Abstract
The prevalence of metabolic dysfunction-associated fatty liver disease (MAFLD) is significant, impacting almost one-third of the global population. MAFLD constitutes a primary cause of end-stage liver disease, liver cancer and the need for liver transplantation. Moreover, it has a strong association with increased mortality rates due to various extrahepatic complications, notably cardiometabolic diseases. While MAFLD is typically correlated with obesity, not all individuals with obesity develop the disease and a significant percentage of MAFLD occurs in patients without obesity, termed lean MAFLD. The clinical features, progression and underlying physiological mechanisms of patients with lean MAFLD remain inadequately characterized. The present review aims to provide a comprehensive summary of current knowledge on lean MAFLD and offer a perspective on defining MAFLD in individuals with normal weight. Key to this process is the concept of metabolic health and flexibility, which links states of dysmetabolism to the development of lean MAFLD. This perspective offers a more nuanced understanding of MAFLD and its underlying mechanisms and highlights the importance of considering the broader metabolic context in which the disease occurs. It also bridges the knowledge gap and offers insights that can inform clinical practice.
Introduction
The increasing worldwide incidence of obesity and type 2 diabetes mellitus (T2DM) has led to the expansion of non-communicable chronic diseases, resulting in a significant strain on healthcare systems in developed and developing countries. 1 The aforementioned increases can be attributed to numerous factors, including the ageing population and the prevalence of an obesogenic environment. Such factors promote physical inactivity and high-calorie food intake in relation to expenditure.2,3 According to a recent study, the estimated proportion of inadequate levels of physical activity was approximately 23.3% in 2010 4 and has shown an upwards tendency over the years. 5
Metabolic dysfunction-associated fatty liver disease (MAFLD) is one of the non-communicable diseases that affect nearly 38.77% of the global population.6–8 The prevalence of MAFLD has significantly increased over the past few decades, fuelled by the increase in obesity and diabetes. MAFLD is the fastest-growing contributor to the development of cirrhosis and liver cancer and is an indication for liver transplantation. Roughly 20 million individuals are susceptible to mortality resulting from liver disease.6–8 MAFLD is not only linked to adverse liver-related consequences but also encompasses extrahepatic manifestations, including diabetes, cardiovascular diseases, extrahepatic cancers, chronic kidney disease, osteoarthritis, obstructive sleep apnoea, gallbladder disease and psoriasis.9,10
Nevertheless, not all individuals exposed to this obesogenic environment develop MAFLD, and not all patients with the disease have obesity. Despite significant advances in understanding the pathogenesis of MAFLD, one of the key lacunas is identifying and defining characteristics and mechanisms of MAFLD development among lean individuals, referred to as ‘lean MAFLD’ or ‘MAFLD in individuals of normal weight’. Lean patients with MAFLD are known to have distinct pathophysiological characteristics. 11 However, these characteristics have yet to be thoroughly specified to guide clinical practice in terms of therapy choice.
This review aims to summarize the current knowledge about lean MAFLD and provide a perspective on the definition of MAFLD as an ideal framework for understanding the disease phenotype in individuals with normal weight. This process incorporates the concept of metabolic health and flexibility, which links states of dysmetabolism to lean MAFLD. By providing an overview of the disease and its characteristics, this review seeks to bridge the gap in knowledge and offer insights that can inform clinical practice.
Diagnosis of MAFLD in lean individuals
The use of body mass index (BMI) as a proxy for adiposity in the diagnosis and categorization of obesity has been prevalent due to its strong association with adiposity data obtained from population studies. The conventional criteria for defining overweight and obesity rely on BMI thresholds that vary according to ethnicity. In Western societies, those falling in the BMI range of 18.5–24.9, 25–29.9 and 30 kg/m2 or more are categorized as having normal weight, overweight and obese, respectively. By contrast, in the Asian community, persons whose BMI falls in the range of 18.5–22.9, 23.0–24.9 and above 25.0 kg/m2 are considered as having normal weight, overweight and obese, respectively.
In 2020, the concept of MAFLD was introduced, accompanied by a set of practical and easily applicable positive diagnostic criteria.12–15 According to the international consensus recommendation for the definition of MAFLD, patients who exhibit hepatic steatosis and fulfil one of three criteria for being overweight or obese as determined by ethnic-specific BMI standards or have diabetes are categorized as having MAFLD. Conversely, lean individuals with hepatic steatosis must exhibit clinical evidence of metabolic dysfunction to be diagnosed with MAFLD,12–14 based on the presence of at least two out of the seven identified metabolic risk factors. The factors include waist circumference, blood pressure, levels of plasma triglycerides, levels of plasma high-density lipoprotein (HDL-C), presence of prediabetes, the homeostasis model evaluation of insulin resistance score and levels of plasma high-sensitivity C-reactive protein (hs-CRP), as a marker of systemic low-grade inflammation.
