Abstract
Background:
Epigenetic biomarkers may provide novel insights into long-term health risk. DNA methylation (DNAm) predicted leptin has been proposed as an epigenetic proxy related to metabolic regulation, but its association with mortality remains underexplored.
Objectives:
To examine the association between DNAm-predicted leptin levels and all-cause, cardiovascular, and cancer mortality among US adults using The National Health and Nutrition Examination Survey (NHANES) data.
Design:
Population-based cohort analysis with linkage to the National Death Index.
Methods:
We analyzed 2531 participants from NHANES 1999–2002 with available DNAm-predicted leptin derived from whole-blood methylation profiles. Cox proportional hazards models were used to estimate hazard ratios (HRs) and 95% confidence intervals (CIs) for mortality outcomes across quartiles of DNAm-predicted leptin, with sequential adjustment for potential confounders. Subgroup and sensitivity analyses were performed to evaluate robustness.
Results:
Over a mean follow-up of 17.1 years, 1360 deaths occurred. DNAm-predicted leptin showed a J-shaped association with all-cause mortality. In fully adjusted models, HRs for all-cause mortality were 1.28 (95% CI: 1.00–1.62) for Quartile 1, 1.00 (reference) for Quartile 2, 1.32 (95% CI: 1.01–1.73) for Quartile 3, and 1.35 (95% CI: 1.11–1.64) for Quartile 4. After full adjustment, DNAm-predicted leptin was not statistically significantly associated with cancer mortality, while cardiovascular mortality risk was higher in Quartile 4 compared with Quartile 2.
Conclusion:
DNAm-predicted leptin was associated with all-cause mortality in a J-shaped pattern, with elevated risk at both lower and higher levels. Associations with cancer mortality were not statistically significant after adjustment, and evidence for cardiovascular mortality was limited to the highest quartile. Larger studies are needed to validate these findings and clarify cause-specific associations.
Trial registration:
Not applicable.
Plain language summary
To examine the association between DNAm-predicted leptin levels and all-cause, cardiovascular, and cancer mortality among U.S. adults using NHANES data. Design: Population-based cohort analysis with linkage to the National Death Index. We analyzed 2,531 participants from NHANES 1999–2002 with available DNAm-predicted leptin derived from whole-blood methylation profiles. Cox proportional hazards models were used to estimate hazard ratios (HRs) and 95% confidence intervals (CIs) for mortality outcomes across quartiles of DNAm-predicted leptin, with sequential adjustment for potential confounders. Subgroup and sensitivity analyses were performed to evaluate robustness. Over a mean follow-up of 17.1 years, 1,360 deaths occurred. DNAm-predicted leptin showed a J-shaped association with all-cause mortality. In fully adjusted models, HRs for all-cause mortality were 1.28 (95% CI: 1.00–1.62) for Quartile 1, 1.00 (reference) for Quartile 2, 1.32 (95% CI: 1.01–1.73) for Quartile 3, and 1.35 (95% CI: 1.11–1.64) for Quartile 4. After full adjustment, DNAm-predicted leptin was not statistically significantly associated with cancer mortality, while cardiovascular mortality risk was higher in Quartile 4 compared with Quartile 2. DNAm-predicted leptin was associated with all-cause mortality in a J-shaped pattern, with elevated risk at both lower and higher levels. Associations with cancer mortality were not statistically significant after adjustment, and evidence for cardiovascular mortality was limited to the highest quartile. Larger studies are needed to validate these findings and clarify cause-specific associations.
Introduction
Leptin, an adipocyte-derived hormone, plays a critical role in regulating energy balance, appetite, and metabolic processes.1,2 Beyond its well-documented role in obesity and metabolic disorders, accumulating evidence suggests that leptin may influence cardiovascular and overall mortality.3–6 However, the biological mechanisms underlying these associations remain unclear, where the interplay between metabolic and cardiovascular health is pivotal.
DNA methylation (DNAm), an epigenetic modification affecting gene expression without altering the underlying DNA sequence, has emerged as a potential mediator linking environmental and genetic factors to disease outcomes.7,8 Recent studies have explored the utility of DNA methylation as a predictive biomarker for various physiological traits.9–11 The National Health and Nutrition Examination Survey (NHANES) has just released DNAm data among US adults since July 2024, which offers a unique opportunity to investigate the relationship between DNAm predicted biomarkers and mortality, given its comprehensive dataset encompassing biomarker measurements, health outcomes, and longitudinal follow-up. Despite the growing body of research, the contribution of DNAm predicted leptin to all-cause and cardiovascular mortality remains insufficiently studied, especially in diverse populations. In this study, we aimed to evaluate the association between DNAm-predicted leptin and the risk of all-cause and cardiovascular mortality among US adults aged 50 years and older.
