Abstract
Background:
Limited studies have explored the relationship between surrogate markers of insulin resistance (IR) with both cardiovascular disease (CVD) risk and CVD mortality in individuals with metabolic dysfunction-associated fatty liver disease (MASLD). This study aimed to assess the associations of various IR surrogates with CVD risk and mortality and to identify effective predictors of cardiovascular outcomes in this population.
Design:
This study constituted a population-based cross-sectional investigation, utilizing data derived from six cycles of the National Health and Nutrition Examination Survey (NHANES) spanning from 2005 to 2016. The data were linked to the NHANES public-use linked mortality files up to December 31, 2019, to facilitate follow-up on CVD mortality. Following predefined exclusion criteria, participants diagnosed with MASLD were identified and incorporated into the study. The research assessed the associations between surrogate markers of IR and both the prevalence of CVD and CVD mortality.
Methods:
The primary outcome of this study was the incidence of CVD in individuals with MASLD, and the secondary outcome was CVD mortality. Weighted multivariate logistic regression was utilized to examine the relationship between surrogate markers of IR with total CVD and other subtypes. Weighted multivariate Cox regression and Kaplan–Meier analysis were used to examine the relationship between surrogate markers of IR and CVD mortality. Restricted cubic spline (RCS) was used to explore potential nonlinear relationships. Receiver operating characteristics (ROC) and calibration curves were plotted to evaluate the discriminability and accuracy of IR surrogate markers in predicting CVD risk. Furthermore, mediation analysis was conducted to determine whether surrogate markers of liver fibrosis play a mediating role in the association of surrogate markers of IR with the risk of total CVD and other subtypes.
Results:
This study suggests that in the fully adjusted model, estimated glucose disposal rate (eGDR) was significantly negatively correlated with total CVD, congestive heart failure (CHF), heart attack (HA), and CVD mortality. Metabolic score for IR and triglyceride-glucose-body mass index were significantly positively correlated with CVD mortality, total CVD, and CHF. eGDR was best correlated with total CVD (odds ratio (OR) = 0.40, 95% confidence interval (CI): 0.25, 0.63), CHF (OR = 0.15, 95% CI: 0.06, 0.38), and HA (OR = 0.40, 95% CI: 0.22, 0.71). Weighted multivariate Cox regression indicated that for each unit increase in eGDR, the risk of CVD mortality decreased by 27%. RCS analysis showed that all surrogate markers of IR were linearly related to CVD and CVD mortality (
Conclusion:
In conclusion, eGDR outperformed other IR surrogates in predicting both CVD risk and CVD mortality in MASLD. Incorporating eGDR into prediction models may enhance risk stratification and facilitate early identification of high-risk individuals in this population. Further studies are warranted to validate these findings in external cohorts.
Plain language summary
For people with metabolic dysfunction-associated fatty liver disease (MASLD), which simple blood-based markers of insulin resistance best flag a higher risk of heart and blood-vessel problems–and which one does the best job of predicting future deaths from these conditions?
We analyzed data from 3919 U.S. adults with MASLD who took part in the NHANES surveys (2005–2016). We compared several common markers of insulin resistance, including the estimated glucose disposal rate (eGDR)–a calculation where higher values mean better insulin sensitivity–as well as METS-IR and TyG-BMI. We looked at who had cardiovascular disease (CVD) such as heart failure, heart attack, or stroke, and who later died from CVD. We also tested how well each marker could sort people into higher- vs lower-risk groups.
What it means People with higher eGDR had lower chances of having CVD, including heart failure and heart attack, and were also less likely to die from CVD over time. METS-IR and TyG-BMI were linked to some CVD outcomes (like overall CVD and heart failure) but did not predict CVD death as well. Overall, eGDR outperformed the other markers in identifying who was at risk. When we added eGDR to a basic risk model, its ability to tell apart high- and low-risk patients improved. We also found that signs of liver scarring (fibrosis) may partly explain how eGDR relates to heart problems.
For people with MASLD, eGDR is a strong, easy-to-use signal of heart risk and future CVD death. Including eGDR in routine assessments could help clinicians spot high-risk patients earlier and act sooner. These results should be confirmed in other groups to make sure they apply widely.
Introduction
Metabolic dysfunction-associated fatty liver disease (MASLD) is a chronic liver disease linked to metabolic dysfunction, predominantly driven by hepatic fat accumulation resulting from IR, lipotoxicity, and oxidative stress, has become one of the most common chronic liver diseases in the world, affecting more than one-third of adults worldwide. 1 MASLD contributes substantially to the burden of liver disease and serves as a hidden driver of the increasing risk of CVD. 2 CVD is the leading cause of mortality among patients with MASLD. 3 Excessive and dysfunctional adipose tissue is a key factor linking hepatic and cardiovascular comorbidities, increasing CVD risk through metabolic and inflammatory pathways. The underlying pathophysiological mechanisms include chronic inflammation, endothelial dysfunction, insulin resistance (IR), and lipid metabolism disorders.4,5
IR is defined as impaired insulin signaling pathway function resulting in diminished biological response to insulin in target organs/tissues, particularly adipose tissue and liver. 6 Extrahepatic effects of IR, including dyslipidemia, low-grade inflammation, and endothelial dysfunction, markedly increase CVD risk in individuals with MASLD. 7 To date, the hyperinsulinemic euglycemic clamp (HIEG) technique remains the gold standard for evaluating IR. However, its invasiveness and high cost limit its large-scale application in clinical practice. 8 The typical characteristics of IR are hyperglycemia and compensatory hyperinsulinemia. At the same time, IR is also related to dyslipidemia, obesity, and hypertension.9–11 Therefore, according to the different characteristics of IR, multiple surrogate markers of IR have been introduced, including the homeostasis model assessment of IR (HOMA-IR), metabolic score for IR (METS-IR), the triglyceride-glucose (TyG) index and its derivatives, as well as the estimated glucose disposal rate (eGDR). eGDR was initially used to assess the IR in type 1 diabetes, and was subsequently validated in type 1 diabetes patients using HIEG technique, becoming a reliable IR index. 12 In addition, IR is one of the important risk factors for the progression of MASLD to liver fibrosis. At the same time, increasing evidence shows that there is a relationship between liver fibrosis and CVD,13,14 yet no noninvasive fibrosis biomarker has received endorsement in clinical guidelines for CVD diagnosis or prognosis. The Metabolic Dysfunction Associated Fibrosis 5 (MAF-5) Score is a novel tool used to assess the risk of fibrosis in the general population with metabolic dysfunction related fatty liver disease. This score is age-independent and based on simple anthropometric indicators, it may be a better alternative to the Fibrosis 4 Index score (FIB-4). 15 In order to strengthen the prevention of CVD in MASLD individuals, it is necessary to identify high-risk groups with increased CVD risk as early as possible, achieve early identification and early intervention, and promote cardiovascular-liver-metabolic health.
