Abstract
Background:
As a part of metabolic syndrome, hyperuricemia has a higher incidence in patients with diabetes than in the general population owing to various underlying factors.
Objectives:
The objective of the present study was to investigate the prevalence of hyperuricemia among patients with diabetes and identify associated factors.
Design:
A cross-sectional study.
Methods:
Herein, we included patients with diabetes managed at nine healthcare centers in Chenghua District, Chengdu, from February 2021 to November 2021. Clinical data, lifestyle habits, and laboratory data were collected to determine the prevalence and factors associated with hyperuricemia.
Results:
In total, we included 1577 patients with diabetes (males, 50.35%; females, 49.65%). The median serum uric acid level was 337.9 μmol/L, and the prevalence of hyperuricemia in patients with diabetes was 21.24%. The prevalence of hyperuricemia in male patients was significantly higher than in females (29.35% in males
Conclusion:
Hyperuricemia is highly prevalent in patients with diabetes, especially in males. In addition to traditionally associated factors, fenofibrate and SGLT-2 inhibitors were also associated with the risk of hyperuricemia.
Registration:
The study protocol was registered in the Chinese Clinical Trial Registry (http://www.chictr.org.cn/), and the registration number was ChiCTR 2100042742.
Introduction
Serum uric acid (SUA), a final product of purine metabolism, is known to be associated with the development of gout and kidney stones. 1 The prevalence of hyperuricemia varies substantially across regions and populations. A cross-sectional survey conducted in 31 Chinese provinces has revealed that the prevalence of hyperuricemia from 2018 to 2019 was 14% (24.4% in males and 3.6% in females), with the highest prevalence documented in East China (17.1%), followed by West China (13.0%), and Middle China (10.4%). 2 Likewise, the prevalence of hyperuricemia among adults in the United States is 11.9%. 3 Furthermore, hyperuricemia has been documented among 23.1% of the older Polish population. In Japan, the prevalence of hyperuricemia among adult fluctuates between 1% and 30% with significant differences between males and females.4,5
Based on cohort studies, it is highly probable that patients with elevated SUA will develop insulin resistance and diabetes in several years to several decades, and every 1 mg/dL increase in SUA increases the risk of type 2 diabetes mellitus (T2DM) by 17%, which may be attributed to high SUA reducing the level of nitric oxide (NO) in endothelial cells, which, in turn, induces insulin resistance.6–8 Jia
Conversely, insulin significantly increased the expression of uric acid transporter 1 (URAT1) and decreased the expression of adenosine triphosphate-binding cassette G member 2 (ABCG2) in the renal cortex, resulting in reduced uric acid secretion and elevated uric acid reabsorption.
13
In patients with T2DM, insulin resistance can be positively correlated with SUA levels.
14
Chen
Few studies have reported the prevalence of hyperuricemia in patients with diabetes in China. Furthermore, the prevalence of hyperuricemia varies considerably across different regions. Therefore, we conducted the present study to determine the prevalence of hyperuricemia in patients with diabetes in Southwest China.17,18 Moreover, the common drugs for patients with diabetes, such as antihypertensive, hypoglycemic, and lipid-lowering drugs, may have impacts on the occurrence of hyperuricemia and SUA levels. However, few studies have comprehensively analyzed their effects, so in addition to lifestyle habits and biomarkers, we included common drugs as many as possible to assess the potential effects of common drugs on hyperuricemia and SUA levels and provide possibilities for future researches.
Methods
Study population
This cross-sectional study was conducted at nine healthcare centers (Third People’s Hospital, Wannian Community Health Center, Tiaodeng River Community Health Center, Longtan Community Health Center, Shuangqiaozi Community Health Center, Erxian Bridge Community Health Center, Fuqing Road Community Health Center, Baohe Community Health Center, and Qinglong Community Health Center) in Chenghua District, Chengdu, between February and November 2021. All patients with diabetes (⩾18 years) who completed the relevant assessments and signed an informed consent form were enrolled. To obtain accurate information regarding lifestyle, patients with aphasia, deafness, dysgnosia, or mental disorders were excluded. Given that high sex hormone levels and chemotherapy drugs may affect uric acid levels, we excluded pregnant women and patients with malignant tumors. Patients with severe heart, hepatic, or renal failure were unstable and unsuitable for investigation.
