Abstract
Metabolic dysfunction–associated fatty liver disease (MAFLD) has appeared as the leading liver disease worldwide. Whereas the terminology nonalcoholic fatty liver disease (NAFLD) mainly reflected a negative selection and exclusion of alcohol-related liver disease (ALD), the new definition made its focus on the association of MAFLD with overweight/obesity, type 2 diabetes and metabolic risk factors especially also in normal weight/lean subjects. Several studies from the past 2 years have now used the new definition and have provided substantial information that this new definition might be accurate. Studies from the past 2 years have provided evidence that the new definition might be especially advantageous in the characterization and identification of patients with significant fibrosis. This has also been demonstrated in the well-known Rotterdam study in which the MAFLD-only group showed a higher rate of fibrosis and liver stiffness. MAFLD might also be able to predict all-cause mortality as demonstrated in the Third National Health and Nutrition Examination Survey. Furthermore, MAFLD might improve characterization of the cardiovascular risk of this patient population. As the term MAFLD has not yet been accepted universally, it remains important to coordinate efforts globally to adapt to this new definition and especially involve all specialities dealing with metabolic disorders such as diabetologists to further improve its definition and to prepare the medical community for its future use. The aim of this review is to summarize and critically address evidence emerging over the past 2 years that usage of the term MAFLD could be helpful in daily clinical practice.
Keywords
Introduction
Nonalcoholic fatty liver disease (NAFLD) has appeared as the most common liver disease globally in the past years, and this disease is affecting public health substantially.1,2 This disease includes a wide spectrum of clinical presentations such as simple steatosis, nonalcoholic steatohepatitis (NASH), fibrotic NASH, liver cirrhosis and hepatocellular carcinoma (HCC). Furthermore, NAFLD is more part of a syndrome as it is frequently associated with metabolic disturbances. Therefore, not surprisingly, cardiovascular, renal and malignant complications within and outside the liver substantially contribute to morbidity and mortality of this disease.3–5 Whereas initially NAFLD has been characterized mainly as a fatty liver disease with histological features reminiscent of alcohol-related liver disease (ALD) and diagnosed by exclusion criteria, 6 knowledge from the last two decades has clearly demonstrated that NAFLD is part of a systemic metabolic disorder in which liver manifestation is only one part of this syndrome. 7 Metabolic features dominate this syndrome as it is a disease that is highly linked to obesity and type 2 diabetes (T2D)3,8 and lean NAFLD is on average observed in less than 10% of the overall NAFLD population. 9 But not only the clinical phenotype of NAFLD but also substantial knowledge from the past years regarding genetic associations has supported the metabolic nature of this disease. 10 In addition, pathophysiologic insights strongly support the importance of metabolic pathways that trigger in many affected NAFLD subjects liver inflammation, fibrosis and immunological processes. 11 Furthermore, caloric excess as common basis of this disease affects the gut microbiome that is frequently disturbed potentially contributing to disease pathogenesis. 12 Overall although initially described as NAFLD, knowledge from the past two decades has strongly put pressure on the medical community to consider all these aspects and potentially move towards a new definition of this disease.