However, the performance of different IR surrogate markers in predicting CVD risk and CVD mortality in individuals with MASLD remains underexplored and compared. The purpose of this study was to investigate the association between different insulin replacement indicators and the risk of CVD incidence rate and cardiovascular mortality in MASLD individuals. Simultaneously assessing the mediating role of liver fibrosis degree in these associations.
Methods
Study population and design
The National Health and Nutrition Examination Survey (NHANES) is a continuous cross-sectional survey designed to provide nationally representative data through a complex, multistage sampling strategy. It incorporates interviews, examinations conducted at Mobile Examination Centers, and laboratory analyses. The study protocol was approved by the National Center for Health Statistics Research Ethics Review Board, and all participants provided informed consent. To create this cross-sectional study, data from six NHANES cycles (2005–2016) were merged. Exclusion criteria: (1) under 20 years old; (2) lacked USFLI data; (3) heavy drinking (⩾30 g/day for men and ⩾20 g/day for women); (4) positive for hepatitis B surface antigen, hepatitis C antibody, or hepatitis RNA; (5) participants with malignancy; (6) participants with missing data required for calculating the TyG-body mass index (TyG-BMI), HOMA-IR, METS-IR, and eGDR; (7) participants with ineligible CVD data and follow-up data; (8) without other key covariates. The final sample consisted of 3919 MASLD participants drawn from NHANES data collected between 2005 and 2016. This study is reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 16 The STROBE checklist is provided in Table S9.
Covariates
All variables were derived from the NHANES database, and comprehensive details are provided in the Supplemental Materials. The present study collected the following covariates: age, gender, race, education level, physical activity, marital status, smoking status, the ratio of family income to poverty (PIR), waist circumference (WC), BMI, systolic blood pressure (SBP), diastolic blood pressure (DBP), glycated hemoglobin (HbA1c), fasting insulin, fasting plasma glucose (FPG), total cholesterol (TC), triglyceride (TG), high density lipoprotein cholesterol (HDL-C), low density lipoprotein (LDL), albumin (Alb), alanine aminotransferase (ALT), aspartate aminotransferase (AST), gamma-glutamyl transferase (GGT), total bilirubin (TBIL), estimated glomerular filtration rate (eGFR), uric acid (UA), Healthy Eating Index 2015 (HEI-2015), CVD, type 2 diabetes mellitus (T2DM), and hypertension.
Definition of MASLD and IR
In accordance with the Delphi method consensus criteria, MASLD was defined as steatotic liver disease (SLD) with at least one cardiometabolic risk factor, while excluding viral hepatitis, autoimmune liver disease, genetic liver disorders, drug-induced liver disease, or alcohol-related liver disease (alcohol intake ⩾30 g/day for men or ⩾20 g/day for women). A USFLI ⩾30 has demonstrated greater validity for SLD diagnosis than the FLI, this criterion demonstrated an area under the ROC curve of 0.80 (95% confidence interval (CI): 0.77, 0.83) in predicting ultrasonographically confirmed SLD.17,18 Compared to liver biopsy, USFLI provides a simple, non-invasive way to assess MASLD risk, particularly suitable for screening large populations.
USFLI = (e−0.8073 × non-Hispanic black + 0.3458 × Mexican American + 0.0093 × age + 0.6151 × ln (GGT) + 0.0249 × WC + 1.1792 × ln (insulin) + 0.8242 × log (FPG) − 14.7812)/(1 + e−0.8073 × non-Hispanic black + 0.3458 × Mexican American + 0.0093 × age + 0.6151 × ln (GGT) + 0.0249 × WC + 1.1792 × ln (insulin) + 0.824 × log (FPG) − 14.7812) × 100. Participants who self-identified as non-Hispanic Black or Mexican Americans were coded as 1, while all others were coded as 0. 19
The specific adult cardiometabolic criteria are defined as follows:
(1) BMI ⩾25 kg/m2 (23 kg/m2 for Asians) or WC >94 cm in men, >80 cm in women; (2) FBG ⩾5.6 mmol/L (100 mg/dL) or 2-h post-load blood glucose ⩾7.8 mmol/L (140 mg/dL) or HbA1c ⩾5.7% (39 mmol/L) or a diagnosis of type 2 diabetes or type 2 diabetes treatment; (3) blood pressure ⩾130/85 mmHg or specific antihypertensive treatment; (4) TG ⩾ 1.70 mmol/L (150 mg/dL) or lipid-lowering therapy; (5) HDL-C ⩽1.0 mmol/L (40 mg/dL) in men, ⩽1.3 mmol/L (50 mg/dL) in women, or lipid-lowering therapy.
IR was assessed using the commonly used alternative indices derived from readily available clinical parameters. The primary index used was the eGDR index. In addition, three other commonly used IR indices were included for comparison, including TyG-BMI, HOMA-IR, and METS-IR. The detailed calculation procedure is described in the Supplemental Materials. According to the quartiles of each index, participants were divided into four groups (
Outcome definitions
The primary outcome of this study was the incidence of CVD in individuals with MASLD, and the secondary outcome was CVD mortality.