Methods
Study design and participants
We used data from NHANES, which offers comprehensive data from a nationwide cross-sectional study meticulously conducted by the Centers for Disease Control and Prevention. This program was designed to monitor the health and nutritional status of the US population, and the data was publicly available for researchers to study interesting topics in terms of chronic diseases, which cause a heavy burden in public health among the US population.12–14 Our analysis utilized data from two NHANES cycles (1999–2000 and 2001–2002) with available DNAm data. The original dataset comprised 4983 participants, no less than 50 years. The availability of DNAm data in NHANES 1999–2002 was determined by a predefined subsampling design implemented by NHANES, rather than post hoc exclusion by the investigators. DNAm assays were conducted on a subsample of eligible participants selected using a race/ethnicity-stratified sampling strategy, in which half of non-Hispanic White participants and all eligible participants from non-Hispanic Black, Mexican American, other Hispanic, and other racial/ethnic groups were included for DNAm profiling. This subsampling approach was designed by NHANES to enhance analytic efficiency and representation of minority populations. We excluded individuals either lacking demographic data or without adequate information on DNA methylation and sample weights (
Hazard ratios of all-cause mortality by different levels of DNAm-predicted leptin. Adjusted for age, sex, ethnicity.
Data collection and variables
The NHANES 1999–2002 cycles utilized a complex, multistage sampling approach to ensure representation of the civilian, non-institutionalized population of the United States. Data collection involved a combination of in-home interviews and physical assessments conducted at mobile examination centers. Trained interviewers collected information on demographics, socioeconomic status, marital status, diet, and health-related behaviors using structured questionnaires.
The physical examinations included various clinical measurements, such as height, weight, and blood pressure, alongside other health evaluations. Biological samples were also obtained for laboratory testing. For this study, we extracted data on variables including age, sex, race/ethnicity, family poverty ratio, education, body mass index (BMI), alcohol use, daily energy intake, education level, smoking habits, C-reactive protein (CRP), blood pressure, total cholesterol (TC), and medical history of chronic conditions including cardiovascular disease (CVD), diabetes, and cancer.
DNAm measurements
In this study, adults aged 50 and older who provided blood samples for DNA purification were eligible for inclusion. Approximately half of the eligible non-Hispanic White participants were randomly selected, alongside all eligible individuals from non-Hispanic Black, Mexican American, other Hispanic, and other racial groups. Genomic DNA was extracted from whole blood samples and stored at −80°C until analysis. DNA methylation assays were conducted in the laboratory of Dr Yongmei Liu at Duke University. For each sample, 500 ng of genomic DNA underwent bisulfite conversion using the Zymo EZ DNA Methylation Kit (Zymo Research, Tustin, CA, USA), following the manufacturer’s standardized protocol to convert unmethylated cytosines to uracil while preserving methylated cytosines. Following bisulfite conversion, DNA samples were subjected to whole-genome amplification according to the Illumina Infinium assay protocol. Amplification consisted of 16 PCR cycles, with denaturation at 95°C for 30 s followed by incubation at 50°C for 60 min per cycle. Subsequently, 4 µL of bisulfite-converted and amplified DNA was processed through the Illumina Infinium HD methylation workflow, which included enzymatic fragmentation, precipitation, resuspension, and hybridization to the Illumina HumanMethylationEPIC BeadChip (v1.0) (Illumina, San Diego, CA, USA). Hybridization was performed for 16–24 h under controlled conditions. After hybridization, BeadChips underwent post-hybridization washing, single-base extension, and fluorescent labeling, followed by scanning using the Illumina iScan system to generate raw intensity data for downstream analysis. To account for cellular heterogeneity in whole blood samples, cell type proportions were estimated using a regression-based deconvolution approach. This method employed the IDOL (Identifying Optimal Libraries) probe subset and the FlowSorted.Blood.EPIC_ref reference dataset. Cell type proportions were derived using the estimateCellCounts2 function from the immunomethylomics/FlowSorted.Blood.EPIC R package.16–18 Finally, DNAm-predicted leptin levels were calculated using previously validated regression models incorporating CpG-specific methylation values, chronological age, and sex. Detailed information on the CpG sites included in the prediction models is restricted and available through the Centers for Disease Control and Prevention upon request.
