Abstract
Background:
Sepsis remains one of the leading causes of neonatal morbidity and mortality, particularly among premature infants. Blood culture is the ‘gold standard’ for the diagnosis of neonatal sepsis but is associated with several pitfalls.
Aim of the work:
We aim to evaluate the validity of measuring serum (S.Hep) and urinary hepcidin (U.Hep) concentrations as diagnostic biomarkers for late-onset sepsis (LOS) in preterm infants.
Patients and Methods:
The current case-control study included 73 cases of clinically and laboratory confirmed late-onset sepsis as the ‘case group’ and 50 nonseptic premature infants of comparable age and sex as the ‘control group’. S.Hep and U.Hep concentrations were evaluated at enrollment (acute sample) and after 1 week of treatment (convalescent sample).
Results:
Patients had a statistically significant higher concentration of both S.Hep and U.Hep as compared with nonseptic controls (p = 0.0001). Similarly, a significant reduction of both S.Hep and U.Hep was detected after 1 week of treatment (p = 0.001). A cut-off value of ⩾ 94.8 ng/ml of S.Hep and ⩾ 264 ng/mg of U.Hep/urinary creatinine showed high sensitivity, specificity, and positive predictive value in the diagnosis of neonatal LOS.
Conclusions:
Both S.Hep and U.Hep can function as promising accurate and rapid surrogate tests for the diagnosis of LOS. U.Hep measurement has the advantage of being noninvasive, with no hazards of phlebotomy, and is less variable throughout the day.
Introduction
Sepsis remains one of the leading causes of neonatal morbidity and mortality particularly, among premature infants. 1 It affects an estimated 3 million neonates worldwide with mortality of 11% to 19% and unquantified long-term neurological deficits. 2 Premature infants have a three to 10 times higher risk of developing late-onset sepsis (LOS) than full-term infants due to low levels of maternally acquired immunoglobulin G, and exposure to invasive procedures in the neonatal intensive care units (NICUs) setting. 3 Classification of neonatal sepsis based on the time of onset of symptoms helps to guide empiric antibiotic therapy as it implies a difference in the presumed mode of transmission and predominant causative organisms. 4 Clinical manifestations of neonatal sepsis are nonspecific and range from ‘not doing well’ to ‘severe focal or systemic disease’. 5
Timely diagnosis and prompt initiation of antimicrobials is paramount to mitigate the deleterious effects of sepsis and its associated morbidities and high case fatality. 6 The vexing challenge in managing neonatal sepsis is the inability to differentiate truly septic from nonseptic infants by rapid accurate tests. 7 Blood culture is the ‘gold standard’ for diagnosis of neonatal sepsis, but is associated with several pitfalls; first is the long turn-around time for results even with the updated techniques, 8 and second is the high false-negative results due to small inoculum size and intermittent nature of bacteremia. 9 Accordingly, adjunctive diagnostic tests including hematologic indices, scoring system, and biological markers should be used to aid in the antibiotic-starting decision in the presumed septic preterm infant until culture results are available. 10
Hepcidin is a 25-amino acid peptide that is produced by the liver (Hep) 11 and proved to have bactericidal properties (cidal) and accordingly labeled as hepcidin. 12 It is well reputed as a negative feedback master regulator of iron hemostasis in humans. 13 Hepcidin lowers serum iron by mediating iron sequestration inside enterocytes, macrophages, and monocyte through downregulation of ferroportin expression on iron export channels. 11 As iron is an essential element for both host and pathogen, hepcidin may play a significant role in combating infection through hypoferremia that impairs bacterial survival. 14 An additional role of hepcidin in innate immunity is by altering macrophage immune response involving cytokine and nitric oxide production. 15 Moreover, in vitro studies described hepcidin as having direct antibacterial and antifungal effects. 16 Hepcidin expression is induced by IL-6 and its urinary excretion is increased 7.5 folds within 2 hours of IL-6 infusion 13
Both 20 and 22 amino acid metabolites of hepcidin exist in urine. 17 However, only 5% of hepcidin filtered by the kidney is found in urine. This is explained by the reabsorption and degradation of hepcidin in proximal tubules similar to other small peptides. 18
This work aims to evaluate the validity of measuring serum hepcidin (S.Hep) as a diagnostic biomarker for LOS in preterm infants and to quantify its cut-off value that differentiates truly septic from nonseptic symptomatic premature infants. In addition, to examine the correlation between S.Hep and urinary hepcidin (U.Hep) to find out if measuring U.Hep can be used as an alternative safe, noninvasive biomarker for LOS diagnosis without exposing preterm infants to phlebotomy and its risks.
Patients and methods
The current case-control study was carried out in the NICU of Zagazig University Children’s Hospital–Egypt during the period from July 2019 to December 2020.
