Abstract
Objectives:
Type 1 diabetes mellitus is a chronic autoimmune disease caused by insufficient production of insulin. Many studies have linked type 1 diabetes mellitus to vitamin D3 deficiency. We investigated the prevalence of vitamin D deficiency in Sudanese children and adolescents with type 1 diabetes mellitus and assessed the impact of vitamin D deficiency treatment on their glycemic control.
Methods:
In 2019–2022, we conducted a quasi-experimental study on 115 children with type 1 diabetes mellitus (1–19 years old) at the Sudan Childhood Diabetes Center. Vitamin D supplements were given orally to deficient patients for 3 months. The concentrations of hemoglobin A1c, fasting blood glucose, insulin dosage, and vitamin D (25-hydroxyvitamin D (25(OH)D)) were measured before and after vitamin D3 administration. One-way ANOVA and paired sample t-tests were used to evaluate the effect of supplementation.
Results:
Only 27% of type 1 diabetes mellitus children were deficient in vitamin D, whereas 31.1% were inadequate and 40.9% were sufficient. The administration of vitamin D supplements slightly improved hemoglobin A1c levels in 67.7% of the patients, but the difference was not significant (mean 10.8 ± 2.1% before, 10.1 ± 2.5% after, p0.05 = 0.199). However, there was a significant decrease in the fasting blood glucose level (mean: 174.978.5–136.759.1 ng/ml; p0.05 = 0.049). Vitamin D levels were significantly increased after treatment (mean = 49.6 ng/mL; t-test = −11.6, 95% CI 40.8–(−28.6); p0.05 = 0.000). After vitamin D3 supplementation, 25.8% of individuals changed their insulin dosage; however, there was no significant variation in insulin needs.
Conclusions:
The prevalence of vitamin D deficiency in children and adolescents with type 1 diabetes mellitus in Sudan is relatively high; incorporating vitamin D supplements in their treatment plan may improve their glycemic control.
Introduction
Diabetes mellitus is a globally challenging chronic disease. It can be classified into three main types based on the causes (i.e., type-1, type-2, and gestational diabetes). The term “type 1 diabetes mellitus” (T1DM) refers to insulin-dependent diabetes mellitus, which is caused by the autoimmune destruction of insulin-producing β-cells in the pancreas with subsequent requirement of exogenous insulin intake for survival. 1 Over 1.5 million children and adolescents between 0 and 19 years of age were reported to have type 1 diabetes (approximately 18% of the global prevalence worldwide), with a rapid expected increase, especially in countries with limited resources. 2 Statistics in Sudan in 2015 showed that the incidence of T1DM among children and young people <20 years old was 10.1/100,000, and the prevalence was 0.74/1000. 3 Many factors contribute to the etiology of T1DM. Genetics, viral infectious agents, intestinal microbiota, vaccines, dietary factors, toxins and chemical compounds, and psychosocial and socioeconomic factors are the leading causes of T1DM. 4
Vitamin D3 is a fat-soluble secosteroid dietary supplement, and vitamin D3 deficiency can be considered a risk factor for developing T1DM. 5 Vitamin D deficiency appears to be a widespread global issue that affects people of all ages. 6 Several studies have been published on vitamin D (25(HO)D) levels worldwide. However, the data are scarce and outdated in regard to African countries, including Sudan. 7 There is no broad consensus as to what constitutes a vitamin D deficiency. The variation in definitions is attributed to the serum levels of vitamin D 25-hydroxyvitamin D (25(OH)D), that is, deficient (⩽20 ng/mL), insufficient (20–29 ng/mL), and optimal (⩾30 ng/mL). 8 A lack of sunlight exposure plays a major role in widespread vitamin D3 deficiency. Poor status and malnutrition, premature birth, sex, low socioeconomic status, urban livelihood, obesity, and advanced age are considered risk factors.8,9
Vitamin D deficiency contributes to both the initial insulin resistance and subsequent onset of diabetes caused by β-cell death. Vitamin D reduces inflammation, which is a major process in inducing insulin resistance. 10 Insulitis is an inflammatory lesion that is considered the histological hallmark of T1DM and consists of the infiltration of pancreatic islets by macrophages, T helper cells (CD4+ or Th cells), and cytotoxic T cells (CD8+), ultimately resulting in the destruction of β-cells. This inflammation plays an important role in T1DM pathogenesis, contributing to β-cell dysfunction and apoptosis through cytokines and chemokines produced by both β-cells and immune cells.11,12 More recent studies have confirmed the direct effects of vitamin D3 (1,25(OH)2D3) on B lymphocyte homoeostasis, including the inhibition of differentiation, maturation of B lymphocytes into plasma cells, and class-switched memory cell differentiation. Other B lymphocyte targets known to be modulated by 1,25(OH)2D3 include IL-10 and CCR10. These effects support the role of vitamin D in the prevention of autoimmune diabetes.13–15 In this study, we investigated the effect of vitamin D3 supplementation on the diets of children and adolescents with T1DM to evaluate glycemic changes.
