Abstract
Objective
We determined the prevalence of poor glycemic control and associations with sociodemographics, comorbid conditions, and medication adherence among patients with type 2 diabetes mellitus (T2DM) at a tertiary hospital in southwestern Nigeria.
Methods
We conducted a retrospective observational study among 300 patients with T2DM using systematic random sampling. We used a semi-structured questionnaire to collect information on respondents’ sociodemographic profile, lifestyle, comorbid conditions, and antidiabetic medications. Adherence was determined using the Morisky Medication Adherence Scale. Fasting blood samples were tested using a glycated hemoglobin marker. Multivariate logistic regression was used to identify factors associated with poor glycemic control.
Results
Respondents’ mean age was 61.9 ± 11.8 years. The prevalence of poor glycemic control was 40.0% (95% confidence interval [CI]: 34.4%–45.8%). The adjusted odds ratio (95% CI) for factors associated with poor glycemic control was 2.522 (1.402–4.647) for older age, 1.882 (1.021–3.467) for low income, 1.734 (1.013–3.401) for obesity, 2.014 (1.269–5.336) for non-initiation of insulin therapy, and 1.830 (1.045–3.206) for poor medication adherence.
Conclusion
Older age, lower income, obesity, non-initiation of insulin, and poor medication adherence were associated with poor glycemic control. These variables may help clinicians identify patients at high risk of poor glycemic control.
Introduction
Diabetes is a metabolic disorder and a chronic disease, often characterized by a hyperglycemic state of the body.1,2 The development of diabetes is often owing to either a deficiency in insulin secretion or an inadequate response to insulin secretion.1,2 Diabetes has global public health importance as a leading cause of blindness, end-stage renal disease, and stroke.1,2
Type 2 diabetes mellitus (T2DM) is often characterized by sufficient insulin secretion but poor utilization by body cells, resulting in insulin resistance. 3 T2DM constitutes approximately 85% to 95% of all cases of diabetes in developed countries, with a growing proportion in developing countries as the result of increases in urbanization, sedentary lifestyles, aging populations, and unhealthy behavioral patterns. 4 The main therapeutic goal for all patients with T2DM is to maintain good control so as to prevent the risk of complications associated with poor control. 5 The burden of poor glycemic control in developing countries has increased despite increased awareness of its complications, mainly because of a lack of accessible and affordable health care. 3
In a longitudinal survey by Ali et al. in the United States between 2007 and 2010, the prevalence of poor glycemic control was 12.9%. 5 A cross-sectional study by Sheleme et al. in southwest Ethiopia found a prevalence of poor glycemic control of 72.0%. 6 Previous studies in Nigeria have revealed that the prevalence of poor glycemic control ranges from 34% to 45% in southeast and southwest Nigeria, respectively.7,8 Research has indicated that glycemic control remains poor, even among patients who undergo treatment.5,6
Sociodemographic factors, comorbidities, unhealthy lifestyles, and duration of diabetes have been found to be associated with poor glycemic control.5–8 Obesity, physical inactivity, cigarette smoking, and alcohol intake are common unhealthy lifestyle factors linked with poor glycemic control.5–8 Nonadherence to hypoglycemic medication has been reported to be a significant independent risk factor of poor glycemic control.6,9
Most relevant studies have been conducted in urban settings of Nigeria, which leaves out most of the population residing in rural areas. Findings involving rural populations would contribute to better management of patients with T2DM in rural areas and provide a template for further interventional studies. Therefore, in the present research, we aimed to determine the prevalence of poor glycemic control and its association with sociodemographic profiles, unhealthy lifestyles, comorbid conditions, and medication adherence among patients with T2DM.
In this study, we aimed to answer the following questions. 1) What is the prevalence of poor glycemic control among patients with T2DM in rural Nigeria? 2) What is the relationship between the sociodemographic profile of these patients and their glycemic control? 3) What is the relationship between unhealthy lifestyles in these patients and their glycemic control? 4) What is the relationship between comorbid conditions in these patients and their glycemic control? 5) What is the relationship between the number of hypoglycemic medications used by these patients and their glycemic control? 6) What is the relationship between medication adherence in these patients and their glycemic control?