Data collection
A specialist collected data from medical records, including sex, age, phone number, height, and weight, as well as levels of alanine transaminase (ALT), aspartate transaminase (AST), total cholesterol (TC), triglyceride (TG), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), SUA, HbA1c, creatinine, and estimated glomerular filtration rate [eGFR = 186 × (creatinine/88.4)−1.154 × (age)−0.203 × (1 (male) or 0.74 (female))]. Blood biochemical parameters were measured using an automatic biochemical analyzer, and HbA1c was detected using high-performance liquid chromatography (1366 cases) and affinity chromatography (211 cases). Laboratory tests of all participants were subject to quality control at the Department of Laboratory Medicine, West China Hospital, Sichuan University.
In addition, we recorded information regarding hypoglycemic, antihypertensive, lipid-lowering, antiplatelet, and uric acid-lowering drugs used at the time of blood collection, including sulfonylureas, thiazolidinedione, other insulin secretagogues, metformin, α-glucosidase inhibitors, dipeptidyl peptidase 4 inhibitors, glucagon-like peptide 1 (GLP-1) receptor agonists, sodium-glucose cotransporter 2 (SGLT-2) inhibitors, insulin, angiotensin-converting enzyme inhibitors (ACEIs), angiotensin II receptor blockers (ARBs), calcium channel blockers (CCBs), adrenergic α-antagonists, adrenergic β-antagonists, diuretics, statins, fibrates, aspirin, clopidogrel, benzbromarone, febuxostat, and allopurinol.
Finally, a well-trained interviewer collected information regarding lifestyle habits at the time of blood collection. We defined variables as follows: (1) current drinking: drinking at least once weekly in the last year 19 ; (2) current smoking: smoking in the past 30 days, including daily smoking and occasional smoking 20 ; (3) exercise: at least 150 min of moderate intensity physical activity, or at least 75 min of high-intensity physical activity weekly, or at least 150 min of moderate and high-intensity physical activities 21 ; (4) staying up late: sleeping after 11:00 pm greater than or equal to five times weekly 22 ; (5) regular lunch break: greater than or equal to three times weekly and ⩾30 min each time; (6) red meats: pork, beef, mutton, and rabbit meat; (7) fishes: fresh water fish; (8) fried foods: foods cooked in oil at a high temperature for rapid maturation; (9) seafood: edible sea-dwelling creatures; (10) strong teas: tea leaves accounting for half or more of the total volume after brewing 23 ; and (11) broths: chicken, duck, fish, bone, or rib soup.
Definitions
Diabetes was diagnosed based on the 1999 World Health Organization diagnostic criteria. 24 Type 1 diabetes mellitus was determined based on young onset age, acute onset mode, insulin-dependent treatment, poor islet function, positive islet autoantibodies, or gene testing. 25 T2DM was established based on relative (rather than absolute) insulin deficiency, peripheral insulin resistance, and inessential insulin treatment. 25 According to Chinese guidelines, hyperuricemia was defined as SUA > 420 μmol/L or a prior diagnosis of hyperuricemia and use of uric acid-lowering drugs (e.g. benzbromarone, allopurinol, and febuxostat) within 1 week prior to blood collection. 26
Statistical analyses
Non-normal data were expressed as median [interquartile range (IQR)], and categorical variables were shown as number (percentages). Differences among groups were analyzed using Chi-squared test (dichotomous variables), Mann–Whitney
Results
Baseline characteristics
In total, 1577 patients with diabetes (6 patients with T1DM and 1571 patients with T2DM) were included, with a median age of 64 years (IQR 56–71 years, range 26–93 years), and males accounted for 50.35%. Among them, lifestyle habits and body mass index (BMI) data were unavailable for 358 patients (22.70%), primarily attributed to patient refusal. Therefore, 1577 patients were included to evaluate the prevalence of hyperuricemia and SUA levels, with only 1219 patients (4 patients with T1DM and 1215 patients with T2DM) enrolled in the analysis of associated factors.
Table 1 summarizes the baseline characteristics of 1219 participants included in the analysis of the associated factors. The median age was 63 years (IQR 56–71 years, range 29–91 years), with males accounting for 52.09%. Females were older than males (median 66
Demographic and laboratory characteristics of included patients.