The diagnosis of total CVD and CVD subtypes was established by self-reported physician diagnoses using a standardized medical condition questionnaire. The participants were asked, “Has a doctor or other health professional ever informed you/SP that you/he/she. . . had congestive heart failure (CHF), coronary heart disease (CHD), angina pectoris, heart attack (HA), stroke, etc.?” A person was considered to have total CVD if they answered “yes” to any of the aforementioned questions. 20 In addition, we analyzed events related to CHD, CHF, angina pectoris, HA, and stroke separately. CVD mortality specifically refers to deaths attributed to heart disease and cerebrovascular diseases. Mortality data for the follow-up population were sourced from the NHANES Public-use link mortality files, with updates extending through December 31, 2019. These records were matched to the National Center for Health Statistics and the National Death Index using probabilistic algorithms. Mortality outcomes were coded using the standardized International Classification of Diseases, Tenth Revision (ICD-10) system. The observation time was defined as the duration between baseline assessment (initial interview) and the subsequent occurrence of either mortality or study completion. 21
Statistical analysis
Statistical analyses were performed using R (version 4.4.2, http://www.R-project.org). Missing data were imputed by the MissForest method based on random forests. Due to the complexity of the sampling design of NHANES, we selected appropriate weights for data analysis. The study population was divided into four groups according to the quartiles (
Receiver operating characteristic (ROC) curves were generated for each marker, and the area under the curve (AUC) was calculated to evaluate its predictive performance. In addition, we calculated the C-statistic, net reclassification index (NRI), and composite discrimination improvement index (IDI) to assess the incremental performance of surrogate markers of IR relative to conventional risk factors for total CVD, CHD, CHF, angina pectoris, HA, and stroke in individuals with MASLD. The “timeROC” package was used to assess the accuracy of these indices in predicting survival outcomes at different time points. The C-index is a discrimination index, while the IDI and NRI assess whether the new model provides improved risk stratification compared with the base model. The interactions of gender, age status (<40, 40–59, and ⩾60), race, education level, marital status, PIR status, BMI status (⩾30 and <30), smoking status, physical activity, and diabetes with CVD and CVD mortality were assessed by stratified analysis and interaction tests. Mediation analyses were performed to assess the mediating role of liver fibrosis degree in the associations of surrogate markers of IR with total CVD, CHD, CHF, angina pectoris, HA, and stroke.
Results
Baseline characteristics of study participants
In total, 3919 participants with MASLD were included in this study from 2005 to 2018. The mean age was 53.76 ± 16.26 years, with 44.68% female and 55.32% male. The baseline mean values ± standard deviation of eGDR, TyG-BMI, HOMA-IR, and METS-IR were 5.26 ± 2.35, 307.59 ± 61.73, 7.58 ± 10.17, and 53.71 ± 11.65, respectively. During a median follow-up of 101 months, 610 total CVDs and 122 cardiovascular deaths were observed. A flowchart of participant selection is provided in Figure S1.
Table 1 summarizes the baseline characteristics of the study population stratified by eGDR quartiles. No significant differences in gender, education, TG, AST, and GGT were observed across the quartiles. However, participants with a lower eGDR tended to be older, obese, widowed/divorced/separated or never married, non-Hispanic Black, non or former smoker, physically inactive and have a history of hypertension, diabetes, and CVD compared to those in the highest quartile. Additionally, the higher quartiles of eGDR were significantly associated with elevated levels of ALT, eGFR, TBIL, TC, LDL.
Baseline characteristics of the study population stratified by quartiles of the eGDR.
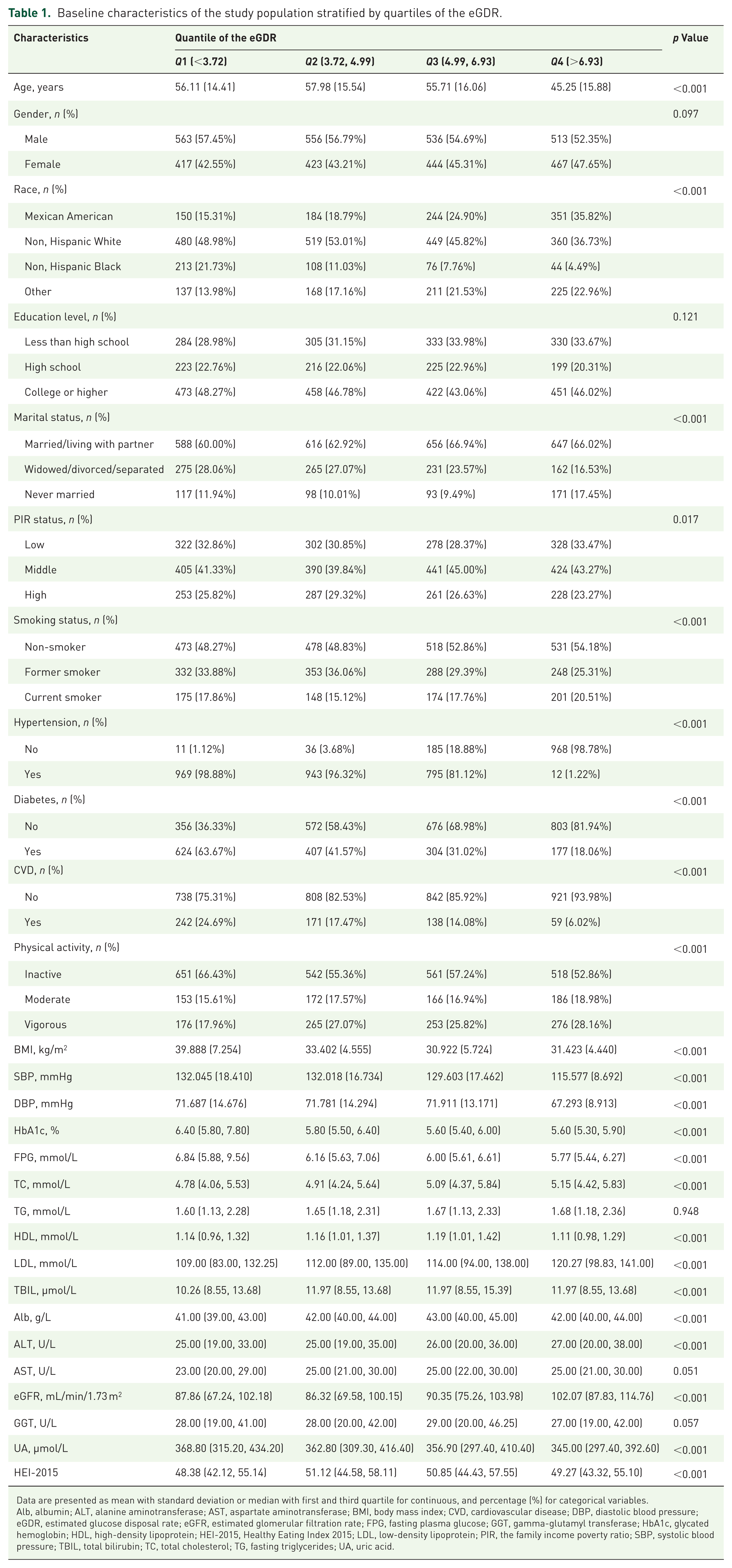
Data are presented as mean with standard deviation or median with first and third quartile for continuous, and percentage (%) for categorical variables.