Methods
Study area
The present study was conducted between August and November 2020 at the family medicine clinic of a tertiary hospital located in Ido Ekiti in southwestern Nigeria. Ido Ekiti is a rural community in Ekiti State and is where the headquarters of the local Ido-Osi government is located. Ido Ekiti is approximately 15 km from Ado Ekiti, the capital of Ekiti State. Ido Ekiti has a total land area of 332 km2 and a total population of 159,114 inhabitants, according to the most recent population census conducted in 2006. The annual population growth rate is 3.2%, with the population in 2019 is estimated to be 225,305 inhabitants. 10 Residents of Ido Ekiti are mainly farmers and traders in the informal sector, with a relatively small proportion comprising the working population and retirees in the formal sector. 10 The study hospital serves as a referral center for both privately and government-owned hospitals and is accredited for residency training by both the National Postgraduate Medical College of Nigeria and the West African College of Physicians. The family medicine clinic offers primary and specialist care for a wide array of acute and chronic medical conditions to individuals in its catchment and in the surrounding area. Presently, the department of family medicine has 11 consultant specialists who are responsible for all outpatient cases.
This was a retrospective observational study and the study population were all patients with T2DM. The inclusion criteria were patients who were 40 years and above, in follow-up treatment for T2DM for at least 6 months, and who consented to the study. The exclusion criteria were patients who were critically ill or had a major psychiatric illness and could not follow the study protocol.
Sampling
We determined the sample size using the following: 11
n = Z2P(1−P)/d2 and nf =
A systematic random sampling technique was used to select study participants. A review of the medical records of patients with T2DM in follow-up visits at the family medical clinic revealed that during 2019, an average of 10 patients were seen daily. This translated to 50 per week (Monday to Friday) and 850 (sampling frame) patients over a period of the 17-week study period. Using k =
Data collection
Eligibility for the study was determined using the inclusion criteria. The data were collected using a data collection form and a pretested, semi-structured interviewer-administered questionnaire. The adopted part of the questionnaire has been validated for face validity and content validity. Pretesting of the questionnaire was carried out to ensure construct validity, conducted among 15 patients with T2DM at a follow-up visit in the family medicine clinic of another tertiary hospital in southwestern Nigeria. This was done to assess the applicability of the instrument and the procedure. The outcome from pretesting and validity assessment led to some modifications in the questionnaire. The time needed to complete the questionnaire was approximately 10 to 15 minutes.
The questionnaire was grouped into five sections (A–E), representing the independent variables. Section A assessed respondents’ sociodemographic characteristics including the duration of T2DM. Section B addressed participants’ lifestyles, considering alcoholic intake, tobacco use, and level of physical activity. These were self-reported. Response options regarding alcoholic intake were yes or no. If the answer was yes, then a follow-up question was asked regarding the frequency of alcohol consumption, with response options of daily, weekly or monthly. Tobacco smoking was grouped into smoker (both passive and active) and never smoker. Physical activity was assessed according to the number of minutes per day and number of days per week spent doing physical exercise. Exercise could also be in the form of farming, community hawking (itinerant trading), or trekking (traveling from one place to another). Respondents were assessed as being physically active with more than 30 minutes of physical activity per day, at least five times per week; otherwise, they were assessed as being physically inactive. Section C addressed comorbid conditions, as documented in the medical records. Section D queried the types and number of hypoglycemic medications taken by patients. Section E assessed respondents’ medication adherence level using the eight-item Morisky Medication Adherence Scale. 13 This scale consists of eight items designed to evaluate medication adherence in patients with T2DM and has been validated and found to be reliable in a number of studies on medication adherence.13,14 Responses consistent with adherence were scored as 1 point. Points were added and adherence categorized into high (0–2), medium (3–5), and low adherence (6–8). For the purpose of this study, the score was dichotomized into adherent, encompassing patients with high adherence (scores 0–2), and nonadherent, comprising patients with medium and low adherence (scores 3–8).
