Abstract
Background
Emerging evidence suggests that elevated serum uric acid levels may be associated with adverse pregnancy outcomes.
Objectives
To investigate the association between serum uric acid levels and adverse pregnancy outcomes among pregnant women.
Materials and methods
A prospective cohort study was conducted among 200 pregnant women attending Vinh Long General Hospital, including 100 women with hyperuricemia and 100 without hyperuricemia. Hyperuricemia was defined as a serum uric acid level >360 µmol/L. Pregnancy-, delivery-, and postpartum-related complications were recorded and compared between the two groups.
Results
The mean maternal age was 25.06 ± 5.79 years. In the hyperuricemia group, 45.0% were primigravida, whereas 22.0% had multiple pregnancies. Adverse pregnancy outcomes were more frequent in women with hyperuricemia than in those without hyperuricemia (28.0% vs. 15.0%; odds ratio = 2.20; 95% confidence interval: 1.09–4.44). Within the hyperuricemia group, smoking was more common among women who developed complications than among those without complications (42.9% vs. 18.1%), along with lower platelet counts and higher creatinine levels (p < 0.05). Multivariable logistic regression analysis identified alcohol consumption (odds ratio = 3.85), serum creatinine (odds ratio = 1.19), and hyperuricemia (odds ratio = 1.65) as independent predictors of adverse pregnancy outcomes (p < 0.05).
Conclusion
Hyperuricemia was independently associated with an increased risk of adverse pregnancy outcomes.
Introduction
Despite substantial advances in obstetric care, serious pregnancy complications, including preeclampsia (PE), preterm birth, intrauterine growth restriction, postpartum hemorrhage, and perinatal mortality, remain major challenges to maternal and neonatal health worldwide. These conditions are associated with significantly increased maternal and neonatal mortality, particularly in developing countries, and impose a substantial economic burden on healthcare systems.1–3 The prevalence of gestational hypertension and adverse pregnancy outcomes is often higher among women with underlying metabolic disorders. 4 Uric acid (UA), a product of purine metabolism, contributes to oxidative stress, endothelial dysfunction, and low-grade inflammation. Accumulating evidence suggests that elevated UA levels are associated with the severity and prognosis of preeclampsia and may predict adverse maternal and fetal events during pregnancy. In a cohort study by Yue et al., 5 women with serum UA (SUA) ≥240 µmol/L demonstrated a 1.25-fold higher risk of developing preeclampsia. Hyperuricemia has also been linked to increased rates of gestational hypertension, gestational diabetes, lower birth weight, and obstetric complications such as postpartum hemorrhage and eclampsia.6–8 Although UA levels may rise physiologically during late pregnancy, 2 early detection of elevated UA concentrations could serve as an important marker for identifying women at risk for pregnancy complications. Therefore, this study aimed to evaluate the association between SUA levels and pregnancy complications among Vietnamese pregnant women, providing additional evidence on this topic.
Materials and methods
Study design and population
The study was conducted using a prospective cohort design and included pregnant women attending routine antenatal care at Vinh Long General Hospital between March 2023 and March 2025. SUA levels were measured during the first trimester, prior to the occurrence of obstetric complications. At this stage of pregnancy, UA levels are less affected by physiological changes such as reduced glomerular filtration rate or placental dysfunction, which are more commonly observed in later trimesters. Participants were classified into two cohorts based on the SUA concentrations: a hyperuricemia group and a nonhyperuricemia group. Hyperuricemia was defined as a SUA >360 µmol/L in women. 9 The two cohorts were selected to be comparable in gestational age at the start of follow-up to minimize bias related to differences in pregnancy stage. Pregnant women were excluded if they met any of the following criteria: (a) a history of gout or congenital purine metabolism disorders; (b) a diagnosis of chronic kidney disease prior to pregnancy; (c) hematologic disorders affecting SUA levels (e.g. acute leukemia, lymphoma, and polycythemia); (d) use of medications, including thiazide diuretics or furosemide, that may influence the UA levels. Additionally, women who discontinued monthly follow-up visits were excluded. Convenience sampling was applied, and all eligible pregnant women meeting the inclusion criteria during the study period were consecutively recruited. A total of 200 participants were enrolled, including 100 pregnant women in the hyperuricemia group and 100 in the nonhyperuricemia group.
Data collection
Clinical and demographic data were obtained through structured interviews and medical records. Risk factors, including smoking and alcohol consumption, were documented. Prepregnancy body mass index (BMI) was calculated within 3 months prior to conception, and overweight/obesity was defined as BMI ≥23 kg/m2 according to Asian criteria. 10 All participants underwent monthly antenatal checkups. Pregnancy-related complications, including gestational hypertension, gestational diabetes, preeclampsia/eclampsia, urinary tract infection, placental abruption, polyhydramnios, and oligohydramnios, were recorded. Gestational hypertension was defined as new-onset hypertension after 20 weeks of gestation without proteinuria or target-organ dysfunction. Preeclampsia was diagnosed as gestational hypertension accompanied by proteinuria (≥300 mg/24 h or protein/creatinine ratio ≥0.3). 11
Adverse events occurring during delivery and the postpartum period may be influenced by obstetric interventions, mode of delivery, and postpartum care; therefore, they differ from the complications arising during pregnancy. Delivery- and postpartum-related complications, including hematoma, surgical site infection, postpartum infection, and postpartum hemorrhage, were also recorded. Postpartum hemorrhage was defined as blood loss of ≥500 mL after vaginal delivery or ≥1000 mL after cesarean section, occurring within the first 24 h after delivery (primary postpartum hemorrhage) or after 24 h up to 6 weeks postpartum (secondary postpartum hemorrhage). 12 At the end of follow-up, the incidence of adverse outcomes was documented in two categories: (a) complications occurring during pregnancy and (b) complications occurring during labor and the postpartum period.