Alb, albumin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BMI, body mass index; CVD, cardiovascular disease; DBP, diastolic blood pressure; eGDR, estimated glucose disposal rate; eGFR, estimated glomerular filtration rate; FPG, fasting plasma glucose; GGT, gamma-glutamyl transferase; HbA1c, glycated hemoglobin; HDL, high-density lipoprotein; HEI-2015, Healthy Eating Index 2015; LDL, low-density lipoprotein; PIR, the family income poverty ratio; SBP, systolic blood pressure; TBIL, total bilirubin; TC, total cholesterol; TG, fasting triglycerides; UA, uric acid.
In contrast, participants in the higher quartiles of TyG-BMI, HOMA-IR, and METS-IR indices were more likely to be male, younger, married/living with partner, higher education, physically inactive, lower PIR, obesity, and hypertension. The laboratory characteristics of the enrolled participants showed similar trends in all indices except eGDR. Those in the highest quartile showed significantly increased FPG, TG, eGFR, DBP, and HbA1c levels, accompanied by significantly decreased HDL, TBIL, and Alb levels (Tables S1–S3).
Associations of different IR alternative markers with CVD mortality, total CVD, CHF, CHD, angina pectoris, HA, and stroke
Table S4 shows that higher eGDR levels were consistently associated with lower risks of total CVD, CHF, CHD, angina, HA, and stroke in model 1. This association remained significant when eGDR was categorized into quartiles. After adjusting for age, gender, race, education level, marital status, smoking status, PIR status, physical activity, HEI-2015, diabetes, SBP, DBP (model 2), we still found that eGDR was significantly inversely associated with total CVD, CHF, HA, and stroke, and this inverse trend remained even after dividing eGDR into quartiles (Table S5). After adjusting for Alb, AST, ALT, eGFR, TBIL, TC, LDL, GGT, UA (model 3), we found that eGDR was significantly inversely associated with total CVD, CHF, HA, and stroke. This inverse trend remained after dividing eGDR into quartiles (Table S6). Table 2 illustrates the association of IR alternative markers with CVD in model 4. After fully adjusting for covariates, weighted logistic regression analysis showed that each 1-SD increase in eGDR was significantly associated with a reduced risk of total CVD (odds ratio (OR) = 0.85, 95% CI: 0.79, 0.92;
Association of IR alternative markers with CVD (adjusted for model 4).
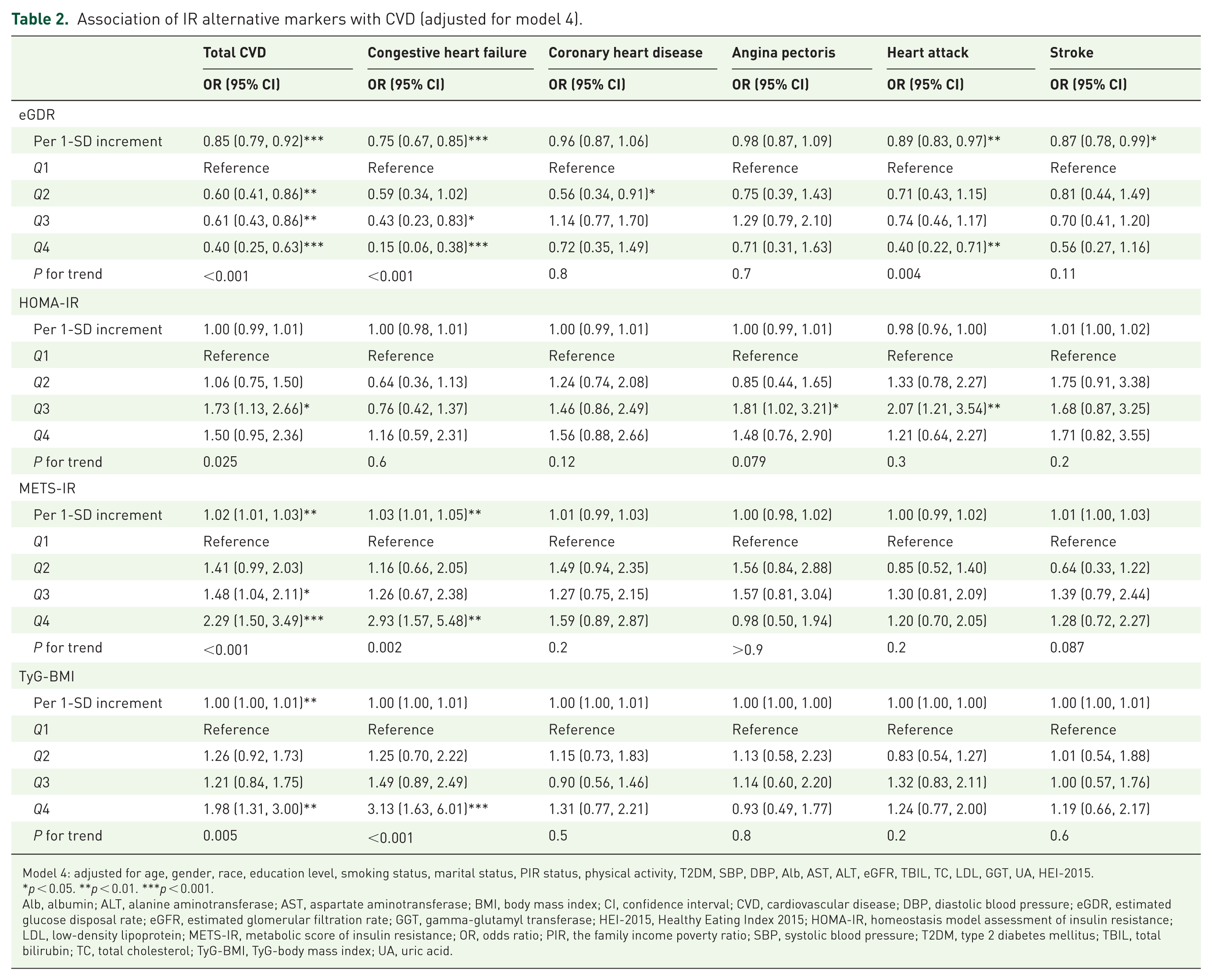
Model 4: adjusted for age, gender, race, education level, smoking status, marital status, PIR status, physical activity, T2DM, SBP, DBP, Alb, AST, ALT, eGFR, TBIL, TC, LDL, GGT, UA, HEI-2015.