One notable characteristic of the MAFLD criteria is its provision of an operational description for the condition, focusing on its defining features rather than its exclusions. As a result, MAFLD can coexist with several other liver diseases, hence influencing their clinical presentations and natural progression. 16 Since the incorporation of MAFLD into the diagnostic lexicon, numerous studies have employed the lean/normal-weight MAFLD classification. They have presented compelling evidence that supports the robustness and potential advantages of the MAFLD classification over Nonalcoholic fatty liver disease (NAFLD) in terms of complications, prognosis and disease outcome. 17 The implementation of this change represents a significant advancement in adopting a patient-centric approach to addressing this condition as well as in shaping ideas on pioneering trial designs for MAFLD.18,19
Clinical characteristics of MAFLD in lean individuals
Epidemiology
One of the initial reports on the prevalence of lean MAFLD among 932 Korean participants showed that 23.4% of the studied population had MAFLD, and 16.1% of normal-weight people (BMI <25 kg/m2) showed evidence of the disease. 20 Subsequently, the data on the prevalence of lean MAFLD have expanded, and MAFLD in lean individuals has been reported in several populations worldwide. The frequency of lean MAFLD varies significantly, ranging from 5% to 26% of the population, and accounts for 15–50% of MAFLD cases 21 (Figure 1). Another study using data from 13,640 adults derived from the third National Health and Nutrition Examination Survey conducted between 1988 and 1994 revealed that the collective occurrence of MAFLD was 19%, of which 46% were categorized as non-obese. 22 In a meta-analysis and comprehensive review including a pooled analysis of 3,320,108 people, the collective prevalence of MAFLD was found to be 38.77%. Among the study population, 5.37% of lean persons and 29.78% of individuals without obesity were diagnosed with MAFLD. 23 An age and gender preference in lean MAFLD was reported, which was more common in males over 65 years of age compared with their younger female counterparts. 22 MAFLD prevalence among lean individuals varies by race/ethnicity, with higher rates in Hispanics and lower in African Americans, similar to the overall MAFLD pattern.24,25 Lean MAFLD is prevalent and also associated with disease severity in people with human immunodeficiency virus (prevalence 14% and representing 35% of all MAFLD cases in 1511 patients with HIV mono-infection). 26

Global prevalence of non-obese MAFLD among the MAFLD population and the general population.
Data on the incidence of MAFLD among lean individuals are limited, with a reported incidence of 24.6 [95% confidence interval (CI): 13.4–39.2] cases per 1000 person-years in populations without obesity based on pooled data from five studies. 27 Collectively, although our current knowledge of the epidemiology of lean patients with MAFLD has limitations, MAFLD among individuals without obesity still represents a significant proportion of the MAFLD population.
Histological characteristics
The histopathological features of MAFLD in patients with normal weight are widely believed to resemble those observed in the general MAFLD population. However, they may be milder in severity. 11 A recent study of 1339 participants with biopsy-confirmed MAFLD (14.4% of them are lean) in Australia, Italy, the United Kingdom and Spain showed that lean MAFLD patients had milder histological disease, including lower steatohepatitis, less advanced fibrosis and lower prevalence of T2DM compared with non-lean patients. 28 Nevertheless, limited number of other cross-sectional studies have shown that individuals with lean MAFLD exhibit more severe liver histology, higher rates of advanced fibrosis, balloon formation, lobular inflammation and steatohepatitis compared with patients with non-lean MAFLD.29–31
A meta-analysis comprising eight studies encompassing 1441 individuals revealed that 39% of individuals with lean MAFLD exhibited steatohepatitis, whereas 29.2% displayed clinically significant fibrosis at stage 2 or higher. In contrast, obese MAFLD individuals demonstrated higher rates of steatohepatitis (52.9%) and clinically significant fibrosis (38.3%). 27 A comparable observation of MAFLD was also documented in another meta-analysis demonstrating that compared with lean individuals, patients with overweight or obesity had a notably higher likelihood of developing metabolic steatohepatitis and advanced fibrosis scores. 32
Variabilities were noted among studies, likely attributable to the heterogeneity of diagnostic criteria employed in individual reports as well as the differing study subject selection criteria. However, the preponderance of available evidence suggests that lean patients with MAFLD tend to have more favourable histological and metabolic characteristics when compared with their obese counterparts.