Outcome measurements
The primary outcomes of this study were all-cause mortality. Secondary outcomes included cancer and cardiovascular mortality. Mortality information for NHANES participants was determined by linking their records to the National Death Index (NDI). The follow-up period for each participant was calculated as the time between the baseline survey date and the earlier of either the most recent mortality status update or the last update of the NDI database (December 31, 2019). Causes of death were classified using the International Classification of Diseases, 10th Revision (ICD-10). Cardiovascular mortality was identified with the following ICD-10 codes: I20–I51, I13, I11, I00–I09 and I60–I69. Cancer mortality was identified with the following ICD-10 codes: C00–C97.
Statistical analysis
Statistical analyses were performed using survey-specific procedures to account for the complex sampling design of NHANES. Since the study utilized a subset of the overall dataset, appropriate sample weights were applied to ensure accurate representation. Cox proportional hazards models with weights were employed to calculate hazard ratios (HRs) and 95% confidence intervals (CIs) for the relationship between DNAm-predicted leptin levels and mortality outcomes. Three models were used in the analysis: Model 1 adjusted for age, sex, and ethnicity; model 2 further adjusted for family poverty ratio, education, marital status, status of smoking, status of alcohol drinking, physical activity meeting recommendations, and healthy eating index; model 3 further adjusted for TC ⩾5.2 mmol/L, elevated blood pressure, eGFR <60 mL/min/1.73 m2 or urinary albumin-to-creatinine ratio >30 mg/g; Model 4 further adjusted for log-transformed CRP.
For further analysis, DNAm-predicted leptin levels were categorized into quartiles (
Subgroup analyses were carried out by stratifying participants based on various factors, including age (<65 or ⩾65), BMI (<25, 25.0–29.9, or ⩾30 kg/m²), sex, ethnicity, family poverty ratio (0.0–1.0, 1.1–3.0, >3.0), educational attainment (less than high school, high school, or more), smoking status (never, former, or current smoker), alcohol consumption (never or former, occasional, or heavy), and the presence or absence of diabetes, hypertension, CVD, chronic kidney disease, or cancer. These stratification factors were considered potential effect modifiers. Interaction terms and likelihood ratio tests were applied to evaluate variations in associations across subgroups.
Three sensitivity analyses were performed, excluding participants who died during the first 2 years of follow-up, with missing data, as well as with a history of cancer, heart failure, or ischemic heart disease at baseline visit.
All statistical analyses and figure generation were conducted using R version 4.3.3 (R Foundation for Statistical Computing, Vienna, Austria). Statistical significance was defined as a
Results
Baseline characteristics
We divided the study participants into four groups based on the quartiles of DNAm predicted leptin levels. The baseline characteristics are shown in Table 1. The mean age of participants was 64 ± 10 years old, with a significant increase across quartiles (
Baseline characteristics by sex-specific quartiles of DNAm-predicted leptin.

Mean (SD);
Design-based Kruskal Wallis test; Pearson’s χ2: Rao and Scott adjustment.
DNAm, DNA methylation.
The association between DNAm predicted leptin and all-cause mortality
Over a mean follow-up of 17.1 years, 1360 deaths occurred. In Cox models accounting for the complex NHANES survey design, higher DNAm-predicted leptin levels were associated with increased all-cause mortality risk compared with the reference quartile, and this association remained after sequential adjustment for sociodemographic factors, lifestyle behaviors, and cardiometabolic comorbidities (Table 2) resulting in HRs of 1.28 (95% CI: 1.00–1.62), 1.00, 1.32 (95% CI: 1.01–1.73), and 1.35 (95% CI: 1.11–1.64) across quartiles of DNAm-predicted leptin levels. Restricted cubic spline analyses suggested a non-linear relationship, with elevated risk at both low and high DNAm-predicted leptin levels (Figure 1). The overall pattern was consistent across models, including the additional CRP-adjusted model.
HRs of all-cause mortality and cause-specific mortality by quartiles of DNAm-predicted leptin.

Model 1 adjusted for age, sex, and ethnicity; model 2 further adjusted for family poverty ratio, education, status of smoking, status of alcohol drinking, Physical activity meet recommendation, healthy eating index, and Marital status; model 3 further adjusted for history of cardiovascular disease, history of cancer, total cholesterol ⩾5.2 mmol/L, elevated blood pressure, and eGFR <60 mL/min/1.73 m2 or urinary albumin-to-creatinine ratio >30 mg/g; model 4 further adjusted for log-transformed C-reactive protein.
DNAm, DNA methylation; HR, hazard ratio.
