Abstract
Objective
Genetic defects and endocrine-related factors are the leading causes of male infertility. This study was performed to analyze the genetic characteristics and sex hormone levels in different types of male infertility.
Methods
A total of 423 men with infertility underwent genetic and sex hormone analysis at The Sixth Affiliated Hospital of Guangzhou Medical University.
Results
The incidences of abnormal karyotypes in patients with male infertility, azoospermia, and oligoasthenozoospermia were 6.94%, 22.40%, 15.09%, respectively. Among men with azoospermia, Klinefelter syndrome (47,XXY) was identified in 60.71% (17/28) of those with abnormal karyotypes. Additionally, the levels of follicle-stimulating hormone and human luteinizing hormone were significantly higher in men with azoospermia showing abnormal karyotypes than in men of the other study groups. The serum testosterone level in men with azoospermia showing abnormal karyotypes was lower than that in men of the other study groups.
Conclusions
Azoospermia is closely associated with chromosome abnormalities. The levels of testosterone, human luteinizing hormone, and follicle-stimulating hormone in men with azoospermia showing abnormal karyotypes provide a clinical reference for genetic counseling and assisted reproduction.
Keywords
Introduction
Infertility is defined as the inability to conceive or produce offspring after 12 months of regular unprotected intercourse. A male-associated factor contributes to 50% to 60% of overall infertility but is solely responsible in only 10% to 20% of couples. 1 Although the causes of male infertility may vary with the geographical region and ethnic makeup of the population, approximately 20% of cases of infertility can be explained by genetic factors. The prevalence of numerical and structural chromosomal abnormalities in the infertile population reportedly ranges from 2% to 10%.2,3 Klinefelter syndrome is the most common genetic disorder that causes infertility and is responsible for approximately 1.8% of cases of male infertility. 4
Abnormal hormone levels in patients with male infertility can be used to assist in the diagnosis of spermatogenesis disorders. Numerous studies have shown that sex hormones can be used to treat male infertility.5,6 In this study, we investigated the genetic characteristics and sex hormone levels in different types of male infertility with the aim of better understanding the different obstacles to spermatogenesis influenced by genetic and endocrine factors.
Materials and methods
Clinical data
We retrospectively analyzed infertile men who had been diagnosed and treated at the Andrology Clinic of the Reproductive Center at The Sixth Affiliated Hospital of Guangzhou Medical University from January 2014 to September 2018. Venous blood was drawn from the infertile men for chromosome karyotype analysis. Men who had lived with their wives for at least 1 year but had been unable to conceive or produce offspring were defined as infertile. In this study, patients with male infertility had a normal sperm amount and activity. After strict screening and exclusion of other causes of infertility such as infection, seminal fluid was collected from the infertile patients and analyzed. According to the seminal fluid analysis criteria of the World Health Organization, 7 patients whose seminal fluid was found to have no sperm in three consecutive tests were initially diagnosed with azoospermia. These patients were then further investigated by a physical examination, laboratory tests, or imaging in some cases and then eventually diagnosed with non-obstructive azoospermia. In this article, azoospermia refers to non-obstructive azoospermia. Patients with a low sperm concentration and a <32% ratio of progressive sperm motility were diagnosed with oligoasthenozoospermia. All patients provided written informed consent. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research and ethics committee of The Sixth Affiliated Hospital of Guangzhou Medical University.
Chromosome karyotype analysis
Chromosome karyotype analysis was performed using the G-banding technique. Peripheral blood samples were collected, and lymphocytes were cultured in RPMI 1640 medium with fetal bovine serum for 72 hours. Cells were harvested after treatment with demecolcine for 2.5 hours. G-banding revealed more than 20 metaphase chromosomes for each patient. Chromosomal disorders were described according to the International System for Human Cytogenetic Nomenclature. 8
Measurement of serum sex hormones
Blood samples were collected by venipuncture and allowed to clot at room temperature before centrifugation. Serum was separated and immediately stored frozen at −20°C until assayed. The levels of several sex hormones [follicle-stimulating hormone (FSH), human luteinizing hormone (hLH), testosterone, estradiol, progesterone, and prolactin] were measured using a chemiluminescence immune assay (Roche, Basel, Switzerland).
