were de-prescribed by others and those given as ‘new medications’ following baseline visit, by the author or other doctors. The main ‘new drugs’ prescribed by the author were antidepressants particularly mirtazapine and duloxetine, when depression was diagnosed. In all participants, the most prescribed drugs were statins, aspirin, benzodiazepines and proton pump inhibitors.
De-prescribing rate of different drugs/drug groups.
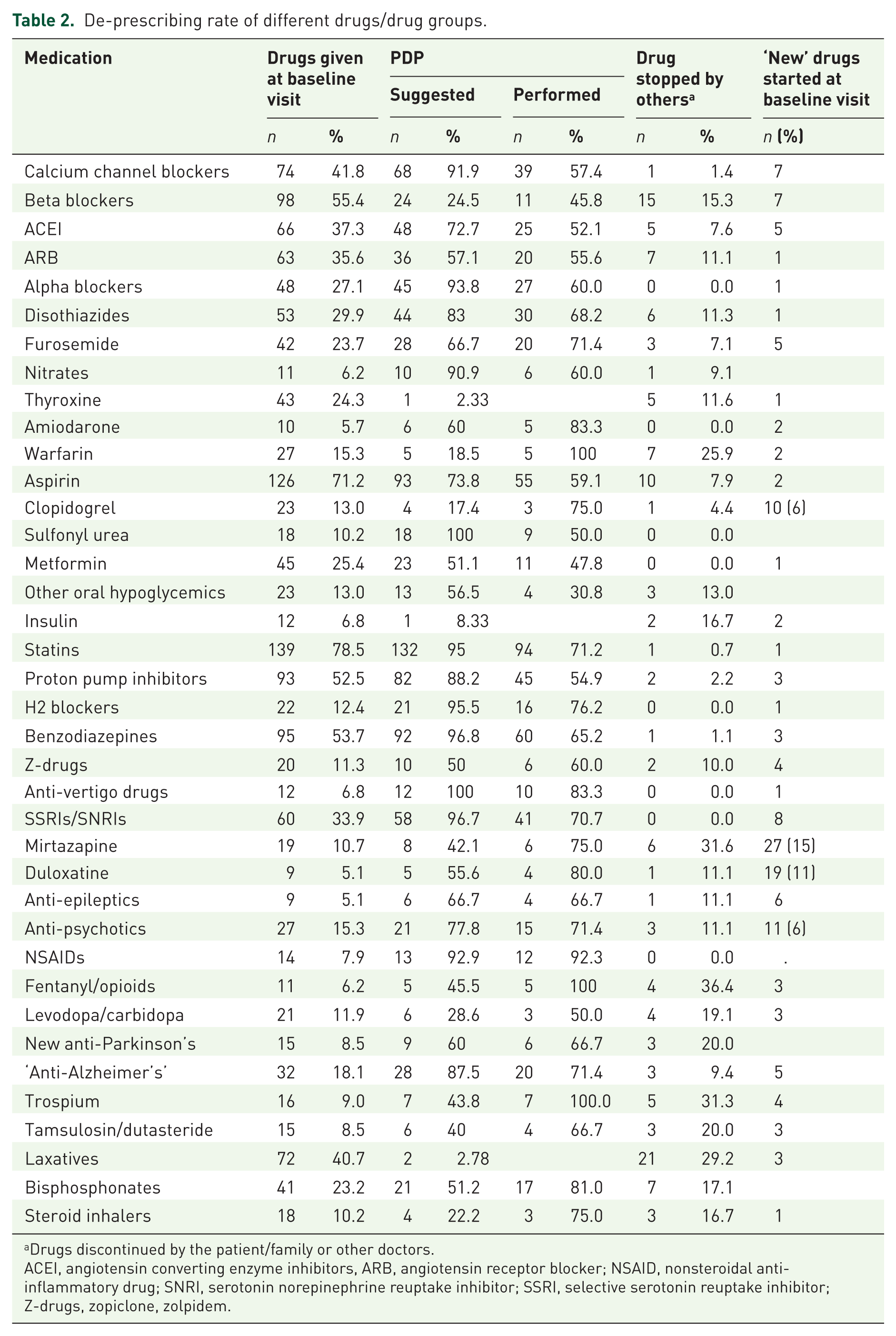
Drugs discontinued by the patient/family or other doctors.