Alb, albumin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BMI, body mass index; CI, confidence interval; CVD, cardiovascular disease; DBP, diastolic blood pressure; eGDR, estimated glucose disposal rate; eGFR, estimated glomerular filtration rate; GGT, gamma-glutamyl transferase; HEI-2015, Healthy Eating Index 2015; HOMA-IR, homeostasis model assessment of insulin resistance; LDL, low-density lipoprotein; METS-IR, metabolic score of insulin resistance; OR, odds ratio; PIR, the family income poverty ratio; SBP, systolic blood pressure; T2DM, type 2 diabetes mellitus; TBIL, total bilirubin; TC, total cholesterol; TyG-BMI, TyG-body mass index; UA, uric acid.
In Table S7, for each unit increase in eGDR, the risk of CVD mortality decreased by 27% in model 4. This relationship persisted even after dividing eGDR into quartiles, the risk of CVD mortality in the highest quartile decreased by 93% (hazard ratio (HR) = 0.07, 95% CI: 0.03, 0.18;
RCS analysis investigating the relationship between IR alternative markers and CVD mortality, total CVD, CHF, CHD, angina pectoris, HA, and stroke
In Figure 1, we visualized the associations between eGDR, HOMA-IR, METS-IR, and TyG-BMI and CVD mortality, total CVD, CHF, CHD, angina pectoris, HA, and stroke using RCS functions. After adjusting for all covariates in model 4, there was a linear association between eGDR and CVD mortality, total CVD, CHF, CHD, angina pectoris, HA, and stroke (

Associations between eGDR, HOMA-IR, METS-IR, and TyG-BMI with cardiovascular mortality, total cardiovascular disease, congestive heart failure, coronary heart disease, angina, and stroke were evaluated by the RCS after adjustment for model 3. The blue solid line of the RCS curve represents the fitted smooth curve, while the shaded area indicates the 95% confidence interval.
The ability of the IR alternative markers to predict CVD mortality, total CVD, CHF, CHD, angina pectoris, HA, and stroke and incremental predictive value of eGDR
As illustrated in Figure 2, eGDR demonstrated the strongest diagnostic capability across multiple cardiovascular outcomes. For total CVD, the AUC for eGDR was 0.650 (95% CI: 0.628, 0.672), exceeding that of HOMA-IR (AUC = 0.530, 95% CI: 0.505, 0.556). In the assessment of CHF, eGDR again achieved the highest AUC at 0.678 (95% CI: 0.643, 0.713), followed by METS-IR (AUC = 0.563, 95% CI: 0.521, 0.606). For CHD, eGDR yielded an AUC of 0.604 (95% CI: 0.571, 0.636), with TyG-BMI ranking second (AUC = 0.543, 95% CI: 0.508, 0.578). Regarding angina pectoris, eGDR reached an AUC of 0.628 (95% CI: 0.588, 0.669), whereas HOMA-IR achieved 0.541 (95% CI: 0.492, 0.591). For HA, the AUC of eGDR was 0.633 (95% CI: 0.600, 0.666), compared with 0.529 (95% CI: 0.492, 0.567) for TyG-BMI. Finally, for stroke, eGDR again performed best with an AUC of 0.629 (95% CI: 0.592, 0.667), surpassing HOMA-IR (AUC = 0.556, 95% CI: 0.513, 0.600). Collectively, these findings highlight eGDR as the most effective IR surrogate for identifying CVD risk in individuals with MASLD.

ROC curve for evaluating the diagnostic power of eGDR, HOMA-IR, METS-IR, and TyG-BMI.