Statistical analysis
Data were analyzed using Statistical Package for Social Sciences (SPSS) v20.0. Continuous variables were expressed as mean ± SD, whereas categorical variables were expressed as frequencies and percentages. The Kolmogorov–Smirnov test was used to assess normality. Between-group differences were evaluated using Chi-square or Fisher’s exact test for categorical variables and independent t-tests for continuous variables. A p-value <0.05 was considered statistically significant. All variables related to the SUA levels and pregnancy outcomes were completely collected for all 200 participants, with no missing data. Therefore, all participants were included in the final analysis, ensuring a comprehensive assessment of the association between the SUA levels and pregnancy outcomes.
Ethical approval
All pregnant women participating in the study were fully informed about the study objectives, content, and procedures, and they provided written informed consent prior to enrollment. Participants were allowed to withdraw from the study at any time without any impact on their medical care or treatment. All personal information and data collected were kept strictly confidential, were used solely for research purposes, and were not disclosed publicly without the participant’s consent. The study was approved by the Institutional Ethics Committee for Biomedical Research (Approval Number: 08/BVDKVL, dated 08 February 2023) and was conducted in accordance with the ethical principles of the revised 2024 Declaration of Helsinki and in compliance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 13
Results
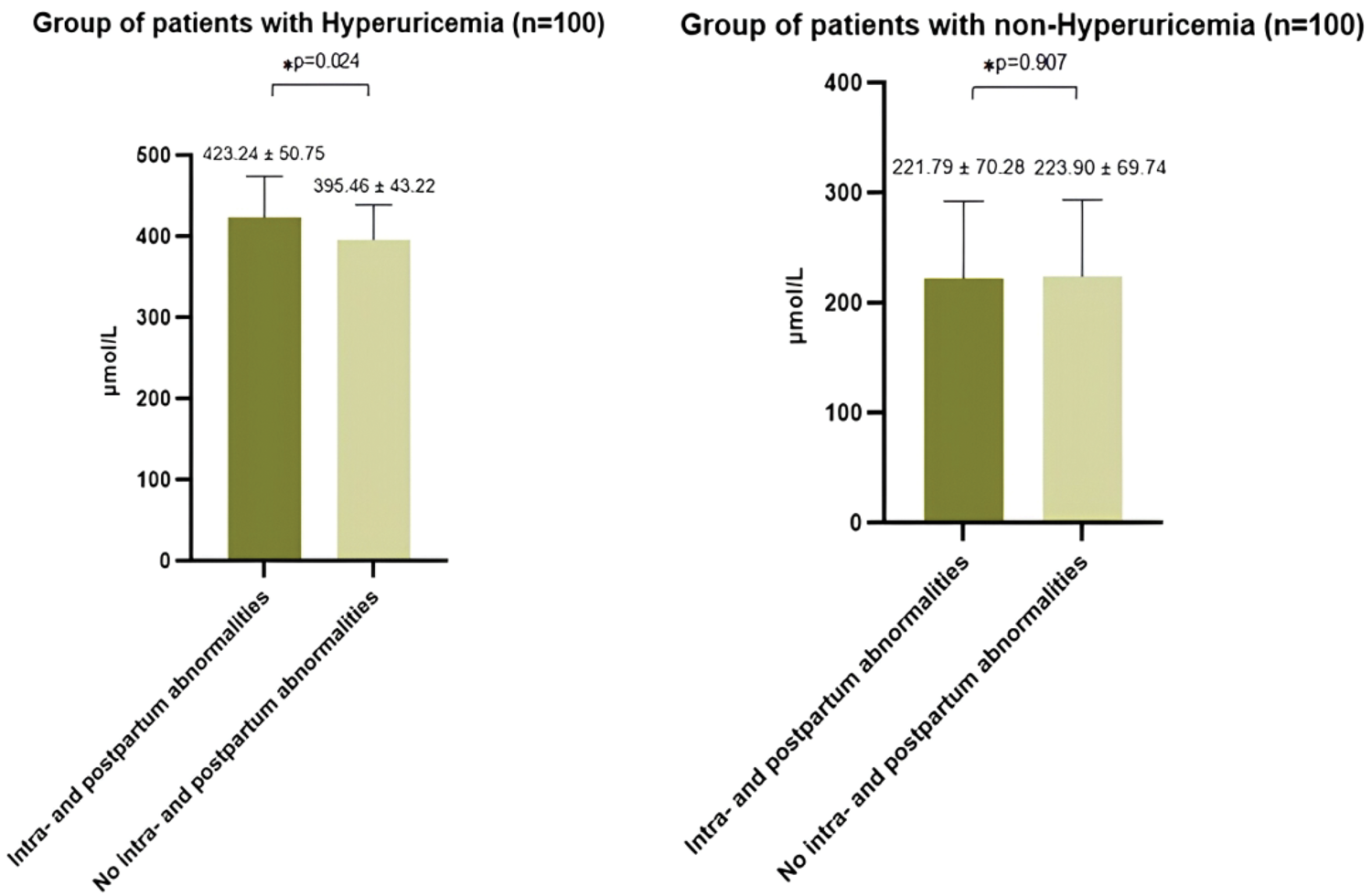