The association between DNAm-predicted leptin and cancer mortality
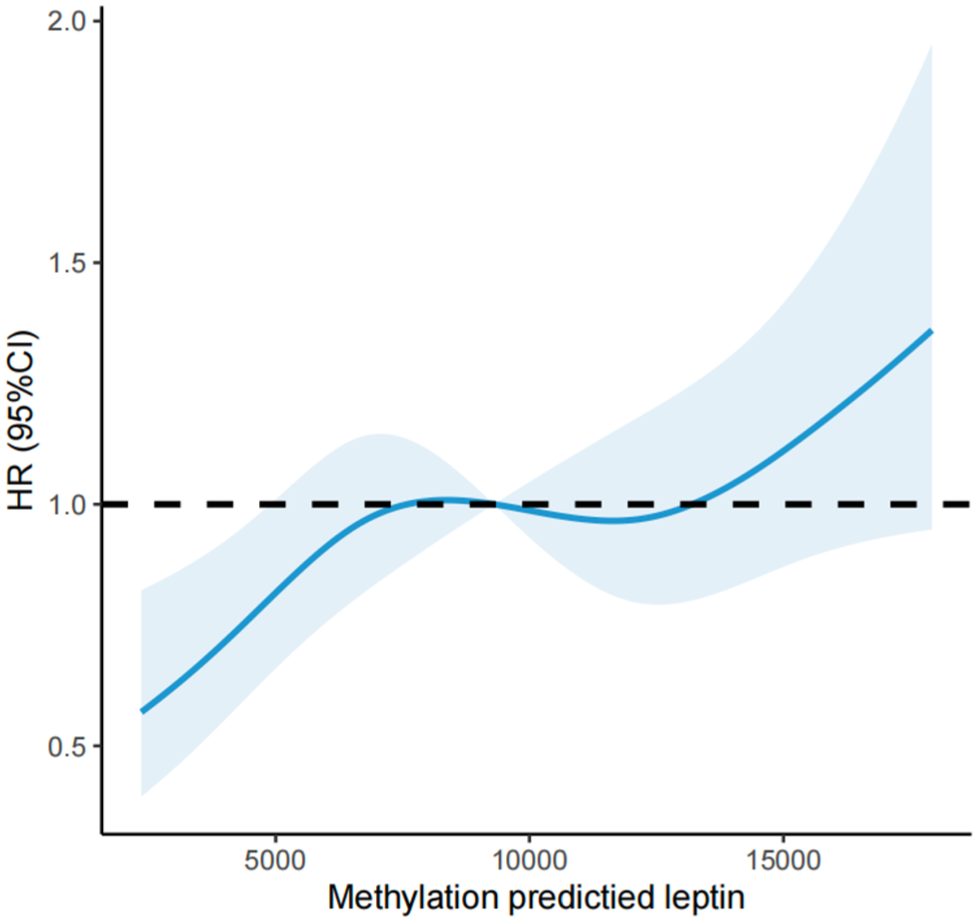
For cancer mortality, point estimates across quartiles were generally above the reference level in partially adjusted models; however, after full multivariable adjustment, the associations were not statistically significant (Table 2). Spline analyses did not provide evidence of a clear non-linear dose-response relationship for cancer mortality (Figure 2).

Hazard ratios of cancer mortality by different levels of DNAm-predicted leptin. Adjusted for age, sex, ethnicity.
The association between DNAm-predicted leptin and cardiovascular mortality
For cardiovascular mortality, higher DNAm-predicted leptin quartiles were associated with increased risk in age-, sex-, and race/ethnicity-adjusted models. After further adjustment for socioeconomic factors, lifestyle variables, and cardiometabolic comorbidities, the associations were attenuated and only Quartile 4 showed a significant result in the fully adjusted model (Table 2). Spline analyses suggested potential non-linearity for cardiovascular mortality (Figure 3), although estimates were imprecise.
