Abstract
Objective
Evidence on the impact of the systemic immune-inflammation index on mortality risk among individuals with circadian syndrome is scarce. This study aimed to examine the association of the systemic immune-inflammation index with all-cause mortality, cardiovascular disease mortality, and cancer mortality among individuals with circadian syndrome.
Methods
We included adult participants with complete information on systemic immune-inflammation index and circadian syndrome from the National Health and Nutritional Examination Survey 2005–2018. Weighted Cox proportional hazards models and restricted cubic splines were used to estimate hazard ratios and 95% confidence intervals for all-cause, cardiovascular disease, and cancer mortality.
Results
This cohort study included 6010 participants in the analysis (mean (SD) age, 57.97 (15.40) years; 3132 participants aged ≥60 years; 2804 men). Over a median follow-up of 8.25 years, 1280 deaths were recorded, comprising 366 deaths from cardiovascular disease and 278 from cancer. Elevated systemic immune-inflammation index levels were strongly predictive of increased mortality risk relative to lower levels, with the multivariable-adjusted hazard ratios of 1.46 (95% confidence interval: 1.17–1.82) for all-cause mortality (p for trend < 0.001), 2.23 (95% confidence interval: 1.56–3.18) for cardiovascular disease mortality (p for trend < 0.001), and 1.03 (95% confidence interval: 0.70–1.50) for cancer mortality (p for trend = 0.791). We revealed evident nonlinear associations between the natural logarithm of systemic immune-inflammation index and all-cause (p < 0.001), cardiovascular disease (p = 0.009), and cancer (p < 0.001) mortality.
Conclusion
We demonstrated that higher systemic immune-inflammation index levels may be associated with an increased risk of all-cause and cardiovascular disease mortality among individuals with circadian syndrome. There was no evident association between systemic immune-inflammation index and the risk of cancer mortality.
Keywords
Introduction
Circadian syndrome (CircS) encompasses a range of health problems and symptoms resulting from disturbances in the biological clock. It includes a spectrum of chronic diseases such as obesity, hypertension, dyslipidemia, type 2 diabetes, depression, sleep disorders, and nonalcoholic fatty liver disease, all of which are closely associated with circadian rhythms. 1 The prevalence of CircS among Chinese adults is 39.0%, making it a stronger predictor of cardiovascular disease (CVD) than metabolic syndrome (MetS). 2 Chronic circadian disruption, which may be caused by shift work or irregular light exposure, contributes to cardiometabolic disorders, 3 dementia, 4 systemic inflammation, lung cancer, 5 sleep disturbances, cognitive impairments, and affective disorders. Given the significant association between circadian rhythm disruption and various diseases, it is imperative to mitigate the disease burden of CircS.
Studies have shown that the occurrence of CircS and its symptoms is correlated with inflammatory responses, including elevated levels of cytokines such as C-reactive protein and interleukin-6 (IL-6).6,7 Additionally, sleep disturbances exacerbate inflammation through the activation of nuclear factor-κB. 8 Thus, these findings suggest a relationship between CircS and systemic inflammation.
The systemic immune-inflammation index (SII), a biomarker combining neutrophil, lymphocyte, and platelet counts, provides an integrated measure of inflammatory responses and immune status. This index may accurately reflect the chronic, systemic inflammation underlying CircS progression. Recent studies have elucidated that SII is closely linked to a multitude of diseases and is indicative of unfavorable prognoses, such as hepatocellular carcinoma, 9 lung cancer,10,11 and gastric cancer. 12 It can predict the risk of certain adverse cardiovascular events.13,14 In addition, higher SII levels have been associated with an increased likelihood of MetS, hyperglycemia, reverse-dipper hypertension, and cerebrovascular diseases.15–17 CircS is associated not only with the fundamental etiology contributing to MetS but also with its related comorbidities. 18 Exploring the role of SII in CircS-related mortality may provide innovative perspectives for risk stratification.
There are currently no studies assessing the association between SII and the risk of mortality. Consequently, this study aimed to examine the association between SII and mortality risk in individuals with CircS using a nationally representative sample of US adults. Our findings could guide the development of targeted anti-inflammatory interventions and improve risk management strategies in this high-risk population.