Following these concepts, Eslam et al.13,14 have started an important process to redefine this disease as metabolic dysfunction–associated fatty liver disease (MAFLD). In their initial two reports, the authors proposed not only the new name but also tried to define this syndrome with its metabolic components. These authors proposed a definition leaving the historical ‘exclusion diagnosis’ towards a definition with a clear set of ‘positive’ criteria: presence of hepatic steatosis (detected either by any imaging technique including ultrasound, liver histology or blood biomarkers), overweight or obesity [body mass index (BMI) >25 kg/m2 in Caucasians or >23 kg/m2 in Asians], T2D or lean/normal weight subjects with at least two metabolic risk factors. Metabolic risk factors were defined as follows: waist circumference >102/88 cm in Caucasian men and women or >90/80 in Asian men and women; blood pressure >130/85 or specific drug treatment; plasma triglycerides >150 mg/dl or specific drug treatment; plasma high-density lipoprotein (HDL) cholesterol <40 mg/dl for men and <50 mg/dl for women or specific drug treatment; prediabetes [i.e. fasting glucose levels 100–125 mg/dl, or 2-h post-load glucose levels 140–199 mg/dl or haemoglobin A1c (HbA1c) 5.7–6.4%], homeostasis model assessment of insulin resistance score >2.5 or plasma high sensitive C-reactive protein levels >2 mg/l (Figure 1). Eslam et al. 15 did also induce an adaption of the MAFLD definition for paediatric patients, using age-dependent percentiles. Although the new definition MAFLD is currently not accepted globally and has raised some criticisms, the new name ‘MAFLD’ has been endorsed in the last 2 years by many international societies and patient groups.16–26 The need for a such a new definition is also strongly supported by many pathophysiological aspects. 27
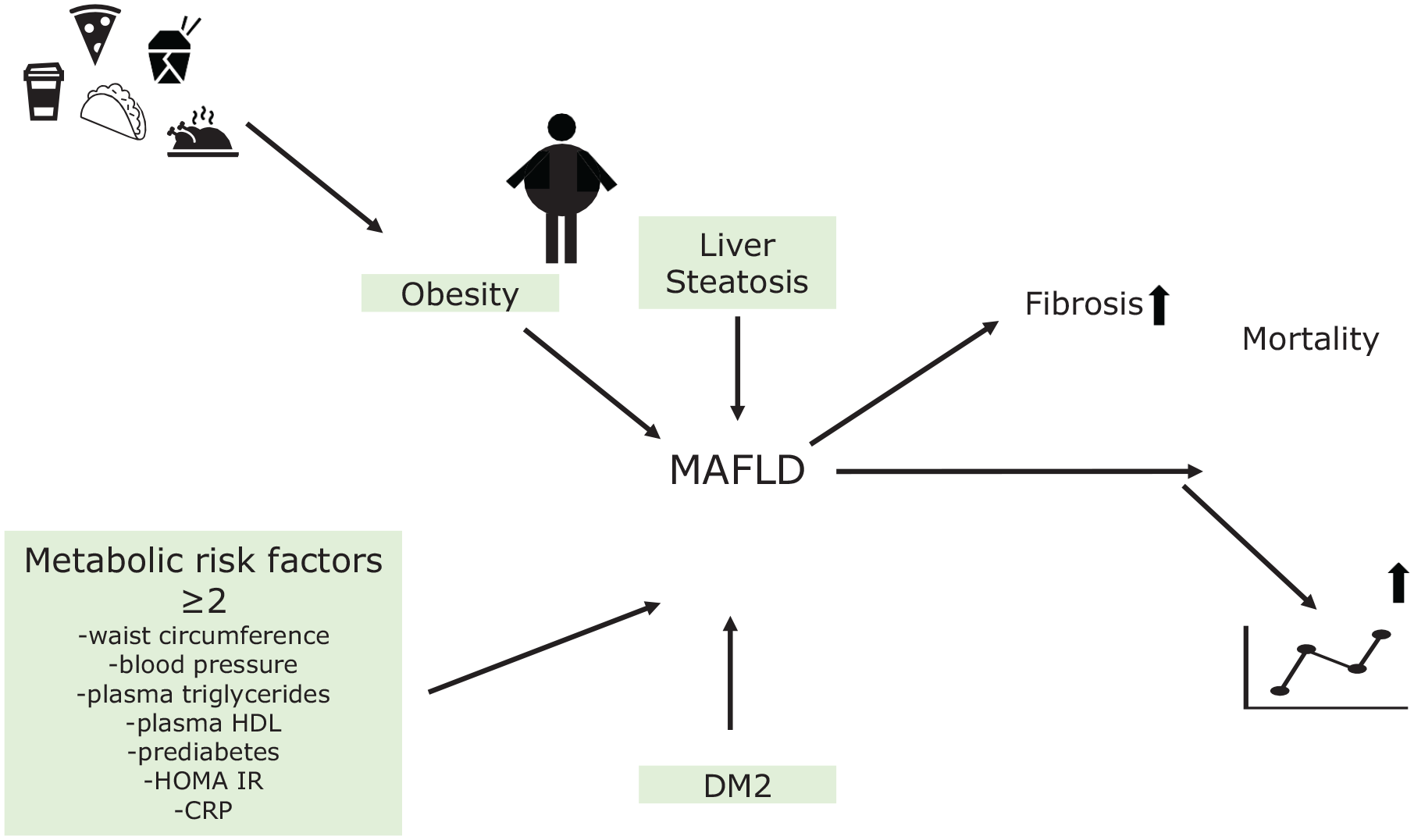
A holistic metabolic view on clinical features relevant in MAFLD: as a paradigm systemic disease MAFLD is defined by hepatic steatosis, overweight/obesity, type 2 diabetes or at least two of the following risk factors: waist circumference >102/88 cm (>90/80 in Asian), blood pressure >130/85, plasma triglycerides >150 mg/dl, plasma HDL <50 mg/dl (<40 mg/dl for men), HOMA-IR >2.5, and plasma CRP >2 mg/l or prediabetes. Individuals with MAFLD have a higher probability of liver fibrosis and an increase in all-cause mortality; thus, the inclusive definition can help clinicians to better identify patients at risk.