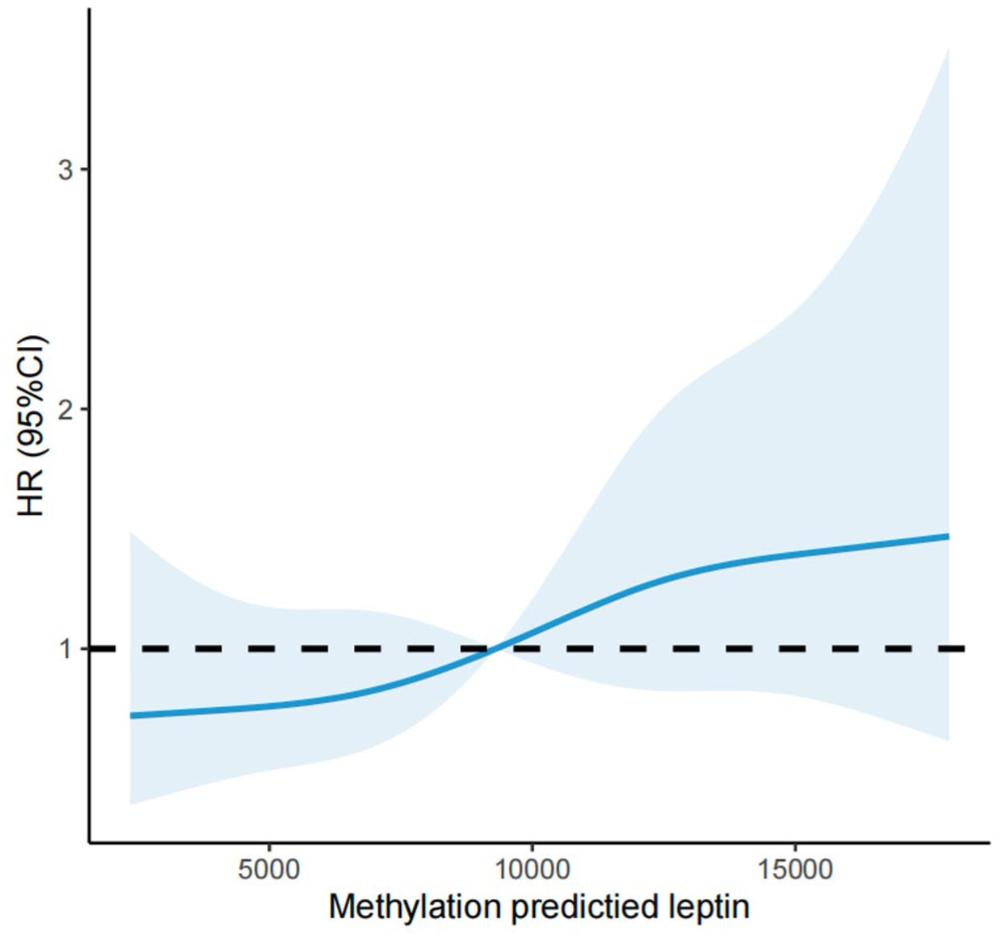
Hazard ratios of cardiovascular mortality by different levels of DNAm-predicted leptin. Adjusted for age, sex, ethnicity.
Subgroup and sensitivity analysis
Further subgroup analysis among participants with different baseline characteristics was performed and confirmed the above findings (Tables 3–5). Three sensitivity analysis confirmed the consistent J-Shape association (Figure 4) between DNAm predicted leptin and all-cause mortality excluding participants who died during the first 2 year of follow-up (Supplemental Table 2), with missing data (Supplemental Table 3), as well as with a history of cancer, heart failure, or ischemic heart disease at baseline visit (Supplemental Table 4).
Hazard ratios of all-cause mortality by quartiles of DNAm-predicted leptin among various subgroups.

Model adjusted for age, sex, ethnicity, family poverty ratio, education, status of smoking, status of alcohol drinking, physical activity meeting recommendations, and healthy eating index, other than the stratifying variable.
DNAm, DNA methylation; HEI, healthy eating index.
Hazard ratios of cancer mortality by quartiles of DNAm-predicted leptin among various subgroups.