Patients
Two hundred seventy-four (274) preterm neonates were evaluated for clinically suspected LOS during the study period. They presented with variable and nonspecific manifestations. All were subjected to thorough history taking, full clinical evaluation, and routine sepsis workup as per unit protocol. Only 102 neonates fulfilled ‘proven sepsis’ criteria both clinically and laboratory, of them 23 neonates who had significant anemia and recent history of blood transfusion, or a recent history of blood transfusion, within the previous 2 weeks, had been excluded from the study. The remaining 79 cases represented the ‘case’ group, and 50 nonseptic (negative cultures) preterm infants were included as the ‘control’ group. Unfortunately, six extremely low birth weight mechanically ventilated infants succumbed before obtaining the convalescent samples. Seventy-three (73) infants completed the study (the case group), and their data were analyzed accordingly, as displayed in the study flow chart (Figure 1). All neonates in (the control group) were selected throughout the study to be of comparable demographic characteristics to our enrolled cases. They were admitted to NICU or intermediate unit with clinical manifestations that were proved to be ‘non-infectious’ by both clinical and laboratory [complete blood count (CBC), C-reactive protein (CRP), and cultures] profiles. Most of them presented with neonatal jaundice, seizures, metabolic disorders, postoperative care, noninfectious respiratory symptoms (transient tachypnea of newborn, pulmonary hypertension, polycythemia, metabolic acidosis, and diaphragmatic hernia), preterm infants staying for weight gain, among others. Blood and urine samples were taken at enrollment to rule out sepsis and to aid in their diagnosis. After family consent hepcidin levels (serum and urine) were measured from such samples.

Flow chart of the study.
Exclusion criteria
Term infants.
Premature babies who presented with suspected sepsis earlier than 7 days old.
Premature infants with significant anemia or a history of recent transfusion.
Clinically suspected septic premature but with negative culture.
Premature infants with multiple congenital anomalies.
Definitions
Late-onset sepsis describes cases presenting after the seventh day of life. 19
Suspected sepsis: clinical and nonspecific laboratory findings of sepsis are present, but the failure to show the causative microorganism in cultures. 20
Proven sepsis: clinical and laboratory findings are present, and demonstration of the pathogenic causative microorganism in the cultures taken from sterile fields. 20
Clinical sepsis
Signs and symptoms of sepsis are generally nonspecific ranging from just ‘not doing well’ to severe focal or systemic manifestations. Accordingly, sepsis workup has been performed on all symptomatic neonates with any of the following:
Body temperature: greater than 38.5°C or less than 36°C and/or temperature irregularities; cardiovascular instability: bradycardia or tachycardia and rhythm irregularity, urine amount less than 1 ml/kg/h, hypotension, impaired peripheral perfusion; skin and subcutaneous lesions: petechiae, sclerema; respiratory instability: apnea or tachypnea or increased oxygen demand or increased need for ventilation support; gastrointestinal: nutritional intolerance, insufficient breastfeeding, or abdominal distention; nonspecific: irritability, lethargy, and hypotonia. 5
Laboratory finding: several findings in CBC [high or low total leukocyte (<4000/mm3or >20,000 lmm3), neutropenia, high immature/ total neutrophil ratio (0.2), low platelets < 100,000/mm3], arterial blood gases (ABG) (metabolic acidosis, base deficit > 10 mEq/L), abnormal blood glucose (hyper/hypo glycemia > 180 mg/dl or <45 mg/dl) values or high acute phase reactants (high CRP > 1.5 mg/ml or procalcitonin ⩾ 2 ng/ml) may support sepsis diagnosis. However, isolation of causative pathogens from sterile body fluid (e.g. blood and urine) is the cornerstone for ‘proved sepsis’ diagnosis. 5
Methods
All presented cases were subjected to full history taken, thorough clinical examination, and laboratory sepsis workup as per unit protocol, which includes CBC, CRP, blood culture, urine culture, cerebrospinal fluid (CSF) analysis and culture, and variable radiological modalities such as X-ray and ultrasonography as needed.
All participants (cases and controls) were subjected to the evaluation of S.Hep. For cases, two samples were evaluated, one at initial presentation (acute sample) and the second on the seventh day after starting treatment (convalescent samples), while for healthy control, only a single sample at presentation was evaluated. Concurrent measurements of U.Hep levels for all participants, at the same phases, were also carried out.
