Abstract
Background:
There is limited data about the risk factors of clinically significant glycosylated hemoglobin (HbA1c) change and post-transplant diabetes mellitus (PTDM) in the first year post-kidney transplantation (KT), especially in the Middle East.
Objectives:
To determine the trends of HbA1c levels, the risk factors associated with HbA1c increases, and predictors of clinically significant HbA1c change and PTDM in the first year post-KT.
Design:
Retrospective chart review.
Methods:
We included all KT recipients (KTRs) at our center from 2017 until 2020. The study focused on reviewing the patients’ demographic information, cardiovascular risk factors, and HbA1c values at baseline, 6 months, and 12 months.
Results:
A total of 203 KTRs were included. The mean age of the participants was 44.7 ± 15.5 years, 59.1% were men, and 80.3% received living donors. Eighty-two (40.4%) KTRs had pre-KT diabetes. At 12 months post-KT, the total HbA1c change was 0.87 ± 1.6. In total, 130 (64.04%) KTRs demonstrated clinically significant HbA1c change, and 19 (15.7%) nondiabetics developed PTDM. Pre-KT diabetics suffered greater increases than their nondiabetic counterparts (0.8 vs 0.6,
Conclusion:
The incidence rate of PTDM in Saudi KT patients is similar to that of other populations. Several risk factors, including low baseline HbA1c and pre-KT hypertension, predict a clinically significant change in HbA1c. Patients with these risk factors may require stricter monitoring and control of HbA1c.
Plain language summary
Post-kidney transplant diabetes mellitus (PTDM) is one of the most frequent complications of kidney transplantation (KT). However, risk factors for PTDM and clinically significant changes in HbA1c levels (>0.5%) are still limited. In this article, we retrospectively reviewed the medical files of 203 Saudi KT recipients and the changes in HbA1c levels during the first year post-KT. Age, weight gain, pre-KT hypertension, and lower baseline HbA1c were independently associated with clinically significant HbA1c change. Age was the only predictor of PTDM.
Introduction
Diabetes mellitus (DM) is the primary etiology of end-stage kidney disease (ESKD) for at least 25% of kidney transplant (KT) candidates in Saudi Arabia. 1 Additionally, glycemic control worsens in many KT recipients (KTR) during their first few months post-KT,2,3 resulting in approximately 27% of nondiabetic KTRs developing post-transplant DM after KT (post-transplant diabetes mellitus, PTDM). 2 PTDM ranks among the most frequent complications of KT. 4
Poor glycemic control in KTRs has been linked to unfavorable graft outcomes, including major adverse cardiovascular events, 5 and decreased graft and patient survival.6–10 Glycated hemoglobin (HbA1c) reflects the impact of blood glucose levels on circulating red blood cells over 120 days and is a key laboratory indicator of longer-term glycemic control.11,12 Elevated HbA1c levels have been associated with adverse outcomes, making regular HbA1c monitoring and control essential. 12 Indeed, a 1% reduction in HbA1c value corresponds to a 37% decrease in microvascular complications and a 21% reduction in DM-related mortality. 13 A meta-analysis based on 2057 KTRs from 6 studies found an HbA1c cut-off point of 6.5% in early post-KT months was highly specific (96%) for the diagnosis of PTDM. 14 According to the American Diabetes Association (ADA), an increase of 0.5% in HbA1c from baseline is considered a clinically significant change in glycemic control (clinically significant HbA1c change), prompting intervention in diabetic patients.15,16
Although studies about PTDM in KTRs are limited in the Middle Eastern region, some reports have reported increased prevalence in Kuwait and Qatar.17,18 Given the scarcity of data on post-KT DM in Saudi Arabia, investigating PTDM and shedding light on its associated risk factors in the country have become paramount. Furthermore, identifying risk factors for clinically significant changes in HbA1c may help classify high-risk patients who require more intensive post-KT glycemic control. Therefore, this study aims to analyze the changes in HbA1c during the first year post-KT in Saudi KT patients and define risk factors for PTDM and clinically significant HbA1c change.