Methodology
Study design
This was a quasi-experimental design, hospital-based study without a control group (one-group pretest–posttest design). The study was conducted at the Sudan Childhood Diabetes Center, Khartoum, Sudan. The Center serves approximately 3000 children serving in Khartoum State and supervises 25 diabetes clinics in other states of Sudan. It is considered one of the largest diabetes centers in Africa. The study was performed between 2019 and 2022 and included all children (⩽19 years old) with an established diagnosis of T1DM according to the International Society for Pediatric and Adolescent Diabetes (ISPAD) Consensus Guidelines of 2018 (https://www.ispad.org/page/ISPADGuidelines2018) and vitamin D levels below 20 ng/mL who were followed up at the center during the period of the study.
Sample size and study setting
The sample size for the study was calculated using the adequate sample size in the prevalence study formula without a finite population 16 :
where n = sample size, Z = statistics at the 95% confidence level, d = precision (5% or 0.05), and P = expected prevalence or proportion = 0.075% based on the prevalence calculated by Saad et al. 3 for children (aged 6 months–19 years) with T1DM in Khartoum state (2015). The total number of participants was 117 children and adolescents (aged ⩽ 19 years), keeping in mind that this sample size is very limited and would produce a smaller vitamin-D-deficient subgroup with limited demographic and socioeconomic characteristics that do not provide a comprehensive analytical view. The study reports no selection bias of samples.
The 117 T1DM study subjects were divided into three groups based on their serum levels of vitamin D (25(OH)D). Group A = 47 participants (vitamin D sufficient or optimal 30–100 ng/mL), Group B = 37 participants (vitamin D insufficient 20–29 ng/mL), and Group C = 33 participants (vitamin D deficient ⩽ 20 ng/mL). Two participants in Group C withdrew from the study, and 31 participants were included in Group C. Participants were screened for diseases such as diabetes with celiac disease malabsorption (effect on the absorption of vitamin D supplementation) and/or other diseases such as thyroid disease, chronic renal disease, liver disease, or immune deficiency. Patients may also take medications such as vitamin D supplements for the previous 3 months, antiepileptic drugs such as rifampicin, antiretroviral drugs, and steroid therapy that might interact with vitamin D supplementation. These diseases and treatments were considered reasons for exclusion from the study.
Treatment and monitoring of T1DM-vitamin D deficiency
Group C received vitamin D treatment for a period of 3 months at a precalculated daily single dosage, with tolerable upper intake limits (ULs) for age stratification as follows: 1–3 years (2.500 IU), 4–8 years (3.000 IU), and 9–18 years (4.000 IU). 17 Vitamin D was purchased from Vitabiotics (London, UK). The effectiveness of the treatment was ensured by direct monitoring of the correct dose intake and assessing the presence/absence of possible side effects. All the children were monitored for symptoms and signs of vitamin D toxicity (vomiting, polyuria, and gastritis) by the study PI at each follow-up. Most reports suggest a toxicity threshold for vitamin D between 10.000 and 40.000 IU/day; however, symptoms of toxicity are unlikely at daily intakes below 10,000 IU/day. 18 Adequate dietary calcium intake (e.g., milk, yogurt) and a diet rich in vitamin D were ascertained through dietary recall and a checklist. The PI also monitored, advised, and managed participants’ lifestyles, 17 physical activities, and school adherence based on a preprepared checklist (Supplemental File S1) and reported no changes throughout the study that might have affected the outcome after vitamin D supplementation.