The mean maternal age was 25.06 ± 5.79 years. The prevalence of primigravidity (45.0%) and multiple pregnancies (22.0%) did not differ significantly between the groups. However, the mean serum creatinine levels (SCr) were higher among women with hyperuricemia (92.94 ± 55.11 µmol/L) than in those without hyperuricemia (67.96 ±10.54 µmol/L, p <0.001) (Table 1). Overall, 21.5% of the participants experienced complications, with a higher rate observed in the hyperuricemic group (28.0%) than in the nonhyperuricemia group (15.0%) (odd ratio (OR) = 2.20; 95% confidence interval (CI): 1.09–4.44; p = 0.025) (Table 2). The incidence of complications during and after delivery was comparable between the two groups (16.0% vs. 19.0%; p > 0.05) (Table 3). In the hyperuricemia group, the proportion of pregnant women with pregnancy-related complications who reported smoking was 42.9%, which was significantly higher than 18.1% among those without complications. Additionally, these women exhibited lower platelet counts (194.96 ± 72.19 g/L vs. 226.08 ± 63.49 g/L) and higher SCr levels (115.15 ± 73.14 µmol/L vs. 84.29 ± 43.94 µmol/L) (Table 4). Among pregnant women with hyperuricemia, those who experienced intrapartum or postpartum complications had a higher prevalence of smoking and alcohol consumption than those without complications (50.0% vs. 20.2% and 50.0% vs. 17.9%, p < 0.05) (Table 5). In this group, pregnant women who developed complications during pregnancy or in the intrapartum and postpartum period exhibited higher mean SUA levels than those without complications. A similar trend was observed in the group with normal UA levels (Figure 1 and Figure 2). Analysis of the correlation between the UA levels and selected parameters (Table 6) revealed