ACEI, angiotensin converting enzyme inhibitors, ARB, angiotensin receptor blocker; NSAID, nonsteroidal anti-inflammatory drug; SNRI, serotonin norepinephrine reuptake inhibitor; SSRI, selective serotonin reuptake inhibitor; Z-drugs, zopiclone, zolpidem.
For these same drugs, de-prescribing was recommended in 95%, 73.8%, 96.8% and 88.2%, respectively; successful discontinuation was achieved in 71.2%, 59.1%, 65.2% and 54.9%, respectively. Anti-hypertensive drugs (AHDs) were also highly recommended for discontinuation; in some patients gradual de-prescribing of several AHDs was achieved while keeping average BP below 150/90. Less common drugs taken by less than 7% of participants do not appear in Table 2 but are included in the total number of drugs consumed (e.g. anti-allergic, antispasmodic, anti-arrhythmic, erythropoietin, nitrofurantoin).
Table 3 compares the changes in clinical outcomes between the two groups. The median number of medications taken in the baseline visit was 10 (IQR 8–12) in both the PDP and NR groups (
Comparative change in the main clinical outcomes and quality of life parameters a between the NR and the PDP groups.
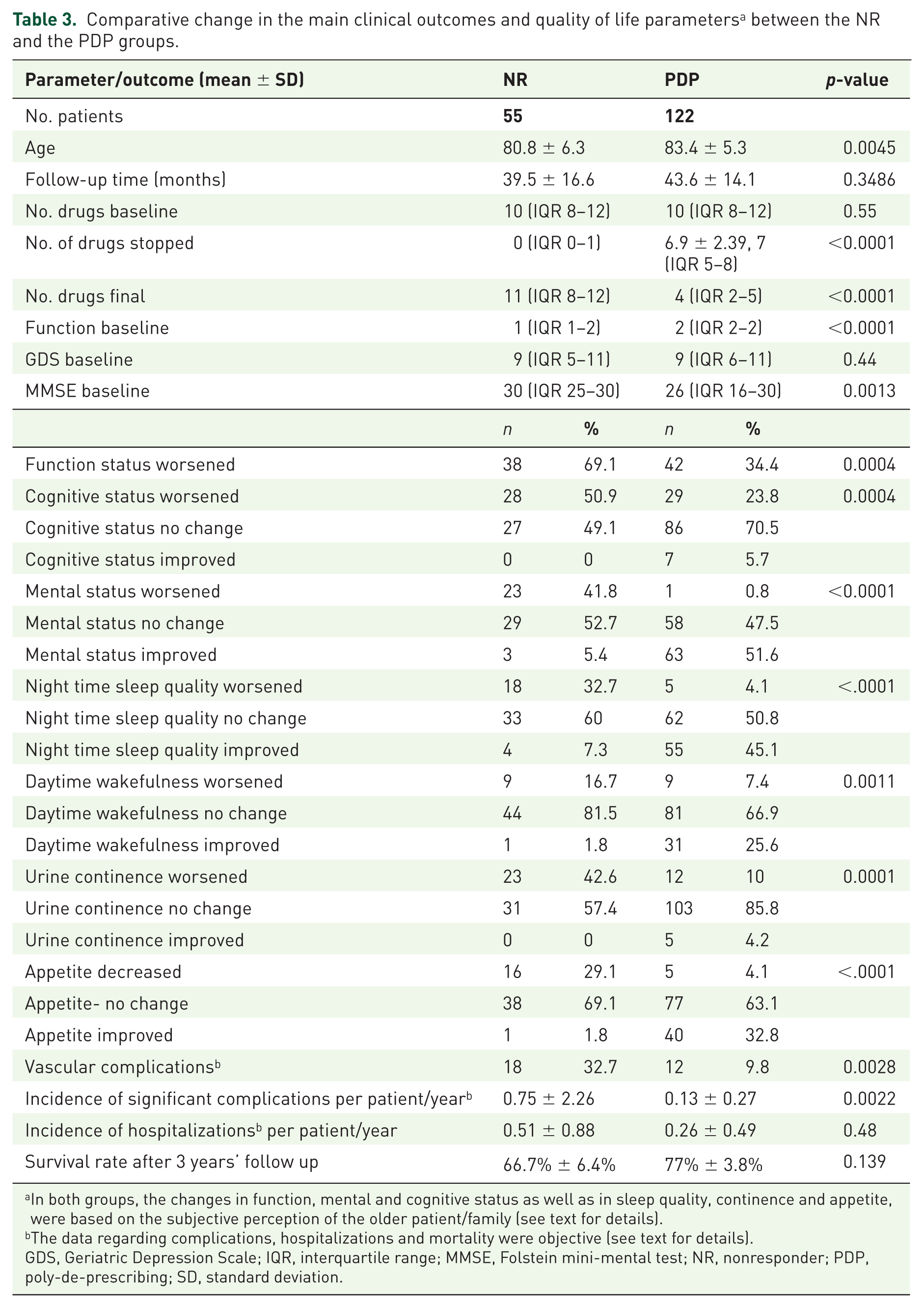