The criteria for measuring the effectiveness of the treatment were evaluated based on the concentrations of hemoglobin A1c (HbA1c), fasting blood glucose (FBG), insulin dose based on the type of insulin, adherence to insulin intake, adherence to vitamin D intake, duration of diabetes, glycemic control, and frequency of hospital admission.
Blood sample and data collection
Three-milliliter blood samples were collected from participants and processed based on the manufacturer’s protocol for each test. A chemiluminescence immunoassay was used for the quantification of vitamin D2 and D3 (25(OH)D) in serum samples using a protocol described in the commercially available VIDAS kit (Biomeriéux, Marcy-l’Étoile, France). Whole blood samples were used for quantitative determination of HbA1c using a MISPA-i2 kit (Agappe Diagnostics Ltd., Mumbai, India). FBG was measured using biosystem blood glucose (Biosystem Corp, Crediton, UK). Insulin compliance was measured according to the Morisky Medication-Taking Adherence Scale (MMAS) (4 items). 19 Further data, such as age, sex, weight, and duration of diabetes, were collected from the Center’s patient records.
Statistical methods
Descriptive statistics (Chi-square test for associations) were used to test the significance of associations between demographic parameters and different statuses of vitamin D deficiency, and Pearson correlation, paired sample t-tests, and analysis of variance (one-way ANOVA) were used to analyze the effect of supplementation on vitamin D, HbA1c, and FBG levels at a statistically significant level (p < 0.05). The SPSS software package version 20 (IBM SPSS Statistics for Windows, Version 20.0, IBM Corp, Armonk, NY, USA) was used for the analysis.
Ethical considerations
Legally authorized representatives of the participating children and adolescents were told about the study’s goals and objectives, and they were granted both verbal and written agreement, allowing the use of the obtained data for scientific reasons (Supplemental File S2). The study was approved by the ethical committees of the Ministry of Health, General Administration of Strategic and Information, Innovation, Development, and Scientific Research, Khartoum, Sudan (No: 217) (Supplemental Files S3 and S4). The STROBE statement checklist was used for reporting (Supplemental File S5).
Results
Demographic characteristics of T1DM participants
There were 117 adolescent T1DM participants in this quasi-experimental study conducted between 2019 and 2022; two of them withdrew, and their data were omitted from the study. Fifty-nine (51.3%) of the participants were males. All participants’ ages ranged between 1 and 19 years (mean = 11.6 ± 4.0 years; median = 12 years; 75th percentile = 14 years). Their mean weight was 33.3 ± 14.6 kg, and the 75th percentile was 44 kg. Their families’ socioeconomic status was as follows: 85.2% resided in urban areas, 75.6% owned a house or a flat, and the rest were on rent. Approximately 54.8% of the fathers owned businesses, and 12.2% were not working. A total of 58 (50.5%) of the participants’ mothers held high degrees from universities, while 5.2% were illiterate; however, most of them were stay-at-home mothers (89, 77.4%). Only 51 (44.3%) of the participants had health insurance, and 81.7% of the children went to school. Upon monitoring the children’s physical activity, 52 (45.2%) were moderately active, 43.5% were highly active, and 4.4% showed low physical activity or none. All our participants were asked to identify the frequency with which they consumed certain nutrients. The percentages of participants who regularly consumed dairy products were 66.1% for milk, 68.7% for yogurt, and 53.9% for cheese. Moreover, the intake of vegetables and fruits was 60.9% and 52.2%, respectively, while the consumption of fish (20.9%) was lower than the consumption of 56.5%. Throughout the study, no changes were observed in lifestyle or dietary intake.