Materials and methods
After the approval of the local IRB, we conducted a retrospective study of KTRs at King Abdulaziz Medical City in Riyadh, Saudi Arabia. The study encompassed patients who underwent KT between 2017 and 2020. In total, 287 patients received KT at our center. In our analyses, only patients with complete HbA1c measurements during the first year post-KT were included. Therefore, 84 patients were excluded from the analyses due to loss of follow-up during the first year post-KT, and 203 KTRs were included in our final analysis. Excluded patients were younger on average (44.7 ± 15.5 vs 39.7 ± 15.8,
DM was defined according to the ADA guidelines as HbA1c ⩾ 6.5, or a fasting plasma glucose level of ⩾126 mg/dL on repeated measurements, or a random plasma glucose level of ⩾200 mg/dL in a patient with symptoms of hyperglycemia. 25 PTDM was defined as the development of DM post-KT. An increase of 0.5% in HbA1c level from baseline to the 12-month mark was considered clinically significant HbA1c change, a threshold widely accepted for the adjustment of hypoglycemic agents.15,16,26–30 Accordingly, our patients were divided into two groups based on each of the following: clinically significant HbA1c change and PTDM. Our primary study outcome was to assess the trends of HbA1c within the first year post-KT. Additionally, we aimed to identify risk factors associated with clinically significant HbA1c change and PTDM among KTRs at our center.
Statistical analysis was performed using SPSS software version 26 (Armonk, NY: IBM Corp). All continuous variables were initially tested for normal distribution using both visual inspection of histograms and
Results
General characteristics and cardiovascular disease risk factors
We reviewed the medical files of 203 patients who underwent KT at our center between the years 2017 and 2020 (Table 1). The mean age of the participants is 44.7 ± 15.5 years. A total of 120 (59.1%) KTRs were male, and 163 (80.3%) received living donors. 157 (77.3%) patients received hemodialysis pre-KT, 24 (11.8%) received peritoneal dialysis, and the remainder (10.8%) were transplanted preemptively. The average dialysis vintage was 2 (1–4) years.
