Abstract
Objective
To compare the growth of preterm infants fed standard protein-fortified human milk with that containing human milk fortifier (HMF) with a higher-than-standard protein content.
Methods
Published articles reporting randomized controlled trials and prospective observational intervention studies listed on the PubMed®, Embase®, CINAHL and Cochrane Library databases were searched using the keywords ‘fortifier’, ‘human milk’, ‘breastfeeding’, ‘breast milk’ and ‘human milk fortifier’. The mean difference with 95% confidence intervals was used to compare the effect of HMF with a higher-than-standard protein content on infant growth characteristics.
Results
Five studies with 352 infants with birth weight ≤1750 g and a gestational age ≤34 weeks who were fed human milk were included in this meta-analysis. Infants in the experimental groups given human milk with higher-than-standard protein fortifier achieved significantly greater weight and length at the end of the study, and greater weight gain, length gain, and head circumference gain, compared with control groups fed human milk with the standard HMF.
Conclusions
HMF with a higher-than-standard protein content can improve preterm infant growth compared with standard HMF.
Introduction
Human milk provides benefits to general health, growth and development, while significantly decreasing the risk for a large number of acute and chronic diseases. 1 It also has positive effects on mental and motor development, intelligence quotient and visual acuity of infants compared with milk formula feeding. 2 Therefore, human milk is preferred for feeding neonates and premature infants. 3 However, the concentrations of protein, sodium and zinc decline in human milk as lactation continues over time, while the nutrient needs of very low birth weight infants remain high. 4 Therefore, human milk fortifier (HMF) may be used when human milk provides insufficient amounts of the nutrients and energy required at the usual feeding volumes. 5 There is evidence that the growth of infants fed human milk fortified with standard HMF was slower than that of infants fed preterm formula. 6 The authors of this study concluded that the protein content of human milk with standard fortification failed to meet the requirements of preterm infants, thereby affecting their growth. 6 Several studies have demonstrated that HMF containing an increased protein level is beneficial to infant growth.7–10 A study undertaken at eight US hospitals demonstrated that HMFs containing both standard and higher protein levels were well-tolerated; and that the blood chemistries were normal and acceptable for the infants’ ages.9 The aim of the current meta-analysis was to determine whether HMF with a higher-than-standard protein content would benefit preterm infant growth.
Materials and methods
Data sources and keywords
Eligible studies published up to 31 May 2014 were identified by a systematic search of the electronic literature databases PubMed®, Embase®, CINAHL and the Cochrane Library. The following keywords were used in the search for published randomized controlled trials and prospective observational intervention studies: ‘fortifier’, ‘human milk’, ‘breastfeeding’, ‘breast milk’ and ‘human milk fortifier’.
Selection criteria
Eligible studies were identified initially by reviewing the abstracts. If the abstract was consistent with the inclusion criteria, then the full-text article was obtained. The intervention groups were feeding infants fortified human milk that contained a higher-than-standard protein level. The current dose of extra protein provided by HMF is between 0.7 and 1.1 g/100 ml depending on the brand. 7 Inclusion criteria were as follows: (i) randomized controlled trial or prospective observational intervention study; (ii) infants with birth weight ≤1750 g, gestational age ≤34 weeks and fed with human milk; (iii) control group included infants fed with human milk fortified with standard HMF, while experimental group included infants fed with human milk fortified containing a higher-than-standard protein level; (iv) outcome measures were growth characteristics, including weight (g), length (cm) and head circumference (cm).
Data extraction
Several investigators (T-T.L., D.D., X-M.L.) independently extracted the data and reached a consensus on all the data to be included in the meta-analysis. The following data were extracted from each study: name of first author, year of publication, sample size in each group, gestational age and growth characteristics of the different study groups.