a positive association between UA and systolic blood pressure, diastolic blood pressure, and total cholesterol in both groups. In the hyperuricemia group, SUA showed a significant positive correlation with serum creatinine (r = 0.268, p < 0.05). In the multivariable logistic regression analysis, after adjustment for anthropometric characteristics (age and BMI), primigravida status, multiple pregnancy, smoking, alcohol consumption, and biochemical markers, alcohol use (OR = 3.85; 95% CI: 1.42–5.45; p < 0.05), serum creatinine level (OR =1.19; 95% CI: 0.98–1.32; p < 0.05), and hyperuricemia (OR = 1.65; 95% CI: 1.12–3.81; p < 0.05) were identified as independent factors associated with adverse pregnancy outcomes. In contrast, for complications occurring during labor and the postpartum period, only smoking (OR = 2.76; 95% CI: 1.03–7.41; p < 0.05) and aspartate aminotransferase (AST) levels (OR = 0.97; 95% CI: 0.95–0.99; p < 0.05) were found to be independently associated with these outcomes. No statistically significant interaction was observed between hyperuricemia and maternal age or prepregnancy BMI (all interaction p-values >0.05), indicating that the association between hyperuricemia and adverse pregnancy outcomes was relatively consistent across the maternal age groups and prepregnancy BMI levels. (Table 7).
Baseline characteristics.
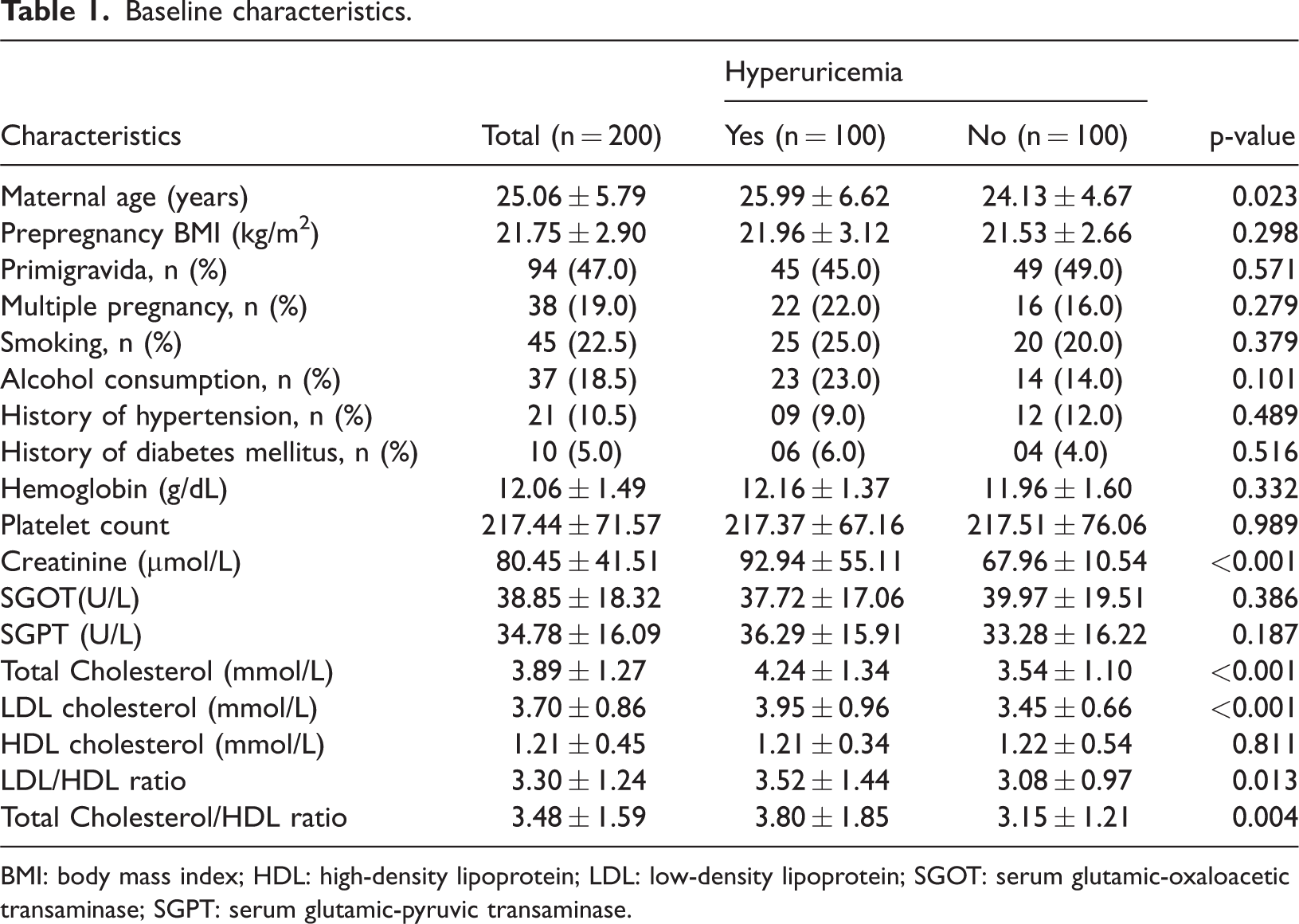
BMI: body mass index; HDL: high-density lipoprotein; LDL: low-density lipoprotein; SGOT: serum glutamic-oxaloacetic transaminase; SGPT: serum glutamic-pyruvic transaminase.
Prevalence of pregnancy complications among study participants.
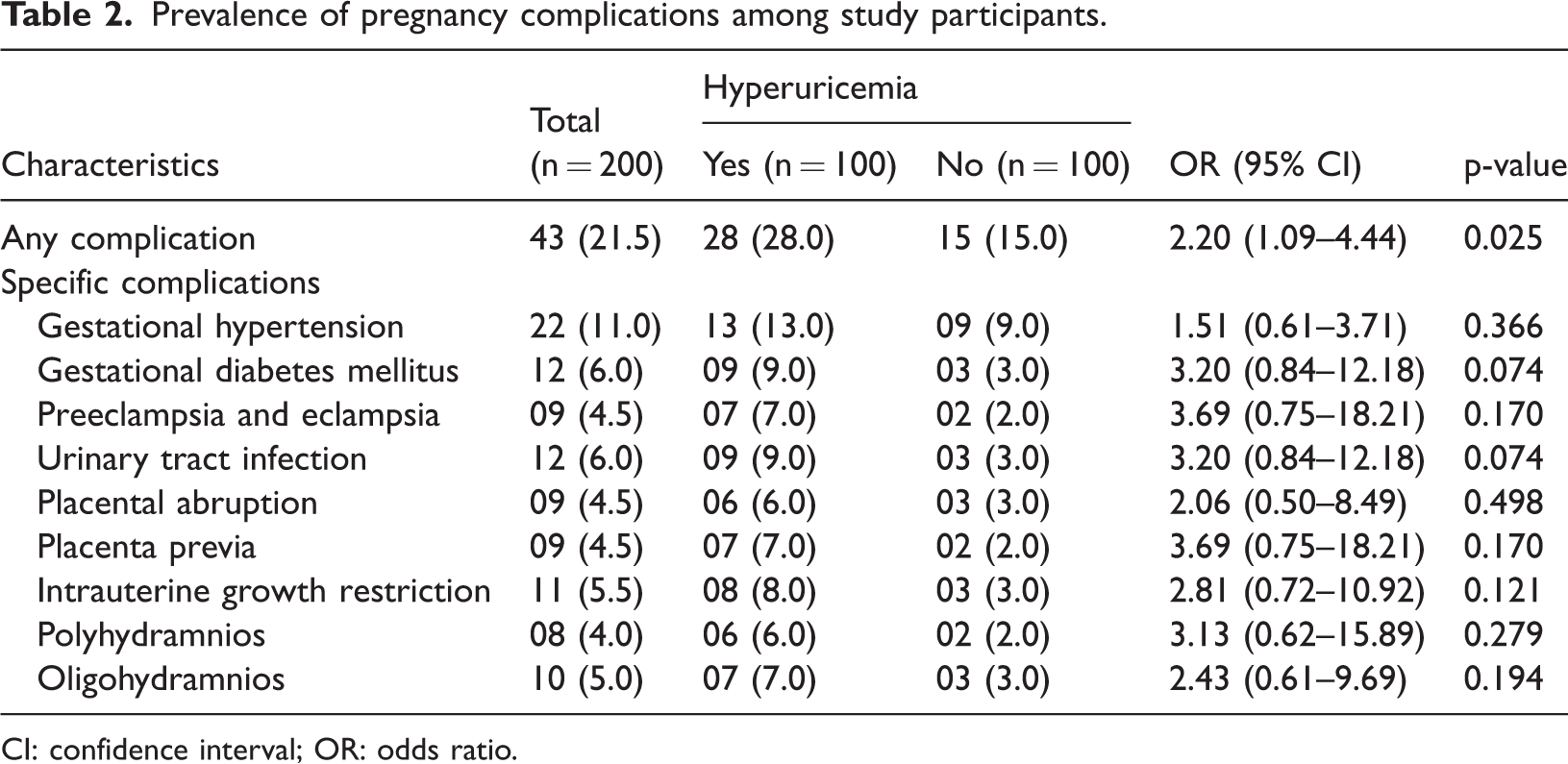
CI: confidence interval; OR: odds ratio.