General characteristics of KTRs.
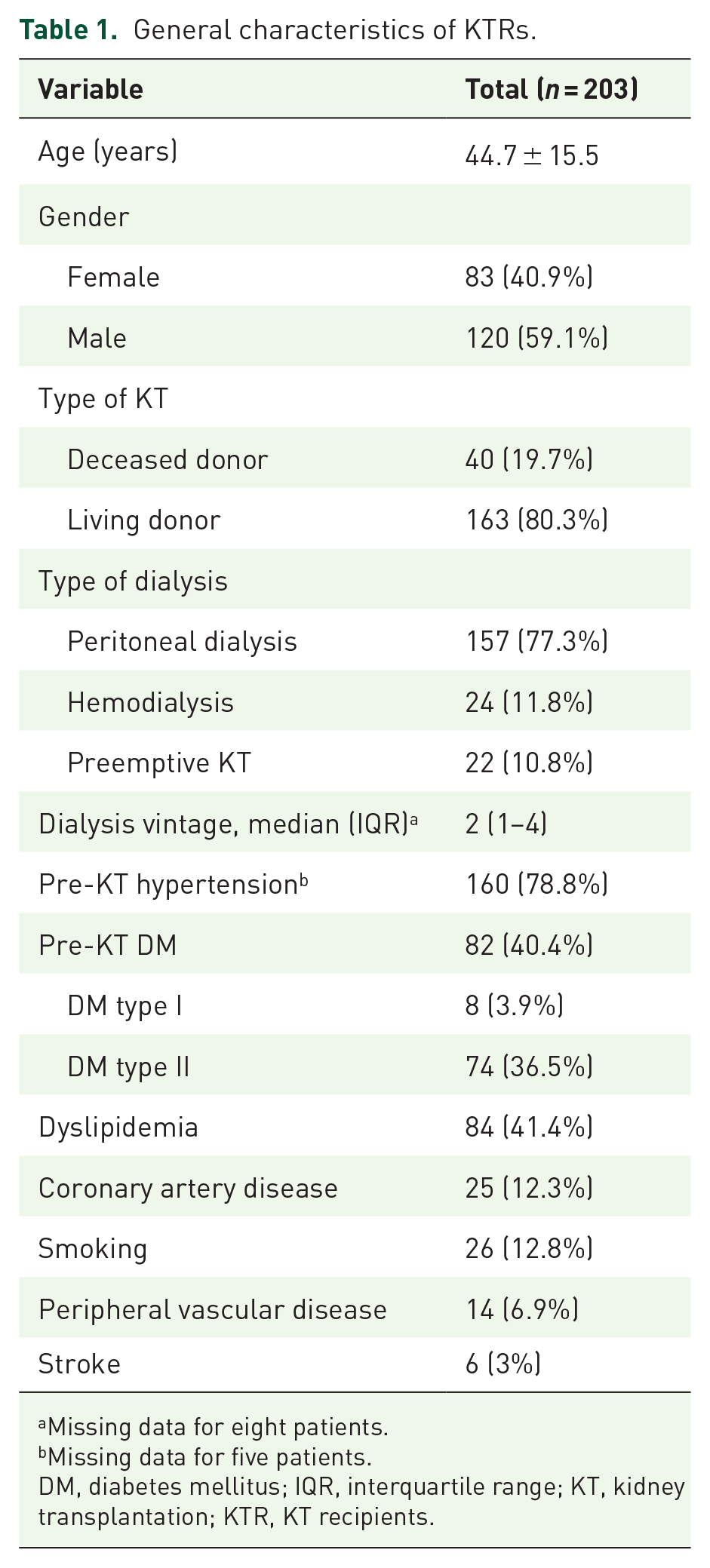
Missing data for eight patients.
Missing data for five patients.
DM, diabetes mellitus; IQR, interquartile range; KT, kidney transplantation; KTR, KT recipients.
The average weight at baseline was 69.5 ± 17.1 kg and increased to 75.8 ± 17.5 kg at 12 months post-KT. Eleven (5.4%) KTRs had obesity stage 2, 46 (22.8%) had obesity stage 1, 64 (31.7%) were overweight, and 66 (32.7%) had normal weight at baseline. 160 (78.8%) KTRs had pre-KT hypertension, 84 (41.4%) had dyslipidemia, and 25 (12.3%) had coronary artery disease.
Eighty-two (40.4%) patients had pre-KT DM, of which 74 had type 2 DM and 8 had type 1 DM. The mean HbA1c increased from 5.9 (41 mmol/mol) ±1.6% at baseline to 6.7 (50 mmol/mol) ±1.8% at 12 months post-KT (Figure 1). The total HbA1c change was 0.87% ± 1.6% after 12 months of follow-up. 130 (64.04%) KTRs demonstrated clinically significant HbA1c change, and 19 (15.7%) nondiabetic patients developed PTDM.

Trends in HbA1c levels within the first year post-KT in our patient population.
Patient demographics and general HbA1c trends
Both genders experienced significant increases in mean HbA1c levels from baseline to 12 months post-KT (

Trends in HbA1c levels in the first year post-KT in males versus females.
Patient demographics and clinically significant HbA1c change
In total, 130 (64.03%) KTRs developed clinically significant HbA1c change in the first year post-KT. By definition, clinically significant HbA1c change patients had greater increases in HbA1c levels post-KT than HbA1c change <0.5% patients (1.6% ± 1.4% vs −0.4% ± 1.2%,

Trends in HbA1c levels in the first year post-KT in clinically significant HbA1c change patients versus HbA1c change <0.5%.
Characteristics of KTRs with clinically significant HbA1c change versus HbA1c change <0.5% patients.