Methods
Study design and participants
We utilized data from the National Health and Nutrition Examination Survey (NHANES), which employs a multistage, probability-based sampling framework to obtain nationally representative estimates of health parameters among the civilian, noninstitutionalized population. All participants provided written informed consent upon enrollment (https://www.cdc.gov/nchs/nhanes/index.html). This study was conducted in accordance with the principles of the Declaration of Helsinki. The reporting of this study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 19
This study extracted data from adults aged 20 years or older who participated in seven NHANES cycles (2005–2018). The study flowchart is presented in Figure 1. Exclusion criteria were as follows: (a) age <20 years or pregnancy; (b) no diagnosis of CircS; and (c) missing data on SII, covariates, and mortality status. Of the 70,190 participants, 6010 were diagnosed with CircS.

The flowchart of participant selection.
CircS and SII definition
CircS was defined as the presence of four or more of the following seven components 20 : (a) increased waist circumference (≥102 cm in men and ≥88 cm in women); (b) hypertension (systolic blood pressure ≥130 mmHg and/or diastolic blood pressure ≥85 mmHg) or current drug treatment for hypertension; (c) low high-density lipoprotein cholesterol (HDL-C) level (<40 mg/dL in men and <50 mg/dL in women) or current drug treatment for dyslipidemia; (d) high triglyceride level (≥150 mg/dL) or existing drug treatment for dyslipidemia; (e) elevated fasting glucose level (≥100 mg/dL) or taking medication for hyperglycemia; (f) self-reported short sleep duration, less than 6 h per day; and (g) depression, a score of five or higher on the Patient Health Questionnaire.
SII was computed based on lymphocyte, neutrophil, and platelet counts measured using the Uni Cel DxH 800 analyzer. According to previous research,
9
the formula was set as follows:
Mortality ascertainment
Follow-up and mortality status were determined by linking NHANES data to the National Death Index records, which were available until 31 December 2019. Cause-specific death was classified using the International Classification of Diseases, Tenth Revision (ICD-10). All-cause mortality was defined as death from any cause. Additionally, we delineated CVD mortality (I00–I09, I11, I13, I20–I51, or I60–I69) and cancer mortality (C00–C97).
Covariate assessment
Sociodemographic and lifestyle data were collected using standardized questionnaires. Variables collected in this study included sex (male or female), race (Mexican American, non-Hispanic Black, non-Hispanic White, other Hispanic, or other races), age (≤39, 40–59, or ≥60 years), educational level (lower than high school, high school, or college graduate or above), family poverty-to-income ratio (PIR ≥ 1, not poor; PIR < 1, poor), smoking status (never, former, or current smoker), alcohol consumption status (yes or no), body mass index (BMI) (underweight (<18.5 kg/m2), normal weight (18.5–24.9 kg/m2), overweight (25–29.9 kg/m2), or obese (≥30 kg/m2)), and physical activity (inactive, moderate, vigorous, or both moderate and vigorous). In addition, the following continuous variables were assessed in cases where participants provided baseline blood samples: lymphocyte count, neutrophil count, platelet count, red blood cell (RBC) count, hemoglobin level, mean platelet volume, aspartate aminotransferase (AST) level, alanine aminotransferase (ALT) level, uric acid (UA) level, serum potassium level, serum sodium (Na) level, serum creatinine level, fasting blood glucose level, triglyceride level, low-density lipoprotein cholesterol level, and HDL-C level. Blood pressure and waist circumference were measured during physical examination. Information regarding congestive heart failure, myocardial infarction, angina pectoris, coronary heart disease, and tumor history was also collected from self-reported questionnaires.
Statistical analysis
Continuous variables were expressed as mean ± standard deviation, while categorical variables were presented as frequencies and percentages. Due to the right-skewed distribution of SII, the data were log-transformed prior to statistical analysis. We computed the the natural logarithm of SII (lnSII) for each participant and subsequently categorized them into four quartiles. The first quartile of lnSII (Q1) was set as the reference.
Cox proportional hazards regression models were employed to calculate hazard ratios (HRs) and 95% confidence intervals (CIs), exploring the associations between SII and the risk of all-cause mortality, CVD mortality, and cancer mortality. Person-time was calculated from the NHANES interview date to either the date of death or the end of follow-up, whichever occurred first. The following three models were constructed: Model 1 was the crude model; Model 2 was adjusted for age, sex, and race; and Model 3 was adjusted for age, sex, race, educational level, PIR, alcohol consumption status, smoking status, physical activity, congestive heart failure, coronary heart disease, angina pectoris, myocardial infarction, stroke, and cancer.