In both groups, the changes in function, mental and cognitive status as well as in sleep quality, continence and appetite, were based on the subjective perception of the older patient/family (see text for details).
The data regarding complications, hospitalizations and mortality were objective (see text for details).
GDS, Geriatric Depression Scale; IQR, interquartile range; MMSE, Folstein mini-mental test; NR, nonresponder; PDP, poly-de-prescribing; SD, standard deviation.
Evaluating all interacting/combined influences of baseline characteristics (e.g. mental, functional, and cognitive status), on the extent of success of PDP regarding specific clinical outcomes, is beyond the scope of this manuscript. PDP patients with relatively preserved cognition (MMSE > 20) had 85.6-times [± standard error (SE) 1.85] greater chance of having improved mental status as compared with NR patients with comparable cognitive status (
Figure 2(a) shows the Kaplan–Meier survival curves for the entire cohort stratified by study group. Although the long rank test was not significant, there is a trend of better survival for the PDP group. Figure 2(b) represent only patients who died in both groups. The Kaplan–Meier survival curves reveals survival advantage to the PDP intervention group over NR group (
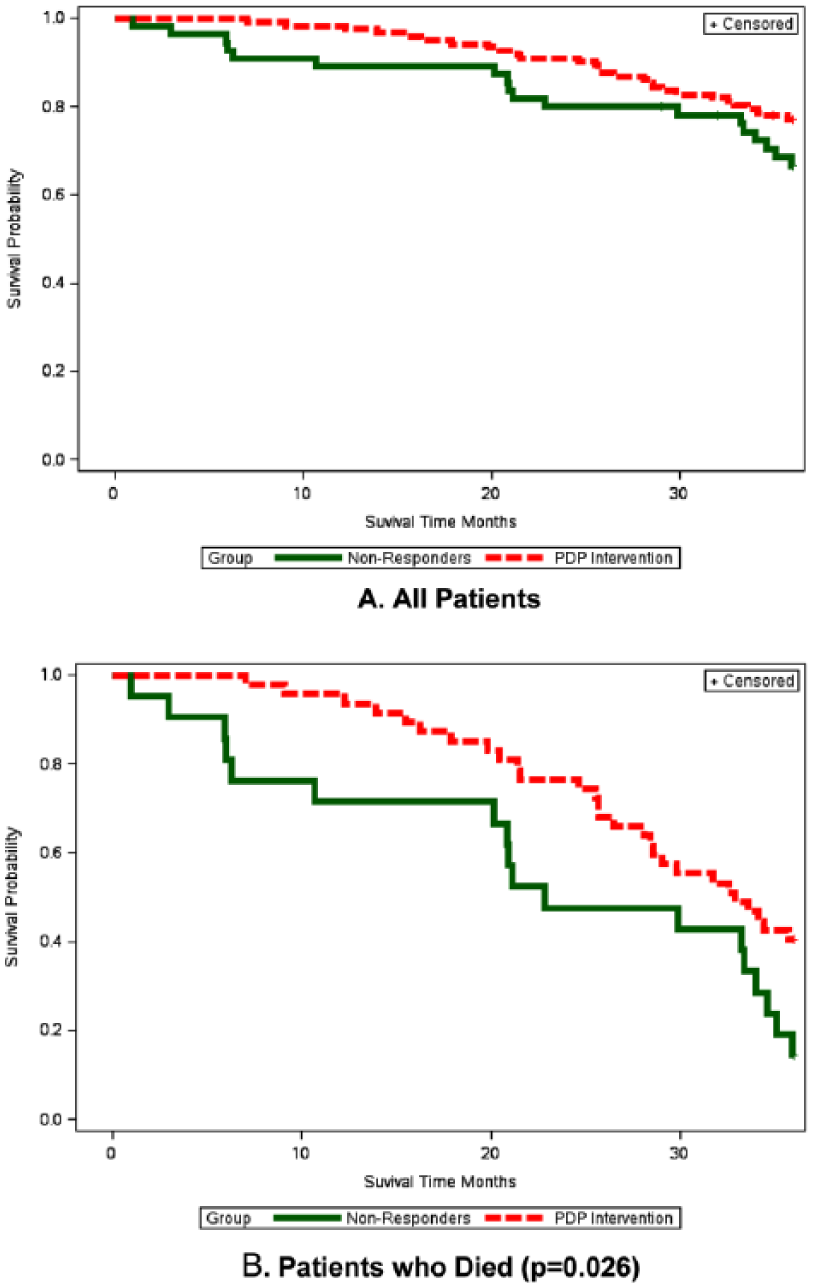
Survival time at 36 months in PDP and NR groups.
A comparison of clinical outcomes and QoL was performed in all patients who died (Table 4). In their last period of life, PDP and NR groups had a comparable rate of hospitalizations (RR = 0.63, 95% CI: 0.36–1.09) and decline in functional (OR = 3.55, 95% CI: 0.82–15.29) and cognitive decline (OR = 2.41 95% CI: 0.72–8.09). However, PDP was associated with significantly less deterioration (sometimes improvement) in mental status (OR = 60.52, 95% CI: 6.58–556.44), sleep quality (OR = 5.26, 95% CI 1.28–21.52), appetite (OR = 11.75, 95% CI: 2.37–58.25), and sphincter control (OR = 9.39, 95% CI: 1.77–49.82), and lower incidence of major complications (RR = 0.35, 95% CI: 0.17–0.73) 95% of PDP participants who eventually died experienced at least 1 year, 61.5% experienced ⩾2 years defined as ‘improvement’ or good QoL, before death. Most PDP patients who died have not experienced much suffering shortly before death (i.e. sudden death, very short hospitalization before death).
