Abstract
Understanding the hypothalamic–pituitary–gonadal (HPG) axis is essential for grasping human responses under extreme physiological and pathological conditions. The HPG axis regulates reproductive and gonadal hormone activities and significantly impacts the body’s response to acute and chronic illnesses. This review explores the fundamental functions of the HPG axis, modifications under critical conditions, and impacts on disease progression and treatment outcomes. In addition, it examines interactions between sex hormones and biomolecules like cytokines and gastrointestinal microorganisms, highlighting their roles in immune response regulation. Clinically, this knowledge can enhance patient prognoses. The review aims to provide a comprehensive framework, based on existing research, for understanding and applying the functions of the HPG axis in managing critical diseases, thereby broadening clinical applications and guiding future research.
Plain language summary
Our review systematically analyzes the alterations in sex hormone levels during critical illness, delves into the underlying mechanisms, and evaluates the current clinical applications of sex hormone therapies in critical care. In females, critical illness is associated with the reduction of follicle-stimulating hormone (FSH) and luteinizing hormone (LH) levels, alongside the increase in testosterone, estradiol, and progesterone levels. This elevation in estrogen is attributed to two primary factors: (1) a by-product of adrenal activation, and (2) increased aromatization, which is a chemical structural change that converts testosterone into estradiol. The rise in testosterone and progesterone is predominantly due to adrenal and peripheral tissue activity. In males, there is the decrease in FSH, LH, and testosterone levels, coupled with increased levels of estradiol and progesterone. The decline in testosterone is primarily due to: (1) chronic low-level endotoxin exposure and inflammation impairing androgen synthesis, and (2) increased aromatization in peripheral tissues. The rise in progesterone is attributed to stress-induced stimulation of the adrenal glands. These hormonal changes are closely linked to the patient’s critical condition, with hormone levels generally returning to reference ranges as the condition improves.
Keywords
Introduction
Critical diseases, including a series of traumas such as sepsis and severe traumatic brain injury (TBI) in this review, create significant adversities that challenge patients’ overall health, especially in their endocrine system. The hypothalamic–pituitary–gonadal (HPG) axis, in particular, crucially responds to and reflects endocrine stress responses under severe physiological strains. The typical functioning of the HPG axis involves coordination of gonadal hormone secretion via pulsatile secretion of the gonadotropin-releasing hormone (GnRH). However, under extreme conditions like severe infections, multiple organ failures, and long-lasting inflammatory responses, the functioning of this axis tends to be disrupted, which is closely linked with both the severity of the disease and the prognosis of the patient.1–4
Various factors, such as elevated levels of stress hormones like cortisol, metabolic disorders, endotoxemia, and cytokine storms, might cause the inhibition of HPG axis inhibition in critically ill patients. These changes could influence hormone levels and even worsen the patient’s condition or recovery process by interfering with immune and inflammatory pathways. It has been proven that variations in sex hormones, such as increased or decreased levels of estrogen and testosterone, can serve as indicators for disease progression and have direct impacts on treatment responses and disease prognoses.
The review provides an exhaustive analysis of the structure and functionality of the HPG axis, its regulatory mechanisms, and perturbations under critical conditions. We will further elucidate the role of sex hormones in immune regulation, inflammatory responses, and metabolic processes, as well as their interactions with other notable biomolecules such as cytokines and gastrointestinal microbiota. These collectively influence disease progression and treatment outcomes.
Through this comprehensive exploration, we aim to enhance the understanding of the critical role of the HPG axis and sex hormones in critically ill patients and reveal their potential applications in clinical treatment strategies, particularly in devising targeted hormone regulation treatments. This knowledge is crucial for optimal management of critically ill patients, which will not only improve prognoses but also increase survival rates.
The HPG axis and sex hormones under normal condition
The HPG axis, one of the three classical feedback mechanisms, functions under normal conditions through a series of procedures involving multiple hormones. The GnRH, typically characterized by its pulsatile nature, is secreted by the hypothalamic arcuate nucleus. Following its release, it stimulates anterior pituitary gonadotropin cells to release luteinizing hormone (LH) and follicle-stimulating hormones (FSH) via the pituitary portal vein system.5–7
In males, LH primarily prompts testicular Leydig cells to generate androgens, while FSH collaboratively works with testosterone to act on Sertoli cells, ultimately accelerating sperm formation. FSH also has a role in activating the aromatase in Sertoli cells, which enhances the conversion of testosterone to estradiol. This induces a reduction in the pituitary gland’s responsiveness to GnRH besides locally modulating testosterone secretion. 7 Furthermore, inhibin, another hormone produced by the testes, exerts a potently inhibitory effect on FSH secretion within the pituitary gland, while the same physiological dose of inhibin has less effect on LH. 8 Other hormones, such as leptin, can promote GnRH secretion, whereas prolactin, corticotropin-releasing hormone (CRH), and various cytokines tend to inhibit GnRH release 9 (Figure 1).