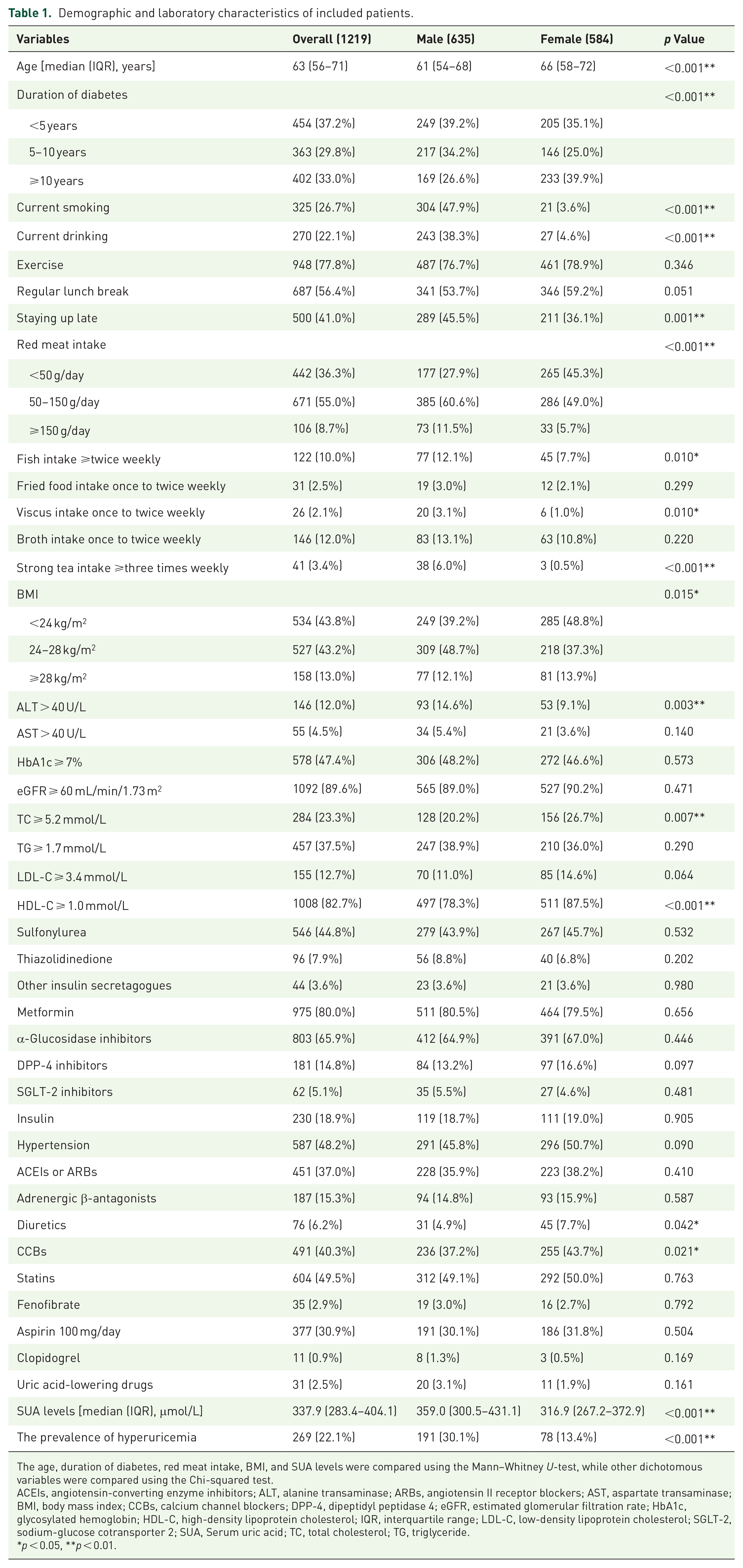
The age, duration of diabetes, red meat intake, BMI, and SUA levels were compared using the Mann–Whitney
ACEIs, angiotensin-converting enzyme inhibitors; ALT, alanine transaminase; ARBs, angiotensin II receptor blockers; AST, aspartate transaminase; BMI, body mass index; CCBs, calcium channel blockers; DPP-4, dipeptidyl peptidase 4; eGFR, estimated glomerular filtration rate; HbA1c, glycosylated hemoglobin; HDL-C, high-density lipoprotein cholesterol; IQR, interquartile range; LDL-C, low-density lipoprotein cholesterol; SGLT-2, sodium-glucose cotransporter 2; SUA, Serum uric acid; TC, total cholesterol; TG, triglyceride.