Clinical parameters of respondents
Body weight was measured to the nearest 0.1 kg using a portable scale and height was measured to the nearest 0.1 cm with the participant in the standing position. Body mass index (BMI) was calculated as weight in kilograms divided by the square of height (in meters). BMI
After an overnight fast, 4 mL venous blood samples were collected from each participant into EDTA tubes and sent to the hospital laboratory where glycated hemoglobin (HbA1c) was measured immunochemically on a DCA 2000 HbA1c auto analyzer, using kits supplied by Boehringer Mannheim (Germany). 15 Commercially prepared standards and control samples were used to ensure accuracy of the test results. Glycemic control was categorized as good with HbA1c ≤7.0% (i.e., ≤53 mmol/mol) and poor with HbA1c >7.0% (i.e., >53 mmol/mol). 16
Definitions
Patients with T2DM were defined as those who had documented treatment with anti-diabetes medication or with a glucose concentration of ≥7.0 mmol/L. 16 Medication adherence was defined as patients with T2DM who took their prescribed medication regularly during the 7 days prior to recruitment. 9
Ethical considerations
The Ethics and Research Committee of Federal Teaching Hospital, Ido Ekiti, Nigeria approved the study. The methods and objectives of this study were carefully explained to each patient individually. All patients were thoroughly informed about the risks and advantages of the procedures. Written informed consent for treatment was obtained from each respondent (signature or thumbprint) before their participation in the study, and enrollment was according to their willingness to participate in this research. Respondents were free to refuse or discontinue participation at any time without losing any benefits of health care, and no additional benefits were granted to those who participated. For respondents who could not read or write, the questionnaire was translated from English language into their local language by an independent interpreter who served as their legal representative; subsequent translation back into English was done to maintain consistency in the responses. All recorded information was kept anonymous. Confidentiality and privacy were ensured throughout the study. The study was administered at no cost to participants. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 17 All patient details have been deidentified.
Data entry and analysis
The data were checked, cleaned, and entered into EPI Info version 7.0 and were then exported to IBM SPSS version 20.0 (IBM Corp., Armonk, NY, USA) for analysis. Quantitative data are expressed as mean ± standard deviation. Significance was assessed using the chi-square test. Repeated-measures analysis of variance was used for comparison between groups. Multivariate logistic regression with the forward stepwise method and likelihood ratio was then used to determine the associations with poor glycemic control. A p-value of <0.05 was considered statistically significant.
Results
A total of 300 patients with T2DM were selected for inclusion in this study. The mean participant age was 61.9
The mean HbA1c was 7.04% ± 2.07 (range 4.3%–16.0%). A total of 120 respondents had HbA1c ≤7.0%. Therefore, the prevalence of poor glycemic control was 40.0% (95% CI: 34.4–45.8), shown in Table 1. There was a statistically significant association between glycemic control and age (p = 0.020), sex (p = 0.022), rural residence (p = 0.010), and low income (p = 0.018), shown in Table 2.
Prevalence of poor glycemic control.

HBA1c, glycated hemoglobin; SD, standard deviation.
Relationship between sociodemographic profile and glycemic control (N = 300).

SD, standard deviation.
Most respondents (n = 240, 80.0%) reported that they did not consume alcohol and nearly all (n = 293, 97.7%) said they did not smoke tobacco. A total of 180 (60.0%) participants were physically inactive and 54 (18.0%) were obese. We found statistically significant associations between glycemic control and alcohol intake (p = 0.003), tobacco use (p = 0.012), and BMI (p = 0.022), as presented in Table 3.
Relationship between lifestyle habits and glycemic control.