Physiological feedback of the hypothalamic–pituitary–gonadal axis. The GnRH, secreted by the hypothalamus, stimulates anterior pituitary gonadotropin cells to release LH and FSH. In males, LH prompts testicular Leydig cells to generate androgens, while FSH works with testosterone to act on Sertoli cells, accelerating sperm formation. FSH also has a role in activating the aromatase in Sertoli cells. Inhibin, another hormone produced by the testes, exerts an inhibitory effect on FSH secretion. In females, estrogen, secreted by granulosa cells, has a feedback effect on the hypothalamus and pituitary gland, including negative feedback and positive feedback before ovulation. LH stimulates the synthesis of androgens by theca cells during the follicular phase, fostering the maturation and ovulation of oocytes. During the luteal phase, LH bolsters the synthesis and secretion of progesterone. Concurrently, FSH fosters the conversion process from androgens to estrogen in the ovaries.
In females, LH stimulates the synthesis of androgens by theca cells during the follicular phase, hence fostering the maturation and ovulation of oocytes. Conversely, during the luteal phase, LH retains the luteal functionality and bolsters the synthesis and secretion of progesterone. This hormonal interaction plays a crucial role in maintaining a regular menstrual cycle and a normal pregnancy. Concurrently, FSH fosters the conversion process from androgens to estrogen in the ovaries, leading to a sustained secretion that propels the growth, maturation, and expulsion of follicles 7 (Figure 1).
The HPG axis and sex hormones during critical illness
Currently, there are variations in defining critical illness across different diseases. This review defines critical illness as a clinical condition characterized by instability, the potential involvement of one or more organs, posing a threat to the patient’s life due to one or more diseases. Critical patients are those with unstable clinical conditions, potential involvement of one or more organs, and requiring respiratory and circulatory support.
At the outset of critical illness, the body, in a self-protective mode, responds to acute stress by heightening the levels of hormones such as adrenaline and glucocorticoids. However, persistent high stress can inhibit the HPG axis, suggestive of the body’s adaptation toward reducing energy expenditure and preserving essential organs. 10 This indicates that the long-term inhibition of the HPG axis in severe sickness conditions results from not only hormonal depletion but also an active response from the body itself.
Among studies focusing on hormone levels,11–27 most reveal that FSH and LH levels are lower in critically ill or deceased patients during the early and middle stages of stress compared to healthy or surviving controls. By contrast, estrogen levels are higher in critically ill or deceased patients, regardless of sex.20,21,25,26,28,29 While male patients in critical conditions have lower testosterone levels than those in the control group,13,25,26,30 female patients in similar conditions tend to have higher levels.17,31,32 Some studies show that after brain trauma, female patients exhibit decreased gonadal function, characterized by diminished FSH, LH, and estradiol. 3 However, most research indicates that the levels of estrogen in critically ill patients, regardless of sex, are significantly higher than in the control groups.14–19,21,28,30 It is worth noting that a prospective cohort study of septic patients indicated that biologically active estrogen levels were higher in non-survivors than in survivors, 19 suggesting that the heightened levels of estrogen are not due to a decrease in hormone-binding proteins (Table 1). Regardless of sex, a sustained decrease in testosterone levels33–35 and/or an increase in estrogen levels within 48 h of stress14,16,18 significantly correlate with an unfavorable prognosis.
Overview of clinical studies reporting changes in sex hormone levels in critically ill patients.

COPD, chronic obstructive pulmonary disease; FSH, follicle-stimulating hormone; IQR, interquartile range; LH, luteinizing hormone.
There are also dynamic changes in hormone levels during patient treatment
Throughout the disease course, patients’ hormone levels undergo dynamic changes, with both similarities and differences observed between males and females. Regardless of sex, FSH and LH levels significantly decrease following injury, while estradiol and progesterone levels progressively increase. However, testosterone shows distinct patterns: in males, testosterone levels decline after injury but may return to normal as the condition improves, whereas in females, testosterone levels initially rise after injury and gradually decrease during recovery. Then we provide a detailed analysis of the sex hormone changes.