Time-dependent ROC curve analysis (Figure S2) showed that eGDR had the best prediction effect on 3-year CVD mortality, followed by HOMA-IR, METS-IR, and TyG-BMI. Meanwhile, eGDR showed better performance in predicting 5- and 10-year CVD mortality. Time-dependent AUC further showed that eGDR showed the highest accuracy in predicting long-term CVD mortality risk, followed by HOMA-IR. We further investigated whether adding IR alternative markers to the basic model could improve the prediction ability of total CVD, CHF, CHD, angina pectoris, HA, and stroke in individuals with MASLD. As shown in Table S8, adding eGDR to the basic model improved the C statistics for all studied outcomes. However, except for the significant improvement in the C statistic for total CVD and CHF, the
Kaplan–Meier survival curves of the IR alternative markers for CVD mortality
During a median follow-up of 101 months, a total of 122 (3.1%) cardiovascular deaths occurred. Figure S3 plots the Kaplan–Meier survival curves for surrogate markers of IR and CVD mortality in individuals with MASLD. In the MASLD population, mortality decreased with increasing eGDR quartiles (
Mediation analysis of eGDR and total CVD, CHF, CHD, angina pectoris, HA, and stroke
Mediation analysis showed that MAF-5 indirectly mediated the associations between eGDR and total CVD, CHF, CHD, angina pectoris, HA, and stroke (Figure S4). The indirect effects of MAF-5 mediated eGDR on total CVD, CHF, CHD, angina pectoris, HA, and stroke accounted for −25.2% (
Stratified analysis of eGDR in relation to CVD mortality, total CVD, CHF, CHD, angina pectoris, CHD, and stroke
Stratified analysis indicated that the association between eGDR and CVD mortality was generally consistent across most subgroups in the MASLD population, including age category, sex, BMI category, educational level, PIR category, smoking status, marital status, physical activity, and diabetes status (all

Stratified analysis of the association between eGDR and CVD in individuals with MASLD. (a) Total CVD. (b) CHF. (c) CHD. (d) Angina pectoris. (e) HA. (f) Stroke. Adjusted for age, gender, race, education level, BMI, smoking status, PIR status, physical activity, diabetes, SBP, DBP, Alb, AST, ALT, eGFR, TBIL, GGT, UA, TC, LDL, HEI-2015, except the subgroup factors themselves.
Discussion
This study represents the first comprehensive evaluation of the associations between multiple IR surrogates and both CVD incidence and CVD mortality in a U.S. MASLD population. Our results showed that eGDR was significantly inversely associated with total CVD, CHF, and HA, whereas METS-IR and TyG-BMI demonstrated positive associations with total CVD and CHD. In addition, eGDR, METS-IR, and TyG-BMI were all significantly associated with CVD mortality. RCS analysis showed that all surrogate markers of IR were linearly related to CVD and CVD mortality. In ROC analysis, eGDR showed the highest efficacy in predicting CVD and long-term CVD mortality, and it was found that eGDR combined with the basic model had better accuracy and discrimination ability than the basic model. Collectively, these results suggest that IR surrogates—particularly the cost-effective and readily accessible eGDR—may serve as practical tools for cardiovascular risk monitoring and management in individuals with MASLD.
Given the complex interplay between various pathophysiological mechanisms of IR and MASLD, early detection of individuals at risk of CVD is crucial. Currently, the commonly used IR surrogate markers in clinical practice include TyG and TyG-related parameters, METS-IR, HOMA-IR, and eGDR. Numerous studies have examined the associations between these IR markers and both CVD incidence and mortality. Evidence from prior research has consistently demonstrated that IR is strongly associated with the risk of CVD, all-cause mortality, and cardiovascular mortality in diverse populations, including patients with chronic kidney disease, hypertension, diabetes, and MASLD.22–25 At present, most studies have proved that TyG and related parameters are excellent indicators for predicting the risk of CVD, all-cause mortality, and cardiovascular mortality in NAFLD/MASLD individuals.26–29 However, some research results are still contradictory. Some studies have shown that TyG-WHtR is the best predictor of CVD and mortality in this group, 29 while other studies suggest that TyG-BMI is more accurate. 27 In addition, in patients with diabetes, higher HOMA-IR quartiles are significantly positively correlated with all-cause mortality, CVD mortality, and major adverse cardiovascular events (MACE).30,31 A recent study showed that the HOMA-IR index was not significantly associated with all-cause mortality and cardiovascular mortality in MASLD patients, which is consistent with our research findings. 27 A recent meta-analysis showed that elevated TyG levels in the general population significantly increased the risk of myocardial infarction, cardiovascular mortality, all-cause mortality, stroke, and the need for revascularization. Subgroup analyses revealed a significant positive correlation between TyG levels and MACE risk and all-cause mortality in both diabetic and non-diabetic individuals. However, only the diabetic group showed significantly higher outcomes in terms of stroke, myocardial infarction, and cardiovascular mortality. Conversely, in the non-diabetic group, the risk of requiring revascularization was only significantly increased. 32 It has been reported that METS-IR is a powerful predictor of mortality and CVD risk in the general population, metabolic syndrome (MetS) patients, hypertensive patients, and patients who have undergone coronary artery bypass grafting/percutaneous coronary intervention.33–36 The eGDR index represents a novel, noninvasive IR surrogate that evaluates IR in multiple dimensions, incorporating WC, HbA1c, and hypertension status. 37 In the current study, other IR surrogates exhibited comparatively weak or nonsignificant associations with CVD risk and CVD mortality in MASLD, with lower predictive performance relative to eGDR.