Prevalence of hyperuricemia in patients with diabetes
The overall prevalence of hyperuricemia was 21.24% (335/1577), among which 233 (69.55%) were males and 102 (30.45%) were females. Males exhibited a significantly higher prevalence of hyperuricemia than females (29.35% in males
Participants were categorized into three or four groups to assess differences in hyperuricemia prevalence and SUA levels according to age and BMI. Hyperuricemia prevalence and SUA levels were lowest in males and females aged 55–65 years [Figure 1(a) and (b)]. With increasing BMI, the prevalence of hyperuricemia and SUA levels in males and SUA levels in females also gradually increased [Figure 1(c) and (d)]. The highest prevalence of hyperuricemia was documented when the BMI ranged between 24 and 28 kg/m2, with the lowest noted when BMI was <24 kg/m2 in females [Figure 1(c)]. Notably, hyperuricemia rates and SUA levels in males were significantly higher than those in women in each age and BMI group.

Hyperuricemia prevalence and SUA levels at different ages and BMI. Panel (a) Hyperuricemia prevalence at different ages. Panel (b) SUA levels at different ages. Panel (c) Hyperuricemia prevalence at different BMI. Panel (d) SUA levels at different BMI (*
Factors associated with hyperuricemia in patients with diabetes
Univariate logistic regression analysis revealed that in males, current smoking, current drinking, red meat intake 50–150 g/day, strong tea intake greater than or equal to three times weekly, overweight, obesity, TG ⩾ 1.7 mmol/L, hypertension, ACEIs or ARBs, adrenergic β-antagonists, and diuretics were positively associated with the risk of hyperuricemia in patients with diabetes. In addition, hyperuricemia was negatively associated with advanced age, eGFR ⩾ 60 mL/min/1.73 m2, HbA1c ⩾ 7%, and HDL-C ⩾ 1.0 mmol/L (Table 2).
Univariate logistic analysis of hyperuricemia in patients with diabetes.

ACEIs, angiotensin-converting enzyme inhibitors; ALT, alanine transaminase; ARBs, angiotensin II receptor blockers; AST, aspartate transaminase; BMI, body mass index; CCBs, calcium channel blockers; CI, confidence interval; DPP-4, dipeptidyl peptidase 4; eGFR, estimated glomerular filtration rate; HbA1c, glycosylated hemoglobin; HDL-C, high-density lipoprotein cholesterol; IQR, interquartile range; LDL-C, low-density lipoprotein cholesterol; SGLT-2, sodium-glucose cotransporter 2; TC, total cholesterol; TG, triglyceride.
In male patients with diabetes, overweight, obesity, and TG ⩾ 1.7 mmol/L were positively correlated with the presence of hyperuricemia, whereas HbA1c ⩾ 7%, eGFR ⩾ 60 mL/min/1.73 m2, and SGLT-2 inhibitors were negatively associated with the risk of hyperuricemia. In female patients with diabetes, hyperuricemia showed a significant positive correlation with overweight, obesity, ALT > 40 U/L, AST > 40 U/L, TG ⩾ 1.7 mmol/L, hypertension, ACEIs or ARBs, CCBs, adrenergic β-antagonists, and diuretics, while eGFR ⩾ 60 mL/min/1.73 m2 and HDL-C ⩾ 1.0 mmol/L showed significant negative correlations with hyperuricemia (Table 2).