Males
In response to stressful events like injury or surgery, estrogen concentrations accelerate above the normal physiological range, then subsequently decrease gradually and essentially normalize once the patient’s condition improves. 17 Progesterone levels exceed that of the healthy control group within 48 h post-injury. As time progresses, the levels of serum progesterone significantly decrease, gradually equal to the control group. 17 By contrast, the levels of testosterone, FSH, and LH rapidly decline in the initial few days, eventually nearing original levels as the patient’s condition improves. 13 However, testosterone levels fail to recover if the patient’s condition worsens.36–38 Looking at myocardial infarction patients, the critical illness treatment results in a rapid decline in FSH and LH in male patients over the initial 2 days. Hormone levels drop to the lowest point on the 4th and 5th day before gradually recuperating and are somewhat lower than pre-injury levels upon discharge when the condition significantly improves. To illustrate with coronary artery bypass grafting (CABG) male patients, FSH and LH levels fall to their lowest on the 3rd day post-surgery before gradually recovering, essentially returning to the levels of the second day after surgery by the 5th week post-surgery. This study also evaluated pituitary responsiveness to physiological doses of GnRH before and after CABG. Preoperative administration of a physiological dose of GnRH showed that neither LH nor FSH responses were suppressed postoperatively. Interestingly, the LH response was slightly higher after surgery compared to before, suggesting that FSH secretion was more sensitive to suppression by acute illness than LH. This indicated that the hypothalamus, which regulates pituitary function, may be impacted by acute illness. Furthermore, despite markedly low circulating testosterone levels, there was no significant increase in the FSH response and only a minimal increase in the LH response after CABG. This suggested that pituitary responsiveness to GnRH may be impaired. These findings implied that both the hypothalamus and the pituitary gland were affected by acute illness 39 (Figure 2(a)).
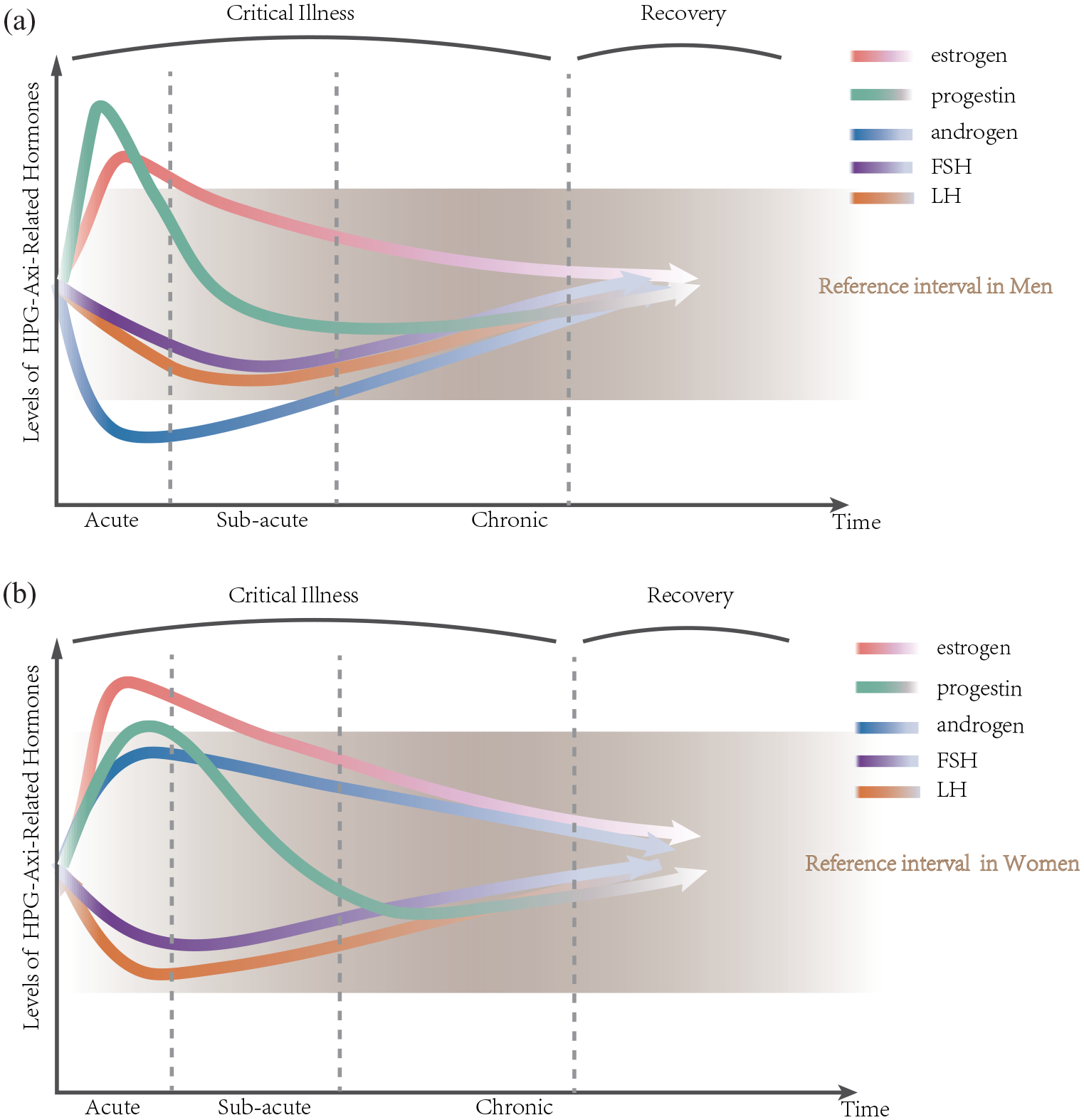
Time- and dose-dependent changes in plasma concentrations during critical illness. These figures show the dynamic changes in blood FSH, LH, estrogen, androgen, and progestin after the onset of critical illness. (a) In men, FSH, LH, and androgens are all reduced during the acute phase, and the decrease in androgens occurs earlier than FSH and LH. Androgen is even lower than the reference range. Progesterone and estrogen have increased levels, even exceeding the reference range. As the condition improves, hormone levels gradually return to their initial state. (b) In women during the acute phase, FSH and LH show a decreasing trend within the reference range, while progesterone, estrogen, and androgens increase. As the condition improves, hormone levels gradually return to their initial state.