Evidence that the new definition ‘MAFLD’ might advance the field of fatty liver diseases
The term ‘MAFLD’ has been introduced in 2020 and has been increasingly used in the medical literature in the past 2 years (Table 1). Almost 10% of all articles published on this topic in 2022 so far have preferred and used MAFLD and not NAFLD, a similar trend was observed for the search term ‘MAFLD + metabolism or diabetes’. The observed trend indicates a rising acceptance of the new definition, and one could interpolate that in a few years, MAFLD could be used more often than NAFLD. Several studies have now demonstrated that MAFLD is not only the proper name for this disease, but its use might also offer advantages and allow a better characterization of this disease. Yamamura et al. 28 studied the effect of the new definition compared with NAFLD in the identification of patients with significant fibrosis. These authors investigated 765 Japanese patients with a median age of 54 years resulting in 79.6% MAFLD patients versus 70.7% with NAFLD. Overall, 55.4% patients exhibited overlapping NAFLD and MAFLD. Importantly, they also assessed the relevance of mild alcohol consumption as defined by <20 g/day. Fibrosis was defined by the Fibrosis-4 (FIB-4) Index (>1.3) and >6.6 kPa by transient elastography. MAFLD, NAFLD and alcohol intake were all associated with fibrosis, but MAFLD appeared with a significantly higher fibrosis detection. Interestingly, even intake of moderate alcohol caused an increase in the prevalence of significant fibrosis. Yamamura et al. 28 concluded that the new MAFLD definition might be superior in the identification of advanced disease using noninvasive fibrosis tests. Importantly, a recent study by Trauner’s group demonstrated that at least 25% of NAFLD patients consume substantial alcohol as detected in hair which is assumed to have a major impact on NAFLD/MAFLD phenotype. 29 The authors showed that in 28.6% of NAFLD patients and in 25% of MAFLD patients, alcohol-related liver injury might be relevant. As proposed by Yamamura’s study, the observed ‘mild alcohol’ consumption in Staufer’s study might be clinically highly relevant and in many NAFLD/MAFLD cases associated alcohol consumption might play a major role in disease phenotype and prognosis. Therefore, in many NAFLD/MAFLD cases, we might see ‘combined fatty liver disease’.
Use of term NAFLD versus MAFLD and number of articles published in PubMed 2020–2022 (search 17 June 2022), and percentage of similar NAFLD search term.

MAFLD, metabolic dysfunction–associated fatty liver disease; NAFLD, nonalcoholic fatty liver disease.