Comparison of the change in the main clinical outcomes and quality of life of NR
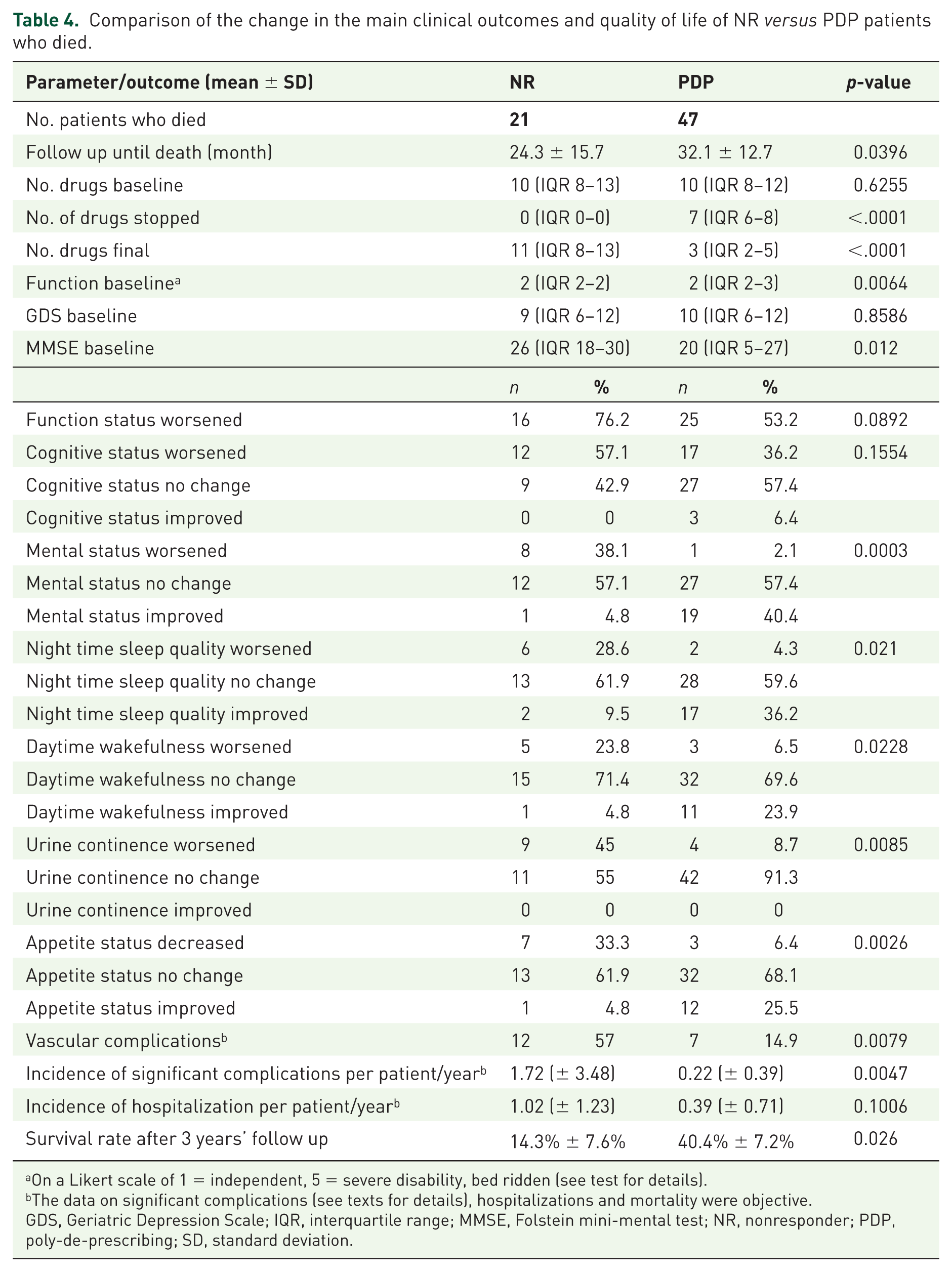
On a Likert scale of 1 = independent, 5 = severe disability, bed ridden (see test for details).
The data on significant complications (see texts for details), hospitalizations and mortality were objective.
GDS, Geriatric Depression Scale; IQR, interquartile range; MMSE, Folstein mini-mental test; NR, nonresponder; PDP, poly-de-prescribing; SD, standard deviation.
Discussion
Addressing the problem
The dilemma addressed here is whether, while adhering to all ‘traditional’/ existing clinical guidelines, we are not paradoxically though unintentionally, achieving worse QoL in older people. The Bible (Genesis) contains the first mention of a medical intervention where God took a rib out of Adam and created Eve (Genesis II, 21–22). Having not mentioned side effects, this invasive procedure may be defined as the best ever intervention with an infinite benefit–risk ratio. The second intervention recommended by the first medical consultant (the serpent) relates to ‘Fruit of the tree of knowledge’, an apparently innocent ‘food additive’ that in fact, got us all evicted from the Garden of Eden while ruining any chance for perfect health. Going fast forward, we must admit modern medicine has not reached the stage of divine intervention. Furthermore, all too often specialists who treat patients ‘by their book’ (the serpent way) have but one aim, to deal with their one aspect of the disease spectrum; no in depth consideration of the ultimate effects of medications they prescribe combined with other consultant’s interventions on patients’ overall welfare.