Prognosis
There is a scarcity of data about the extended-term prognosis of MAFLD in patients who have a normal body weight (Table 1). A preliminary investigation, including a cohort of 483 individuals diagnosed with biopsy-confirmed MAFLD and with a median duration of observation spanning 11 years, revealed that patients with a normal weight exhibited greater rates of liver transplantation compared to their obese counterparts. 33 According to recent research, it was shown that non-obese individuals with MAFLD had a greater cumulative 20-year incidence of all-cause death (33.2%) compared to their obese counterparts (28.8%). 22 Similarly, a separate investigation including a substantial sample size of 169,303 individuals from the comprehensive French countrywide Constances cohort revealed that lean individuals with MAFLD exhibit a heightened severity in terms of fibrosis, the advancement of hepatic ailments, chronic renal disease and overall mortality. 34 Consistently, a meta-analysis of 10 cohort studies involving 109,151 patients with MAFLD showed a higher risk of liver-related mortality in patients with lean MAFLD than those with non-lean MAFLD and comparable risks for all-cause mortality and cardiovascular mortality. 35 Notably, a retrospective analysis of adult patients with MAFLD who underwent liver transplantation was conducted on data obtained from the United Network for Organ Sharing. It revealed that individuals classified as having a normal BMI had a higher probability of being removed from the wait list compared with their obese counterparts. 36
Studies on lean MAFLD prognosis compared to their non-lean counterpart.

BMI, body mass index; MAFLD, metabolic dysfunction-associated fatty liver disease.
MAFLD in lean individuals is linked with an increased risk of various extrahepatic complications. In a prospective cohort study using UK Biobank that included 325,129 participants with a median follow-up of 12.8 years, lean patients with MAFLD had a higher risk of myocardial infarction and stroke, similar to MAFLD patients with diabetes or obesity. 43 A similar outcome was also seen in a large Korean cohort, revealing that those with lean MAFLD or diabetic MAFLD had a greater susceptibility to cardiovascular diseases compared with those with overweight/obese MAFLD, regardless of the presence of metabolic abnormalities or comorbidities. 44
A meta-analysis of eight studies involving a sample size of 56,745 people diagnosed with MAFLD classified 11% as lean. It revealed a significant association of the presence of lean MAFLD with an increased risk of hepatic [relative risk (RR): 1.77; 95% CI: 1.15–2.73], pancreatic (RR: 1.97, 95% CI: 1.01–3.86) and colorectal malignancies (RR: 1.53; 95% CI: 1.12–2.09) compared with non-lean MAFLD. 45 Moreover, a recent investigation including 124 consecutive individuals who had colonoscopies during their health check-ups revealed that MAFLD was the only independent predictor linked to the occurrence of colorectal adenoma [with an odds ratio (OR) of 3.19; 95% CI: 1.494–7.070]. The presence of colorectal adenoma was shown to be significantly linked with non-obese MAFLD, which emerged as the only independent predictor with an OR of 3.35 among the three subtypes of MAFLD. 46 Another study of 147 patients found that the presence of MAFLD is associated with more than a twofold increase in the risk of recurrence of oesophageal squamous cell carcinoma, independent of other risk factors. The cumulative incidence of recurrence was significantly higher in those without obesity than in the patients with obesity and MAFLD. 47
In summary, lean patients afflicted with MAFLD tend to experience a worse long-term outcome compared with non-MAFLD patients, as well as those with overweight or obesity. The interesting aspect is in the seeming paradox of heightened liver-related morbidity and death in individuals with normal weight and with less pronounced initial liver impairment. Therefore, further studies are required to clarify this clinically relevant paradox. A better understanding of the pathophysiology of this condition, rather than relying solely on BMI, is necessary to address this matter.
Pathophysiology of MAFLD in lean patients
Genetic contribution
The precise pathophysiological mechanisms underlying lean MAFLD remain uncertain. The interindividual variability of risk for MAFLD within a common environment and how individuals react to these environmental stimuli are influenced, suggesting that, at least in part, genetic profile plays a crucial role in the development and progression of this condition.48,49 MAFLD is a heritable disease, and its estimated heritability is 50% based on findings from twin studies. 50 Although genome-wide association studies (GWAS) have been successful in identifying gene loci linked to the risk of developing and progressing MAFLD, the specific variant(s) associated with lean people affected by MAFLD still provides a challenge to pinpoint. 51
In a recent GWAS, a cohort of 1275 metabolically healthy Japanese individuals with fatty liver disease, who were of normal weight, was compared with a control group of 1411 individuals without fatty liver disease. The analysis was adjusted for factors such as age, sex and alcohol consumption. The findings of this study indicated that the human leukocyte antigen locus may be a potential genetic region associated with susceptibility to fatty liver disease in lean patients. Furthermore, it was suggested that alterations in the gut microbiota may influence this association. 52 A subsequent investigation utilizing whole-exome sequencing was conducted on a limited group of six lean individuals of Indian origin diagnosed with MAFLD, along with two control subjects who did not have MAFLD. The findings from this study were further confirmed by replicating the experiment on a larger validation cohort consisting of 191 lean patients with MAFLD and 105 control subjects. The study identified a specific variant in the phosphatidylethanolamine N-methyltransferase (PEMT) gene that is associated with the development of MAFLD in lean individuals. 53 Nevertheless, reproducing these results in more extensive and ethnically diverse cohorts is essential. Mice lacking the PEMT gene and subjected to a diet rich in fat and fructose exhibit susceptibility to fatty liver and steatohepatitis. However, these mice are resistant to obesity and insulin resistance. 54 Consequently, they may be used as genetic models for lean MAFLD.