Cardiovascular complications crucially affect the outcome of patients with NAFLD. 30 Another study investigated whether the new definition MAFLD might affect the identification of patients with progressive atherosclerosis and cardiovascular complications. 31 The authors studied 2306 patients with fatty liver disease (MAFLD 80.7%; NAFLD 63.4%). Atherosclerotic cardiovascular assessment was performed using the Suita score 32 and the Framingham risk score. 33 The Suita score also includes metabolic assessment with evaluation of glucose intolerance and serum low-density lipoprotein cholesterol levels. When comparing NAFLD with no metabolic dysfunction versus overlapping and MAFLD with moderate alcohol consumption, nonmetabolic NAFLD patients exhibited a lower rate of cardiovascular complications. Patients with MAFLD and alcohol consumption showed a worsening Suita score. MAFLD might be superior in identifying cardiovascular complications, although the effect of alcohol in this patient population might be important. Many more studies into this association, however, are needed to demonstrate superiority of the new MAFLD definition, although the authors concluded that the benefit of the positive association of MAFLD with the rate of cardiovascular complications was mainly because of metabolic dysfunction and not alcohol consumption. 31 Another important study supporting the strength of the new definition was published by Kim et al. 34 The authors used data from 7761 participants of the Third National Health and Nutrition Examination Survey (NHANES III) investigating effects of the new definition on all-cause mortality. Patients were followed over median 23 years and subjects with MAFLD showed a 17% higher risk of all-cause mortality and an increase in cardiovascular mortality whereas NAFLD subjects did not show such an association. Furthermore, again highlighting the importance of fibrosis, patients with MAFLD and advanced fibrosis showed higher mortality compared with those with advanced fibrosis and NAFLD. Authors concluded that MAFLD definition might better reflect metabolic risk factors, and the new definition might overall improve better classification of prognosis and risk factors. 35 Probably, the most compelling study in support of the new definition has been recently presented by van Kleef et al. 36 Here, the authors used data from the Rotterdam study to find out whether MAFLD might improve detection of high liver stiffness. The Rotterdam study reflects an ongoing prospective cohort study that had been started in 1990 in Rotterdam targeting aetiology, natural history and course of chronic diseases in mid- and late-life including assessment of chronic liver diseases and metabolic risk factors. 37 Patients were included between 2009 and 2014 and diagnosis included ultrasound and transient elastography and patients were allocated to overlap fatty liver disease, NAFLD-only or MAFLD-only. Fibrosis was defined by transient elastography >8.0 kPa. The authors included 5445 subjects, and again the MAFLD group was larger (1866, 34.3%) versus NAFLD (1604; 29.5%). Numbers were corrected after publication of MAFLD criteria.13,14 Importantly, the MAFLD-only group was convincingly associated with liver fibrosis independent of demographic and life style factors. Importantly, the authors concluded that use of the new MAFLD definition might not lead to exclusion of patients with fatty liver disease at substantial risk of fibrosis. This study with several studies discussed before therefore presents substantial evidence that indeed MAFLD definition might be superior in detection of the key prognostic feature in this disease, that is, liver fibrosis. A new definition, however, might have additional benefits as suggested by Fouad et al. 38 They contacted 161 physicians in Egypt experienced in the management of liver disease demonstrating that the new definition increases awareness on the management of patients with fatty liver diseases as 73.3% of physicians became more familiar with fatty liver disease when being aware of the MAFLD and its definitions. Data from Mexico showed a similar trend in which an increased awareness using the MAFLD definition in paediatric and adult patients was raised compared with the NAFLD definition.39,40 Furthermore, after introduction of the term MAFLD, several sessions and lectures at numerous international meetings were dedicated to the topic. 41 This is a very important aspect as proper management of this disease in the future throughout the world will only be possible when primary care physicians, diabetologists and gastroenterologists/hepatologists are perfectly aware of the relevance and importance of this disease. Another report into this direction was recently presented by Farahat et al. 42 proposing that such a definition might enhance awareness globally in primary care. Authors concluded that improvement of fatty liver disease management, reduction of overdiagnosis and overtreatment in primary care, the key area in which most of the patients are seen, and more importantly reduction of underdiagnosis might be enabled by a better disease definition. Zhang et al. 43 investigated 11,000 participants from the NHANES III stratifying patients into alcoholic fatty liver disease, no MAFLD or MAFLD. During a follow-up of 23.2 years, 3240 deaths were observed and MAFLD patients exhibited an increased mortality, and this was especially true for MAFLD and associated T2D. Underlining the importance of liver fibrosis as prognostic marker, even in MAFLD negative patients, mortality was associated with increased fibrosis scores. The authors concluded that new MAFLD definition and fibrosis score were superior to NAFLD definition with respect to clinical outcomes. In support of the new definition is also a study comparing epidemiological and clinical features of MAFLD versus NAFLD. 