Females
Current literature suggests that the tendency in estrogen levels is fundamentally the same for pre- and post-menopausal women in critical illness, thereby a unified description is provided in the article without requiring further subdivision. During the initial few days of intensive care unit (ICU) admission, there is an upsurge in estrogen levels, which gradually decline below the baseline levels thereafter. In studies of TBI, compared to healthy women, female patients exhibit elevated levels of estradiol on the day of injury, even exceeding the regular luteal-phase range. Progesterone levels waver between the luteal and follicular phases of the menstrual cycle. Subsequently, as time passes, estrogen and progesterone levels diminish gradually, with estrogen levels on the 6th day of observation being lower than the day of injury. Progesterone levels equate to those of women in the follicular or post-menopausal stages. 17 In contrast to male patients, female patients experience a surge in testosterone levels post-injury. As their condition stabilizes, testosterone levels gradually dwindle but consistently remain higher than the control group. 17 Unlike estrogen, progesterone, and androgens, LH, and FSH levels witness a rapid plunge after injury, steadily reverting to initial levels as the patient’s condition improves. According to the literature, LH and FSH levels drop rapidly post-injury, hitting their lowest on the 4th–6th days. As the disease condition improves, the hormone levels slightly exceed the admission levels and can be restored to admission levels post-discharge 13 (Figure 2(b)).
Regardless of sex, comparing three groups—myocardial infarction, postoperative, and brain trauma patients—postoperative patients exhibit the most rapid recovery of hormone levels. This could be attributed to the fact that elective surgery inflicts less harm on the body than the other two conditions and is considered a therapeutic “injury.” 3
Causes of sex hormone changes during critical illness
Within critically ill patients, corticotropin-releasing factor (CRF) inhibits the secretion of GnRH, thereby suppressing pituitary hormone secretion. 40 In addition, a rise in cortisol levels incites a feedback suppression of the secretion of hypothalamic and pituitary hormones, resulting in a similar HPG axis reduction, namely a drop in FSH, LH, and testosterone. 33 Furthermore, the secretion suppression of gonadotropin trials behind the diminished testosterone, 13 which further indicates that the inhibition of the gonads is more severe than the central. The treatment of critically ill patients involves drugs such as dopamine, 41 opioid drugs, 42 and cytokines like interleukin (IL)-1, 43 which could potentially inhibit LH secretion by reducing the amplitude of LH secretion pulses. Clinical studies have revealed that hypogonadism in long-term critically ill male patients is central, originating from the hypothalamic–pituitary–gonadal junction, which can only be partially counteracted via exogenous iv GnRH pulses. 26 Functional or relative adrenal insufficiency may exist in critically ill patients. Studies have discovered that elevated cortisol levels and diminished dehydroepiandrosterone sulfate levels are closely tied to critical illnesses in patients, despite both being produced by the adrenal gland and ideally should alter in harmony. But low-dose adrenocorticotropic hormone (ACTH) stimulation test and Thorn test reveal that the hypothalamic pituitary adrenal (HPA) axis was at full stimulation. Furthermore, any additional stimulation to ACTH appears relatively absent.44,45
The rise in estrogen levels is not only a by-product of adrenal activation but may also be the outcome of elevated aromatase activity catalyzed by high-level cytokines like IL-6 or tumor necrosis factor (TNF), leading to the conversion of peripheral androgens into estrogen through aromatization46,47 (Figure 3).