Other studies have examined the involvement of previously known genetic variants of MAFLD in individuals belonging to the lean subgroup. For instance, certain studies have investigated the rs738409 C > G allele, which encodes the patatin-like phospholipase domain-containing protein 3 (PNPLA3) I148M variation. This genetic variant has been widely recognized as the most robust genetic risk variant for MAFLD across various ethnicities and geographic regions. 49 Reports have suggested a higher prevalence of rs738409 G allele among lean MAFLD patients compared with obese MAFLD patients. However, this finding has not been confirmed in other studies.55,56 Additionally, the PNPLA3 risk allele is associated with the risk of steatohepatitis as well as the progression to more advanced fibrosis stages (stage 2 or beyond) among lean individuals with MAFLD.28,55,56
The presence of the rs58542926 C > T variant in the transmembrane 6 superfamily member 2 (TM6SF2) gene locus has also been found to be of a greater prevalence in lean than in obese MAFLD patients.56,57–59 A more recent study of 1007 individuals diagnosed with MAFLD indicated an association between the presence of TM6SF2 rs58542926 T and PNPLA3 rs738409 G variants and lean MAFLD. 60
The rs641738 C > T variant of the membrane-bound O-acyltransferase domain-containing 7 (MBOAT7) gene has been identified as a genetic factor that is related to an increased risk of developing MAFLD, hepatic inflammation and fibrosis. This variant is also associated with an elevated risk of progression to hepatocellular carcinoma and the regulation of toll-like receptor response not only in individuals with MAFLD but also in those with other liver diseases.61–63 Nevertheless, a study in Austria revealed no statistically significant difference in the prevalence of the MBOAT7 rs641738 C > T variant between lean MAFLD patients and those diagnosed with obesity-related MAFLD. 30 A separate study including 6939 Korean adults undergoing health examinations confirmed the association of PNPLA3 and TM6SF2 genetic variants with the presence of fatty liver among lean patients. Nevertheless, no correlation was observed between the occurrence of fatty liver and the genetic variants MBOAT7 (rs641738), HSD17B13 (rs72613567), MARC1 (rs2642438) or AGXT2 (rs2291702) among lean patients. 64
The association between interferon lambda 3/4 variants with fibrosis severity in patients with viral hepatitis has been discerned. Subsequently, these variants are also found to be linked to histological severity in patients with MAFLD, particularly in lean people.65–67 A genetic risk score composed of five genetic variants (PNPLA3 rs738409 C/G, TM6SF2 rs58542926 C/T, GCKR rs1260326 T/C, MBOAT7 rs641738 C/T and HSD17B13 rs72613567 T/TA) was reported to amplify the impact of MAFLD of intrahepatic and extrahepatic events. However, the genetic risk score did not influence the risk in lean patients with MAFLD. 68 A recent study suggested that lean MAFLD could be stratified into two broad subtypes: Type 1, which occurs in subjects with visceral adiposity and insulin resistance, and type 2, which occurs in subjects with hepatic steatosis due to monogenic diseases. 69
Epigenetics
The human epigenome serves as a connection between an individual’s genetic makeup and environmental stimuli, and it undergoes dynamic regulation throughout the individual’s lifespan. 70 Epigenetic modifiers that have been identified include DNA methylation, histone changes, chromatin remodelling and non-coding RNAs. 70 The investigation of epigenetic alterations implicated in the progress of MAFLD in individuals with a lean phenotype remains scarce.