To investigate the dose–response association between SII and various mortality outcomes, we conducted a 4-knot (at the 5th, 35th, 65th, and 95th percentiles) restricted cubic spline regression with the multivariable adjustments specified in Model 3. The likelihood ratio test was used to evaluate nonlinearity. Subgroup analysis was performed using multiple stratification variables, including age, sex, race, physical activity, BMI, CVD, and cancer. We evaluated p-values for interaction in these subgroups. Statistical software (R version 4.1.1) was used for performing statistical analyses. A two-tailed p-value of <0.05 was considered to indicate statistical significance.
Results
Baseline characteristics
This study included 6010 individuals diagnosed with CircS. The mean age of the participants was 57.97 years, with an SD of 15.40; 3132 participants were aged 60 years or older, constituting 52.11% of the entire cohort. The sample comprised 2804 (46.67%) men and 3206 (53.33%) women. The ethnic distribution was as follows: 846 (14.08%) individuals were Mexican American, 1468 (24.43%) were Non-Hispanic Black, 2795 (46.51%) were Non-Hispanic White, and 901 (14.99%) belonged to other ethnic groups. The baseline characteristics of all participants with CircS, stratified by quartiles of lnSII levels, are shown in Table 1. The quartile cutoffs were as follows: Q1 (n = 1505): lnSII ≤ 5.82; Q2 (n = 1502): 5.83 ≤ lnSII ≤6.16; Q3 (n = 1504): 6.17 ≤ lnSII ≤ 6.50; and Q4 (n = 1499): lnSII > 6.51.
Baseline characteristics of participants with CircS according to quartiles of lnSII in the NHANES from 2005 to 2018.
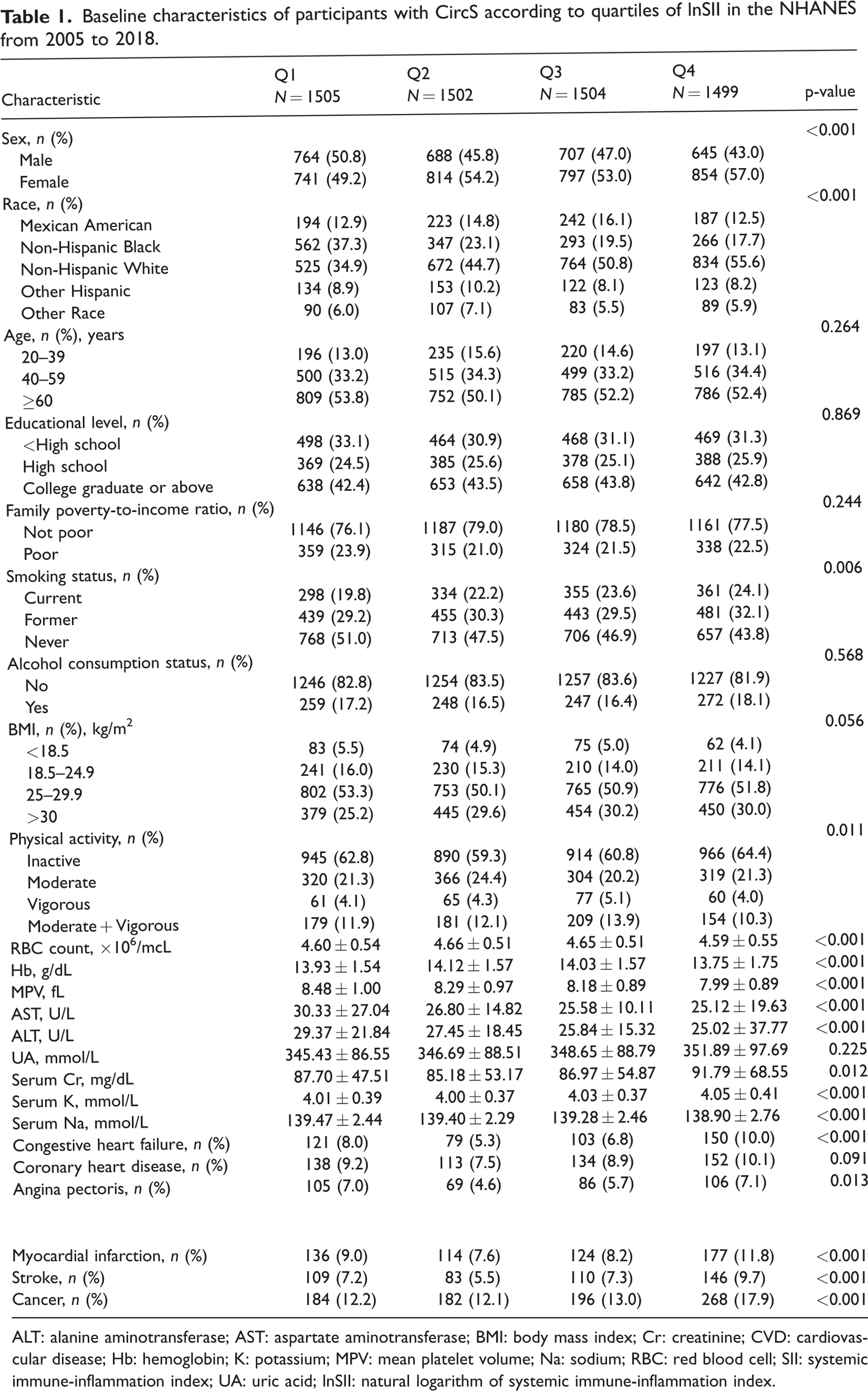
ALT: alanine aminotransferase; AST: aspartate aminotransferase; BMI: body mass index; Cr: creatinine; CVD: cardiovascular disease; Hb: hemoglobin; K: potassium; MPV: mean platelet volume; Na: sodium; RBC: red blood cell; SII: systemic immune-inflammation index; UA: uric acid; lnSII: natural logarithm of systemic immune-inflammation index.
Participants with higher SII were more likely to be female, Non-Hispanic White, and physically inactive; had lower RBC count, mean platelet volume, AST level, ALT level, and Na level; and had higher levels of serum potassium. Moreover, these participants were more predisposed to conditions including congestive heart failure, angina pectoris, myocardial infarction, stroke, and cancer (all p < 0.05).