The mechanism of hormone changes in critically ill patients. In critically ill patients, in addition to the self-adjustment of the HPG axis, high cortisol levels incite a feedback suppression of the secretion of hypothalamic and pituitary hormones. Drugs such as dopamine, opiates, and cytokines could potentially inhibit LH secretion. LPS, TNF-a, IL-6, and IL-2 curtail testosterone secretion, possibly inhibiting GnRH and/or LH secretion by the hypothalamus and pituitary gland. The CRF, produced by the hypothalamus and peripheral tissues such as gonads and immune cells, restricts the secretion of GnRH in the hypothalamus. In addition, glucocorticoids inhibit the secretion of LH, estrogen, and progesterone. E2 amplifies the inhibition of LH pulse induced by CRF.
For male patients, the decrease in testosterone levels stems from relative adrenal insufficiency, combined with chronic low-level endotoxin exposure and inflammation, damaging androgen synthesis. 48 Experiments have suggested that external administration of endotoxins reduces testosterone production by 30% within 6 h of exposure, potentially due to cytokine damage to testicular Leydig cells. This brief exposure to endotoxins helps clarify the common phenomenon of decreased testosterone levels in males during acute infections. Furthermore, the increase in the peripheral conversion of testosterone to estradiol during critical illness leads to a further decline in testosterone levels. In addition, lipopolysaccharides (LPS), TNF-α, IL-6, CRH, and IL-2 curtail testosterone secretion by Leydig cells in various experimental models, possibly inhibiting GnRH and/or LH secretion by the hypothalamus and pituitary gland49,50 which can be explained by a reduced LH feedforward drive toward testosterone secretion coupled with heightened testosterone feedback to LH secretion. 51 Importantly, this combined hypogonadism mechanism is intensified by IL-2. 50 The increase in progesterone arises from the stress-induced stimulation of the adrenal gland. 32
For post-menopausal women in critical conditions, there is a notable decline in FSH and LH, yet an increase in testosterone, estradiol, and progesterone. At this point, the upsurge in testosterone and progesterone is entirely derived from the adrenal gland and peripheral tissues, encompassing the liver, kidneys, pancreas, peripheral T cells, macrophages, adipocytes, etc. 52 The uptick in estradiol results from the aromatization of testosterone in peripheral tissues. Studies have indicated that the rise in progesterone is more noticeably prominent than cortisol in female patients, 32 which could be attributed to variations in adrenal gland dysfunction or the responsiveness of hormone receptors to stimuli. Moreover, the interplay between the HPG and HPA axis contributes to hormonal disruptions. During stressful conditions, the CRF, produced by the hypothalamus and peripheral tissues such as gonads and immune cells, restricts the secretion of GnRH in the hypothalamus. In addition, glucocorticoids inhibit the secretion of LH, 53 estrogen, and progesterone. 54 Furthermore, research has suggested that estradiol (E2) amplifies the inhibition of LH pulse induced by CRF in rats 55 (Figure 3).
In addition, in critical or advanced stages of illness, medications such as dopamine and opioid analgesics are commonly used. These drugs may inhibit the pituitary gland and suppress gonadotropin function.42,56
Overall, gonadotropin decrease in critical illness stems centrally, influenced by the adrenal axis, cytokines, and medications. Elevated estrogen levels result from adrenal activation and increased peripheral androgen aromatization. Testosterone decline is attributed to chronic low-level endotoxin exposure, inflammatory damage hindering synthesis, and heightened peripheral aromatization. Elevated progesterone in critical states solely emanates from adrenal glands and peripheral tissues.
Interactions of sex hormones with other factors
The interplay between sex hormones and various other factors is multi-faceted. From a broader perspective, there are complex relationships between sex hormones and various diseases. In the context of tumors, androgens can induce the depletion of CD8+ T cells, resulting in accelerated tumor cell growth. 57 For chronic obstructive pulmonary disease (COPD), estradiol has been shown to significantly mitigate COPD-like lung damage in murine models. 58 Regarding Alzheimer’s disease (AD), FSH acts directly on hippocampal and cortical neurons to accelerate amyloid-β and Tau deposition and impair cognition in mice displaying features of AD. 59 In chronic kidney disease (CKD), while the incidence is higher in females, males tend to have a worse prognosis. 60 Inflammatory bowel disease activity during pregnancy is influenced by fluctuations in hormone levels. 61 In heart failure (HF), among men with chronic HF and reduced left ventricular ejection fraction, both high and low concentrations of estradiol compared with the middle quintile of estradiol are related to increased mortality. 62 Changes in estrogen metabolism and disrupted receptor expression may contribute to the pathophysiology of chronic HF.63,64 The mechanisms through which hormones impact these diseases involve various factors, including oxidative stress, inflammation, apoptosis, and cellular aging. Building on these macro-level effects, it is crucial to explore how sex hormones also interact with the immune system and microbiota at a more granular level. Further investigation into the role of sex hormones in modulating cytokine production and gut microbiota composition may offer deeper insights into their broader physiological impact.