In a study of 53 individuals with normal weight diagnosed with MAFLD, a reduction in the levels of serum histone variants macroH2A1.1 and macroH2A1.2 was observed in lean patients with MAFLD. These alterations were not observed among non-lean patients, suggesting that these specific histone variants may potentially function as non-invasive biomarkers in lean individuals diagnosed with MAFLD. 71 The involvement of dysregulated miRNA expression patterns has been suggested in the pathogenesis of MAFLD, and various miRNA expression profiles have been linked to the disease. A recent study shows that the levels of serum miR-4488 expression were higher in lean patients with MAFLD than in obese MAFLD patients and healthy controls. 72 Another study has demonstrated that Escherichia fergusonii contributes to the development of non-obese MAFLD by disrupting the hepatic lipid metabolism of the host organism via the action of its messenger RNA molecule, namely msRNA 23487. 73
Epigenetics has considerable adaptability in response to dietary and stress-related factors. Consequently, an adverse intrauterine environment can initiate foetal metabolic programming, thereby augmenting the susceptibility to developing MAFLD throughout adulthood. 48 The occurrence of intrauterine growth retardation is linked to heightened levels of insulin resistance and disease activity on histology, irrespective of BMI. The majority of children diagnosed with fatty liver, approximately 80%, who were born with short gestational age, exhibit insulin resistance at an average age of 11 years. This condition occurs despite having a normal BMI and a notably low occurrence of metabolic abnormalities. 74
Metabolic health: Not all fats are the same
Although no universal agreement has been reached over the specific criteria and threshold levels used to establish metabolic health, numerous studies have provided unequivocal evidence of the fundamental role of metabolic health in the pathogenesis of MAFLD. It is one of the key factors that drive the transformational redefinition of fatty liver disease and the generation of MAFLD criteria, recognizing the pivotal role of metabolic health status in this context. 75 The predominant definition of metabolic health is characterized by the lack of insulin resistance, the absence of subclinical inflammation as designated by hs-CRP and the presence of just one of the criteria for the metabolic syndrome as defined by the Adult Treatment Panel III criteria.76,77
The likelihood of developing steatohepatitis and severe fibrosis escalates in a dose-dependent manner as the number of metabolic risk factors increases. 78 A study of over 1000 patients with biopsy-confirmed MAFLD has consistently shown that metabolic health has a more substantial influence on the likelihood of developing metabolic steatohepatitis and advanced fibrosis, regardless of BMI. 78 This finding suggests that the impact of metabolic health on the advancement of liver disease is more profound than that of BMI. This impact is likely mediated by unfavourable distribution of body fat and/or unidentified factors coupled with a sustained phase of subclinical systemic inflammation. 78 Comparable results have been seen in populations of Asian and Mexican descent.79–81
Moreover, the presence of MAFLD may potentially contribute to, or at least demonstrate an association with, the progression of an individual’s metabolic health from a state of metabolic health to a state of metabolic ill-health, irrespective of factors such as age, gender, BMI, lifestyle choices, insulin resistance and other components of metabolic syndrome. The observed effects are more profound in those with a lower BMI and body fat mass in contrast to those with a higher BMI and body fat mass. 82
Mechanisms for regulation of metabolic health
The exact mechanisms in the maintenance or alteration of metabolic health remain incompletely understood. A multitude of components, encompassing genetic and epigenetic, enterohepatic circulation and gut microbiota, and lifestyle factors such as dietary quality and quantity, the pattern of alcohol intake and physical activity, are anticipated to engage in intricate and dynamic interactions. The outcome of the interaction of the aforementioned factors significantly determines an individual’s nature of metabolic health status, affecting their susceptibility to MAFLD, even in persons with a lower BMI.
The influence of lifestyle factors on metabolic health in individuals with normal weight lacks comprehensive understanding. However, these factors seem to have a substantial contribution to the variability in metabolic health across individuals. The widely accepted definition of a healthy lifestyle often encompasses the incorporation of four fundamental behaviours that contribute to overall well-being: moderate alcohol use, abstaining from smoking, participating in 30 min of physical activity on a daily basis and consuming a minimum of five servings of fruits and vegetables each day. 83 The high occurrence of metabolic anomalies in lean individuals with MAFLD indicates that, in addition to the total calorie intake, the quality of one’s diet may serve as an independent predictor of metabolic well-being.