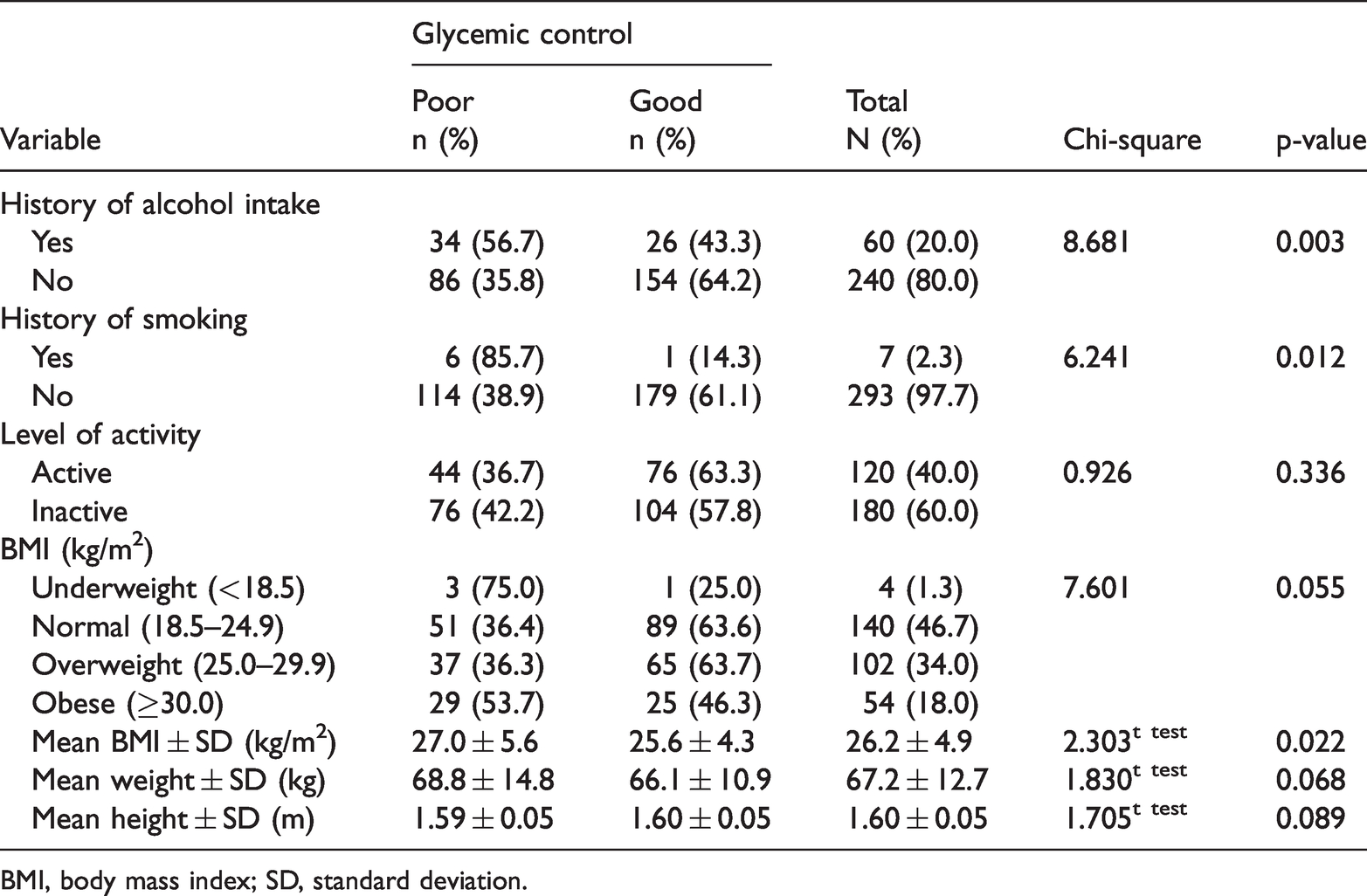
BMI, body mass index; SD, standard deviation.