Serum and urinary hepcidin: The concentrations of 25- amino acid hepcidin in serum and urine were determined using a quantitative double antibody sandwich ELISA [kits provided by CUSABIO Life Science; China, Catalog Number (CSB-E13062 h)] according to the manufacturer’s instructions. Hepcidin being a polypeptide that consists of 25 amino acids, it is very fragile and labial to proteases. Accordingly, both blood and urine samples were collected in an ethylenediamine-tetraacetic acid (EDTA) tube containing proteases inhibitor (aprotinin) to prevent coagulation and action of proteases. To avoid frequent phlebotomy and invasive maneuvers, an acute serum sample was taken at the same time of the initial blood culture, centrifuged, and stored at −80°C for analysis. Similarly, acute urine samples were taken, by a catheter or suprapubic aspirate at the same time of urine evaluation as part of late-onset sepsis workup 21 and all samples were stored at −80°C. The minimum detectable concentration of human hepcidin is less than 6.25 ng/ml. Urinary creatinine was also measured in urine samples by the Jaffe reaction using the creatinine parameter assay (R&D Systems, Minneapolis, MN) to evaluate urinary hepcidin/creatinine concentration that was expressed as ng/mg.
This research has been approved by the Institutional Review Board (IRB) of Zagazig University and informed consent was taken from the parents or legal guardians of each child before enrollment in the study (Institutional Review Board Number: 2991/18-9-2018).
Statistical analysis
Data were statistically analyzed using the SPSS program (Statistical Package for Social Science) version 22.0 (SPSS Inc., 2007). Qualitative (categorical) data were represented as frequencies and percentages. The Chi-square test (χ2) test was carried out for testing the association between the qualitative data frequencies. Quantitative (numerical) data were represented as mean and standard deviation (SD) and Student’s t-test was used to detect the difference between groups, which were normally distributed. Nonparametric variables were presented as median and interquartile ranges (IQRs) and the Mann–Whitney U test was used to detect the difference between these groups. To assess the presence of significant dynamic changes in S.Hep, U.Hep, and CRP after 7 days of treatment as compared with acute concentrations, paired t-test was performed to determine this issue. The Pearson correlation coefficients were used to estimate the association between acute S.Hep with clinical and laboratory data, namely, CRP, U.Hep, and different hematological indices. To assess the utility of S.Hep and U.Hep as biomarkers for LOS diagnosis, we compared the areas under receiver operating characteristic (ROC) curves between hepcidin values and blood culture. Several clinical and laboratory parameters were used in constructing a prediction model for LOS using logistic regression analysis and the results were reported as odds ratio (OR) with 95% CI. All variables were tested for normality using the Shapiro–Wilk test and considered normally distributed if the p-value is greater than 0.05. The test results were considered significant when the p-value was ⩽ 0.05, and all p-values are two-tailed.
Results
A total of 123 premature infants were involved in this study, as 73 cases with clinical and laboratory-proven LOS ‘case group’ (Figure 1) and fifty (50) nonseptic premature infants of comparable age and sex as ‘control group’. 72/123 (58.5%) of them were male, majority of them had gestational age (GA) ranging from 28 to 35 weeks (60/123 infants − 48%), while 32 % (39/123 infants) were late preterm (GA > 35 weeks). All participants of the case group were hospitalized at enrollment to the study (65% still at hospital since birth, 35% readmitted after developing symptoms) while the majority (35/50 – 70%) of the selected controls were discharged from hospital and are following up regularly at the neonatal outpatient clinic. Cases were screened to rule out LOS at a mean age of 18.6 ± 6.3 (range: 11–28) days versus 19 ± 5.9 (10–28) days for controls’ enrollment in the study. No statistically significant difference could be detected between participants in the case and control groups regarding age at presentation, gestational age, birth weight, or sex (Table 1). The majority of preterm infants needed respiratory support ranging from low flow nasal cannula to mechanical ventilator; cases need much more intensive support than controls. Eighteen percent of septic premature infants were on mechanical ventilation compared with only 6% of nonseptic comparable (p = 0.05). Forty (80%) nonseptic preterm are feeding enterally, while 63% of septic cases are on parenteral nutrition (p = 0.0001). None of the enrolled infants received erythropoietin; however, as per unit protocol, any infant who reached full enteral feeding is supplemented with iron as shown in Table 1.
Demographic and clinical characteristics of the studied group.

HHHNC, high humidified heated nasal cannula; CPAP, Continuous Positive Airway Pressure; MW, Mann–Whitney U test; SD, standard deviation; t, Student’s t-test; χ2, chi-square.
Leading isolated causative pathogens among our septic preterm infants were gram-negative organisms (43/73, 59%) [Klebsiella pneumonia (35/73, 47%) and Pseudomonas aeruginosa (4/73, 6%), Citrobacter freundii (4/73, 6%)] followed by several subtypes of gram-positive cocci, namely, Staphylococcus subtypes (30/73, 41%) [Staphylococcus hemolyticus (16/73, 20%), Staphylococcus hominis (10/73, 15%), and Staphylococcus warneri 4/73, 6%]. Nonsignificant higher values of S.Hep were reported among cases with gram-negative septicemia as compared with those with gram-positive isolates (p = 0.39).