Furthermore, multivariate logistic regression was used to explore independently associated factors. The results showed that male sex, overweight, obesity, TG ⩾ 1.7 mmol/L, and diuretics were positively associated with the presence of hyperuricemia in all patients with diabetes, while eGFR ⩾ 60 mL/min/1.73 m2, HbA1c ⩾ 7%, and fenofibrate were negatively associated with the risk of hyperuricemia (Table 3). Considering male subjects, patients with obesity or TG ⩾ 1.7 mmol/L had a higher risk of hyperuricemia, whereas hyperuricemia was negatively associated with eGFR ⩾ 60 mL/min/1.73 m2, HbA1c ⩾ 7%, fenofibrate, and SGLT-2 inhibitors (Table 4). In female subjects, overweight, ALT > 40 U/L, and TG ⩾ 1.7 mmol/L showed a significant positive correlation with hyperuricemia, and eGFR ⩾ 60 mL/min/1.73 m2 was negatively associated with the risk of hyperuricemia (Table 5).
Multivariate logistic analysis of hyperuricemia in patients with diabetes (

BMI, body mass index; CI, confidence interval; eGFR, estimated glomerular filtration rate; HbA1c, glycosylated hemoglobin; OR, odds ratio; SE, standard error; SGLT-2, sodium-glucose cotransporter 2; TG, triglyceride.
Multivariate logistic analysis of hyperuricemia in male patients with diabetes (

BMI, body mass index; CI, confidence interval; eGFR, estimated glomerular filtration rate; HbA1c, glycosylated hemoglobin; SE, standard error; SGLT-2, sodium-glucose cotransporter 2; OR, odds ratio; TG, triglyceride.
Multivariate logistic analysis of hyperuricemia in female patients with diabetes (

ALT, alanine transaminase; BMI, body mass index; CI, confidence interval; eGFR, estimated glomerular filtration rate; HbA1c, glycosylated hemoglobin; OR, odds ratio; SE, standard error; TG, triglyceride.
Discussion
In the present study, we found that the prevalence of hyperuricemia in patients with diabetes in Chenghua District, Chengdu, was 21.24%, and the prevalence of hyperuricemia and SUA levels in males were significantly higher than those in females. After adjusting for multiple variables, male sex, overweight, obesity, TG ⩾ 1.7 mmol/L, and diuretics were positively associated with the presence of hyperuricemia in all patients with diabetes, while eGFR ⩾ 60 mL/min/1.73 m2, HbA1c ⩾ 7%, and fenofibrate were negatively associated with the risk of hyperuricemia. Male patients with obesity or TG ⩾ 1.7 mmol/L had a higher risk of hyperuricemia, whereas hyperuricemia was negatively associated with eGFR ⩾ 60 mL/min/1.73 m2, HbA1c ⩾ 7%, fenofibrate, and SGLT-2 inhibitors. Considering female subjects, overweight, ALT > 40 U/L, and TG ⩾ 1.7 mmol/L demonstrated a significant positive correlation with hyperuricemia, while eGFR ⩾ 60 mL/min/1.73 m2 was negatively associated with the risk of hyperuricemia. It is worth noting that since only six patients with T1DM were included in the analysis of hyperuricemia prevalence and SUA levels, and only four patients with T1DM were included in the analysis of associated factors, our conclusions mainly represented patients with T2DM.
Combining the results of two published studies on the general population in Southwest China, we found that the prevalence of hyperuricemia in male patients with diabetes (29.35%) was higher than that in the general population (18.26%).27,28 In the above-cited studies, hyperuricemia in females was defined as >360 μmol/L, which was lower than that in the present study. However, the prevalence of hyperuricemia in female patients with diabetes (13.03%) was still higher than that in normal females (9.84%). Thus, the prevalence of hyperuricemia is higher than that in the general population. This phenomenon could be attributed to the direct impact of diabetes on hyperuricemia and elevated BMI and TG levels in patients with diabetes. Menopause is an independent risk factor for hyperuricemia.
29
After excluding the effect of estrogen on SUA, progesterone levels were also negatively associated with SUA.