Obesity is widely acknowledged as a major risk factor for IR, and dysfunction of adipose tissue represents a pivotal initiating event in the IR cascade. 38 Dysfunctional adipose tissue releases excessive free fatty acids (FFAs) into circulation, inducing lipotoxicity, and secretes pro‑inflammatory adipokines such as TNF‑α and IL‑6. These processes trigger oxidative stress and systemic chronic low‑grade inflammation, ultimately damaging vascular endothelial cells, impairing endothelial function, and contributing to the onset and progression of CVD.39,40 Hypertension also constitutes a key risk factor for CVD, playing a crucial role in its initiation and development. 41 In previous studies, eGDR was mostly used to evaluate the association between IR and CVD in diabetic patients. A study involving 19960 T1DM individuals showed that during a median follow-up period of 10 years, for every 1 unit increase in the eGDR index, the summary HR for the composite CVD outcome was 0.83, and the pooled HR for all-cause mortality was 0.84. 23 Another meta-analysis also demonstrated that lower eGDR was associated with an increased risk of MACE, coronary artery disease (CAD), and all-cause mortality in patients with T1DM. 42 In addition, another study based on the NHANES database used a machine learning algorithm to find that eGDR was significantly negatively correlated with the likelihood of CVD, CAD, CHF, and stroke in people with diabetes or prediabetes, and its predictive performance was better than other IR alternative indicators (including TyG, HOMA-IR, and METS-IR). 43 With the accumulation of evidence, it has become clear that the predictive utility of eGDR extends beyond diabetic populations. Interestingly, a retrospective study of 2144 patients who underwent percutaneous coronary intervention showed that higher eGDR was associated with a lower incidence of major adverse cardio-cerebrovascular events (MACCE) in pre-DM and non-DM groups, but not in DM patients. 44 A prospective cohort study found that lower eGDR levels were associated with a higher risk of CVD among non‑diabetic participants, and each 1 unit increase in eGDR reduced CVD risk by 17% (HR = 0.83, 95% CI: 0.78, 0.89). 45 A recent meta-analysis showed that lower eGDR was significantly associated with worse cardiovascular outcomes, including an increased risk of MACCE, CVD, and all-cause mortality in patients with diabetes. Furthermore, subgroup analyses based on diabetes status revealed that in non-diabetic groups, reduced eGDR was strongly associated with a significantly increased risk of stroke and CVD. 46 These results may be attributed to the use of hypoglycemic agents such as sodium-glucose cotransporter-2 inhibitors and glucagon-like peptide-1 receptor agonists in diabetic patients. Furthermore, diabetic patients often have multiple risk factors, such as chronic inflammation, endothelial dysfunction, and dyslipidemia; the combined effect of these factors may reduce the potential of IR to predict cardiovascular risk in diabetic patients. A large cohort study involving the UK Biobank and the China Health and Retirement Longitudinal Study showed that individuals with low-level increases and persistent low levels of eGDR had a higher risk of CVD during follow-up, while individuals with persistently high eGDR levels had the lowest risk of CVD. 47 Recent studies have shown that eGDR is closely related to predicting all-cause mortality and cardiovascular mortality in individuals with cardiovascular-renal-metabolic syndrome stage 0–3. At the same time, eGDR is significantly negatively correlated with the risk of CVD in this population, and its ability to predict CVD is superior to other IR alternative markers (including TyG and its derivative parameters, TG/HDL-C and METS-IR). In addition, incorporating eGDR into the prediction model can significantly improve the prediction ability of the model. 48 Currently, MASLD is frequently described as the hepatic manifestation of MetS, for which IR is a defining characteristic. Evidence from MetS cohorts has shown that eGDR is significantly associated with both all‑cause and CVD mortality and provides superior predictive accuracy for all‑cause mortality in this population. 49 Collectively, these findings support eGDR as a reliable and broadly applicable biomarker for identifying high‑risk individuals for CVD. Consistent with prior research, our study demonstrated that lower eGDR levels were independently associated with increased CVD risk and mortality and revealed a clear inverse linear relationship between eGDR and both outcomes. Overall, these results highlight eGDR as a novel and promising surrogate of IR with substantial clinical potential for predicting CVD risk and long‑term cardiovascular mortality in individuals with MASLD.
Our study further reveals the heterogeneity of eGDR’s predictive value across different subgroups. Our findings indicate that the significant association between eGDR and CHF is more pronounced in women, and women with low eGDR have significantly increased total CVD, CHD, HA, stroke, and cardiovascular mortality. Women, due to changes in the hormonal environment and sex differences in fat distribution, may further exacerbate IR-related hemodynamic and inflammatory stress, thereby increasing susceptibility to CVD.50,51 Simultaneously, we observed that the association between eGDR with CVD mortality, CHD, CHF, angina pectoris, HA, and stroke was more significant in the non-T2DM group than in the T2DM group. An important reason for this may be that diabetes and MASLD themselves remain major risk factors for cardiovascular events, and these patients often have multiple risk factors simultaneously, such as chronic inflammation, endothelial dysfunction, and dyslipidemia. These factors independently drive cardiovascular outcomes, meaning that IR may not have increased predictive value in patients with diabetes and MASLD. This finding is consistent with recent evidence; a meta-analysis showed that lower eGDR was significantly associated with an increased risk of MACCE in non-diabetic individuals. 46 This finding suggests that for MASLD patients, strategies for monitoring IR may be more effective in non-diabetic populations than in diabetic populations, and early IR monitoring should be considered for individuals with normal glycemia and additional CVD risk factors. Our study also found that the significant association between eGDR with total-CVD, CHF, and stroke was more pronounced in individuals with low PIR. This is consistent with previous research indicating an increased risk of cardiovascular events and death in low-income populations. This may be attributed to factors such as a lack of education/health literacy, unhealthy lifestyles, inability to afford medications, and decreased accessibility to preventive medical services among low-income populations, which reduce their ability to resist metabolic and vascular damage and amplify the impact of IR on adverse outcomes. 52 Furthermore, the high prevalence and mortality of CVD in low-income groups may also be attributed to psychosocial stressors and coping behaviors, such as substance or alcohol abuse. Therefore, exploring targeted interventions for low-income patients with MASLD—such as lifestyle modifications, enhanced health education, and pharmacological treatments to improve insulin sensitivity—may provide valuable insights into reducing mortality in this high-risk population.