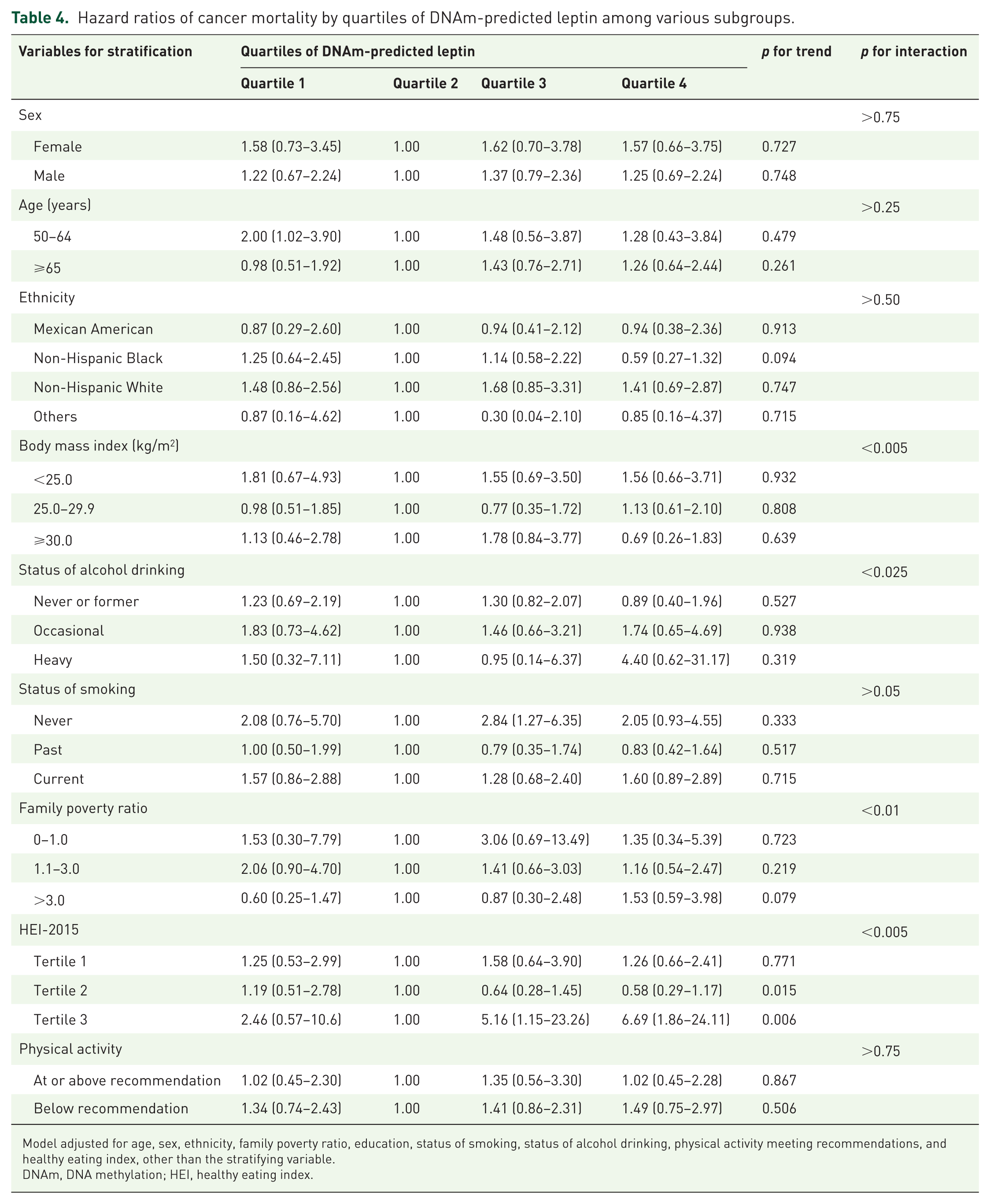
Model adjusted for age, sex, ethnicity, family poverty ratio, education, status of smoking, status of alcohol drinking, physical activity meeting recommendations, and healthy eating index, other than the stratifying variable.
DNAm, DNA methylation; HEI, healthy eating index.
Hazard ratios of cardiovascular mortality by quartiles of DNAm-predicted leptin among various subgroups.

Model adjusted for age, sex, ethnicity, family poverty ratio, education, status of smoking, status of alcohol drinking, physical activity meeting recommendations, and healthy eating index, other than the stratifying variable.
DNAm, DNA methylation; HEI, healthy eating index.

The cumulative incidence curve of time to all-cause mortality (a), time to cancer death (b), and (c) cardiovascular death according to different levels of DNAm-predicted leptin. Adjusted for age, sex, ethnicity.
Mediation effect of BMI on the association between DNAm-predicted leptin and mortality
Mediation analyses were performed to assess whether BMI mediated the associations between DNAm-predicted leptin and mortality outcomes. For all-cause mortality, BMI accounted for only a small and inverse proportion of the association, while the direct effect of DNAm-predicted leptin remained statistically significant, indicating minimal mediation through BMI. For cancer mortality, no significant mediation effect through BMI was observed, and the association was primarily driven by the direct effect. For cardiovascular mortality, although a modest mediation effect through BMI was detected, neither the direct nor total effects reached statistical significance. Overall, these findings suggest that BMI does not meaningfully mediate the relationship between DNAm-predicted leptin and mortality, and that the observed associations are largely independent of adiposity as captured by BMI.
Discussion
In this study, we demonstrated a significant J-shaped association between DNAm predicted leptin and all-cause mortality, while a moderate association between DNAm predicted leptin and cancer and cardiovascular mortality. Our findings contributed to the growing body of evidence on the role of leptin in mortality risk, highlighting the utility of epigenetic markers like DNAm-predicted leptin in assessing long-term health outcomes.