Prevalence of intrapartum and postpartum complications.

CI: confidence interval; OR: odds ratio.
Factors associated with pregnancy complications.
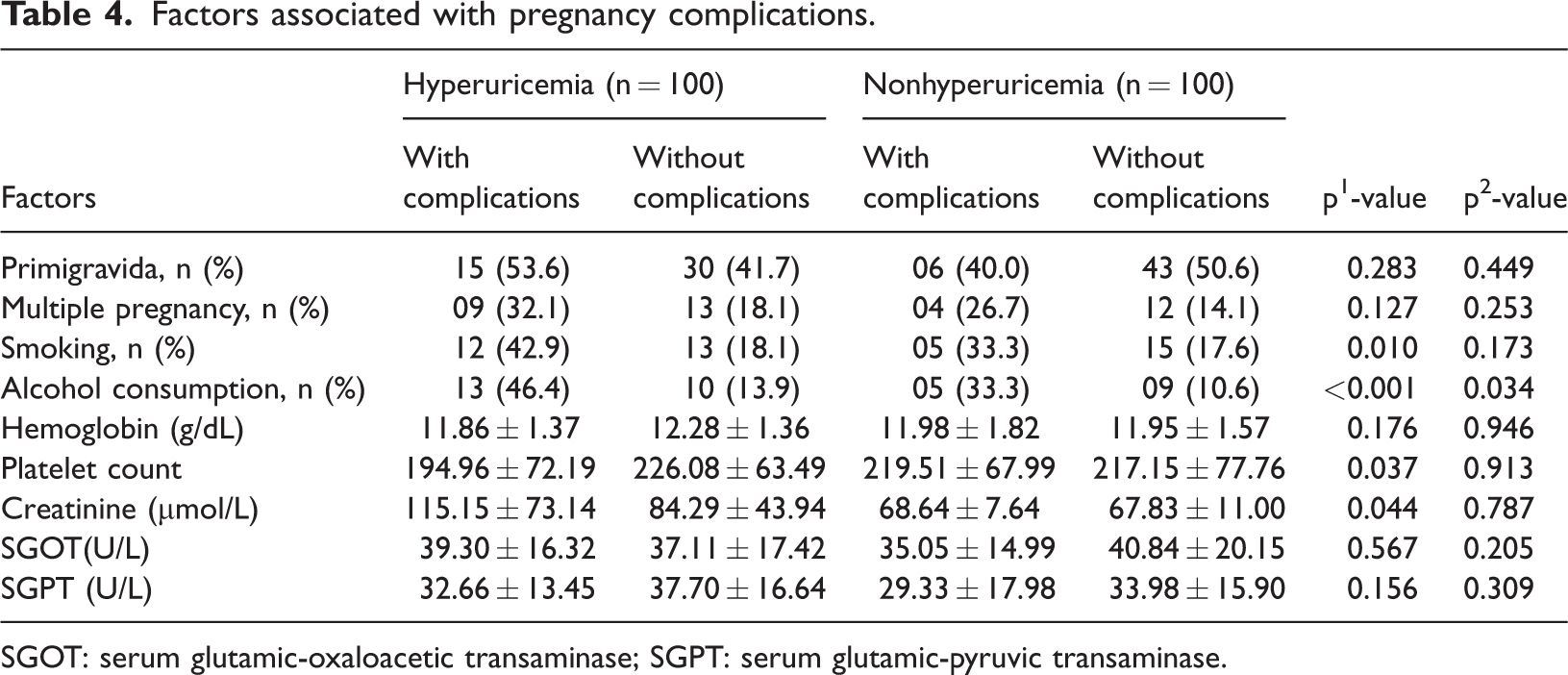
SGOT: serum glutamic-oxaloacetic transaminase; SGPT: serum glutamic-pyruvic transaminase.
Factors associated with abnormalities during and after delivery.

SGOT: serum glutamic-oxaloacetic transaminase; SGPT: serum glutamic-pyruvic transaminase.

Comparison of serum uric acid levels according to the presence of pregnancy complications.
Comparison of serum uric acid levels according to the presence of intrapartum and postpartum complications.
Correlation between serum uric acid levels and selected parameters.