44 In this report, 17 studies including 9808.677 individuals were studied. MAFLD appeared in 33.0% of the studied population whereas NAFLD was present in 29.1%. In patients with overlap fatty liver disease, only 4.0% were missed by MAFLD criteria and had MAFLD only while 15.1% were identified by MAFLD criteria. Again bringing the fibrosis aspect into attention, the MAFLD-only group had a significantly higher risk of fibrosis. The authors concluded that the new definition is better to identify patients at risk for liver disease, and less patients are missed using MAFLD definition proposing that MAFLD is superior to NAFLD at the population level. An Egypt study demonstrated impairment of health-related quality of life in MAFLD patients but not NAFLD-only patients compared with healthy controls. Although this study has limitations (small sample size of NAFLD only), it also supports patient-based evidence arguing for the use of the new definition. 45 A similar viewpoint is stated by Fouad et al. 46 in a recent review, in which they argue that diagnosing MAFLD in hepatitis C virus (HCV) patients after sustained virologic response (SVR) will increase quality of life in these patients.
Overall, as discussed in this article, substantial evidence has been generated in the past 2 years supporting the strength and potential superiority of the new definition, although many more studies are needed to show indeed superiority of MAFLD to NAFLD with respect to complications, prognosis and outcome of this disease. This opinion is also shared by Fouad et al. 47 and Eslam et al. 48 who also provide perspectives on innovative trial design for MAFLD. A review published a year after the new definition by Alharthi et al. 49 also concluded that the transformation is an important step towards a more patient-centred approach for tackling this disease.
Potential pitfalls of the new definition: consensus not yet achieved
Critical comments on the new MAFLD definition substituting NAFLD terminology appeared from several groups around the world especially from the United States. 50 Younossi et al. 50 claimed that such a terminology is currently premature. These authors estimate that the current definition of NAFLD overemphasizes the absence of alcohol use and certainly underemphasizes the relevance of metabolic disturbances and risk factors. They also comment that the term NASH adds complexity as it needs liver histology and is associated with a high intra-/inter-observer variability. In our opinion, two of the most crucial points are that both alcohol consumption and assessment of liver inflammation reflect demanding issues of this disease whatever definition will be used today and in the future. This is also supported by the recent study from Vienna as discussed before demonstrating that approximately 25% of also MAFLD patients consume too much alcohol. 29 Another major problem discussed by Younossi et al. 50 reflect current and past clinical trials in this field with well-established clinical and histological endpoints which would need to be adapted when using the new definition. How should endpoints of clinical trials be defined when using the new MAFLD definition? NAFLD-associated HCC might reflect another difficult topic. 51 Vitale et al. 52 recently presented data from the Italian Liver Cancer (ITA.Li.CA) group. Vitale et al. 52 reported not only that in the next years MAFLD will become the main underlying disease in HCC and also showed that single aetiology MAFLD might have a better prognosis compared with non-MAFLD HCC. This, however, could also be because of various confounders such as improved management of metabolic disturbances in the past years. In the accompanying commentary, Hernaez and Peck-Radosavljevic critically discussed the new term MAFLD when considering HCC. 51 Especially they were criticizing why alcoholic liver disease should not be a separate disease but is now defined as a subgroup within the unspecific term MAFLD. Another challenge with the new definition appears that the new MAFLD definition has a very low specificity for the detection of patients with a higher HCC risk, making surveillance in this population impossible. Chen et al. 53 observed when assessing adults from the NHANES III study (1988–1994) that there were no differences between normal controls and patients with non-MAFLD steatosis or MAFLD with respect to clinical outcome. In this study, MAFLD was not associated with all-cause mortality, but subjects with lean MAFLD and diabetic MAFLD had a higher risk of all-cause mortality proposing that further subclassification of MAFLD might be needed. Semmler et al. 54 also found that MAFLD was not associated with an increase in mortality. Interestingly, they also observed that rather lean MAFLD had a worse prognosis, but again this association was probably confounded by age and associated metabolic disorders such as T2D.