Sex hormones and cytokines
First, the interaction between cytokines and estrogen is complex and subtle. Concerning cytokines, a reciprocity is identified with estrogen. Studies indicate that under critical conditions, cytokines may suppress the pulsatility of GnRH, thereby leading to a decrease in gonadotropin hormone levels. Investigations involving primates have found that the infusion of IL-1α suppresses gonadotropin secretion, with CRH counteracting this effect. 21
In terms of estrogen’s influence on cytokines, E2 has been shown to promote the expression of inflammatory mediators by LPS-activated tissue-resident macrophages through estrogen receptor (ER) α signaling. A study found that chronic E2 administration to ovariectomized mice significantly increased IL-1β, IL-6, and TNF-α. 65 In addition, the interferon-γ (IFN-γ) promoter region contains estrogen response elements, 66 inferring theoretically that E2 upregulates the IFN-γ production by mediating ERα, ERβ, and mERs. A study involving coronavirus disease 2019 patients substantiates this theory—estradiol levels positively correlate with IFN-γ levels. 36
Sex hormones and gut microbiota interactions
Sepsis, as a critical condition, triggers significant shifts in both the gut microbiota and sex hormone levels. During sepsis, the intestinal microbiota undergoes a collapse, transitioning from commensal to pathogenic species. 67 This dysbiosis is marked by reduced diversity and structural alterations, with a notable increase in Candida and a decrease in beneficial fungal groups.68,69 These microbiota changes are associated with worsened outcomes.70,71 Notably, the interactions between sex hormones and gut microbiota could play a critical role in the pathophysiology of sepsis.
The impact of sex hormones on gastrointestinal microbiota is worth paying attention to. First, estradiol demonstrates complex, contrasting effects on inflammation. On the one hand, β-estradiol conducts the generation of IL-12 and IFN-γ in dendritic cells, activating pro-inflammatory cytokine pathways in the process. It also prolongs B-cell survival and stimulates polyclonal B cells, increasing intestinal permeability and facilitating gut microbiota migration to the lamina propria, thereby intensifying inflammatory processes. 72 On the other hand, estrogen, or compounds with similarities, can instigate an upsurge in targeted intestinal alkaline phosphatase levels, causing a reduction in LPS production and decreased intestinal permeability, thereby reducing metabolic endotoxemia and systemic chronic inflammation.73,74 These opposing effects highlight the dual role of estradiol in modulating inflammatory responses. Moreover, studies suggest that supplementing small quantities of estrogen and progesterone to the brain can ameliorate menopause symptoms by reducing serum FSH levels and maintaining diversity within gut microbial communities in rats. 75 Second, the influence of androgens on gut microbiota is just as significant as that of estrogen. The gut microbiota in juvenile mice is generally indistinguishable between sexes. However, in adult females, the gut microbiota mirrors that of their juvenile counterparts. This suggests a significant influence of androgens on gut microbiota composition, hinting that changes in androgen levels could trigger subsequent shifts in gut microbiota. For instance, male mice display increased susceptibility to type 1 diabetes (T1DM). To counter this, modifications in the gut microbiota of mature male mice enhance their resistance against T1DM. 76 In addition, the correlation between variations in androgens and conditions like polycystic ovary syndrome further underscores the significance of the interplay between sex hormones and gut microbiota.77,78
Lastly, gut microbiota can influence the levels of estrogen, androgens, and their precursors via various mechanisms, thereby impacting the hormone levels. Importantly, via the action of β-glucuronidase secreted by microorganisms, estrogen gets metabolized from its bound state to an unbound state, which exhibits biological activity. The unbound estrogen then penetrates the bloodstream and binds to ERα or ERβ to exert biological effects.79,80 Dysbiosis in the microbiome can therefore engender estrogen imbalance, leading to various conditions such as obesity, cognitive changes, and cancer. 81 In terms of androgens, certain species of the Clostridium genus found in gut microbiota can transform glucocorticoids into androgens. 82 Animal-based studies have also highlighted that a healthy microbiome is critical for maintaining normal estrogen cycles, testosterone levels, and the reproductive function of both sexes. Mouse models of T1DM have demonstrated that colonization by commensal microbes can elevate serum testosterone levels, thereby shielding males from T1DM. Moreover, a transfer of gut microbiota from adult males to immature females induces increased testosterone and triggers metabolomic modifications in the recipients, lending strong resistance against T1DM. 83 These studies have established that alterations in gut microbiota are crucial not only for regulating the cycles of estrogen and testosterone but also hold potential value in preventing the development of certain diseases.