SGOT: serum glutamic-oxaloacetic transaminase; SGPT: serum glutamic-pyruvic transaminase.
Multivariable logistic regression analysis of factors associated with adverse outcomes during pregnancy, at delivery, and in the postpartum period.
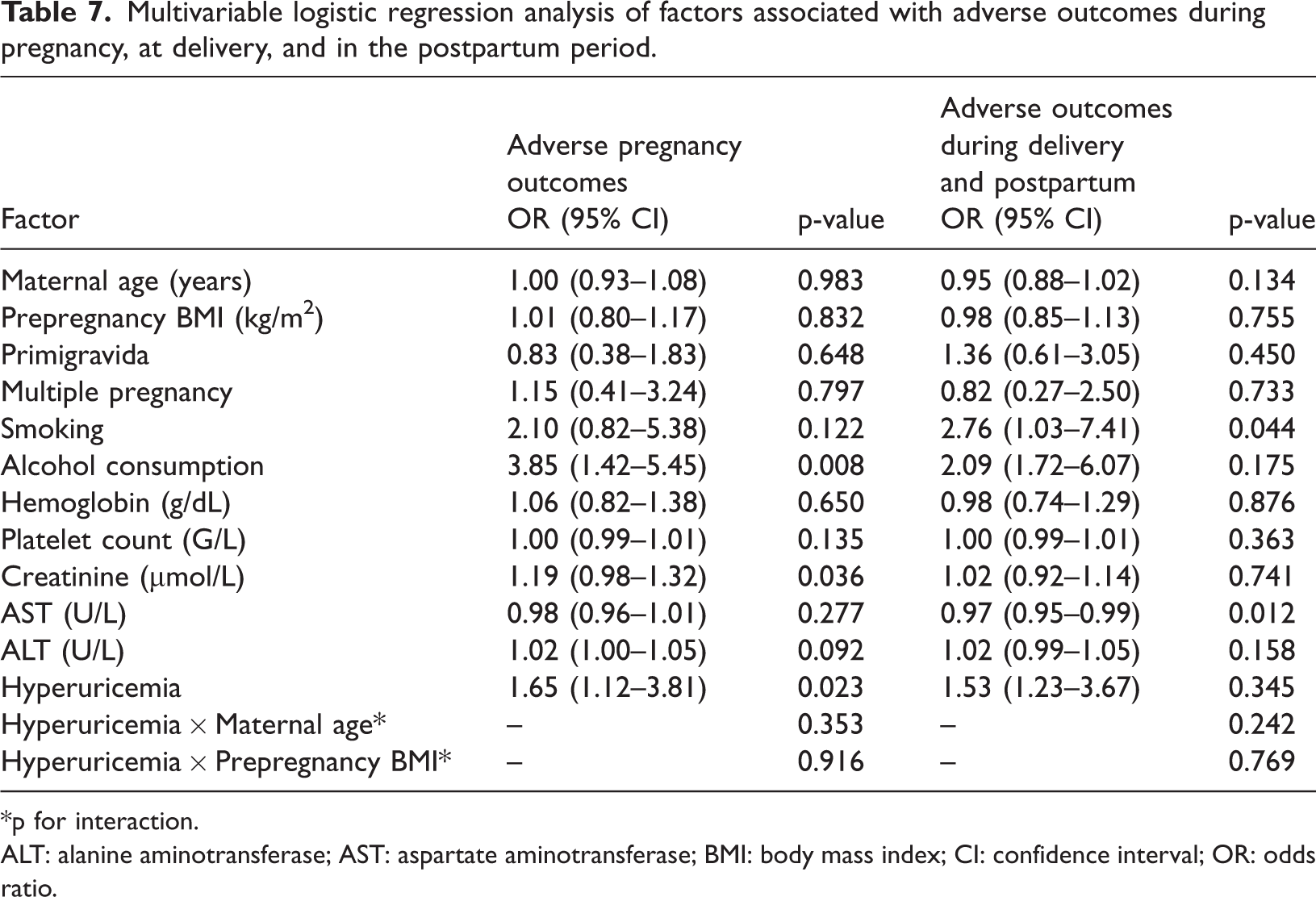
*p for interaction.
ALT: alanine aminotransferase; AST: aspartate aminotransferase; BMI: body mass index; CI: confidence interval; OR: odds ratio.
Discussion
Among the 200 pregnant women followed, we found that, in the hyperuricemia group, the proportion of women who smoked was significantly higher among those who developed adverse pregnancy outcomes compared with those without complications (42.9% vs. 18.1%). Similarly, platelet counts were lower and SCr levels were higher in women with adverse pregnancy outcomes (p < 0.05). In the multivariable regression analysis, alcohol consumption, SCr levels, and hyperuricemia were identified as independent factors associated with adverse outcomes during pregnancy. In contrast, for complications occurring during labor and the postpartum period, only smoking and AST levels were found to be significantly associated (p < 0.05). Maternal hyperuricemia has been observed to be associated with a higher incidence of pregnancy complications, including gestational hypertension and preeclampsia. SUA levels are generally elevated in women with preeclampsia compared with those with uncomplicated pregnancies. Although not all women with preeclampsia develop hyperuricemia, the proportion of patients with preeclampsia who exhibit elevated UA levels ranges from approximately 40% to 90% across studies, partly due to inconsistent definitions of hyperuricemia during pregnancy and physiological variations in the UA levels according to gestational age. 14 Hyperuricemia has been shown to predict the development of preeclampsia in early pregnancy. In a retrospective cohort study including 4725 women, with preeclampsia as the primary outcome and preterm birth and low birth weight as secondary outcomes, Yue et al. 5 reported that women with UA ≥240 µmol/L demonstrated a higher risk of preeclampsia (OR = 1.25; 95% CI: 0.96–1.65) than those with UA <240 µmol/L. Elevated UA levels before 20 weeks of gestation were associated with the subsequent development of preeclampsia, particularly during the early gestational period (8–12 weeks), with the effect gradually attenuating with increased gestational age.