Hypertension (n = 116, 38.7%), chronic kidney disease (CKD; n = 10, 3.3%), dyslipidemia (n = 48, 16.0%), obesity (n = 54, 18.0%), chronic osteoarthritis (n = 114, 38.0%) and HIV/AIDS (n = 4, 1.3%) were the most common comorbid conditions reported by respondents. There was a statistically significant association between glycemic control and obesity (p = 0.023), as can be seen in Table 4.
Relationship between comorbid conditions and glycemic control.

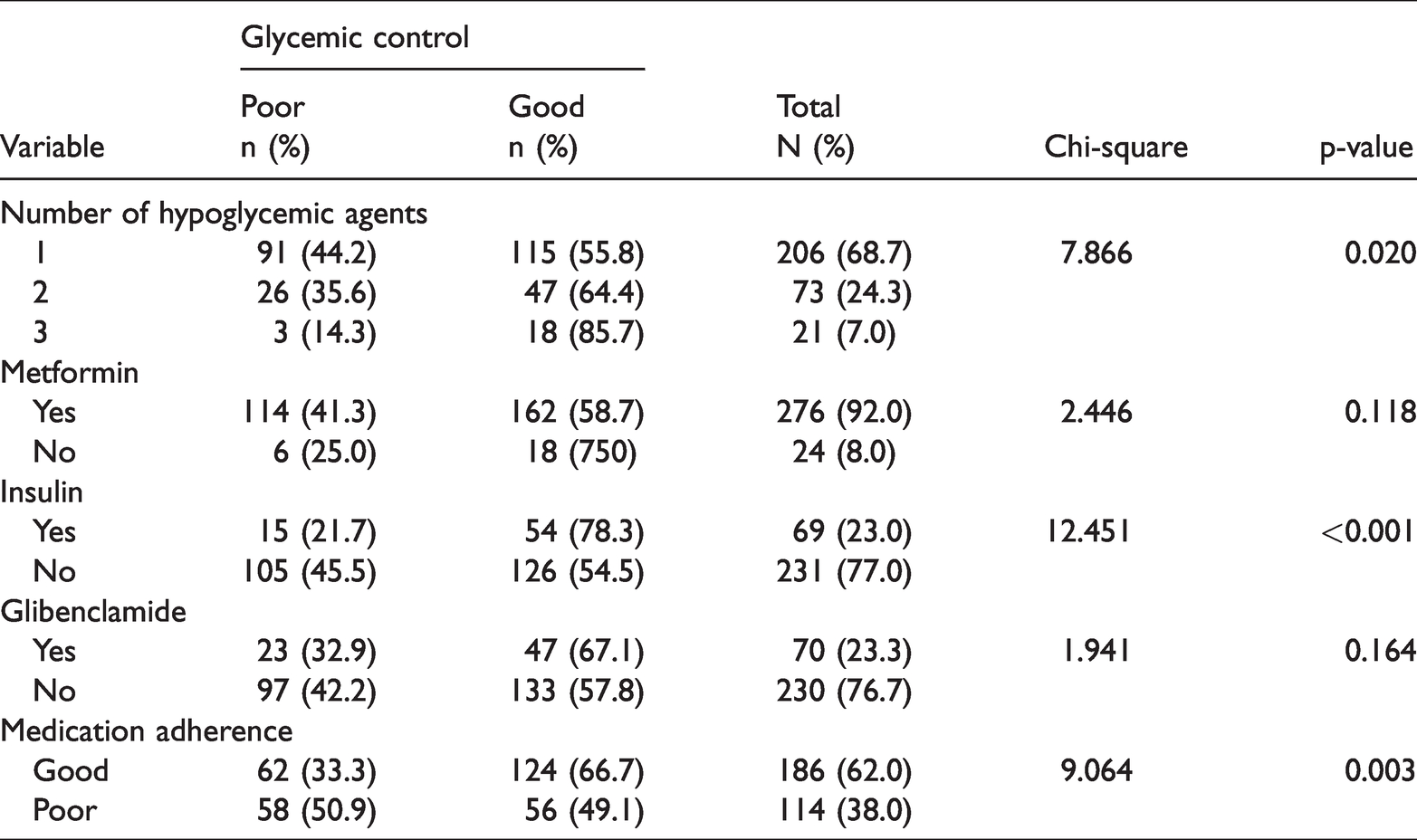
A total of 206 (68.7%) patients were taking a single oral hypoglycemic agent (OHA) and 276 (92.0%) were on metformin. Sixty-nine patients (23.0%) were on insulin therapy and 70 (23.3%) were taking glibenclamide. Most respondents (n = 186, 62.0%) had good adherence to their medications. There was a statistically significant association between glycemic control and the use of hypoglycemic agents (p = 0.020), insulin therapy (p<0.001), and medication adherence (p = 0.003), as shown in Table 5.