Recent research has provided evidence indicating that certain aspects of dietary intake, such as increased sugar consumption and decreased consumption of cereals, fish and root vegetables, are linked to a condition known as normal-weight obesity. This condition is characterized by a relatively high body fat percentage and compromised metabolic health. 84 Another research revealed that those classified as having normal-weight obesity had greater total calorie intake, lower fibre consumption, reduced levels of antioxidant chemicals and a fewer servings of fruits and nuts, legumes and seeds compared with the lean group. 85 The consumption of cholesterol is comparatively greater in individuals with a lean body composition compared with those with obesity and MAFLD.86–88 Another research revealed that lean people with MAFLD exhibit a larger proportion of carbohydrate energy consumption in comparison with those without the condition who are in good health. 89 In contrast, a separate study revealed that disparities in physical fitness, rather than dietary quality, were seen when comparing persons with normal-weight obesity with lean individuals. 90
Sarcopenia, the age-related decrease in muscle mass and function, is closely associated with MAFLD and various studies suggest that lean MAFLD had a significant association with sarcopenic obesity, independent of metabolic confounders. 91 The presence of sarcopenia identifies a special subgroup of lean MAFLD with an increased risk of cardiovascular disease and liver fibrosis.92,93
The composition of microbiota is mostly influenced by diet, regardless of the genetic makeup of the host. 94 The investigation of the impact of gut microbiota on metabolic health and MAFLD is a field of study that is now gaining significant attention. According to a new study, individuals diagnosed with lean MAFLD have a unique composition of gut microbiota characterized by an abundance of species that have been linked to the development of hepatic steatosis. 11
A growing body of research suggests a potential association between variations in overall fat mass and fat distribution in specific regions of the body and the development of metabolically healthy or unhealthy phenotypes. 95 The location (visceral versus subcutaneous, upper body versus lower body) and composition (brown versus white) of adipose tissue have a more substantial influence on an individual’s metabolic well-being compared with the overall quantity of fat mass. 96 The constrained storage capacity of peripheral adipose tissue, namely subcutaneous adipose tissue, which has little metabolic consequences, may result in the accumulation of ectopic fat in organs such as the liver and skeletal muscles. This accumulation is associated with an elevated risk of cardiometabolic complications. 95 As suggested, fibrosis in the adipose tissue may have a possible function in determining the storage capacity of peripheral adipose tissue, therefore impacting metabolic health. Recent research has shown an association between MAFLD and a rise in the fibrogenesis of adipose tissue, whereas no evidence has shown a reduction in expandability.97,98
In a similar vein, research examining the distribution of adipose tissue among different ethnic populations indicates that individuals of South Asian and Chinese descent tend to possess higher levels of visceral adipose tissue compared with Europeans, even when accounting for overall body fat. Furthermore, the negative consequences of weight gain are more pronounced among Asians compared with adults of other races/ethnicities, including Hispanics, non-Hispanic blacks and non-Hispanic whites. A positive correlation between an increase in BMI by one unit and an elevated likelihood of developing hypertension and diabetes is more profound among individuals of Asian descent.1,99 Consequently, the World Health Organization has suggested a reduced BMI threshold as a catalyst for health intervention among Asians who exhibit a greater susceptibility due to having low BMI and higher body fat percentage in comparison with non-Hispanic whites. 100 The precise mechanisms responsible for these racial disparities have yet to be fully elucidated.
Moreover, the existence of a significant heritable component in relation to metabolic health is likely. Despite the absence of any GWAS conducted particularly to investigate this matter, specific studies have shown the presence of genetic variations that influence the distribution of body fat.101,102 A GWAS has successfully discovered 53 genetic loci that are associated with an increased susceptibility to cardiometabolic diseases. 95 These loci are characterized by lower levels of peripheral adiposity as well as greater insulin resistance phenotypes. These aspects manifest as elevated fasting insulin and triglyceride levels and reduced HDL-C levels. 95 Previous research has shown the existence of genetic variants, often referred to as ‘favourable adiposity genes’, which is positively correlated with increased subcutaneous fat deposition. However, the same genetic variants are inversely related to the accumulation of liver fat as well as a reduced susceptibility to developing T2DM, hypertension and cardiovascular diseases. Notably, among the discovered genes are PPARG and LYPLAL1.103–105