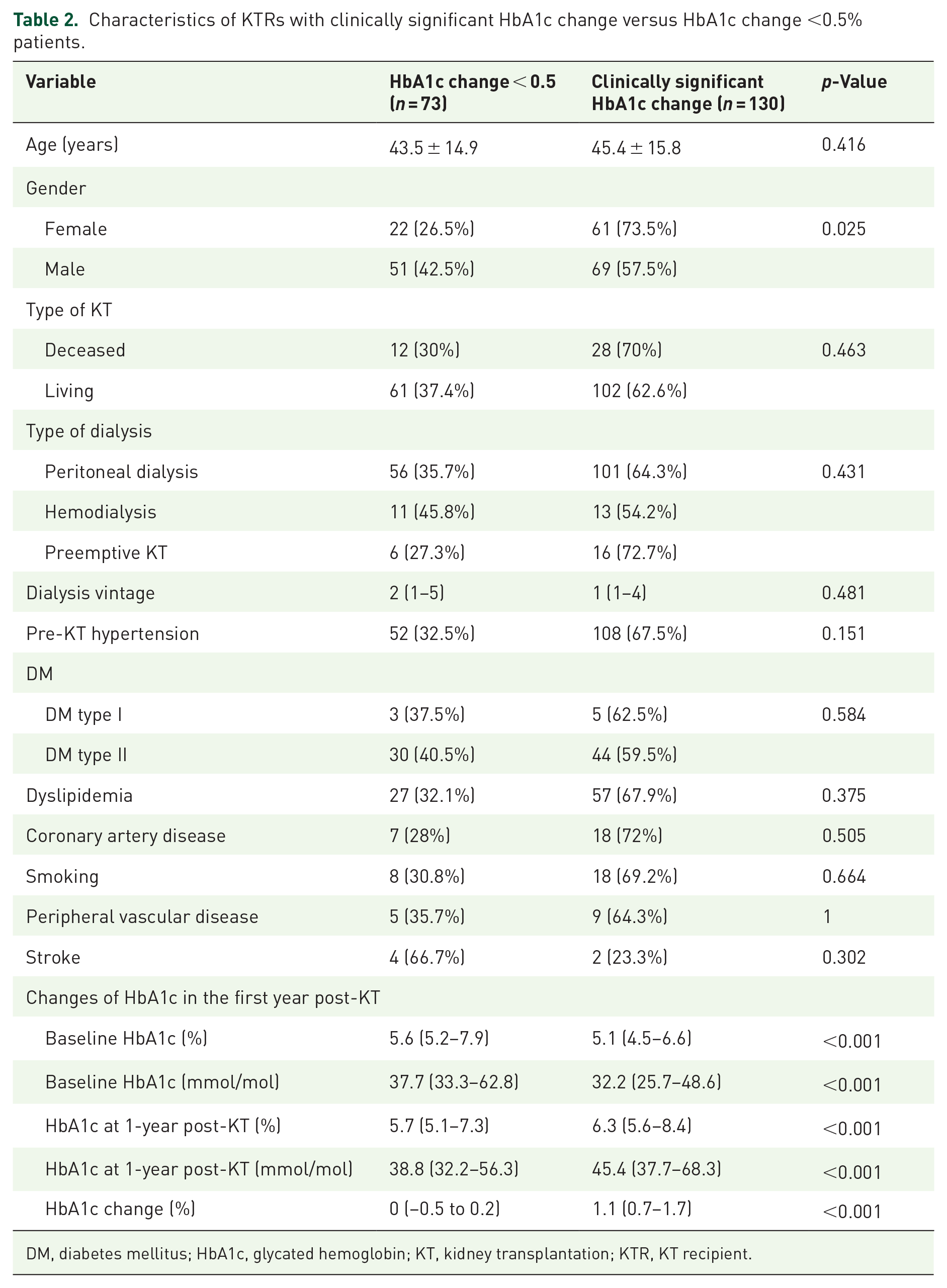
DM, diabetes mellitus; HbA1c, glycated hemoglobin; KT, kidney transplantation; KTR, KT recipient.
Patients with clinically significant HbA1c change had lower pre-KT weight (67.4 ± 17.1 vs 73.3 ± 16.6 kg,
Multivariate analysis of clinically significant HbA1c change among KTRs.

CI, confidence interval; DM, diabetes mellitus; HbA1c, glycated hemoglobin; KT, kidney transplantation; KTR, KT recipient; OR, odds ratio.
Patient demographics and PTDM
After excluding KTRs with pre-existing DM, 19 (15.7%) patients developed PTDM during the first year post-KT. PTDM patients were older (45.7 ± 15.2 vs 36.1 ± 12.7 years,
Multivariate analysis for PTDM.

CI, confidence interval; aOR, adjusted odds ratio; PTDM, post-transplant diabetes mellitus.