In septic preterm infants there were significantly higher total leukocyte count increased immature/total neutrophil ratio of (>0.20), and significantly more patients who had of neutropenia as compared to nonseptic peers. On the contrary, they had significantly lower hemoglobin levels and platelets count as compared with nonseptic participants (Table 2).
Laboratory finding of the studied groups.

CRP, C-reactive protein; IQR, interquartile range; MW, Mann–Whitney U test; SD, standard deviation; t, Student’s t-test; t * paired, t test, between acute and convalescent sample of the same variable; χ2, chi-square.
CRP, S.Hep, and U.Hep were significantly higher among septic premature infants as compared with nonseptic participants (Figures 2 and 3).Convalescent samples taken from septic cases 1 week after starting treatment showed significantly lower CRP, S.Hep, and U.Hep as displayed in Table 2. Two extreme preterm infants had a progressive increase of both S.Hep and U.Hep. This increase was concomitantly associated with worsening of their clinical status.
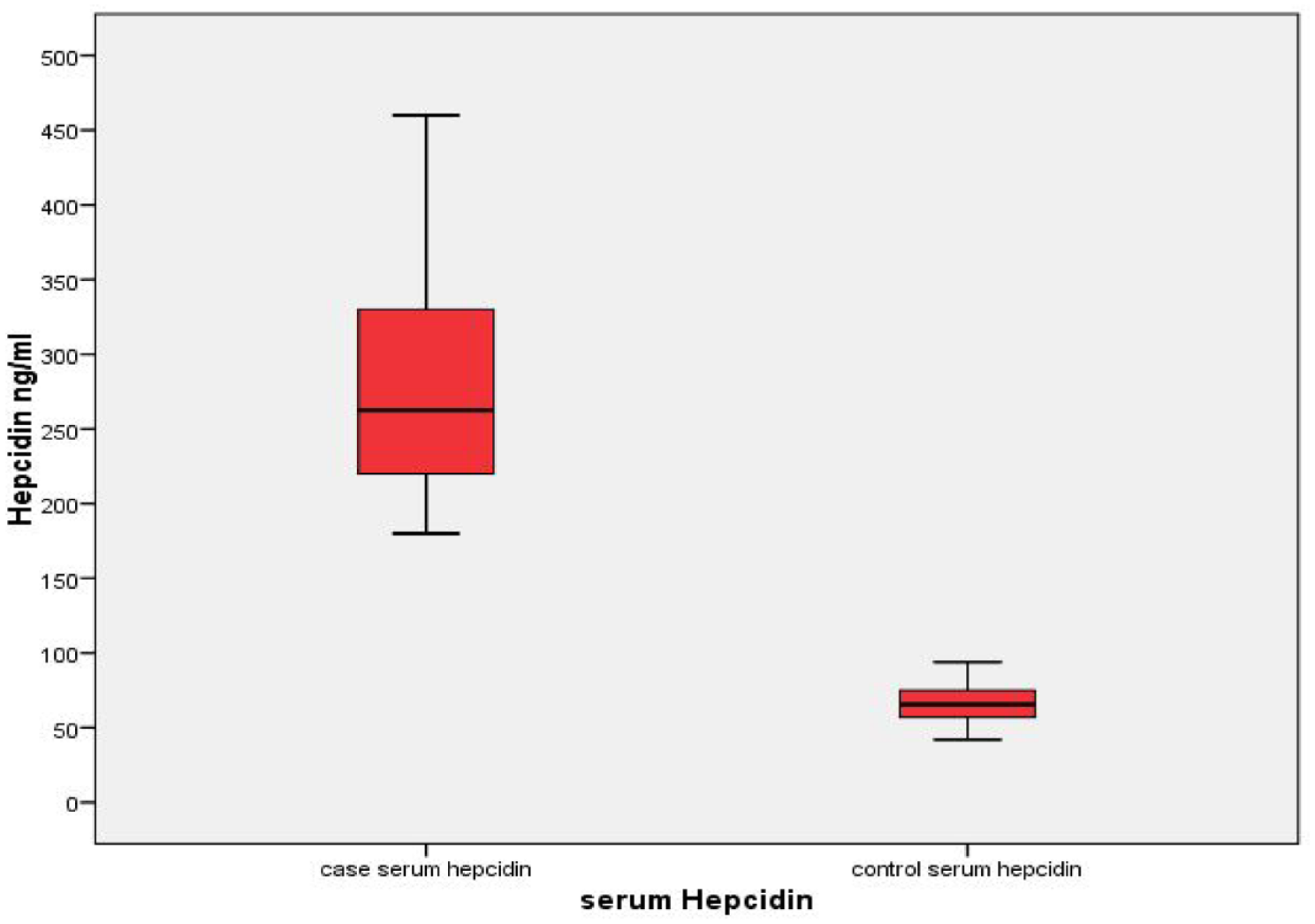
Comparison of serum hepcidin (ng/ml) between septic cases (73) and controls (50).