Statistical analysis
Data were analyzed using GraphPad Prism 5.0 software (GraphPad Software, San Diego, CA, USA). Differences among more than two groups were analyzed by one-way analysis of variance with Tukey’s test. Most data are presented as mean ± standard error of the mean; age is presented as mean ± standard deviation. Statistical significance was set at
Results
Chromosome karyotype analysis
We collected data on 245 patients with male infertility, 125 patients with azoospermia, and 53 patients with oligoasthenozoospermia for chromosome karyotype analysis. The incidences of abnormal karyotypes in patients with male infertility, azoospermia, and oligoasthenozoospermia were 6.94%, 22.40%, and 15.09%, respectively. As shown in Table 1, the number of abnormal karyotypes in men with azoospermia was obviously higher than that in patients with male infertility and oligoasthenozoospermia.
Abnormal karyotypes in different types of male infertility.

The most common type of chromosomal abnormality in patients with male infertility was pericentric inversion of chromosome 9, with an incidence of 17.6% (3/17). Pericentric inversion of chromosome 9 was also found in men with azoospermia and oligoasthenozoospermia. The incidence of Klinefelter syndrome (47,XXY) reached 60.71% (17/28) in men with azoospermia showing an abnormal karyotype. The polymorphism 46,XY,1qh+ was found in all groups. Details are shown in Table 1.
Hormone abnormalities in different types of male infertility
We further collected data of sex hormone levels in 97 men with azoospermia showing normal karyotypes (mean age, 30.47 ± 6.47 years; range, 18–46 years), 28 men with azoospermia showing abnormal karyotypes (mean age, 30.07 ± 5.66 years; range, 25–52 years), 10 patients with male infertility showing abnormal karyotypes (mean age, 35.50 ± 6.79 years; range, 25–46 years), and 54 healthy volunteers with normal physical examination findings as a control group (mean age, 31.91 ± 8.29 years; range, 18–53 years). No significant differences in the estradiol, progesterone, or prolactin levels were found among the four groups. The serum testosterone level in men with azoospermia showing abnormal karyotypes was significantly lower than that in the other study groups (
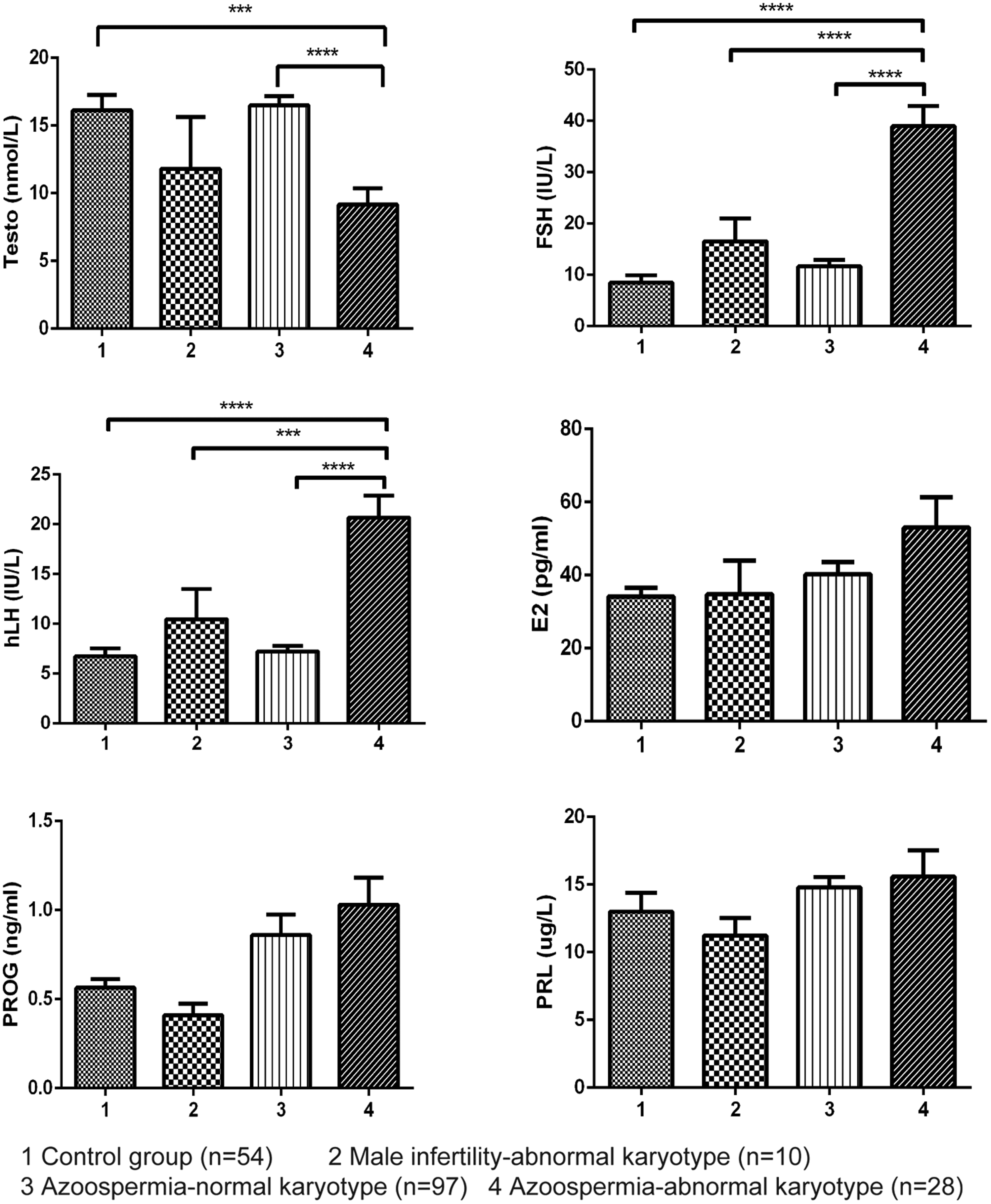
Serum sex hormone levels in different male infertility groups. Error bars indicate the standard error of the mean. ***
Discussion
In the present study, the frequency rates of chromosome abnormalities and sex hormone levels differed among different types of male infertility. We found that the most common type of chromosomal aneuploidy in men with azoospermia was 47,XXY (Klinefelter syndrome). XXY is the only known chromosome abnormality in which a substantial proportion (approximately 50%) of cases arise as a result of non-disjunction at the first paternal meiotic division. 9 The incidence of Klinefelter syndrome was 13.6% among men with non-obstructive azoospermia in this study, which is slightly higher than the rate of 8.05% to 10.56% reported in the literature.10,11 The different sample sizes used in the statistical analyses may be the reason for this deviation.