Abnormal pre-KT screening cardiac imaging was more common in KTRs with PTDM, including greater coronary calcifications (
Discussion
The present study examined a cohort of 203 KTRs at King Abdulaziz Medical Center, Saudi Arabia, to investigate the prevalence of clinically significant HbA1c change and PTDM, and associated risk factors during the first year after KT. Our findings provide valuable insights into the epidemiology of post-transplant hyperglycemia in the region. We observed that 64.0% of KTRs developed clinically significant HbA1c changes. Our findings are consistent with previous research demonstrating a significant increase in HbA1c levels after KT. 6
Age and established pre-KT hypertension were also independent risk factors for incremental HbA1c increase. Patients with clinically significant HbA1c change had a lower pre-KT weight but experienced greater weight gain 12 months after KT and were more likely to develop PTDM. Additionally, lower baseline HbA1c was independently associated with greater HbA1c increases in the first year after KT. These findings highlight the association between dietary alteration and post-KT weight gain with blood sugar control post-KT. ESKD patients typically follow a restrictive renal diet and have a lower appetite due to uremia. These factors are typically elevated by KT which results in improved appetite and significant weight gain.32,33 The findings of our current study could be explained by the younger age of our population and the higher prevalence of living donor KT, which may indicate a healthier study population. To the best of our knowledge, there have been no studies examining the risk factors for significant changes in HbA1c levels in KTRs. Further studies are needed to confirm our results in this regard.
By 12 months post-KT, 15.7% of patients without established DM developed PTDM. The incidence rate of PTDM found by our study aligns with that reported nationally 34 and worldwide.20,35,36 However, studies from the Gulf region have demonstrated a higher incidence of PTDM. For instance, PTDM was reported in 21.2% and 34% of KTRs in Kuwait and Qatar, respectively.17,18
Although only mildly associated, increasing age was the sole predictor of PTDM at 1-year post-KT in our population, consistent with other studies in the Middle Eastern region.17,18,20–24 Pirsch et al. 37 also demonstrated the same in a double-blinded clinical trial. However, other studies have identified additional risk factors, such as prediabetes, weight, BMI, waist circumference, peritoneal dialysis, and smoking status.18,20–24,38–40 Other reported risk factors for PTDM include polycystic kidney disease, acute rejection, hepatitis C virus infection, cytomegalovirus infection, family history of diabetes,21,24,41 and possible genetic predisposition.35,42,43 The influence of different risk factors may vary from one study to another, as seen in our study. Nonetheless, age appears to be a major risk factor for PTDM in KTRs.
Dietary modification and immunosuppression remain the most important modifiable factors of HbA1c increase and PTDM.44,45 Active lifestyle interventions post-KT resulted in a significant weight loss, reduced fat mass, and a suggestion of reduced PTDM incidence compared to passive lifestyles as per the CAVIAR randomized, controlled trial. 44 Mediterranean-style diet has also shown to be associated with a lower risk of PTDM. 45 Calcineurin inhibitors not only prevent organ transplant rejection but also affect insulin production and contribute to the development of PTDM by modifying transcription factors of the nuclear factor NFAT. 46 Tacrolimus is known to be more diabetogenic than cyclosporine. However, it is currently the core calcineurin inhibitor used in KT worldwide due to its superior immunosuppressant properties. 46
The diabetogenic effect of calcineurin inhibitors is dose-dependent, and their diabetogenic effect is increased with higher steroid doses. 4 Corticosteroids also decrease peripheral insulin sensitivity, inhibit pancreatic insulin production and secretion, increase hepatic gluconeogenesis, and promote protein degradation and lipolysis. 4 High steroid maintenance may increase the risk of PTDM; however, steroids at small maintenance dose (such as that used in our protocol) are likely to have only a limited impact on the incidence of PTDM.37,47 In fact, randomized, controlled trials have shown that early corticosteroid withdrawal nor steroid avoidance has an impact on PTDM risk.37,48 Therefore, in patients at high risk of PTDM, a glucocorticoid minimization strategy should be balanced with the immunological risk profile to avoid acute rejection and graft loss.41,49
Notably, our study showed a higher incidence of abnormal pre-KT cardiac workup including, abnormal perfusion tests, and coronary artery disease on coronary angiography in patients who developed PTDM in the first year post-KT. PTDM is a known predictor of major cardiac events 5 and negatively affects patient survival. 10 This intriguing finding deserves further evaluation by prospective studies.