Insulin regimens, storage, and diabetes and vitamin D status
The most common insulin regimen used among the study participants (39.1%) was premix insulin and short-acting insulin. Premixed insulin alone was used by 27% of the participants. However, 24.3% of the participants used long-acting insulin in addition to short-acting insulin. Long-acting insulin alone was used by 6.1% of the participants. Other combinations were also reported. The insulin was mainly stored in refrigerators; however, 7.8% of the participants stored it in ice coolers or at room temperature. The participants were assessed for adherence to insulin therapy, which revealed that 33.9% were careless about dose timing, 33% forgot to take their doses, 6.1% stopped taking their insulin when they felt better, and 2.6% stopped taking it when they felt worse.
Diabetes status among participants was assessed through three measures: the duration of diabetes, the number of hospital admissions, and the HbA1c values. The mean duration of diabetes was 4.1 ± 3.3 years, ranging from 0.1 years to 12 years; the 75th percentile was 6 years. The hospital admission frequency ranged between 0 and 30 admissions, with a mean of 1.8 ± 4.0 hospital admissions (75th percentile = 2 times the number of admissions). The mean HbA1c level was 10.4 ± 2.2%, ranging from 5.7% to 15% (75th percentile = 12%). Based on the baseline levels of HbA1c, the data were divided into two groups (i.e., controlled and uncontrolled). Eighty percent of the participants had uncontrolled diabetes. The mean baseline vitamin D concentration was 27.6 ± 10.4 ng/mL, ranging between 9.7 and 61.2 ng/mL. The participants were subsequently divided into groups based on the levels of vitamin D: 40.9% (Group A), 32.1% (Group B), and 27% (Group C) had insufficient vitamin D. There was a reverse correlation between vitamin D levels and the duration of diabetes (r = −0.249; P0.05 = 0.007).
The statistical associations between various vitamin D statuses and all previously mentioned demographic characteristics and the HbA1c status of the participants and their nutritional intake are shown in Tables 1 and 2, respectively.
Association between demographic characteristics of the participants and vitamin D status (N = 115).

Bold values represent significance at P0.05.
Association between participant dietary consumption practices and vitamin D levels (N = 115).

Bold values represent significance at P0.05.
T1DM participants with vitamin D deficiency
Thirty-one T1DM patients (27%) were vitamin D deficient. Monitoring and follow-up following vitamin D administration revealed varying levels of adherence to the treatment strategy. A total of 6.5% were negligent with their dose schedule, 3.2% stopped taking their doses when they felt better, and 1.7% stopped when they felt worse.
Prior to and after vitamin D administration, HbA1c and FBG levels were tested in triplicate. Although there were no significant changes in HbA1c levels before or after vitamin D administration (mean = 10.8 2.1 ng/mL, 10.1–2.5 ng/mL; P0.05 = 0.199), 67.7% of patients demonstrated improvement in their HbA1c status, with HbA1c levels decreasing from baseline. However, there was a significant reduction in blood glucose levels upon correcting vitamin D deficiency with supplementation (mean = 174.9 ± 78.5 mg/dL, 136.7 ± 59.1 mg/dL; P0.05 = 0.049), with an obvious significant increase in vitamin D levels (mean = 14.9 ± 3.3 ng/mL, 49.6 ± 16.2 ng/mL; P0.05 = 0.000); however, there was no significant correlation (r = 0.071, P0.05 = 0.704) (Table 3).
Effect of vitamin D on the glycemic control of T1DM-vitamin D deficiency patients (N = 31).

Bold values represent significance at P0.05.