Leptin, a key hormone regulating energy balance and metabolic processes, is increasingly recognized for its broader role in influencing chronic disease, especially metabolic disorders. 2 However, traditional leptin measurements may not fully capture its biological variability or account for its regulation by epigenetic mechanisms. DNAm-predicted leptin could provide a novel approach to bridging this gap by integrating genetic, epigenetic, and environmental influences into a single biomarker. Several other studies reported directly measured leptin and its association with mortality5,6,19,20 and a meta-analysis concluded all their findings. 21 To our knowledge, this is the first study reporting the association between DNAm predicted leptin and mortality outcomes. Its association with mortality outcomes suggests it could serve as a valuable tool for identifying high-risk individuals who might benefit from early preventive measures. Incorporating DNAm-predicted leptin into clinical risk models could improve the accuracy of long-term prognoses, guiding interventions to mitigate risks and improve survival rates.
The epigenetic approach underlying DNAm-predicted leptin allowed us for a more comprehensive understanding of how environmental and lifestyle factors, such as diet, physical activity, and stress, interact with genetic predispositions to influence health trajectories. 22 This integrative perspective was essential for addressing the complex etiology of chronic diseases and mortality. We provided insights into how metabolic dysfunction contributes to cardiovascular and cancer mortality, as demonstrated in this study, since leptin was shown to be a key factor regulating metabolism. DNAm-predicted leptin represents an epigenetic proxy derived from stable DNA methylation patterns rather than a direct measurement of circulating leptin concentrations. As such, it is more likely to reflect long-term epigenetic regulation of leptin expression, integrating cumulative influences of genetic predisposition, metabolic status, and environmental exposures over time. This characteristic distinguishes DNAm-predicted leptin from serum leptin, which is known to exhibit substantial short-term variability in response to acute changes in energy balance, inflammation, and hormonal signaling. Consequently, DNAm-predicted leptin may capture chronic leptin-related biological states that are more relevant to long-term mortality risk, but it does not fully represent the dynamic, moment-to-moment physiological activity of circulating leptin.
Existing evidence linking leptin to cancer pathogenesis through mechanisms including chronic inflammation, angiogenesis, and cell proliferation.23–25 In the present study, DNAm-predicted leptin was not independently associated with cancer or cardiovascular mortality after full multivariable adjustment, and several explanations should be considered when interpreting these null findings. First, the number of cause-specific deaths was substantially smaller than that of all-cause mortality, which likely limited statistical power and resulted in wide CIs, reducing the ability to detect modest associations. Second, the attenuation of effect estimates after adjustment suggests that any observed associations in minimally adjusted models may be partially explained by established cardiometabolic risk factors, comorbidities, and social determinants of health that were included in the fully adjusted models. Third, DNAm-predicted leptin may reflect long-term epigenetic regulation relevant to overall mortality risk, while its contribution to specific disease pathways leading to cancer or cardiovascular death may be weaker or more heterogeneous. Therefore, the absence of statistically significant associations should not be interpreted as definitive evidence of no biological relevance, but rather as an indication that DNAm-predicted leptin does not appear to confer an independent risk for cancer or cardiovascular mortality beyond conventional risk factors in this cohort. The findings also emphasized the need for larger cohorts and more focused studies to confirm and clarify these associations. Similarly, the association between DNAm-predicted leptin and cardiovascular mortality, though more robust in the earlier models, became less pronounced after adjusting for additional covariates. This suggested that some of the observed risks may be mediated through traditional cardiovascular risk factors, such as the comorbidities and the social determinants of health. The nonlinearity observed in these models further pointed to a complex dose-response relationship, where the effects of DNAm-predicted leptin on cardiovascular outcomes may vary depending on the interaction of metabolic, inflammatory, and epigenetic pathways.
The lack of statistical significance in these adjusted models also raised important questions about the role of DNAm-predicted leptin as an independent predictor of cancer and cardiovascular mortality. Meanwhile, the mechanisms underlying the J-shaped association between DNAm predicted leptin levels and mortality are also complex and multifactorial. While it may not act as a standalone risk factor, DNAm-predicted leptin could still be valuable as part of a multifactorial risk assessment framework, particularly when combined with other biomarkers and clinical data. While optimal leptin levels contribute to metabolic balance, both hypo- and hyperleptinemia can disrupt this balance, leading to adverse health outcomes. Elevated leptin levels, on the other hand, often correlated with leptin resistance,2,26 a hallmark of obesity, which is linked to chronic inflammation, insulin resistance, and CVDs, all of which contribute to increased mortality. Low leptin levels may impair the body’s ability to mount an effective immune response, increasing the risk of infections and mortality. 27 Additionally, leptin levels are closely tied to adiposity, with low levels indicating insufficient adipose tissue and high levels reflecting excessive adiposity.28,29 Both extremes are associated with increased mortality, with underweight individuals vulnerable to frailty and infections and obese individuals predisposed to chronic diseases. Our findings also highlighted the importance of considering both the strengths and limitations of DNAm-predicted leptin as a biomarker. Its ability to capture long-term epigenetic regulation of leptin provides unique insights into the biological processes influencing health outcomes. However, its predictive utility may vary across different diseases and populations, emphasizing the need for further validation and refinement of this marker in diverse settings.