Although increasingly more studies are focusing on genetic infertility, the genetic mechanism of male infertility remains uncertain. Many researchers have evaluated not only chromosomal abnormalities but also mutations in genes that are closely associated with infertility. For example, testis-expressed genes 11 and 15 (
The hormone abnormalities in infertile men are caused by hypothalamic-pituitary-testis dysfunction. Identification and characterization of hormonal factors of male infertility are important not only for diagnostic purposes but also for potential pharmacogenetic therapy. Intratesticular testosterone is vital for maintenance of spermatogenesis. In a recent survey, 25% of urologists reported using exogenous testosterone to treat low testosterone levels in patients with male infertility. 15
FSH and hLH are synthesized and secreted by the gonadotropic cells of the anterior pituitary gland. In males, the synergism of hLH and FSH may stimulate production of testosterone by Leydig cells, thus promoting spermatogenesis.16,17 The exact role of FSH in male testosterone production is not entirely clear. A low testosterone level or high LH level is present in an estimated 20% to 30% of patients with male infertility. 18 The effect of LH-stimulated spermatogenesis is mediated through intratesticular testosterone. 17 The release of FSH and LH in gonadotropic cells can be inhibited by testosterone, ultimately inhibiting spermatogenesis. In the present study, the testosterone level in men with azoospermia showing abnormal karyotypes was decreased, while the hLH and FSH levels were increased. However, whether low testosterone and high hLH and FSH can predict an abnormal karyotype in men with azoospermia remains unclear.
Estradiol is the primary female sex hormone responsible for female development and maintenance of secondary sex characteristics. Progesterone is an endogenous steroid involved in the menstrual cycle, pregnancy, and embryogenesis of humans. Prolactin is best known for its role in enabling females to produce milk. Physiologic levels of prolactin in males enhance luteinizing hormone receptors in Leydig cells, resulting in testosterone secretion, which further affects spermatogenesis. 19 This study showed no significant differences in the serum levels of estradiol, progesterone, and prolactin among the four groups.
In conclusion, our results show the genetic and hormonal characteristics of different types of male infertility in Qingyuan district of South China. The findings of this study provide an explicit understanding of the risk factors for male infertility and may thus serve as a clinical reference for genetic counseling and assisted reproduction.
Footnotes
Authors’ contributions
Xiaolei Xie and Xiaoyan Guo designed the study and performed the data analysis. Xiaolei Xie, Fuguang Li, and Weihe Tan collected the data. Weihe Tan and Runqinag Chen performed the clinical diagnosis. Xiaolei Xie and Weiguo Yin prepared the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The research was supported by the Qingyuan Science and Technology Project (No. 2018B036) and Qingyuan People’s Hospital Innovation Fund (No. 20180031).