Hyperuricemia has also been strongly associated with an increased risk of preterm birth. One possible mechanism is that elevated UA reflects placental dysfunction or systemic inflammation, which may contribute to the initiation of early labor. Evidence from a large cohort study in China supports this hypothesis, reporting that the rate of delivery before 37 weeks among women with hyperuricemia (>360 µmol/L) reached 17.3%, more than threefold higher than the overall population rate (5.4%). In the same study, women with hyperuricemia also exhibited higher rates of gestational diabetes mellitus (GDM) (28.2% vs. 16.0%) and low birth weight (<2500 g) (14.8% vs. 3.1%) compared with those with normal UA levels. 15 Unlike our study, which assessed UA during the first trimester, Xiong et al. 15 measured the UA levels during early, mid, and late pregnancy. Nevertheless, findings from both studies suggest that UA levels are associated with adverse pregnancy outcomes. Similarly, Zhang et al. 16 conducted a prospective cohort study involving 33,030 pregnant women. The reported incidence rates of GDM, pregnancy-induced hypertension (PIH), cesarean delivery, preterm birth, large-for-gestational-age (LGA) infants, small-for-gestational-age infants, and low Apgar score were 15.18%, 7.96%, 37.62%, 4.93%, 9.39%, 4.79%, and 0.28%, respectively. SUA was assessed during the first trimester, consistent with our study design. Their results demonstrated that the highest quartile of the SUA/SCr ratio was associated with the greatest risk of GDM (OR = 2.14; 95% CI: 1.93–2.36), PIH (OR = 1.79; 95% CI: 1.58–2.04), cesarean section (OR = 1.24; 95% CI: 1.16–1.33), and preterm birth (OR = 1.30; 95% CI: 1.12–1.51). A largely linear relationship was observed between SUA/SCr and adverse pregnancy outcomes, except for GDM. Subgroup analyses further revealed that the association between SUA/SCr and the risk of PIH and LGA was significantly stronger in younger pregnant women (p = 0.033 and p = 0.035, respectively). In another study, Ahmed et al. 17 compared pregnancy-related outcomes before, during, and after pregnancy between women with elevated UA levels (>6.0 mg/dL) and those with UA <6.0 mg/dL. Their findings indicated that hyperuricemia was a risk factor for maternal complications such as postpartum hemorrhage; eclampsia; placental abruption; and hemolysis, elevated liver enzymes, and low platelet count (HELLP) syndrome. Similarly, Amini et al. 18 reported that hyperuricemia in pregnant women was independently associated with adverse outcomes, including preterm birth (OR = 3.17; 95% CI: 2.10–4.79), low birth weight (OR = 1.28; 95% CI: 1.04–2.57), neonatal intensive care unit admission (OR = 1.65; 95% CI: 1.12–2.94), and neonatal intracranial hemorrhage (OR = 8.14; 95% CI: 1.11–87.1). Notably, this study focused on fetal outcomes and measured maternal UA, creatinine, and blood urea nitrogen levels within 24 h prior to delivery. Of particular interest, a retrospective cohort study of 1027 women undergoing assisted reproductive technology (ART) reported a GDM prevalence of 16.7%. Prepregnancy SUA levels were independently associated with the risk of developing GDM after adjustment for confounders, and this association became stronger when SUA/SCr was used. The authors concluded that UA, particularly when normalized to renal function, may serve as an early marker associated with GDM risk in women undergoing ART. 19 Recent meta-analyses further support the predictive potential of UA. Nikparast et al. 20 evaluated 23 studies involving 105,380 pregnant women and found that elevated UA levels were significantly associated with an increased risk of GDM (OR = 2.58; 95% CI: 1.89–3.52; p < 0.05). Gestational age subgroup analyses demonstrated that higher UA levels before 20 weeks were significantly associated with an increased risk of GDM (OR = 3.26; 95% CI: 2.26–4.71; p < 0.05). Similarly, a meta-analysis conducted in China reported a positive association between elevated the UA levels and the risk of GDM, particularly during the first trimester. 8 Colmenares-Mejia et al. 21 also identified a linear relationship between increasing UA levels and the presence of preeclampsia. After adjustment for confounders, the pooled OR was 1.21 (95% CI: 1.11–1.33; p < 0.05) for each SD increase in UA. Moreover, UA measured before 20 weeks of gestation was associated with a higher risk of preeclampsia, with an OR of 1.46 (95% CI: 1.22–1.75; p < 0.05) when comparing the highest quartile to the lowest. Several studies have measured UA levels in late pregnancy,15,17,18 when concentrations may be influenced by pregnancy complications and physiological adaptations. In contrast, we assessed UA during the first trimester to better reflect baseline exposure. Furthermore, our analytical models were adjusted for renal function and other clinically relevant covariates, which may have reduced potential bias. Nevertheless, the exact mechanisms underlying the association between elevated UA and adverse pregnancy outcomes remain incompletely understood. Bainbridge et al. 22 suggested that UA promotes oxidative pathways, endothelial activation, and oxidative stress, thereby contributing to the development of preeclampsia and GDM. Other studies have proposed that metabolic abnormalities reflected by elevated UA may impair placental angiogenesis and disrupt placental circulation, particularly when elevated UA occurs early in pregnancy.23,24 Collectively, these mechanisms may partially explain the higher risk of complicated pregnancy observed in women with hyperuricemia, consistent with our findings and previous reports.4,6,7,11,22,23