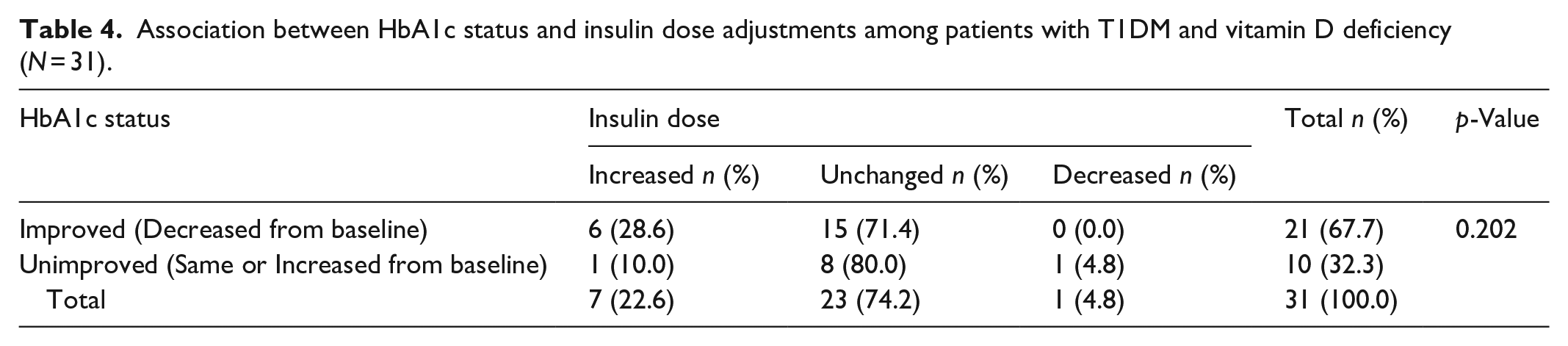
Participants who received vitamin D supplementation had their insulin doses revised and adjusted. The changes introduced provided no significant association between insulin dose and HbA1c status (Table 4).
Association between HbA1c status and insulin dose adjustments among patients with T1DM and vitamin D deficiency (N = 31).
Discussion
Vitamin D affects the etiology of many diseases, such as diabetes. To our knowledge, this is the first study to investigate the relationship between vitamin D levels and pediatric T1DM in Sudan.
A global perspective provided by a comprehensive meta-analysis showed that vitamin D deficiency is prevalent in children and/or adolescents with T1DM. 20 This finding is consistent with the prevalence reported in our study (27%). This percentage is also comparable to the 20% reported by Al Senani et al. 21 and the 14.8% reported by Giri et al. 22 However, it was lower than the prevalence reported in studies conducted in Egypt (53.3%) 23 and India (63%). 24 Moreover, in a study conducted by Al Kharashi and colleagues on 100 Saudi children with T1DM, 70% were reported to suffer from reduced levels of vitamin D. 25
The relatively high prevalence of vitamin D deficiency in Sudan might be attributed to genetic polymorphisms and a predisposition, especially because vitamin D is controlled by approximately 200 genes and is influenced by the dark skin complications of African people due to the increase in melanin pigmentation, which lowers the absorption of UV light, hence decreasing the ability of the skin to synthesize vitamin D.26,27 Nonetheless, a previous study performed among Sudanese women revealed an apparent insufficiency, with more than 80% showing that sunray exposure is inadequate; however, the authors concluded that sun-ray exposure may not be critical, and other factors should be carefully evaluated. 28
One of the important nonskeletal effects of vitamin D is its role in glucose homeostasis. Vitamin D appears to protect pancreatic β-cell function and improve insulin sensitivity in target organs.14,29 While several studies have shown the impact of vitamin D status on glycemic control in adults, 30 few studies have been conducted in children. 22 Poor glycemic control can be indicated by the measurement of HbA1c, which was not associated with the presence or severity of vitamin D deficiency according to our results. Although we reported an improvement in glycemic control by HbA1c measurement (67.7% of participants) after vitamin D treatment where the numbers dropped from baseline, there was no significant difference in HbA1c before and after supplementation. Such results could be attributed to the doses and the duration of supplementation as well as the variation in insulin sensitivity which might require further adjustment. 31 This was consistent with other reports in which improvements in glycemic control measured by HbA1c levels were documented but without statistical significance.23,24,32 In a systematic meta-analysis review conducted in 2022, Nascimento et al evaluated the effect of vitamin D supplementation on HbA1c levels and reported a significant improvement in glycemic control in 50% of the analyzed studies. 33 However, there is yet to be consistent evidence on the effect of vitamin D supplementation on the glycemic control of children/adolescents with T1DM, as other studies have shown that vitamin D supplements fail to improve glycemic control.21,34 FBG was reduced after correcting vitamin D deficiency in our type 1 diabetic patients, whereas daily insulin requirements changed after vitamin D treatment by a high percentage, but the difference was not significant, as in previously published reports.24,35
Although Balla and his colleagues reported a significant improvement in the metabolic control of children and adolescents with type 2 diabetes in Sudan after vitamin D supplementation, such improvements were not observed in our type 1 diabetic subjects. 36 Furthermore, a systematic meta-analysis conducted in 2015 showed that there is no supporting evidence for the notion that vitamin D supplementation can improve hyperglycemia, β-cell secretion, or insulin sensitivity in patients with T2DM. 37 The poor glycemic control reported in our study can also be attributed to carelessness and poor compliance with insulin intake by 33.9% of participants with different vitamin D statuses.