Extensive discussions of all the negative consequences of ‘the epidemic’ of inappropriate medication use and polypharmacy,1,16–20 as well as the many barriers to de-prescribing are beyond the scope of this manuscript.1,2,21–29 Analyzing the main predictors of ‘the epidemic’, the higher the number of diseases the larger the number of specialists that, together with GPs recommend medications based on EBM guidelines supported by randomized controlled trials (RCTs) in their field of expertise. 30 However, most RCTs are based on ‘single disease models’ in younger, healthier populations. Older people certainly VOCODFLEX are excluded from RCTs, and the few trials in older people are nonrepresentative of the general old population.31–34 Therefore, as Boyd and colleagues conclude:”…in elders with several comorbidities…basing standards for quality of care…on existing guidelines could lead to inappropriate judgement of the care provided, create perverse incentives and diminish the quality of their care”. 35 Applying all guidelines in older people with their very different risk–benefit prospects from those in whom testing was conducted, makes ‘the epidemic’ inevitable.36–39 For most medications, the positive benefit–risk ratio becomes nonexistent or reversed, in correlation to the extent of the VOCODFLEX characteristics; the severity of ‘the epidemic’ increases in correlation to exactly the same characteristics and the extent of polypharmacy. The problem is further aggravated due to ‘prescription cascades’ where symptoms resulting from ADEs are perceived as representing ‘new diseases’.40,41 This perpetuates the iatrogenic vicious cycle of over-diagnosis, useless evaluations, hospitalizations, overtreatment and ‘the epidemic’. 42 Possible contributors are for-profit diagnostics and drug companies.43–48 In one study considerable interaction was also found between authors of clinical guidelines and the pharmaceutical industry. 49 Other contributing factors are ‘defensive medicine’ attitudes of automatically ‘following orders’, implementing whatever guidelines we do have in all older people, until death. Many older people do not have one, knowledgeable, reliable ‘all drugs’ case manager’ and a scheduled drug re-evaluation is rarely performed.
There is no global consensus as for the best way to combat ‘the epidemic’. No computer simulation can show all possible interactions among multi-medications, while factoring in the particular characteristics of a specific patient. Structured medication management represent the main approaches suggested. Explicit tools such as Beer’s lists, START/STOPP criteria and many other country specific criteria, usually represent lists of ‘forbidden drugs’.50–56 However, in multidrug situations we are never sure which drugs are causing which reactions, therefore drugs-to-avoid criteria are insufficiently accurate to use as measures of prescribing quality. 57 Stopping ‘inappropriate’ medications may be dangerous and even unethical, because we have no idea of the whole scope of damage caused by interactions between the remaining, apparently ‘appropriate’ drugs.2,9 The author advocates a different, implicit approach that takes into consideration the best evidence-based research data,58–62 but also incorporates individual patient/family particular characteristics and preferences to judge drug appropriateness.
A patient may benefit from stopping only one drug particularly if it is the main contributor to ADEs or interactions. However, as the extent of polypharmacy correlates with the severity of ‘the epidemic’,3–7 the main objective of this study was to prove the beneficial effects of de-prescribing of many drugs, not one or two. Therefore, the PDP group was defined as patients who stopped at least three medications. Similarly, the NR group included not only patients who consumed the same number of drugs or more, but also those who stopped only 1–2 drugs.
Strengths of the study
To the author’s knowledge, this is the first longitudinal study to follow up patients for more than 3 years, comparing clinical outcomes and QoL of older people who have undergone massive drug reduction (PDP group), with those of comparable patients who continued taking medications conventionally.