Systemic inflammation and social factors may partially contribute to the observed associations between DNAm-predicted leptin and mortality. Leptin is known to interact closely with inflammatory pathways, and CRP represents an important downstream marker of chronic low-grade inflammation. In the present study, inclusion of CRP in the multivariable models modestly attenuated effect estimates, suggesting partial confounding or mediation through inflammatory processes. However, the persistence of the J-shaped association after adjustment supports the robustness of our findings. Similarly, marital status, a well-recognized social determinant of health, was independently associated with mortality risk but did not materially alter the observed leptin–mortality relationship.
Limitations
The strengths of this study include the use of a novel biomarker of DNAm predicted leptin and the use of NHANES as a US representative population. However, some limitations should also be acknowledged. First, the observational nature of the study precludes establishing causality between DNAm-predicted leptin and mortality outcomes. While we observed significant associations, the possibility of residual confounding from unmeasured or inadequately controlled variables cannot be ruled out. Second, the precision of the HR estimates for cancer and cardiovascular mortality was limited by the relatively small number of deaths from these specific causes. Third, while DNAm-predicted leptin provided a unique perspective by integrating genetic, epigenetic, and environmental influences, it may not fully capture the dynamic nature of leptin regulation or the direct effects of circulating leptin levels. This limits its ability to reflect acute changes in leptin associated with short-term physiological or pathological processes. Lastly, the study population was derived from NHANES 1999–2002, which may limit generalizability to other populations. Validation of these findings in more diverse populations and settings is necessary to confirm their broader applicability. In addition, although the analytic sample included only a subset of NHANES participants aged 50 years and older, this was primarily due to the NHANES-defined subsampling strategy for DNA methylation assays, rather than investigator-driven exclusion or post hoc missingness. While DNAm-specific survey weights were applied to preserve national representativeness, the possibility of selection bias cannot be entirely excluded.
Conclusion
In conclusion, this study identified a significant J-shaped association between DNAm-predicted leptin levels and mortality among US adults aged 50 years and older. While moderate associations with cancer and cardiovascular mortality were observed, the lack of statistical significance after adjustment highlights the complexity of these relationships and the need for larger cohorts to confirm the findings. DNAm-predicted leptin may represent a promising tool for advancing precision medicine and public health. Future studies in more diverse populations are warranted to validate these findings and refine the utility of this biomarker in clinical practice.
Supplemental Material
sj-doc-1-tae-10.1177_20420188261426713 – Supplemental material for Contribution of DNA methylation predicted leptin to all-cause, cancer, and cardiovascular mortality among US adults aged 50 years and older: findings from NHANES 1999–2002
Supplemental material, sj-doc-1-tae-10.1177_20420188261426713 for Contribution of DNA methylation predicted leptin to all-cause, cancer, and cardiovascular mortality among US adults aged 50 years and older: findings from NHANES 1999–2002 by Yuxin Fan, Li Ding, Junhe Wang, Qi Li, Hua Shu, Shaofang Tang, Baoping Wang, Gang Hu, Yun Shen and Ming Liu in Therapeutic Advances in Endocrinology and Metabolism
Supplemental Material
sj-docx-2-tae-10.1177_20420188261426713 – Supplemental material for Contribution of DNA methylation predicted leptin to all-cause, cancer, and cardiovascular mortality among US adults aged 50 years and older: findings from NHANES 1999–2002
Supplemental material, sj-docx-2-tae-10.1177_20420188261426713 for Contribution of DNA methylation predicted leptin to all-cause, cancer, and cardiovascular mortality among US adults aged 50 years and older: findings from NHANES 1999–2002 by Yuxin Fan, Li Ding, Junhe Wang, Qi Li, Hua Shu, Shaofang Tang, Baoping Wang, Gang Hu, Yun Shen and Ming Liu in Therapeutic Advances in Endocrinology and Metabolism
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