Comparison of urine hepcidin (ng/mg) between septic cases (73) and controls (50).
Pearson’s correlations were examined between acute S.Hep concentrations and relevant quantitative clinical (GA, body weight, hospital stay, and patient outcome) and laboratory variables (CRP, hemoglobin level, platelets count, and acute U.Hep). Significant direct correlations were recognized with CRP (r = 0.23 p = 0.03), acute U.Hep (r = 0.42 p = 0.01), and total leukocyte count (p = 0.001). While significant negative correlation was recorded with platelets count (P = 0.02). No correlation could be detected with birth weight or gestational age. In addition, correlation is maintained between S.Hep and U.Hep after 1 week of treatment, and convalescent sample (r = 0.35 p = 0.023) as displayed in scatter plots (Figures 4 and 5).

Scatter diagram showing significant direct correlation between acute serum (ng/ml) and urine hepcidin (ng/mg) among septic cases (73) (r = 0.4 and p = 0.01).
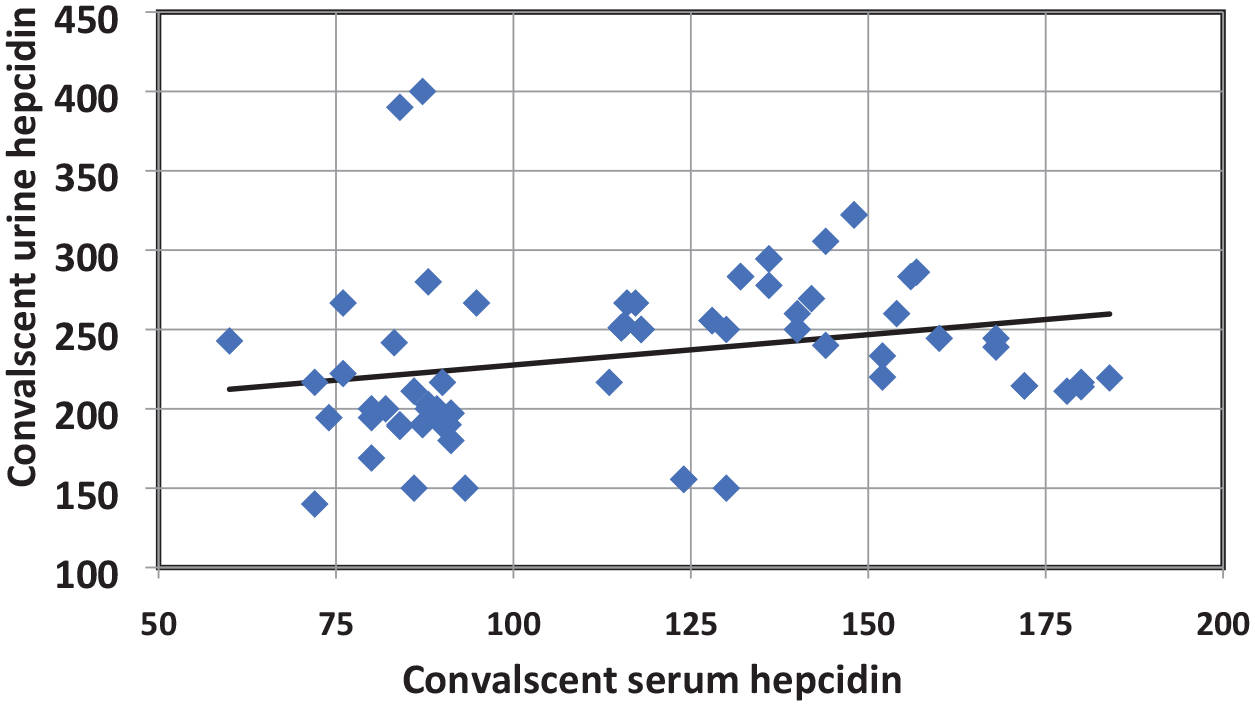
Scatter diagram showing significant direct correlation between convalescent serum (ng/ml) and urine hepcidin (ng/mg) after 1 week of treatment of septic cases (73) (r = 0.35 and p = 0.023).