Relationship between SII and all-cause, CVD, and cancer mortality
Crude and fully adjusted associations between lnSII and all-cause, CVD, and cancer mortality are presented in Table 2. Over a median follow-up period of 8.25 years, a total of 1280 deaths were recorded, including 366 deaths from CVD and 278 from cancer.
Hazard ratios for all-cause, CVD, and cancer mortality among participants with CircS in the NHANES from 2005 to 2018.

Model 1: unadjusted.
Model 2: adjusted for age, sex, and race.
Model 3: adjusted for age, sex, race, educational level, family poverty-to-income ratio, alcohol consumption status, smoking status, physical activity, congestive heart failure, coronary heart disease, angina pectoris, myocardial infarction, stroke, and cancer.
Regarding all-cause mortality, the HR for the fourth quartile in Model 1 was 1.38 (95% CI: 1.10–1.72) compared with the reference group (p for trend <0.001). After multivariable adjustment in Model 3, the HR for all-cause mortality in the fourth quartile was 1.46 (95% CI: 1.17–1.82) relative to the reference group (p for trend < 0.001). Regarding CVD mortality, the HR in the fourth quartile was 1.99 (95% CI: 1.42–2.78) compared with the reference group in Model 1 (p for trend < 0.001). After multivariable adjustment in Model 3, the HR in the fourth quartile was 2.23 (95% CI: 1.56–3.18) compared with the reference group (p for trend < 0.001). With respect to cancer mortality, the HR in the fourth quartile was 1.03 (95% CI: 0.70–1.52) compared with the reference group (p for trend = 0.729). After multivariable adjustment in Model 3, the HR for cancer mortality in the fourth quartile was 1.03 (95% CI: 0.70–1.50; p for trend = 0.791).
The dose–response associations between lnSII and all-cause, CVD, and cancer mortality are shown in Figure 2. The analysis revealed evident nonlinear associations between lnSII and all-cause (p < 0.001), CVD (p = 0.009), and cancer (p < 0.001) mortality. We also generated survival curves for all-cause, cardiovascular, and cancer mortality stratified by SII among patients with CircS (Figure S1).