Role and clinical applications of sex hormones
Sex hormones, including estrogen, progesterone, testosterone, and gonadotropins (FSH and LH), play crucial roles in regulating immune responses, inflammation, and various physiological functions, particularly in critical illness and recovery. For instance, estrogen is known for its anti-inflammatory properties, but it can also promote pro-inflammatory cytokine production under certain conditions, such as septic shock. Progesterone has potent anti-inflammatory and neuroprotective effects, particularly in TBI, while testosterone plays a critical role in muscle recovery but may exacerbate certain cardiovascular and immune-related complications. Gonadotropins, such as LH and FSH, are implicated in the hormonal response to stress and critical illness. Understanding the multifaceted roles of these hormones in disease pathogenesis and recovery provides insights into their potential clinical applications, from acute resuscitation to long-term therapeutic strategies in conditions like sepsis, TBI, and cachexia.
Estrogen
The anti-inflammatory properties of estrogen are well-documented. ERs, found extensively in immune cells, 84 intersect with glucocorticoid receptors. This interaction suggests that estrogen can indirectly suppress inflammatory cytokines such as NF-κB85–87 through glucocorticoid receptors, and may also enhance the secretion of glucocorticoids by the adrenal cortex through negative feedback on the HPG axis, providing anti-inflammatory and immunosuppressive effects. Furthermore, estrogen mitigates stress-induced cardiac injury and inflammation via the GPER-1/PI3K signaling pathway. 88 Furthermore, Mendelian randomization studies have identified that estrogen can reduce markers of mild systemic inflammation such as leukocyte and granulocyte counts. 89
However, estrogen also exhibits pro-inflammatory effects, enhancing the synthesis of nitric oxide by endothelial cells, 90 which reduces peripheral vascular resistance and may exacerbate septic shock. 32 It can also stimulate cytokine production through the Akt/mTOR pathway, 91 leading to immune damage. 92 Long-term administration of E2 to ovariectomized mice in biological experiments has been found to notably increase cytokine (IL-1β, IL-6, and TNF-α) and inducible nitric oxide synthase (iNOS) mRNA abundance in TGC-induced macrophages. The release of IL-1β and IL-6 from LPS-stimulated macrophages also demonstrates the pro-inflammatory effect of E2. This suggests that estrogen enhances the ability of macrophages to produce inflammatory mediators and cytokines upon further TLR activation. 65 Consequently, early inhibition of testosterone-to-estrogen conversion using aromatase inhibitors during septic shock could prove beneficial in disease progression.
Estrogen, showing anti-apoptotic, anti-inflammatory, and antioxidant properties,2,93,94 has strong potential for therapeutic use. It can be an effective drug for acute resuscitation, with favorable impacts during emergencies such as sepsis, renal ischemia, or myocardial ischemia.94,95–99 Moreover, elevating estrogen levels can prove beneficial in rescuing fear extinction observed in post-traumatic stress disorder (PTSD), and it is recommended as an adjunctive therapy in PTSD.100,101 A study showed that hormonal contraceptives, which inhibit estrogen production, impaired fear extinction in both healthy women and female rats. However, this impairment was not permanent and could be alleviated with estrogen treatment. 102 Another study compared sexual assault victims who either received or declined emergency contraceptives immediately after the assault and found that those who took estrogen had significantly lower PTSD symptoms 6 months later 103 (Figure 4(a)).

The role of estrogen and androgen in critical illness. (a) Clinical Application of Estrogen in Critical Illness. (b) Clinical Application of Androgen in Critical Illness.