Quality assessment
The quality of the included prospective observational intervention study was assessed using predefined criteria based on the Newcastle-Ottawa Scale (NOS) criteria; and the randomized controlled trials were assessed using a Modified Jadad score.11,12 This was undertaken by two investigators (D.D., X-M.L.). The NOS criteria are based on three study parameters: (i) subject selection (0–4); (ii) comparability of subjects (0–2); (iii) clinical outcome (0–3). Total NOS scores range from 0 (lowest) to 9 (highest). Studies are classified as low quality (scores 0–6) or high quality (scores 7–9). The Modified Jadad score is based on four study parameters: (i) randomization (0–2); (ii) concealment of allocation (0–2); (iii) double blinding (0–2); (iv) patient withdrawals and dropouts (0–1). The total Jadad scores range from 0 (lowest) to 7 (highest). Studies are classified as low quality (0–3) or high quality (4–7).
Statistical analyses
The mean difference (MD) with 95% confidence intervals (CI) between the study groups was used for analysis of continuous outcomes. Summary statistics were calculated with either a fixed-effects or random-effects model approach, according to the heterogeneity in outcomes across the studies. Heterogeneity was analysed using χ2-test (significance level: P < 0.01) and quantified using the I2 test, where I2 values ≥50% indicated substantial heterogeneity. All analyses were carried out using Review Manager software, version 5.0. 13
Results
Characteristics of the included studies
The study selection procedure is presented in Figure 1. The initial literature search identified 421 articles based on the title and keywords. Review of the abstracts resulted in the exclusion of 402 articles leaving 19 for full-text review. After screening the full-text articles according to the inclusion criteria, the final meta-analysis included five studies.7–10,14 One study was of low quality (NOS score ≤6)
14
and four were of high quality (Modified Jadad score ≥4).7–10 Table 1 presents the characteristics of these five analysed studies.7–10,14 A total of 352 infants were included in the five studies, with 178 in the control groups and 174 in the experimental groups. The experimental groups were fed with human milk supplemented with HMF containing a higher-than-standard protein level; and the control groups were fed with human milk supplemented with HMF containing standard protein. The baseline growth characteristics of the infants in the five studies are shown in Table 2. Table 3 shows the achieved growth characteristics and Table 4 shows the rate of growth of the infants included in the five studies.
Flow diagram of the study selection process in a meta-analysis undertaken to determine whether human milk fortifier with an enhanced protein content would benefit preterm infant growth. Characteristics of studies included in a meta-analysis undertaken to determine whether human milk fortifier (HMF) with an enhanced protein content would benefit preterm infant growth. RCT, randomized controlled trial; BUN, blood urea nitrogen. a≤30 weeks+3 days. Baseline growth characteristics of infants included in five studies in a meta-analysis, undertaken to determine whether human milk fortifier with an enhanced protein content would benefit preterm infant growth. Data presented as mean ± SD. Achieved growth characteristics of infants included in three studies in a meta-analysis undertaken to determine whether human milk fortifier with an enhanced protein content would benefit preterm infant growth.a Data presented as mean ± SD. aOnly three of the five studies evaluated growth by measuring achieved weight, achieved length, and achieved head circumference. Growth rate of preterm infants included in four studies in a meta-analysis undertaken to determine whether human milk fortifier with an enhanced protein content would benefit preterm infant growth.a Data presented as mean ± SD. aOnly four of the five studies evaluated growth by measuring the weight gain, length gain, and head circumference gain. Miller et al.
7
used percentiles to describe the weight gain, length gain and head circumference gain, which was different methodology to the other studies so the data were excluded.