Diabetes is a complicated chronic disease controlled by many factors, including genetics and environmental factors. Socioeconomic factors are key predictors of pediatric T1DM outcomes. Many layers have been identified: (1) diabetes care (i.e., insulin, physical activity, nutrition management, frequent clinic visits, glucose monitoring, and technology used), (2) individual factors (i.e., age, duration of diabetes, family relationships, and genetic factors), (3) social determinants of health (i.e., economic stability, health and health care, education, and social and community context), (4) society, and (5) public policy (i.e., health insurance, value-based care, and research funding).38,39 Many of these factors were investigated in this study in relation to diabetes and vitamin D status. Maternal education is a main element that affects the outcome of diabetes and can be associated with glycemic control through the care and supervision of children and adolescents at home and through follow-ups with schools. Age was also a factor associated with vitamin D deficiency in our T1DM patients. According to Zabeen et al., 40 vitamin D deficiency was more common in older individuals, whereas younger individuals had a greater prevalence of vitamin D sufficiency. It was also reported that vitamin D deficiency is significantly more common in older children who have diabetes for a longer period of time than in nondeficient patients. 41
Although all other demographic and socioeconomic characteristics investigated were not significantly associated with vitamin D status, other studies reported otherwise. For instance, in Saudi Arabia and Egypt, the prevalence of vitamin D deficiency is associated with female sex.25,42 Al-Othman et al. 43 reported that in adolescents with age-matched comparisons, groups with the same amount of sun exposure and moderate or high physical activity had higher vitamin D levels than other groups. In Bangladesh, residential status and location were associated with vitamin D deficiency, and 59.5% of patients residing in urban locations were deficient due to the air pollution that limited their outdoor activity. They also reported that there were significant correlations between vitamin D status and the number of hospital admissions and between vitamin D status and the socioeconomic status of parents. 40 Svoren et al., 41 however, indicated in their study that the duration of diabetes significantly affects vitamin D status, where a longer diabetes duration is significantly associated with vitamin D deficiency. Moreover, parents’ respective jobs and socioeconomic status have no effect. Unlike what was reported in a Saudi Arabian study, the dietary intake of yogurt, fruits, and fish was significantly associated with the various statuses of vitamin D. This might be attributed to the variation in these dietary sources among the various economic classes in Sudan, which affects the regular intake of these nutrients. 25
The outcome contrast observed in relation to studies conducted on vitamin D deficiency among T1DM children and adolescent subjects in regard to the status of serum vitamin D can be explained by the variation in the intervention doses and duration. This may also be related to the variation in study design and sample size, which was evident in Yang et al., 20 who reported that the proportion of patients with vitamin D deficiency in a case-control study tended to be greater than that in other studies, which contributed to the variation in the results after vitamin D supplementation.