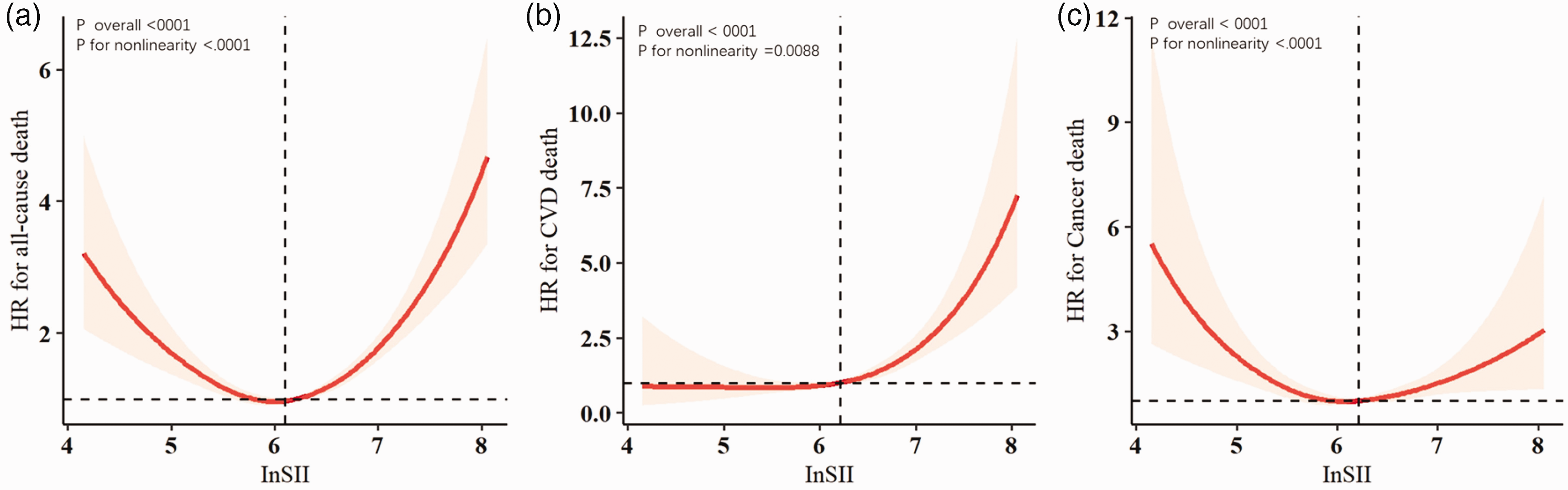
Association between InSII and all-cause (a), cardiovascular disease (b), and cancer (c) mortality in individuals with CircS.
Subgroup analysis
To further investigate the roles of potential confounders in the associations between SII and all-cause mortality among individuals with CircS, we divided the participants into several subgroups stratified by age, sex, race, physical activity, BMI, CVD, and cancer (Figure 3). Notably, elevated SII was associated with increased all-cause mortality, specifically in older adults, those with low BMI, and individuals with CVD. Nevertheless, no significant interactions were observed between SII and any stratified variables related to the risk of all-cause mortality (all p for interaction >0.05). Similarly, there were no significant interactions observed in the subgroup analysis for CVD mortality or cancer mortality (all p for interaction >0.05, Figures 4 and 5).

Subgroup analysis of the association between SII and all-cause mortality.

Subgroup analysis of the association between SII and cardiovascular disease mortality.

Subgroup analysis of the association between SII and cancer mortality.
Discussion
This is the first study to examine the association between SII and the risk of all-cause, cancer, and CVD mortality in individuals with CircS. Our findings indicate that elevated SII levels are associated with an increased risk of all-cause and CVD mortality. After adjusting for multiple confounding variables, this association remained significant. We also identified a nonlinear relationship between SII and the risk of all-cause, cancer, and CVD mortality, which remained significant even after controlling for covariates.
In the subgroup analysis, we demonstrated that elevated SII is associated with increased all-cause mortality, particularly among older adults, individuals with low BMI, and those with CVD. Advancing age is associated with progressive increases in chronic systemic inflammation, which may substantially contribute to adverse prognostic outcomes.21,22 The precise molecular mechanisms warrant further in-depth investigation.