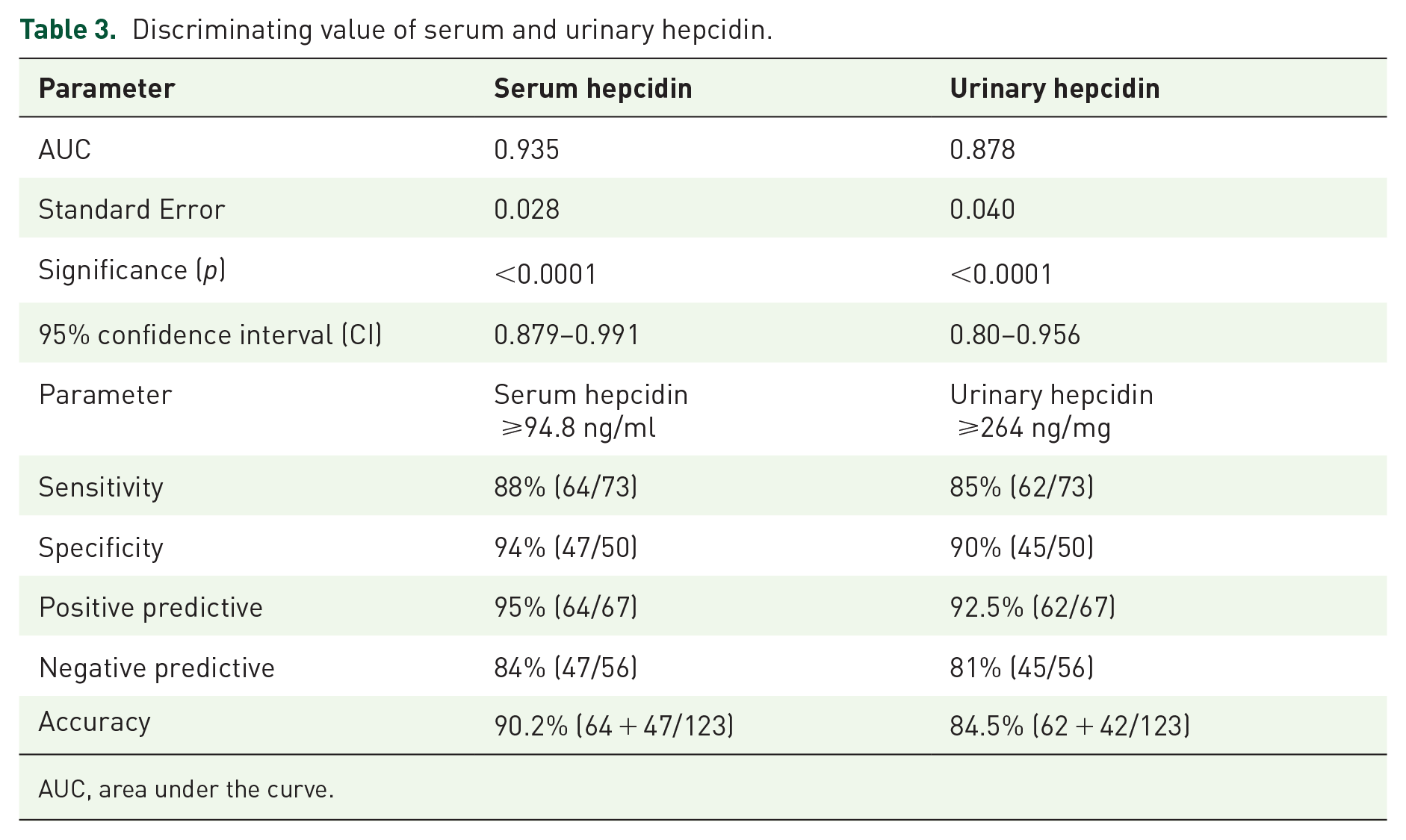
For evaluating the discriminating power of measuring S.Hep and U.Hep in correctly classifying septic from nonseptic infants, ROCs were constructed and the areas under the curves (AUCs) were determined for each as reported in Table 3, and Figures 6 and 7.
Discriminating value of serum and urinary hepcidin.
AUC, area under the curve.

Receiver operating characteristic curve (ROC) for serum hepcidin.

Receiver operating characteristic curve (ROC) for urinary hepcidin.
Highly significant discriminating ability with AUC of S.Hep ROC is 0.93. At a cut-off value of ⩾ 94.8 ng/ml of S.Hep showed sensitivity (88%), specificity (94%), positive predictive value (PPV) (95%), and negative predictive value (NPV) (84%), respectively, with accurately diagnosing 90.2% of presenting cases as septic or not. While U.Hep showed slightly less discriminating ability with an AUC of 0.87. A cut-off value of ⩾264 ng/mg of U.Hep/urinary creatinine showed sensitivity (85%), specificity (90%), PPV (92.5%), and NPV (81%), respectively, with accurately diagnosing 84.5% of presenting cases as septic or not.
The prediction model for neonatal sepsis was calculated by logistic regression analysis to find out real predictors of LOS of premature. High CRP and elevated hepcidin concentration in both serum and urine were reliable predictors as shown in Table 4.
Logistic regression analysis for predictors of neonatal sepsis.

CI, confidence interval; CRP, C reactive protein; SE, standard error.