Beneficial effects of preventive medications may be mitigated by age itself and this research proves the author’s main hypotheses 63 : the cumulative risks of polypharmacy outweigh the potential benefits gained from all specific drugs. Not only is ‘the epidemic’ harmful but its effects are reversible; rational PDP is feasible, efficacious, and well tolerated; it was not associated with increased rate of hospitalizations or mortality, and the prevalence of serious complications was actually decreased. While shattering common myths that underlie patient’s and doctor’s fears of routine de-prescribing, this research is a breakthrough in providing clear answers to overcome these barriers. Another encouraging result is the finding that improvement usually appears quickly, within 3 months following PDP. The improvement due to PDP is sometimes so impressive, that patients/families themselves wish to extend the ‘therapeutic window’ of PDP. This highlights another beneficial medico-legal advantage of the Garfinkel method; ensuring patient/family involvement in decision making reduces doctors’ fear of lawsuits, of specialists and of health systems’ pressures to ‘implement all guidelines in everyone’.
The author entitled the method ‘Good Palliative Geriatric Practice’ because PDP fits well with the palliative approach. The findings presented in Table 4 represents the QoL of participants who died; in the last period of life, the QoL in the PDP group was comparable or better than that of older people who continued taking drugs conventionally. Palliative care at its best combined with drug load reduction of about seven drugs/patient. In other words, although the purpose of this method is aimed at achieving improved health and QoL in older people, it may also represent a potentially important tool for reducing healthcare expenditures in the last years of life.64–66
Study limitations
This study was not a RCT; it represents the experience of one geriatrician (the author) in self-selected aged patients who looked for a second opinion being unsatisfied with their health situation. The fact that the author performed the geriatric assessments, follow up and interviews by phone in all patients may be a potential bias. Furthermore, as the author is the only clinician in this study, it is difficult to distinguish between the benefit of the algorithm and the impact of the author, which is relevant to others who may wish to replicate this work. Also, the method of this study is dependent on the clinician skill as well as the algorithm and is therefore more difficult to define (again for replication). The cohort of patients, who chose to consult the author, were people already unsatisfied by their current treatment. These were therefore people who subjectively identified that they had a problem. This cohort therefore represents a self-selected target group which may have influenced the impact of the PDP. The algorithm used for PDP gives general recommendations leaving the final choice to the GP and patient/family. Therefore, the results may not be simply extrapolated to the whole elderly population.
However, the study definitely demonstrates that PDP in older people with polypharmacy may be highly beneficial at least for periods of 2–3 years. The concept ‘natural history of disease’ is traditionally used to describe the expected course/outcome of specific diseases, if left untreated. However, nowadays most adults/elderly are ‘treated with something’ as recommended by the many guidelines; the ‘natural history of multimorbidity’ reflects not only outcomes of diseases, but also the impact of medical interventions including all iatrogenic effects (the serpent’s impact). Paradoxically, nowadays the vast majority of the older population are patients in whom many drug interventions have been performed thus representing ‘the norm’. Therefore, though not a ‘pure’, randomized control group, older people who did not comply with PDP recommendations (NRs) represent most of the older population with comorbidity who continue taking medications conventionally. It seems reasonable to compare their clinical outcomes with those of the small subpopulation who adopted PDP recommendations.
In the baseline visit, functional, mental and cognitive status were objectively determined by the same geriatrician (the author), using the same scoring tests in all participants (e.g. GDS, MMSE). However, one limitation of this study is that similar objective scorings were not performed in most participants at the last follow-up visit. Changes in functional, mental and cognitive status, sleep quality, appetite and incontinence were assessed using questionnaires and represent subjective feelings and perceptions of patients/families.
Though certainly a limitation, in line with the practical recommendation to ‘treat the patient, not the numbers’, satisfaction of older patients should be the goal of medical research. 11 The significant improvement in several QoL measures represents a real-life proof that PDP is indeed beneficial to at least some older people consuming more than five medications. Obviously, the ‘time to improvement’ parameter was determined in one of the early follow ups; this was the case for most ‘length of time the improvement lasted’ parameter (unless QoL was still good when sudden death occurred). Another practical limitation of the method when trying to disseminate it to larger populations of caregivers is that it is intrinsically time consuming. Nevertheless, at a later stage it should be time saving in cases of patients improvement.