Relationship between hypoglycemic agent use and glycemic control.
Using multivariate logistic regression analysis, we found that older age (above 65 years; adjusted odds ratio [AOR]: 2.522, 95% CI: 1.402–4.647), low income (AOR: 1.882, 95% CI: 1.021–3.467), obesity (AOR: 1.734, 95% CI: 1.013–3.401), use of one OHA (AOR: 5.313, 95% CI: 1.394–20.247), use of two OHAs (AOR: 4.890: 95% CI: 1.208–19.789), non-initiation of insulin therapy (AOR: 2.014, 95% CI: 1.269–5.336), and poor medication adherence (AOR: 1.830, 95% CI: 1.045–3.206) were associated with poor glycemic control in this study (Table 6).
Multivariate binary logistic regression for the predictors of poor glycemic control.
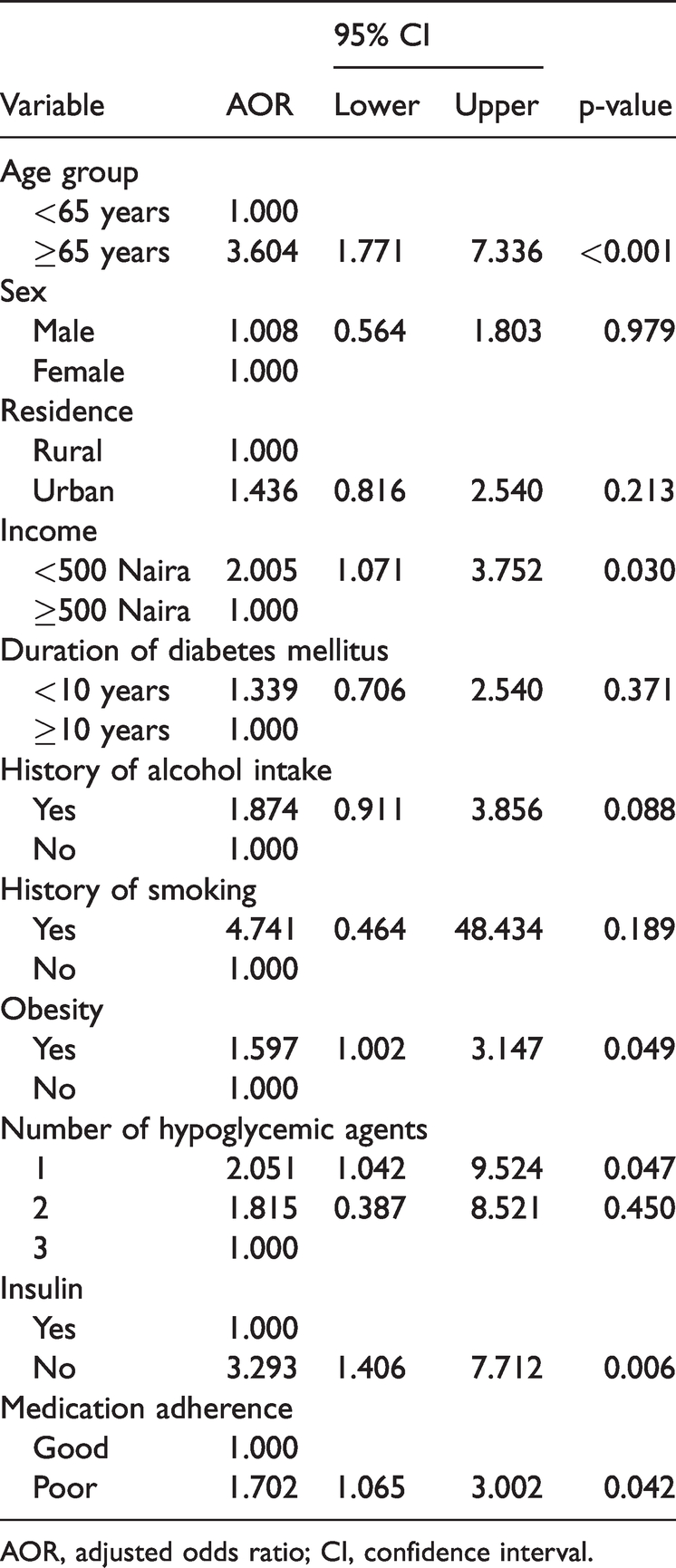
AOR, adjusted odds ratio; CI, confidence interval.
Discussion
In this study, the prevalence of poor glycemic control was 40.0%. This finding is similar to reports of 32.5% by Ajayi et al. 12 and 34.0% and 45.0% in Enugu and Ibadan, Nigeria, respectively.7,8 This could be owing to the similarities in study design, study population, and types of treatment facilities among these studies. Negligible differences may also exist between rural and urban settings, health seeking behaviors, and the purchasing power of rural dwellers in these studies. Our finding regarding the prevalence of poor glycemic control was lower than the prevalence of 50.0% found in Gombe, Nigeria 18 and 72.0% reported in Ethiopia. 6 This discrepancy may be owing to the different settings and the types of index adopted to assess glycemic control. Here, we used HbA1c, which is a more reliable measure than fasting blood glucose used in the above studies. In contrast, our finding was three times higher than the 12.9% prevalence of poor glycemic control in the United States. 5 The lower prevalence in that country could be owing to the high per capital income, functional health insurance scheme, and access to quality health care services.
In this study, the mean age of respondents was 61.9 ± 11.8 years. This is similar to the mean 60.67 ± 13.85 years in the study by Gabriel et al. 19 and 59.8 ± 12.8 years in research by Odusola et al. 20 This similarity may be because most chronic medical conditions like T2DM develop during the middle-age period.
Reports on the influence of sociodemographic profile on glycemic control among patients with T2DM have been inconsistent worldwide.3–6 Both positive and negative relationships have been reported.5–7 In this study, age more than 65 years was associated with poor glycemic control, which is consistent with reports by Bhargava et al. 21 and Chiu et al. 22 This is because pancreatic beta-cell function begins to decline with age, especially in a hyperglycemic state.21,22 This may lead to decreased insulin secretion and poor glucose utilization, and eventually, poor glycemic control.21,23 In contrast, Wahba and Chang found that older patients had better improvement in HbA1c. 24 However, Shani et al. found no relationship between age and glycemic control. 25 In reference to cumulative advantage/disadvantage theory, several explanatory factors affecting health outcomes are subject to modification with aging. 26