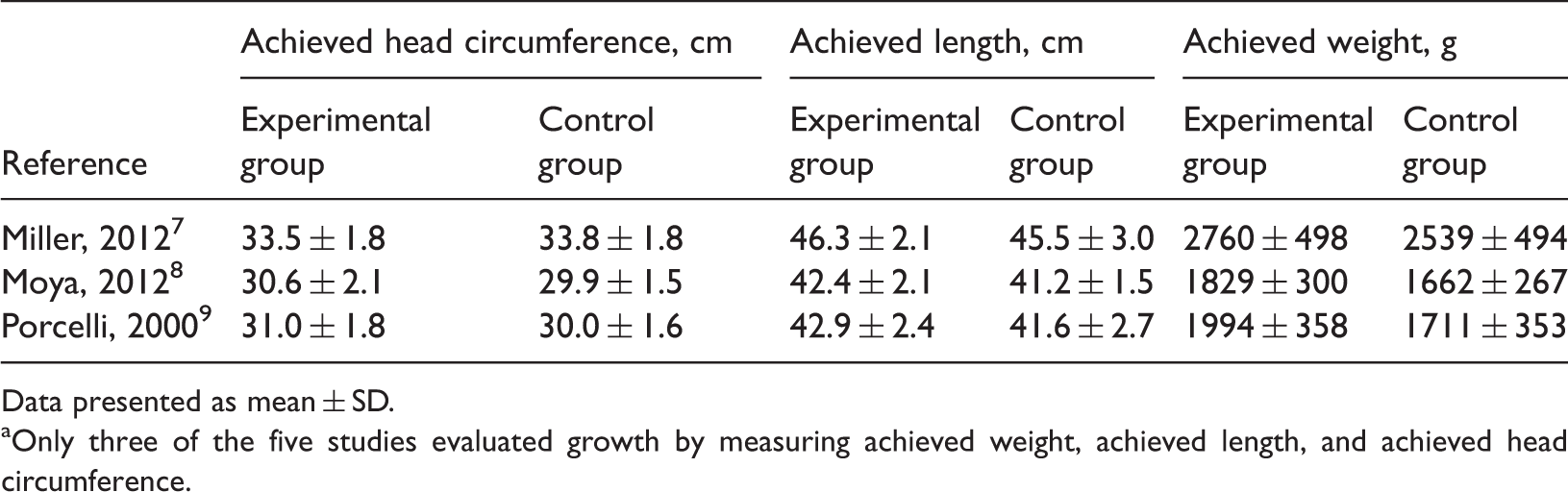

In the study by Miller et al., 7 infants in the higher-protein group achieved a greater weight at the study end (P = 0.03), but no significant differences were observed in the achieved lengths or achieved head circumferences. The length gain, weight gain and head circumference gain, which were described using percentiles, demonstrated no significant differences between these two groups.7 In the report by Moya et al.,8 the experimental group had a significantly higher achieved weight (P = 0.04), achieved length (P = 0.03), achieved head circumference (P = 0.043), and length gain (P = 0.012) compared with the control group, but there were no significant differences in weight gain and head circumference gain between the two groups. In the study by Porcelli et al., 9 infants had significantly greater achieved weight (P = 0.0153), achieved head circumference (P = 0.0043), weight gain (P = 0.0436) and head circumference gain (P = 0.0354) when given the new HMF compared with those given the reference HMF. In the report by Arslanoglu et al., 10 the experimental group had significantly greater weight gain (P < 0.01) and head circumference gain (P < 0.05) compared with the control group. In the study by Alan et al., 14 infants had significantly higher daily growth indices for weight (P = 0.026), for length (P = 0.027) and for head circumference (P = 0.003) when given the adjustable protein fortification with human milk compared with the control group.
Meta-analysis results
In three studies,7–9 the authors evaluated growth by measuring the achieved weight, achieved length, achieved head circumference, weight gain, length gain, and head circumference gain. In the study by Miller et al.,7 the authors used the percentile to describe the weight gain, length gain and head circumference gain, which was different to the other two studies.8,9 In the other two studies analysed in this meta-analysis,10,14 the authors only used weight gain, length gain, and head circumference gain to compare the growth between the two study groups. Therefore, this meta-analysis compared the achieved growth between the two study groups using data from three studies;7–9 and it compared weight gain, length gain, and head circumference gain using data from four studies.8–10,14
Weight
The comparison of the achieved weight and weight gain was carried out as follows. Since no significant heterogeneity in achieved weight (χ2-test = 1.26, I2 = 0%) and weight gain (χ2-test = 4.25, I2 = 29%) was indicated, a fixed-effects model was used to conduct the meta-analysis. As shown in Figures 2 and 3, the results indicated that the achieved weight (MD: 202.94; CI: 119.01, 286.86; P < 0.00001) and weight gain (MD: 1.77; CI: 0.81, 2.73; P = 0.0003) of infants in the experimental groups was greater than those of the control groups.