Progesterone
Progesterone levels rise during the menstrual cycle and pregnancy, demonstrating extensive anti-inflammatory and immunosuppressive effects. Primarily, progesterone signaling operates through progesterone receptors, although some signaling also involves glucocorticoid receptors. Present in diverse immune cells, including NK cells, macrophages, dendritic cells, and T cells, 104 progesterone profoundly influences immune response by dampening leukocyte activation and suppressing the immune system. 105 Abundantly distributed throughout the central nervous system in both sexes, progesterone underscores its crucial neurological role. Following TBI, progesterone modulates genes associated with inflammation and cell apoptosis pathways, thereby exerting neuroprotective effects. 106 It exhibits neuroprotective effects, regardless of whether it is related to TBI107,108 or status epilepticus. 109 These effects include impeding injury-related metabolic cascades, 110 ameliorating neurological deficits, cognitive functions, motor skills, and mitigating anxiety,111,112 infarct size, 113 edema, and neurodegeneration.114,115 Studies administering progesterone to male and female rats post-TBI have demonstrated improvements in brain edema and functional outcomes.114–116 Phase II clinical trials have further indicated its potential to reduce mortality in TBI patients. 108
Testosterone
Testosterone plays a significant role in the recovery of ICU patients, particularly in addressing hypotestosterone, commonly observed in critically ill men. 92 As a synthetic metabolic steroid hormone, testosterone promotes muscle protein synthesis and is crucial for maintaining muscle mass, making it a valuable treatment for nutritional wasting diseases like cachexia, CKD, 117 lung disease, acquired immune deficiency syndrome,118,119 sepsis, 28 burns,120–123 cardiovascular disease,124,125 acute respiratory syndrome, 33 and surgical recovery. 126 Notably, testosterone also elevates hemoglobin levels, 127 and improves the sensitivity of pressure receptors. 125
However, despite its beneficial impact on protein metabolism, testosterone has potential drawbacks for disease recovery. Studies have indicated that it may elevate the risk of infection and atherosclerosis development by inhibiting iNOS. 128 More so, testosterone has been found to amplify the neurotoxic effect of glutamate in vitro and exacerbate ischemia-reperfusion injury in animal models. 129 It also can trigger apoptosis in human vascular endothelial cells through androgen receptor agonists. 130 Studies demonstrated that administering flutamide, an androgen receptor antagonist, post-traumatic bleeding could alleviate the decline in endothelial function and restore organ blood flow. 131 In critical illnesses, testosterone often leads to immune suppression and reduced survival rates. 132 Clinical research on androgens primarily focuses on protein metabolism, cardiovascular, and cognitive function. While supplementing testosterone in HF patients may enhance myocardial motility, there is currently no evidence suggesting an increase in ejection fraction. 124 Some studies found that low-dose exogenous testosterone did not affect functional status or cognition but increased lean weight and decreased fat content in elderly patients. 133 Conversely, other research indicated that exogenous testosterone improved cognitive abilities and musculoskeletal health in postmenopausal women. 134 Despite its anti-inflammatory and immunosuppressive effects, testosterone poses the risk of exacerbating cardiovascular disease and fluid retention. Therefore, routine testosterone supplementation for critically ill patients is not recommended, except for severe wasting diseases 135 (Figure 4(b)).
Moderately supplementing testosterone in specific conditions such as severe malnutrition and cachexia may be beneficial, but the risks and benefits of androgen supplementation therapy should be carefully evaluated for each patient.
FSH, LH, etc
Cortisol was found to inhibit gonadotropin under stressful conditions. In ovariectomized ewes, cortisol lowers the amplitude and frequency of LH pulses by inhibiting the pituitary response to GnRH.53,136,137 Moreover, research has shown that the combined administration of growth hormone-releasing peptide-2 (GHRP-2), thyrotropin-releasing hormone (TRH), and GnRH reactivates the growth hormone, thyroid-stimulating hormone, and LH axis more effectively in long-term critically ill men than the sole infusion of GHRP-2 or combined GHRP-2 and TRH infusion. 138 However, current research mainly focuses on identifying the causes of HPG axis injury in critical conditions, such as direct damage by pathogens or the use of medications like dopamine. 56 Despite these findings, there are no established recommendations for supplementary FSH or LH therapy in critically ill patients. In contrast to the limited evidence in critical illness, recent studies in AD have revealed a potential pathological role for FSH. Rising serum FSH levels during menopause have been shown to exacerbate AD pathophysiology. Blocking FSH action in mice has been found to mitigate AD-like phenotypes, suggesting that FSH may be a novel therapeutic target for AD.59,139 In addition, FSH antibody treatment in mice has demonstrated the ability to increase bone mass, reduce visceral adiposity, and improve energy homeostasis. 140 These findings highlight the potential for targeting FSH in neurodegenerative diseases and metabolic disorders.
Conclusion
Chronic critical conditions often result in suppression of the HPG axis and related hormone levels, illustrating the body’s physiological adjustments aimed at reducing energy expenditure and safeguarding vital organs under extreme stress. Studies have underlined the relationship between alterations in sex hormone levels under stress and patient prognoses, irrespective of the patient’s sex. Typically, low levels of sex hormones such as FSH, LH, and testosterone are associated with severe disease and unfavorable prognosis. Moreover, the interplay between sex hormones, the immune system, cytokines, and gastrointestinal microbiota markedly affects the inflammatory status and recovery process of patients. As understanding deepens, applications of sex hormones, such as estrogen and progesterone, have evolved beyond controlling inflammation, suppressing the immune system, and protecting cells, to potentially providing therapeutic measures for severe infections and other critical situations. However, the usage of androgens like testosterone necessitates a careful evaluation of potential risks and benefits during the recovery process. In summary, understanding and utilizing the complex role of sex hormone fluctuations in critically ill patients is crucial for optimizing treatment strategies and improving outcomes.