Epigenetics plays a significant role in the control of adipose tissue distribution. Recent study showed that modifications in DNA methylation at the IGF2/H19 gene due to an unfavourable prenatal environment were linked to alterations in subcutaneous fat measurements. However, no significant associations were found between these modifications with visceral or central adiposity. 106 Numerous investigations across several species have provided evidence that fluctuations in the nutritional, metabolic and hormonal conditions during intrauterine and early postnatal stages heighten the vulnerability to the onset of metabolic disorders and illnesses throughout adulthood. 107 The phenomenon known as ‘embryonic or foetal programming’ implies that metabolic health has the potential to be inherited throughout generations, highlighting the significant influence of epigenetics in shaping fat distribution starting from the earliest stages of life. 108 (Figure 2).
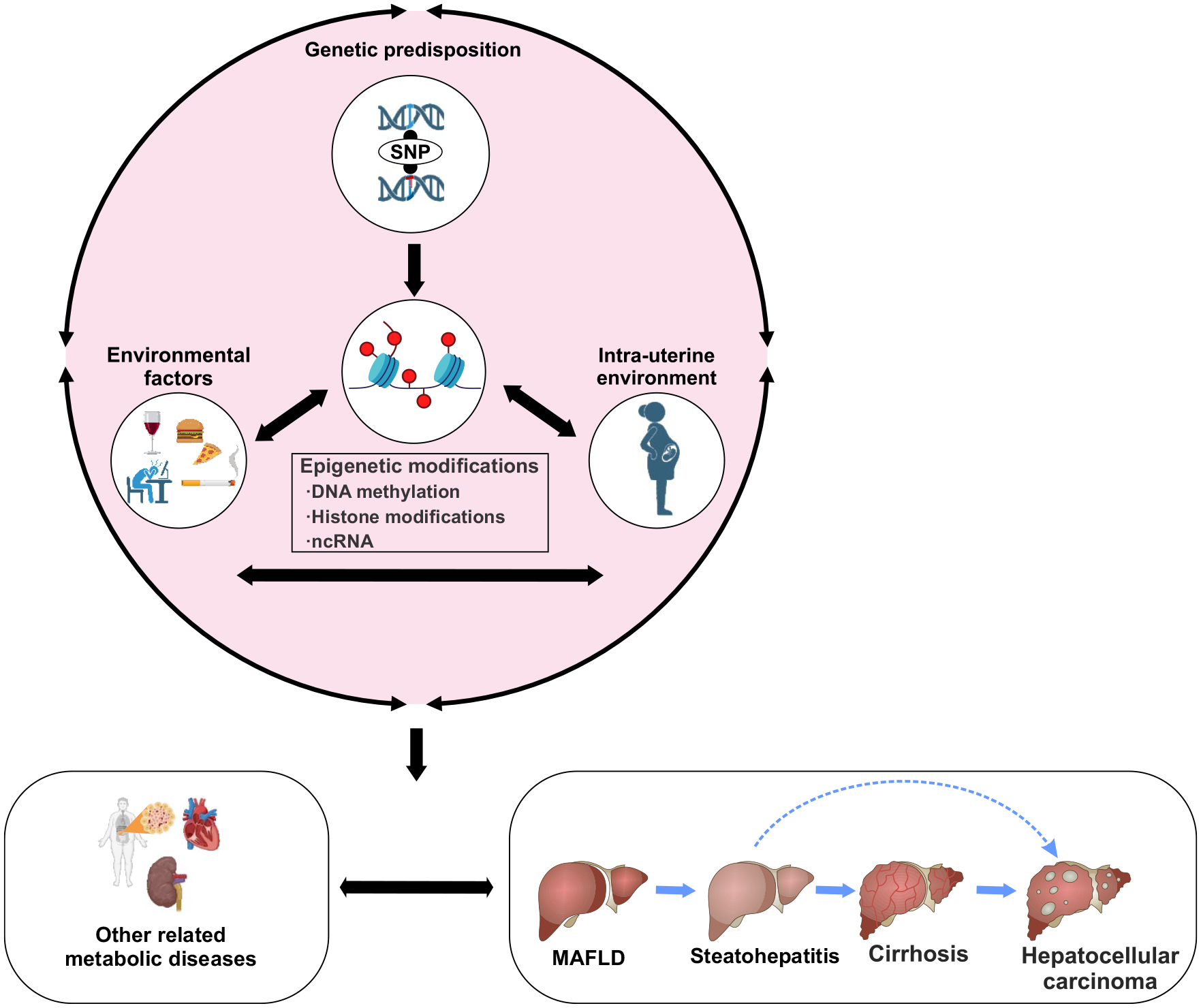
Epigenetics, transgenerational inheritance and gene–environment interactions in MAFLD pathogenesis. MAFLD is a heritable condition, with many genetic variations being discovered as contributing factors to the disease. Various environmental variables, including but not limited to nutrition, smoking, alcohol use, age, exercise and the intrauterine environment, have the potential to interact with an individual’s genetic background, hence exerting an impact on the development of diseases, and this leads to transgenerational inheritance of a phenotype. The epigenome, including DNA methylation, histone modification (methylation/acetylation) and ncRNAs has the potential to play a dual role in the development of MAFLD, as it may both contribute to its onset and facilitate genetic–environment interactions.
Metabolic adaptation
A growing body of data suggests that the maintenance of metabolic health is facilitated by a homeostatic mechanism that regulates metabolic adaptation. 109 Metabolic adaptation pertains to the capacity of the human body to regulate energy expenditure independent of body weight changes. This regulation is achieved by a complex interplay of many regulatory systems, including hormones, chemokine signals and the neuroendocrine axis. 110 Any disturbances in the systems under consideration may lead to a disruption in metabolic adaptation, resulting in an atypical increase in adipose tissue and the development of obesity and insulin resistance. 111
In the population of patients with MAFLD, those with lean MAFLD have a more advantageous metabolic profile than those with overweight or obese MAFLD. 11 This was confirmed in a very recent study comprised 1047 Chinese participants that demonstrated that non-obese MAFLD patients exhibited different metabolic and body composition profiles compared to their obese counterparts. A recent study has found that individuals diagnosed with lean MAFLD have discernible metabolic adaptations. In this model, as illustrated in Figure 3, increases in dietary cholesterol intake within the context of intact metabolic adaptive response (influenced by inherent genetic, epigenetic and gut microbiota characteristics) are linked to certain metabolic adaptations. These adaptations involve an augmentation in the synthesis of bile acids and an increase in the activity of the Farsenoid X receptor (FXR) to regulate serum cholesterol levels and maintain normal body weight. 11 By contrast, a person can have a total absence of metabolic adaptation, leading to a rise in body weight and adipose tissue accumulation. The observed metabolic adaptability elucidates the improved liver histology and metabolic profile seen in these lean MAFLD individuals, particularly during the first phases of their condition. 11 This study suggests a distinct path of metabolic adaptation and flexibility development in obese MAFLD individuals compared with non-obese MAFLD patients. This ability declines over time, leading to the manifestation of metabolic inflexibility. A more recent study has found that the advancement of MAFLD is associated with endotoxaemia, which induces epigenetic alterations. These changes are the triggering factors that reduce bile acid signalling and lead to the transition towards maladaptation throughout the evolution of a disease in lean individuals with MAFLD. 112

The role of metabolic adaptation in the pathogenesis of MAFLD. The schematic figure elucidates the disparities in metabolic adaptation between lean and non-lean people afflicted with MAFLD. The observed variations are impacted by both metabolic and genetic predisposition. Patients with MAFLD who are obese have suboptimal metabolic adaptation, hence contributing to the progression of adiposity and liver disease. In contrast, it is seen that lean individuals with MAFLD have a degree of metabolic adaptation, especially during the first phases of the disease. This adaptation is attributed to many processes, including heightened bile acid synthesis and enhanced activation of FXR, although other mechanisms may possibly be at play. The observed consequence leads to the development of an ‘obesity-resistant’ phenotype, which diminishes with time as the disease advances.