Several critical factors contribute to the development of CircS components, which may partially elucidate the relationship between SII and CircS outcomes. First, systemic inflammation may activate microglial cells and astrocytes in brain regions implicated in sleep and circadian regulation. Activation of glial cells can induce the secretion of proinflammatory cytokines, nitric oxide, and gliotransmitters, which may profoundly influence the expression of pivotal circadian regulators. 23 Previous studies have reported elevated levels of inflammatory markers, including IL-1β, tumor necrosis factor-alpha (TNF-α), IL-6, and IL-17, in the periphery after episodes of acute or chronic sleep deprivation. 24 Second, the circadian rhythm and immune system interact reciprocally. Most immune cells autonomously express clock-regulating genes, which are crucial for orchestrating immune cell phagocytic activity, migration, metabolism, signaling pathway activation, and inflammatory responses. 25 Third, insulin resistance is a key factor in the etiology of the cardiometabolic cluster. CLOCK gene mutations, disrupted sleep, shift work, and social jet lag may significantly contribute to insulin resistance, a primary determinant in the development of type 2 diabetes mellitus and a major factor in morbidity and mortality. 26 Abnormal triglyceride levels and hyperglycemia are also linked to inflammation, contributing to CircS. 27
The circadian rhythm regulates physiological functions through gene expression and immune activity, and its disruption can lead to systemic inflammatory responses and chronic inflammatory conditions. The circadian clock regulates various physiological functions, including the immune system, sleep regulation, and metabolism. Circadian regulation relies on periodic changes in gene expression driven by the transcriptional activity of the CLOCK/BMAL1 complex. 28 Circadian rhythm regulators in innate immune cells modulate the temporal patterns of nuclear factor κB (NF-κB) and TNF-α activities as well as the response to lipopolysaccharide-induced toxic shock, initiating inflammatory responses that may progress into chronic inflammatory conditions. 29 Circadian disruption promotes systemic inflammatory responses through the activation of the hypothalamic–pituitary–adrenal axis, sympathetic nervous system overactivity, or gut microbiota imbalance. 30 Proinflammatory factors such as IL-6 and TNF-α interfere with the expression of core clock genes, including CLOCK and PER/CRY, thereby disrupting the circadian rhythm. 31
From a clinical and public health perspective, we should implement regular monitoring of SII and circadian rhythm indicators in high-risk populations (e.g. night shift workers and individuals with chronic inflammation). Additionally, integrating SII and CircS assessments as a novel risk prediction tool may enable early intervention strategies. Intervention strategies include exploring the potential benefits of anti-inflammatory medications in patients with elevated SII levels, optimizing circadian rhythm–aligned medication timing (e.g. morning glucocorticoid administration), and evaluating the effects of lifestyle modifications—such as adjusted light exposure, regular sleep–wake cycles, and time-restricted eating—on improving CircS and reducing SII. The government should incorporate circadian rhythm protection into occupational health policies (e.g. reducing shift work schedules that disrupt circadian rhythms) and promote public health education to emphasize the importance of circadian rhythm alignment for overall well-being.
Limitations
This study has several limitations. First, the definition of CircS was subjective, as it relied on questionnaire data rather than objective biomarkers. SII values calculated from single time point measurements of lymphocytes, neutrophils, and platelets may lack sensitivity to chronic inflammatory status and are subject to inherent confounding bias in retrospective studies. Second, the NHANES study employs a cross-sectional design, which does not allow for establishing causal relationships between SII and CircS; thus, further experimental studies are required for validation. Additionally, the database predominantly represents the US population, limiting generalizability across race/ethnicity, geographical regions, and disease spectra. Third, although the National Death Index is highly reliable for ascertaining vital status, its accuracy in classifying CVD mortality remains limited. 32 Fourth, our results may have been influenced by confounding variables affecting inflammation, including corticosteroid use, autoimmune disorders, and asthma.
Conclusion
Our findings demonstrated that elevated SII levels were significantly associated with increased all-cause and CVD mortality, with these associations remaining robust even after multivariable adjustment. These observed associations warrant confirmation in large-scale, multi-institutional cohort studies with longitudinal designs.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251384072 - Supplemental material for Systemic immune-inflammation index and mortality among individuals with circadian syndrome
Supplemental material, sj-pdf-1-imr-10.1177_03000605251384072 for Systemic immune-inflammation index and mortality among individuals with circadian syndrome by Yunyan Lu and Tian Lan in Journal of International Medical Research
Footnotes
Acknowledgements
The data used in this study were sourced from the NHANES. We thank all the staff of and participants in the NHANES for their contribution.
Author contributions
Tian Lan and Yunyan Lu designed the study, extracted the data, performed the analyses, interpreted the evidence and wrote the manuscript. Tian Lan revised the article. All authors agreed to be accountable for the reported work.
Declaration of conflicting interests
The authors have declared that no competing interest exists.
Ethics approval and consent to participate
This study did not involve human embryos, gametes, and stem cells. This study received exemption from the Ethics Committee of The First People’s Hospital of Xiaoshan District as no patient, physician, or hospital identifiers were examined.
Funding
No funding was received.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