Overall, many limitations were associated with the study and could be summarized as follows: (1) the use of a nonstandardized insulin regimen, (2) the use of different types of insulin analogs, (3) the relatively small sample size limiting the detailed analytical breakdown of outcomes according to demographic and socioeconomic characteristics and limiting the generalizability of the study based on those characteristics, and (4) the short duration of the study, as such a study might require a longer duration for the treatment to produce more accurate and comprehensive results. This limitation does not negate the strength of this study because it is the first study on the effect of vitamin D supplementation on the glycemic profile of adolescent children suffering from T1DM in Sudan. The continuous monitoring and follow-up with participants throughout the study provided accurate data and close monitoring and anticipation for possible confounders that could present with any lifestyle or dietary changes.
Conclusion
The high prevalence of vitamin D deficiency and insufficiency among T1DM children in Sudan highlights the importance of regular vitamin D screening and maintaining rigorous attention to glycemic control. Moreover, vitamin D deficiency was significantly more common in older children and participants who had illiterate mothers and poor nutrition, which could be addressed through educational and awareness programs that emphasize the importance of nutrition and its effects on children’s health and how to provide proper care for children and adolescents with type 1 diabetes.
Supplemental Material
sj-docx-3-smo-10.1177_20503121241242931 – Supplemental material for Does vitamin D supplementation benefit patients with type 1 diabetes mellitus who are vitamin D deficient? A study was performed at the Sudan Childhood Diabetes Center from 2019 to 2022
Supplemental material, sj-docx-3-smo-10.1177_20503121241242931 for Does vitamin D supplementation benefit patients with type 1 diabetes mellitus who are vitamin D deficient? A study was performed at the Sudan Childhood Diabetes Center from 2019 to 2022 by Hiba Abdelmunim Suliman, Abdalla Omer Elkhawad, Omer Osman Babiker, Yousif Mohammed Alhaj, Kholod Hamad Eltom and Asim Ahmed Elnour in SAGE Open Medicine
Supplemental Material
sj-pdf-1-smo-10.1177_20503121241242931 – Supplemental material for Does vitamin D supplementation benefit patients with type 1 diabetes mellitus who are vitamin D deficient? A study was performed at the Sudan Childhood Diabetes Center from 2019 to 2022
Supplemental material, sj-pdf-1-smo-10.1177_20503121241242931 for Does vitamin D supplementation benefit patients with type 1 diabetes mellitus who are vitamin D deficient? A study was performed at the Sudan Childhood Diabetes Center from 2019 to 2022 by Hiba Abdelmunim Suliman, Abdalla Omer Elkhawad, Omer Osman Babiker, Yousif Mohammed Alhaj, Kholod Hamad Eltom and Asim Ahmed Elnour in SAGE Open Medicine
Supplemental Material
sj-pdf-2-smo-10.1177_20503121241242931 – Supplemental material for Does vitamin D supplementation benefit patients with type 1 diabetes mellitus who are vitamin D deficient? A study was performed at the Sudan Childhood Diabetes Center from 2019 to 2022
Supplemental material, sj-pdf-2-smo-10.1177_20503121241242931 for Does vitamin D supplementation benefit patients with type 1 diabetes mellitus who are vitamin D deficient? A study was performed at the Sudan Childhood Diabetes Center from 2019 to 2022 by Hiba Abdelmunim Suliman, Abdalla Omer Elkhawad, Omer Osman Babiker, Yousif Mohammed Alhaj, Kholod Hamad Eltom and Asim Ahmed Elnour in SAGE Open Medicine
Footnotes
Acknowledgements
We acknowledge the help in participants’ follow-ups and blood testing provided by staff members of the Sudan Childhood Diabetes Center, Khartoum, Sudan, throughout the period of the study.
Authors’ contributions
Conceptualization and writing of the original draft were performed by HAS and AOE. The data collection, investigation, and formal analysis were conducted by HAS, OBO, and YMA. The data were validated and visualized by HAS, AAE, and KHA. The final draft was edited by AAE. All authors revised the manuscript and approved the final draft.
Availability of data and materials
Data from this study will be available upon request and will be provided by Dr. Hiba Abdelmoniem Suliman Elsheikh, Email: hooby1983@gmail.com
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from the Ministry of Health, General Administration of Strategic and Information, Innovation, Development, and Scientific Research, Khartoum, Sudan (No: 217).
Informed consent
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