Length
For the comparison of the achieved length (χ2-test = 0.49, I2 = 0%) and length gain (χ2-test = 1.89, I2 = 0%), a fixed-effects model was used since no significant heterogeneity was detected. As shown in Figures 4 and 5, the results indicated that the achieved length (MD: 1.12; CI: 0.59, 1.64; P < 0.0001) and length gain (MD 0.21; CI: 0.12, 0.29; P < 0.00001) of infants in the experimental groups was longer than those of the control groups.


Head circumference
The comparison of the achieved head circumference (χ2-test = 6.14, I2 = 67%) and head circumference gain (χ2-test = 6.86, I2 = 56%) were carried out using a random-effects model due to the significant heterogeneity. As shown in Figures 6 and 7, the achieved head circumference in the experimental groups demonstrated no significant difference compared with control groups (MD: 0.45; CI: –0.31, 1.22); whereas the results indicated that the head circumference gain of infants in the experimental groups was larger than that of the control groups (MD: 0.19; CI: 0.07, 0.31; P = 0.002).


Discussion
With the rapid development of medical technology, the number of premature newborns with small gestational age and low birth weight continues to rise, and clinicians face challenges in providing proper nourishment for these infants. Research has confirmed that breast milk fortifiers can improve short-term growth, 15 but they result in lower rates of weight gain and increases in length in infants compared with preterm milk formula. 16 This phenomenon may be explained by the fact that the actual intake of protein by preterm infants fed fortified human milk is substantially lower than previously assumed. 17 By consulting the relevant literature, the current meta-analysis attempts to provide a comprehensive evaluation of HMF containing a higher-than-standard level of protein on preterm infant growth.
This current meta-analysis demonstrated that a higher protein intake resulted in a significantly higher achieved weight, achieved length, weight gain, length gain and head circumference gain. As for the achieved head circumference, there was no significant difference between the two groups, but the head circumference gain in the experimental groups was significantly greater than that of the control groups. Taken together, the current results suggest that HMF containing more protein than the standard level can improve preterm infant growth.
Research has demonstrated that there were no significant differences in blood biochemistry data, such as mean blood urea nitrogen, calcium levels, phosphorus levels and alkaline phosphatase, between infants fed HMF containing more protein and control infants fed HMF with prtein at a standard level.10 In addition, the higher protein-containing HMF was well-tolerated by the preterm infants.10 These findings, taken together, suggest that clinicians might find it favourable to use HMF containing an increased protein content when they need to enhance the growth of preterm infants. There are no studies about the long-term growth and neurodevelopmental outcomes associated with the use of HMFs containing higher-than-standard levels of protein, so further research is needed in this area.
In the current meta-analysis, there was significant heterogeneity between the studies when the achieved head circumference and head circumference gain were compared. There are a number of potential explanations for this heterogeneity: (i) differences in the populations at the international research sites; (ii) variations in gestational age; (iii) variations in the HMF product used and its protein content (although the protein content in the experimental groups was higher than the control groups, the actual dose of protein was different in the different HMFs used across the five studies); and (iv) differences in the software and statistical methods used to analyse the data.
This present meta-analysis had several limitations. In the five studies analysed, not only were protein levels in the experimental groups higher than those in the control groups, but the levels of other nutrients, such as calcium and zinc, were also different. Thus, the resulting growth reflected the combined effects of these nutrients, and the exact role of the absolute protein level in the HMF was difficult to interpret. Although the HMF protein level was higher in the experimental group than in the control group of each study, the distribution in each group was not balanced (i.e. differences in the intake of milk when using fortifier), which could have potentially led to bias. Although an extensive search strategy was used to find published studies, no attempt was made to identify unpublished trials. Meta-analyses are inherently limited by the quantity and quality of existing data, which are likely to exhibit heterogeneity. The statistically significant heterogeneity among the studies renders the results of this current meta-analysis less meaningful, but statistical conclusions on determinants of heterogeneity may be flawed, given the small number of included studies. Additional multicentre randomized controlled trials are needed to clarify the long-term effects of HMF containing higher-than-standard levels of protein on preterm infant growth.
In conclusion, the findings of this current meta-analysis of five studies that included 352 infants with a birth weight ≤1750 g and a gestational age ≤34 weeks who were fed human milk suggest that using HMF containing a higher-than-standard protein content can improve preterm infant growth.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