Discussion
Sepsis represents a major contributor to neonatal mortality, particularly among premature infants 22 with mortality rates as high as 40% in very low birth weight (VLBW). 23 Adaption of universal intrapartum antibiotic prophylaxis has resulted in an 80% reduction of early-onset sepsis cases, but the incidence of LOS significantly increased in modern NICUs due to increased survival of extreme preemies with many invasive strategies to support their life.3,24 Frequent screening for LOS, with any change in their clinical status, is an essential strategy for early diagnosis and management of infection as early as possible. 25 Clinical diagnosis is not feasible due to the nonspecific nature of sepsis manifestations. 26 Although blood culture is the cornerstone for sepsis diagnosis, it is associated with several drawbacks that render it not suitable for rapid diagnosis or guiding the antibiotic-starting decision. Critically ill septic premature infants may succumb before the result of culture is available. 14 Also, the high rate of false-negative results is another important challenge for the use of blood culture solely for sepsis diagnosis. 27 In our study, only 37% (102/274) of the evaluated cohort for LOS had positive culture; similarly, Betty and Inderpreet reported a 51% culture positive rate among clinically presumed septic neonates. 28 The risk of morbidities and mortality due to delayed or untreated infection versus the short- and long-term hazards of frequent un-needed exposure to antibiotics, justify the pressing need for an accurate and rapid test for differentiating septic from a non-septic symptomatic premature infant.
In the current research, we hypothesized that, as hepcidin is a well-known first line defender that rapidly increases to militate against pathogenic invasion,14,15 its measurement can point to the septic etiology of nonspecific manifestations in neonates. Coincide with our assumption, statistically significant higher S.Hep values were recorded for culture-proven septic premature infants in our cohort as compared with their non-septic asymptomatic peers. Similarly, Wu and his team affirmed that infection is a potent inducer of hepcidin production in VLBW as it increases 4-fold in newborns with proven infection. 8 Other researchers examined the usefulness of hepcidin level measurement in cord blood and serum as a biomarker of early-onset sepsis and described it as a valid predictor.6,29 S.Hep was dramatically elevated in infants who are bacteremic or even treated for presumed sepsis but with negative culture in the previous report. 16 Infection and inflammation enhance IL-6 production, which in turn induces hepatic hepcidin production to combat the pathogenic invasion. Also, autocrine synthesis of hepcidin can more selectively activate the appropriate cell type, namely, macrophages and monocyte that can fine tune the response to infection. 30 Hepcidin fights pathogens either by depriving them of iron 31 or by direct antimicrobial activity against various pathogens. 32 In follow-up of our septic cohort, S.Hep was significantly lower after 7 days of antibiotic therapy (convalescent sample) as compared with the acute sample, denoting the parallel dynamics of hepcidin with infection and inflammation. It is a worthy note that two mechanically ventilated extreme premature infants in our cohort showed progressive worsening of their clinical condition despite several trials of antibiotics, both of them had a progressive increase in acute phase reactants and also in S.Hep and U.Hep and they died a few days after taking convalescent samples. Their cultures came positive for multidrug-resistant Klebsiella pneumonia. This finding is supporting evidence for the prognostic value of hepcidin; nevertheless, this issue needs to be thoroughly investigated. We excluded the anemic and recently transfused preterm infants to evaluate the changes of hepcidin with variable stages of infection without other interfering factors especially those related to iron hemostasis. The ROC for hepcidin showed an AUC of 0.93 which indicates the high discriminating ability of S.Hep concentration in differentiating septic from nonseptic infants. As compared with several commonly used acute phase reactants such as CRP and procalcitonin which have good NPV but low PPV to diagnose cases of sepsis correctly, 33 hepcidin showed a high diagnostic profile. A cut-off value of ⩾ 94.8 ng/ml of S.Hep showed sensitivity (88%), specificity (94%), PPV (95%), and NPV (84%), respectively, with accurately diagnosing 90.2% of presenting cases as septic or not. Close to our findings, hepcidin was reported to correctly diagnose sepsis among both term and preterm infants by 91% at a cut-off of ⩾ 92 ng/ml. 8 Acute S.Hep values showed a weak positive correlation with CRP levels (r = 0.2, p = 0.03). Even though the production of both of them is IL-6 dependent,30,34 the lack of a strong correlation between both of them may be explained by the different time courses of their production. CRP is delayed for 48 hours to peak, and its half-life time is approximately 24 hours, 35 while hepcidin peaks in a few hours (6 hours). 13
S.Hep levels at initial evaluation (acute) and after 1 week of treatment (convalescent) displayed a significant positive correlation with corresponding U.Hep values (r = 0.42, r = 0.35, respectively). This finding may answer our question ‘Could we find body fluid to diagnose and follow up sick preemies, other than blood?’ to avoid frequent phlebotomy and its hazards. As hepcidin is excreted and can be accurately quantified in urine samples by the same analytic method (ELISA) as S.Hep, and with this positive correlation, U.Hep may replace S.Hep in the initial diagnosis and follow-up of septic preemies, particularly if frequent screening is needed during their NICU stay. U.Hep is significantly higher among clinically and laboratory-proven sepsis cases as compared with nonseptic infants in our series, and its level is significantly reduced with improved cases after 7 days of treatment. All previous data support the idea that similar to S.Hep, U.Hep is dramatically increased in response to IL-6 infusion 13 and its quantification can help in rapid, accurate diagnosis of sepsis in premature infants. In harmony with our data, researchers in pediatrics and adults documented the positive S.Hep and U.Hep correlation.18,36 Simultaneous adult samples usually show a better correlation than those in premature as urine samples may be delayed until involuntary void in urine bag after taking serum sample. As hepcidin follows diurnal variation, 18 such delay may impair correlation.