The contribution of this study in the context of future medicine in older people
The results of this study are in line with the 2012 recommendations of the Institute of Medicine 67 : “Focus on QoL outcome measures, take a more coordinated approach to meeting both health and social needs”. These recommendations display a welcome shift in emphasis to ‘living well’ rather than reducing mortality. 68
Facing this study’s successful results, a common question would be: which of the many de-prescribed drugs caused the improvement in each patient? The nonscientific answer would be that “If the patient improves, why should we care?” The main breakthrough presented in the results is the proof that the absolute number of drugs (polypharmacy) becomes the disease. Something good happens when we reduce drug load and its related symptoms. Current diagnostic tools fail to diagnose these symptoms as ‘the epidemic’; the author’s approach, a therapeutic trial with patients/families consent proving that PDP results in significant QoL improvement, is the only, unequivocal way of proving it. This method was recommended as a leading tool having the strongest evidence of efficacy and clinician acceptability.28,69–71 Quite pragmatically, if ‘poly’-pharmacy is the main etiology of ‘the epidemic’ then ‘poly’-de-prescribing is the cure.1,2,72
There is an issue here as to who takes responsibility for managing or slowing down of ‘the epidemic’. Geriatricians and GPs play a major role (preferably in cooperation with pharmacists) as their hands write prescriptions. We need to change from disease oriented approaches and internalize the fact that for older people, we have no reliable ‘guidelines’ for most diseases/drugs, no proof that prescribing would be better than nonprescribing.1,2 Unfortunately, defensive medicine has turned many doctors into ‘disease technicians’, automatically implementing inappropriate guidelines in everyone without much rethinking. Many ignore two forgotten but essential pillars of the original definition of EBM, clinical judgement and patient/family’s preferences73,74; their combination with ethics are the basis for successful future case managers (weavers), in customizing appropriate therapeutic suits to older people. In the famous Hans Christian Andersen tale, invisible clothes were promoted by crooked weavers as the finest suits possible for the Emperor (the customer). More than clothes, drugs should protect our body and this tale may serve as a fable, the intriguing moral being ‘the epidemic’. Do doctors realize/suspect the new clothes/drugs are useless even harmful, and yet pretend they see benefits for fear of appearing unfit for their positions or stupid, just like the Emperor’s ministers? Doctor’s unawareness or indifference to their own lack of knowledge regarding older people’s therapeutic needs represents the highest level of risk for their patients.
Just like the case of the little boy in Anderson’s tale, we need to shock the system into adjusting to new realities. We are still in search of appropriate educational tools to reduce the GP’s barriers to de-prescribe,1,2,23–28 address ethical issues and increase patients’ involvement in de-prescribing.22,26,29,75–78 Hopefully the author’s approach and this study help promote this section of the war against ‘the epidemic’.
Lastly, the primary goal of this PDP study was improving QoL and not financial incentives. Nevertheless, successful reduction of ‘the epidemic’ has tremendous, potentially beneficial economic consequences representing a triple win–win game: improved health and QoL for millions, and reduced costs of drugs, superfluous evaluations and hospitalizations. 79 Health economists may evaluate the impact of de-prescribing an average of 6–7 drugs in older adults with polypharmacy, on health expenditures. This manuscript applies just as well to the social sphere, to the ethics and legal justification of neglecting patients’ welfare by prescribing drugs without enough knowledge. IGRIMUP, ‘the International Group for Reducing Inappropriate Medication Use and Polypharmacy’, is another step in creating a global body of knowledgeable experts on the topic. However, a successful ‘cure of the epidemic’ requires simultaneous attacks at different points of the vicious cycles perpetuating it. Only an integrated global effort from health professionals, policymakers and consumers, all committed to improving health through optimizing medicines, including involvement from the general press and social networks, can win the war against ‘the epidemic’.
Footnotes
Appendix 1
Questionnaire
—————————————————————————————
—————————————————————————————
—————————————————————————————
Acknowledgements
Statistical analysis was performed by Carmit Rubin and Ilya Novikov, at the Gertner Institute for Epidemiology and Health Policy Research, Israel.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The author declares that there is no conflict of interest.