Our approach to decrease the risk of hyperglycemia and PTDM is to taper tacrolimus dose to a blood level between 5 and 7 ng/ml by 9 months post-KT, along with the reduction in total energy intake and increasing physical activity. 19 We also tend to avoid high steroid dosages during induction and rapidly taper steroids to 5 mg of prednisolone daily by the first 4 weeks post-KT. 19 Additionally, we use early steroid withdrawal for very low immunologic risk KTs.
Till recently, metformin was often employed as a first-line agent in KTRs with PTDM.50,51 However, with the introduction of novel oral hypoglycemic agents, including sodium–glucose linked transporter-2 inhibitors and glucagon-like peptide-1 receptor agonists, multiple retrospective studies have reported no major safety concerns in KTRs. 52 These agents are likely to become first-line hypoglycemic agents for KTRs with PTDM. 52 Further studies are needed about the prophylactic role of these agents in KTRs at high risk to develop PTDM.
Our study has several strengths and weaknesses. Given that our center is one of the largest KT centers in Saudi Arabia, 53 this study has included a large sample size and the examination of multiple variables in an underrepresented population in the literature. Previous studies have reviewed the risk factors of PTDM using an HbA1c cut-off of 6.5. However, this study is the first study to examine incremental changes of HbA1c post-KT using a HbA1c cut-off of 0.5%. Our study also showed an association between HbA1c changes and lower baseline HbA1c which may indicate the importance of dietary factors among younger predominantly living donor KTRs. Additionally, our study also showed a higher incidence of abnormal pre-KT cardiac workup in patients who developed PTDM in the first year post-KT. This may indicate that patients who develop PTDM are a selected group with higher cardiovascular risk at baseline, even before KT. This intriguing finding has not been studied before and it deserves further evaluation by prospective studies. However, the retrospective single-center nature may limit the generalizability of our findings, as it may not reflect the findings of other centers in Saudi Arabia. Furthermore, excluded patients had less risk factors at baseline, which may have overexaggerated the findings seen in our study. Additionally, we did not investigate the influence of dietary factors and family history of diabetes on HbA1c levels, which have a significant role in glycemic control and the development of PTDM. Finally, there is a large debate regarding the optimal diagnostic test for PTDM. Fasting plasma glucose has a low sensitivity for diagnosing PTDM.54,55 Similarly, normal HbA1c may not exclude the diagnosis of PTDM, especially in the early stages of KT.55,56 Nevertheless, HbA1c-based diagnosis is the current recommendation by the International Consensus Meeting on PTDM, due to its convenience in a large clinical setting. 57 On the contrary, recent studies have suggested using oral glucose tolerance tests as the gold standard for PTDM diagnosis since they provide less false negatives than other tests. 57
Conclusion
Our findings underscore the importance of rigorous monitoring and management of HbA1c levels, especially among high-risk groups, during the first year post-KT. These insights can guide clinicians in their early interventions to optimize glycemic control among KTRs, thereby improving the overall outcomes of KT. Future research should consider a multicenter, prospective approach, and incorporate genetic and dietary factors to gain a more comprehensive understanding of the dynamics of glycemic control post-KT.
Supplemental Material
sj-docx-1-tae-10.1177_20420188241301940 – Supplemental material for HbA1c changes in the first year post-kidney transplant and associated risk factors in a Saudi cohort
Supplemental material, sj-docx-1-tae-10.1177_20420188241301940 for HbA1c changes in the first year post-kidney transplant and associated risk factors in a Saudi cohort by Ziad Arabi, Hazim S. Alghamdi, Tarek Arabi, Areez Shafqat, Badr Elwy and Belal Sabbah in Therapeutic Advances in Endocrinology and Metabolism
Supplemental Material
sj-docx-2-tae-10.1177_20420188241301940 – Supplemental material for HbA1c changes in the first year post-kidney transplant and associated risk factors in a Saudi cohort
Supplemental material, sj-docx-2-tae-10.1177_20420188241301940 for HbA1c changes in the first year post-kidney transplant and associated risk factors in a Saudi cohort by Ziad Arabi, Hazim S. Alghamdi, Tarek Arabi, Areez Shafqat, Badr Elwy and Belal Sabbah in Therapeutic Advances in Endocrinology and Metabolism
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