The AUC of U.Hep ROC is 0.87, indicating good discriminating power of measuring U.Hep concentration between septic and nonseptic. A cut-off value of ⩾ 264 ng/mg of U.Hep/urinary creatinine showed sensitivity (85%), specificity (90%), PPV (92.5%), and NPV (81%), respectively, with accurately diagnosing 84.5% of presenting cases as septic or not. U.Hep was normalized for urinary creatinine, and expressed as ng/mg, to adjust for variable urine dilution. The diagnostic profile for U.Hep is reasonable, with less diagnostic power than those of S.Hep but still with accepted accuracy in sepsis diagnosis. Up to our knowledge, no previous study was performed to evaluate the role of U.Hep in sepsis diagnosis either in children or neonates. However, Müller et al. 36 reported a positive correlation between S.Hep and U.Hep and between both of them with iron indices. Accordingly, Researchers were hoping to use U.Hep as a diagnostic and prognostic biomarker for iron deficiency anemia, and their results were promising. 37 Another advantage of using U.Hep as a diagnostic biomarker for neonatal sepsis added to its noninvasive nature is the less impact of diurnal variation on its level 38
Although hepcidin has been discovered as early as 2000, its clinical application was delayed due to the expensive, cumbersome, and invasive nature of its measuring modalities like hepcidin mRNA expression in the liver biopsy sample. In 2008, an enzyme-linked immunosorbent assay for human hepcidin is developed that can accurately, easily, and reproducibly determine physiologic and pathologic changes in hepcidin in variable body fluids. 18
A considerable proportion of our cases were late preterm or near-term infants (39 infants, 32%) with GA 35 to 37 weeks. Usually, those infants are apparently well with safe pulmonary status, thermal control, and nutritional abilities, but indeed they significantly have issues of prematurity like poor immunity, increased risk of feeding problems, and injurious hyperbilirubinemia. 39 These criteria necessitate their close follow-up to avoid such hazards.
Leading isolated causative pathogens among our septic preterm infants are gram-negative organisms [Klebsiella pneumonia (47%) and Pseudomonas (6%), Citrobacter (6%) followed by several subtypes of Staphylococcus]. A previous study from another Egyptian children’s hospital showed the same results. 40 However, coagulase-negative staphylococcus is the most common isolate of LOS of premature in several developed countries (53–78%) and (35–47%) in series from developing countries. The predominant causative pathogen is affected by the geographic areas, and level of health care service, particularly adherence to infection control strategies 41
Higher levels of S.Hep were reported among those with gram-negative infection as compared with peers with gram-positive sepsis but do not reach a significant level. Gram-negative and gram-positive bacteria elicit different patterns of pro-inflammatory cytokines in human monocytes. It was expected to have much higher S.Hep levels among preterm infants with gram-negative sepsis as previously described in an ‘in vitro study’ where gram-negative bacteria induce at least twice as much IL-6 (triggering for hepcidin secretion) as did gram-positive bacteria. 42 Nevertheless, the prematurely functioning immune system of preterm infants and different reactions between in vivo and in vitro situations may explain our findings.
Our study has some limitations; first, a small number of involved premature infants in the case group as inclusion criteria were strict to involve only culture-proven cases who presented almost one-third of screened cases. Second, the dual function of hepcidin as a master iron regulator plus its role in innate immunity forced us to exclude all anemic or recently transfused cases to prevent bias due to disturbed iron status. The majority of extreme premature infants suffer from anemia or receive transfusions somewhere during their NICU stay and thus hepcidin may not be the ideal biomarker for their screening.
Conclusions
All the previous data conclude that hepcidin concentration in different body fluids (e.g. urine) can function as a promising accurate, and rapid surrogate test, with blood culture, that guides empiric antibiotics starting decision or withholding it safely until the culture results are ready in symptomatic presumed septic preemies. U.Hep has advantages over S.Hep as; it is noninvasive, has no hazards of phlebotomy, and is less variable throughout the day.
To widespread its use, especially U.Hep, a wide-scale multicenter study should be performed that involves anemic, transfused, and culture-negative cases to determine the diagnostic cut-off of hepcidin in different situations.