Blood pressure is closely associated with metabolic status and may act as a potential confounder in the relationship between UA levels and adverse pregnancy outcomes. Recent studies have shown that even high-normal blood pressure before pregnancy may be associated with unfavorable reproductive outcomes. Therefore, the absence of data on prepregnancy blood pressure in our study may limit the ability to fully evaluate the independent association between the UA levels and adverse pregnancy outcomes, and this limitation should be considered when interpreting our findings. 25 Additionally, the hyperuricemia group demonstrated significantly higher low-density lipoprotein (LDL)/high-density lipoprotein (HDL) ratios and total cholesterol (TC)/HDL ratios compared with the nonhyperuricemia group (p < 0.05). Evidence from a recent study in China suggested that among women undergoing ART, the prepregnancy lipid ratios such as LDL/HDL and TC/HDL were associated with the risk of GDM and other adverse pregnancy outcomes. These lipid parameters may be useful in estimating the risk of GDM even before ART treatment is initiated. 26 This observation suggests that, in addition to the UA levels, lipid metabolic markers serve as important prognostic indicators for adverse pregnancy outcomes. Collectively, these findings further support the role of metabolic factors and blood pressure in predicting the pregnancy outcomes among women undergoing ART. Liu et al. 26 reported that prepregnancy lipid ratios, particularly LDL/HDL and TC/HDL, may predict the risk of developing GDM, outperforming individual lipid indices. In addition, Ma et al. 25 demonstrated that even systolic blood pressure within the normal range before pregnancy was associated with a lower probability of live birth and an increased risk of pregnancy complications in women undergoing in vitro fertilization/intracytoplasmic sperm injection (IVF/ICSI). Together, these studies highlight the importance of comprehensive evaluation of lipid profiles and blood pressure prior to ART, which may not only help predict adverse outcomes but also provide opportunities for early interventions to improve pregnancy outcomes. Furthermore, Singh et al. 27 reported that SUA levels increased in parallel with the severity of hypertensive disorders of pregnancy (gestational hypertension, preeclampsia, and eclampsia), with the highest levels observed in eclampsia. Elevated SUA levels were also associated with an increased risk of preterm birth and low birth weight; however, no significant association was observed with neonatal intensive care unit admission. These findings emphasize the role of SUA as a risk-stratification marker according to disease severity and suggest that incorporating SUA into predictive models alongside other biochemical indicators may improve the ability to predict maternal and fetal complications.
In the present study, although no a priori sample size calculation or power analysis was performed, the achieved sample size (200 pregnant women equally distributed between the hyperuricemia and nonhyperuricemia groups) was sufficient for detecting a statistically significant association between hyperuricemia and adverse pregnancy outcomes. The hyperuricemia group demonstrated a markedly higher rate of pregnancy complications compared with the nonhyperuricemia group, with an OR of 2.20 (95% CI: 1.09–4.44; p < 0.05), indicating acceptable statistical power for the composite pregnancy outcome. However, for individual complications, the number of events was limited, and the study may have been underpowered to detect statistically significant differences. This limitation should be considered when interpreting results for specific outcomes. Additional limitations should also be acknowledged. Due to resource constraints and implementation conditions, the study was conducted at a single center, which may limit the generalizability of the findings. Moreover, the sample size remained relatively small, particularly for rare pregnancy-related complications, which may further reduce the ability to detect small but clinically meaningful differences. Future multicenter studies with larger sample sizes are warranted to confirm and extend these findings.
Conclusion
Pregnant women with hyperuricemia had a higher risk of developing adverse pregnancy outcomes compared with those without hyperuricemia. After adjustment for potential confounders, hyperuricemia, alcohol consumption, and SCr levels were identified as independent factors associated with adverse pregnancy outcomes. These findings suggest that SUA levels serve as a useful biomarker for risk assessment and monitoring during pregnancy.
Footnotes
Acknowledgments
We would like to express our gratitude to Can Tho University of Medicine and Pharmacy for the support and facilitation that made this study possible.
Authors’ contributions
Hang Thi Thu Ho and Ha Hong Nguyen: Writing-review & editing and Conceptualization. Hang Thi Thu Ho, Ha Hong Nguyen, Le Thi Nguyen, and Sam Phan Hai Nguyen: Writing-review & editing and Writing-original draft. Hang Thi Thu Ho and Dung My Tran: Data curation.
Consent for publication
Not applicable.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare no competing interests.
Funding
None.