Management of patients with lean MAFLD
Currently, no specific management recommendations are available for lean MAFLD patients, mainly due to limited evidence. Similar to non-obese MAFLD people, lifestyle intervention has been shown to have positive effects on lean MAFLD patients, though with potentially lower weight loss goals. The findings of a longitudinal study with a sample size of 16,738 persons who completed several health examinations revealed a significant correlation between weight loss and the resolution of fatty liver in individuals diagnosed with either lean or overweight/obese MAFLD. This correlation was shown to be dependent on the level of weight reduction. 113 Nevertheless, 3–5% weight reduction among lean individuals with MAFLD may be sufficient to achieve histological improvement among them. 114 The incorporation of physical exercise and adherence to a high-quality diet is associated with a significant reduction in the likelihood of developing obese and non-obese MAFLD. 115
Metabolic health deterioration is influenced by poor dietary quality, regardless of an individual’s BMI or waist-to-hip ratio. Poor dietary quality is a notable risk factor that may be modified. Hence, expert nutritional guidance must be provided to those diagnosed with MAFLD, as well as those who are metabolically unhealthy, irrespective of their BMI classification. Promoting physical activity is also advisable for improving metabolic flexibility. In addition, the timely identification and controlling of concomitant metabolic comorbidities must be prioritized. This recommendation may be attributed to the majority of fatalities arising from cardiovascular complications rather than liver-related ailments, including among individuals with lean MAFLD. Whereas several therapeutic agents are being developed and the drug discovery pipeline in MAFLD has been expanding, the specific efficacy of these drugs in lean patients is yet to be well clarified.
Future directions and clinical implications
MAFLD is a disease known to exhibit significant heterogeneity in its presentation, clinical course and outcome. Lean and non-lean subtypes of MAFLD are an exemplar of this variability. Emerging evidence suggests a distinct underlying pathobiology and drivers of these subtypes, which may give rise to potential disparities in their respective outcomes. The lean subtype is characterized by compromised metabolic health but with a comparatively superior ability to adapt metabolically in the short term. These findings indicate that the categorization of patients based on their metabolic health state should be considered and the introduction of the MAFLD framework was a pivotal step on this path. Additionally, future clinical trials should include the categorization of patients into lean and non-lean groups.
To move forward and advance this field, a joint and individual approach is necessary. Healthcare practitioners must be made aware of the potential for underrepresentation of individuals with lean MAFLD who may be erroneously categorized as healthy due to their body composition. Furthermore, these individuals may be neglected in clinical studies. In the current era of precision medicine, individualized management approach is the Holy Grail goal. Achieving this goal requires a comprehensive understanding of genetic and environmental factors that may elucidate individual responses to therapy. Such understanding would facilitate the categorization of individuals into precise subgroups based on specific risk factors and genetic predispositions. Additionally, patient empowerment across all facets of prevention, treatment and care for MAFLD is necessary, leading to improved adherence. Thus, the effectiveness of lifestyle modifications and pharmacological interventions is enhanced.
Limitations
The current data on the pathogenesis of lean MAFLD is considered areas with scanty literature and, therefore, recommended for further investigation using other research strategies. Also, there is paucity of data on the specific management approaches of this group of patients.
Conclusion
Lean MAFLD is characterized by a unique set of features that distinguish it from other forms of fatty liver disease. The onset of MAFLD is significantly influenced by metabolic health status. In cases where lean individuals are affected by this condition, their phenotype can be attributed to several factors, such as genetic and epigenetic factors, gut microbiota composition, bile acid profiles, enterohepatic circulation and lifestyle choices, despite having a normal BMI. Compared with others, lean individuals have a unique and superior ability to adapt to unfavourable metabolic stimuli, allowing them to maintain their lean body mass regardless of heightened cardiometabolic risk. However, the potential long-term costs and trade-offs associated with this partial and transient metabolic adaptation are yet to be identified.