In contrast to prior investigations, this study is one of the first to demonstrate that the eGDR outperforms other widely used IR surrogates in predicting CVD risk and long-term CVD mortality in individuals with MASLD. The superior predictive performance of eGDR underscores its potential as an effective tool for clinical risk stratification. Incorporating eGDR into the basic model notably enhanced its ability to predict total CVD and CHF risk. Furthermore, given that IR is a major factor in the progression of liver fibrosis in patients with MASLD and a driver of CVD, we conducted a mediation analysis to explore the role of liver fibrosis in the relationship between eGDR and CVD, revealing that liver fibrosis partially mediates this association. This finding highlights the complex interaction between IR, liver fibrosis, and MASLD, leading to increased cardiovascular risk. In IR, hepatic selective IR can persistently promote hepatic lipid accumulation, thereby enhancing lipotoxicity, oxidative stress, and inflammatory signaling, activating hepatic stellate cells, and accelerating extracellular matrix deposition, thus exacerbating liver fibrosis. 53 Once fibrosis occurs, systemic low-grade inflammation and endothelial dysfunction are amplified, and a prothrombotic environment may emerge due to coagulation/fibrinolysis dysregulation and increased platelet reactivity. Previous studies have shown that hepatic fetoglobulin A can enhance platelet activation and promote thrombus formation in vivo through the TLR-4 signaling pathway. 54 In addition, altered hepatic factor secretion and gut-hepatic axis disorders may further exacerbate cardiovascular metabolic inflammation and vascular damage, thereby jointly increasing the risk of CVD.55,56 Therefore, interventions that slow the progression of fibrosis may mitigate the harmful cardiovascular effects of IR.
IR represents a central mechanism underlying multiple cardiometabolic disturbances, including abnormal glucose metabolism, obesity, hyperglycemia, dyslipidemia, and hypertension, which are included in the formulas of the eGDR, HOMA-IR, METS-IR, and TyG-BMI. Fasting blood glucose levels primarily reflect hepatic IR. FPG levels primarily reflect hepatic IR. Reduced hepatic insulin secretion and IR lead to excessive glucose production in the liver, resulting in fasting hyperglycemia. 57 Central obesity caused by increased WC is a typical state of IR and is closely associated with CVD and adverse cardiovascular events. 58 Previous studies have shown that a lower eGDR index is significantly associated with a gradual decline in the myocardial function efficiency index, which is an independent predictor of adverse cardiovascular events and heart failure. 59 Furthermore, eGDR has been found to be a suitable indicator of cardiac insulin sensitivity. 60 Meanwhile, disruption of insulin signaling initiates a cascade of metabolic derangements and vascular injury, manifested as endothelial dysfunction, vascular smooth muscle proliferation, increased collagen deposition, mitochondrial oxidative stress, and systemic low‑grade inflammation.9,61 Accumulating evidence indicates that these extrahepatic effects of IR markedly elevate the risk of CVD in individuals with MASLD. Mechanistically, IR diminishes insulin’s biological activity, particularly in adipose tissue and the liver. The compensatory hyperinsulinemia suppresses lipogenesis while promoting lipolysis, resulting in excess FFAs release. This process stimulates the synthesis of TG and cholesterol esters, driving very‑low‑density lipoprotein accumulation and reducing HDL levels, thereby facilitating atherogenesis.62,63 Concurrently, IR enhances the generation of advanced glycation products and free radicals, leading to reduced endothelial nitric oxide production and increased reactive oxygen species, which collectively impair vascular relaxation and contraction, producing peripheral endothelial dysfunction. 64 Moreover, IR‑induced chronic low‑grade inflammation further damages vascular smooth muscle cells and endothelial cells.65,66 Together, these processes accelerate vascular atherosclerosis and functional deterioration, substantially heightening CVD risk. Recognizing IR as a shared pathogenic driver of MASLD and CVD highlights the value of monitoring IR to facilitate early cardiovascular prevention in MASLD, ultimately reducing the associated disease burden.
Limitations
However, our study still has some limitations. First, the data of this cross-sectional study were derived from NHANES, which limits our ability to establish causal relationships between the corresponding indicators and outcomes, and also limits our understanding of how the dynamic changes of these biomarkers affect prognosis. Although we have adjusted for possible confounders, the possibility of residual confounders still exists. Secondly, SLD is diagnosed by USFLI ⩾30 rather than histological or ultrasound assessment, but it is not the best tool for diagnosing SLD. Future research should prioritize the use of gold standard methods, such as histologically confirmed liver biopsy or standardized ultrasound protocols. Thirdly, the collection of CVD outcomes based on self-reporting may lead to reporting bias. Furthermore, the lack of detailed medication data may affect the estimation of IR. Another limitation of our study is that a considerable proportion of samples were excluded from the original dataset, mainly due to missing data or failure to meet the inclusion criteria. This high exclusion rate may introduce selection bias and compromise the external validity of our findings to some extent. Finally, because the study population consisted of adults in the United States, careful consideration is needed when applying the findings to other populations. Despite acknowledging these limitations, our study provides valuable evidence on the association between IR and the risk of CVD and survival outcomes in individuals with MASLD.
Conclusion
This study highlights that multiple IR markers, especially eGDR, showed significant associations with CVD risk and CVD mortality in US adults with MASLD, and may provide superior predictive ability compared with other IR markers such as TyG-BMI, HOMA-IR, and METS-IR. Further studies are needed to validate these findings in different populations and to evaluate the full potential of eGDR in clinical practice.
Supplemental Material
sj-docx-1-tae-10.1177_20420188261435033 – Supplemental material for Association of insulin resistance-related indicators with cardiovascular disease and cardiovascular disease mortality in individuals with MASLD: a population-based study
Supplemental material, sj-docx-1-tae-10.1177_20420188261435033 for Association of insulin resistance-related indicators with cardiovascular disease and cardiovascular disease mortality in individuals with MASLD: a population-based study by Xiwen Yang and Wei Peng in Therapeutic Advances in Endocrinology and Metabolism
Footnotes
Acknowledgements
The authors express their gratitude to the participants and staff of the NHANES for their invaluable contributions to this study.
Declarations
Ethics approval and consent to participate
This research analyzed data downloaded from the National Health and Nutrition Examination Survey public database. The National Center for Health Statistics Ethics Review Committee granted ethics approval. The methods performed in this study were in accordance with relevant guidelines and regulations (Declaration of Helsinki). All individuals signed informed consent before participating in the study.
Consent for publication
Not applicable.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Beijing Bethune Charitable Foundation (GDZL002); 1·3·5 project for disciplines of excellence–Clinical Research Fund, West China Hospital, Sichuan University (2025HXFH015).
Competing interests
The authors declare that there is no conflict of interest.
Availability of data and materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