Low income earners in this study were affected by multiple adverse conditions of poverty, T2DM. and obesity. This is because low income is often associated with poor access to quality health care, utilization, affordability of medication, and good nutrition. 27 Diabetes has been described as a disease that requires large amounts of money to prevent, manage, and treat. 28 Low-income individuals cannot meet this demand, which could lead to worsening disease progression with resultant high morbidity and mortality, especially in rural settings. Although health insurance currently exists in Nigeria, its 5.0% population coverage is still at the lowest level and is most often only accessible to urban dwellers. 29
Obesity was a risk factor for poor glycemic control in this study. This is consistent with the findings of other studies and may be owing to excess storage of fat and a high glycemic index from overconsumption of carbohydrates, as well as a higher risk of insulin resistance in obese individuals.30,31 Prevention of weight gain is a therapeutic goal for patients with T2DM; 32 therefore, motivation of these patients by clinicians and other stakeholders to achieve an optimal body composition through lifestyle modification could serve as a measure of secondary prevention, to reduce cardiovascular risk and improvement in glycemic control among these individuals. 33
In this study, taking one or two OHAs was associated with poor glycemic control in comparison with taking three or more OHAs. This finding is similar to a report by Adisa and Fakeye showing that patients taking multiple medications had better adherence and therefore better glycemic control; a high pill burden could impact the adherence to medication and could therefore lead to better glycemic control. 34
In this study, 76.0% of respondents were not receiving insulin therapy. The cost of insulin for low wage earners may be responsible for its lower uptake in this study. A similar trend was observed in Lagos, Nigeria where many people do not receive insulin therapy, 35 but this finding was opposite to that of a study in Ethiopia, where most (47.0%) respondents were on insulin therapy. 36 Commencement of insulin therapy once OHAs have failed has long been recommended in the treatment guidelines. 37 Yigazu et al. found that initiation of insulin therapy is associated with good glycemic control. 38
Adherence to diabetic treatment in this study was consistent with the findings of other studies.9,39 In contrast, Onwuchuluba et al. found no significant relationship between adherence and glycemic control. 35 Adherence in this study (62.0%) was relatively good given that the reported medication adherence with a chronic medical condition is approximately 30% to 50%. 37 Patients with T2DM and comorbid conditions require multiple medications and a high adherence rate to achieve good glycemic control, as observed in this study and reported in other studies.9,40 Therefore, clinicians and other stakeholders who manage patients with T2DM should screen for adherence in patients who fail to achieve good glycemic control, after controlling other confounding factors.