Do we need a better definition of MAFLD?
Studies discussed before might suggest that the recently proposed MAFLD criteria might be in need of further development and adaptation. Especially also colleagues from the field of metabolism and diabetes have raised some concerns about the terminology and use of MAFLD. 55 These authors stated that use of both waist circumference and BMI adds ‘an unnecessary layer’, and are proposing that BMI is too unspecific for detection of overweight/obesity. They propose therefore rather to use waist circumference or assessment of fat mass by dual-energy X-ray absorptiometry (DXA). They also criticized as several other authors that patients with concomitant disorders, for example, alcoholic or drug-induced liver injury are not excluded. Another point of criticism focused on the classification of MAFLD into certain subtypes of metabolic risk factors and the abolishment of NASH which is currently not only target but also endpoint in many clinical trials. 55 Xie et al. 56 proposed a simpler definition of MAFLD better predicting long-term all-cause mortality in American adults. They were also using data from the NHANES III study and modified MAFLD especially by also including hyperuricemia as a risk factor and observed that when using this simplified MAFLD criteria, patients had a significantly higher risk of all-cause mortality than when using the original MAFLD criteria. All these reports suggest that further discussions and an intense global dialogue will be needed to further develop the quality of MAFLD criteria and its potential global use. There is no doubt that the new name more properly reflects the key nature of this disease. What is needed, however, is a critical consensus how MAFLD should be defined also considering opinions from the diabetes community. Another challenging issue is that there exists no consensus-on-consensus methodology, although for the global acceptance of the term MAFLD, a consensus of many involved disciplines and stakeholders will be needed. 57
Conclusion
The introduction of the new definition of MAFLD has probably been the most disagreement causing issue in the field of hepatology in the past years. It has, however, raised an intensive dialogue between specialists between all medical fields involved in the management of patients with fatty liver disease, and therefore reflects an opportunity to advance the field. As discussed in this article, we have increasingly seen original research articles in the past 2 years supporting the new definition, but we have also seen strong arguments for a need of adaptation of the new definition especially when thinking of HCC and how to deal with overlap of alcohol and metabolically induced fatty liver disease. We need universal acceptance of this definition including opinions from diabetologists/endocrinologists. Whatever name will be chosen in the future for this disease, the challenge of alcohol consumption remains. As nicely shown by Staufer et al. 29 even when using the MAFLD definition, 25% of affected patients consume too much alcohol that very likely affects disease phenotype. Alcohol consumption will always affect presentation of fatty liver disease and all other liver diseases.
NASH remains another challenging aspect to deal with in a future terminology. Inflammation reflects a crucial part of this disease, although finally fibrosis decides on the fate of fatty liver disease. Inflammation might appear smouldering in most patients, and therefore will be frequently missed also by liver biopsy. This aspect can neither be solved by a new name nor by ignoring this crucial aspect of disease. Also patients with prototypic pro-inflammatory diseases such as inflammatory bowel diseases present with intermittent inflammation, and in case of structurizing fibrotic Crohn’s disease, no one would argue that inflammation is not highly relevant in this disorder. In the opinion of these authors therefore it is problematic to eliminate the term ‘steatohepatitis’ in the new definition. Undoubtedly, inflammation reflects a key driving force of fibrosis in fatty liver disease, and it is rather our incapability to detect relevant liver inflammation than its relevance. Although over 1000 signatories representing a plethora of stakeholders endorse the use of MAFLD, 22 a future global consensus will be needed involving not only all major international liver organizations but in addition experts from other disciplines such as diabetologists or primary care physicians. This remains a huge task and challenge for medicine. Despite all potential shortcomings with this new definition, a better reflection of the so frequent metabolic dysregulations needs to be included in a new disease definition. MAFLD seems to be an appropriate name and an important process searching for the best definition of this pandemic disease has been started. This is mandatory for improvement of patient care in the future.