30
Although menopausal female subjects lacked the protective effect of estrogen and progesterone in the present study, the hyperuricemia prevalence in males was still approximately twice that in females, which might result from higher current smoking and drinking rates and more red meat and viscera intake in males than those in females (Table 1). On analyzing 10,141 general subjects, Huang
High TG positively correlated with insulin resistance, and aggravated insulin resistance led to upregulation of URAT1 expression and uric acid reabsorption, clarifying why patients with diabetes exhibiting TG levels ⩾1.7 mmol/L had a high risk of developing hyperuricemia.34,35 Our hypothesis corroborated recently published studies documenting that only TG was positively correlated with SUA among TG, TC, LDL-C, and HDL-C in patients with T2DM.36,37 Therefore, patients with diabetes whose TG level exceeded 1.7 mmol/L should pay more attention to the occurrence of hyperuricemia. Our results revealed that ALT levels >40 U/L were positively associated with the risk of hyperuricemia only in female patients with diabetes. Hyperuricemia is closely associated with nonalcoholic fatty liver disease (NAFLD).
38
ALT, a surrogate marker of NAFLD, is associated with fat deposition in hepatocytes.
38
A similar study has shown that ALT can be positively associated with SUA in female patients with diabetes.
36
Moreover, Klisic
Given that the kidneys excrete approximately 70% of uric acid, reduced eGFR can result in disorganized uric acid excretion and elevated SUA levels. The negative correlation between eGFR and hyperuricemia or SUA levels in patients with diabetes and those at high cardiovascular risk has also been confirmed.17,36,37,41,42 However, the correlation between HbA1c level and hyperuricemia remains controversial. Rafiullah
Notably, overweight or obese patients with diabetes had a high risk of hyperuricemia, who needed further reduction of their weight to avoid the occurrence of hyperuricemia. Adipose tissue can produce and secrete uric acid through xanthine oxidoreductase, with enhanced uric acid production and reduced excretion documented in obese individuals.
45
Previous studies have demonstrated that BMI positively correlates with SUA levels in male and female patients with T2DM.36,37 Ishizaka
The use of diuretics could increase the risk of hyperuricemia in patients with diabetes, given that diuretics can inhibit uric acid secretion from the proximal tubules and reduce blood volume. 47 Hydrochlorothiazide and indapamide have been shown to increase SUA levels in patients with hypertension.48,49 Herein, diuretics were positively associated with the risk of hyperuricemia in patients with diabetes, but the correlation disappeared in male and female subjects, resulting from only 6.2% of subjects using diuretics (81.6% hydrochlorothiazide, 11.8% indapamide, 3.9% furosemide, 6.6% spironolactone, and 1.3% triamterene). In all, we suggested that diuretics should be avoided in patients with diabetes to prevent hyperuricemia. Hyperuricemia was found to be negatively associated with SGLT-2 inhibitors and fenofibrate in male patients with diabetes. SGLT-2 inhibitors can reduce urinary glucose reabsorption, and the increased glucose concentration in renal tubules stimulates glucose reabsorption by GLUT9, which can reverse the transport of glucose and uric acid and promote uric acid excretion. 50 Moreover, empagliflozin has been shown to upregulate ABCG2 expression and promote uric acid secretion in diabetic mice. 51 Fenofibrate promoted uric acid excretion by inhibiting renal URAT1 expression, and this effect was unrelated to lipid parameters. 52 Therefore, SGLT-2 inhibitors and fenofibrate may bring more benefits to male patients with diabetes to prevent hyperuricemia.
The limitations of the present study may need to be addressed. First, the study was cross-sectional; therefore, causal and temporal relationships could not be obtained. Second, we failed to establish lifestyle habits and BMI data for 22.7% of participants, which led to potential selection bias. Proteinuria is an early marker of renal injury; however, this study lacked data regarding proteinuria and cannot reveal the correlation between early renal injury and hyperuricemia. Third, the prevalence of hyperuricemia and SUA levels in the present study only represented the situation of patients with diabetes in Chenghua District, Chengdu. Finally, the sample size was small, and the low utilization rate of some drugs might have impacted our conclusions.
Conclusion
We found a high prevalence of hyperuricemia in patients with diabetes. We suggest that SUA levels should be monitored in patients with diabetes. Moreover, appropriate lifestyle and drug use may reduce the incidence of hyperuricemia and improve SUA levels in patients with diabetes. As the prevalence of hyperuricemia varies significantly in different regions, additional studies should be undertaken in other regions to establish the prevalence of hyperuricemia in patients with diabetes. Furthermore, the relationship between complications of diabetes and hyperuricemia in patients with diabetes remains to be explored.