As a result of our study, interventions have been put in place at our hospital for respondents identified as having poor glycemic control, which include regular follow-up visits and detailed education about the complications of poor glycemic control. Patients with good glycemic control are counseled on ways to maintain and improve their level of glycemic control. The results of the present study can help to guide stakeholders in how best to improve glycemic control in Nigeria.
Limitations
Although, several risk factors for poor glycemic control were identified in this research, the study is limited by having been performed at a single center and among only 300 patients. This sample size was too small to be representative, and the results of this study might not apply to patients in other centers. Additionally, this study was retrospective in nature; long-term follow-up data were not evaluated. Thus, further research from multiple centers is needed to support our study findings. Other factors, like diet and health insurance, also play important roles in glycemic control, but these were not assessed in this study. Large community-based population studies are needed to identify other factors that can adversely affect good glycemic control in the study area and throughout Nigeria.
Conclusion
In this study, the prevalence of poor glycemic control was 40.0%. Older age, low income, obesity, use of one or two OHAs, non-initiation of insulin therapy, and poor medication adherence were associated with poor glycemic control. These variables may help clinicians to identify patients who are at high risk of poor glycemic control.
Footnotes
Acknowledgements
The authors express their profound gratitude to the management of the Federal Teaching Hospital in Ido Ekiti for providing an atmosphere conducive to the conduct of this research. Appreciation goes to the resident doctors and nurses in the Family Medicine Department.
Author contributions
AOI: Conceptualization of the study, design of the study protocol, data acquisition and analysis, and drafting the initial manuscript.
SMA: Critically revised the protocol for methodological and intellectual content.
OTE, WOI, TAA, TAO: Literature review and review of the manuscript for intellectual content.
This manuscript has not been submitted for prior publication and the requirements for authorship have been met. All the authors have read and approved the final version of the manuscript prior to submission.
Declaration of conflicting interest
The authors declare that they have no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for- profit sectors.
