Abstract
Although concurrent chemoradiotherapy (CRT) is recommended as standard of care in patients with locally advanced, unresectable, stage III non-small cell lung cancer (NSCLC), many patients who refuse or are not eligible for chemotherapy received radiotherapy (RT) alone with 5-year overall survival (OS) rate of about 5–6%. Immune-checkpoint inhibitors have demonstrated objective antitumor responses in patients with advanced NSCLC, but it is unclear how these agents can be used in the curative therapy with concurrent radiation. We report three cases of stage III unresectable NSCLC patients who refused chemotherapy received radiation and pembrolizumab immunotherapy. All patients had no local-regional recurrence with acceptable tolerance.
Introduction
Lung cancer is the leading cause of cancer-related incidents and mortality worldwide. Approximately 30% of non-small-cell lung cancer (NSCLC) patients are diagnosed as stage III (locally advanced), unresectable NSCLC. 1 Concurrent platinum-based chemoradiotherapy is recommended as standard of care in patients with stage III unresectable NSCLC.2,3 However, many patients refuse or are not eligible for chemotherapy in clinical practice. Some studies reported that about 50% patients with stage III NSCLC were ineligible for concurrent chemoradiotherapy (CRT).4,5 The patients who refuse or are not eligible for chemotherapy often receive radiotherapy (RT) alone and the 5-year overall survival (OS) rate is about 5–6%. 6 This addressed a common clinical challenge of treating these patients. New therapeutic approaches are needed to improve survival for these patients.
Immunotherapy has changed treatment paradigms in recent years. Immunotherapy targeting the PD-1/PD-L1 axis has developed new potential strategies for treatment of patients with advanced NSCLC. 7 Although around 20% patients with advanced NSCLC achieve durable responses to checkpoint blockade, most patients do not experience such benefits.8,9 The growing evidence support the combination of immunotherapy with chemotherapy and RT. 10 Preclinical evidences have demonstrated that combining radiotherapy with anti-PD-1 treatment could enhance the anti-tumor activity and produce long-term survival. 11 The PACIFIC trial showed that anti-PD-L1 (durvalumab) as consolidation therapy significantly prolonged progression-free survival (PFS) and OS in patients with stage III, unresectable NSCLC. 12
Immune checkpoint inhibitors have improved outcomes mainly in patients with advanced NSCLC, but it is unclear how these agents can be used in the curative setting concurrent with radiation. Herein, we present three patients with stage III unresectable NSCLC who refused chemotherapy received radiation concurrently with anti-PD-1 agent (pembrolizumab).
Case presentation
A total of three patients had pathologically documented stage III, locally advanced NSCLC according to the 8th edition of American Joint Committee on Cancer (AJCC) staging system. PET/CT was used to exclude distant metastases. All patients refused chemotherapy and received radiation concurrently with pembrolizumab between May 2018 and August 2018 in Hangzhou cancer hospital. The pembrolizumab was administered at a fixed dose of 200 mg starting on day 1 and given every 21 days for up to 5 cycles according to the patient’s wishes and their economic condition. Intensity-modulated radiation therapy (IMRT) started on the first day of pembrolizumab. Conventionally fractionated radiation therapy delivered a total dose of 60 Gy given in 30 fractions over 6 weeks. PD-L1 expression was immunohistochemically assessed with the use of the Dako 22C3 in the archived tumor tissue specimens obtained before treatment. Follow-up scans with CT, or PET/CT, were obtained every 12 weeks (± 4 weeks) to evaluate response to therapy. The tumor response was assessed according to the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. 13 Adverse events and abnormal laboratory findings were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) version 4.0. 14 Informed consent was obtained for each patient for the preparation and publication of this study.
Case 1
A 61-year-old male former smoker initially presented in May 2018 with 1 month of cough and neck pain. Chest computed tomography (CT) revealed a 3.5 × 3.0 cm left lung mass (Figure 1(a)) as well as mediastinal, and supraclavicular lymphadenopathy. There was no positive finding on the brain magnetic resonance imaging (MRI). Biopsies of the supraclavicular masses showed lung squamous cell carcinoma (Figure 1(b)). No mutations were detected for EGFR or ALK. The PD-L1 expression (tumor proportion score, TPS) was negative (Figure 1(c)). His disease was characterized as stage IIIB (T2N3M0). The patient was unsuitable for surgery after a careful multidisciplinary discussion. The patient had an Eastern Cooperative Oncology Group (ECOG) score of 1 and adequate organ and pulmonary function. The patient initially refused chemotherapy. Thus, the combination of local radiation and pembrolizumab was recommended. The treatment was well tolerated. The patient experienced grade 2 radiation pneumonitis and grade 1 esophagitis. After administration of oral steroids, the patient’s symptoms improved significantly and no treatment interruption happened. One month after the treatment, the patient was deemed to show a partial response (PR) on July 2018. There was a 70% local tumor reduction based on contrast-enhanced CT analysis (Figure 1(a)). Over the course of 32 months, the patient developed long-term disease stabilization and is alive at his last visit without local recurrence or metastasis (March 2021).
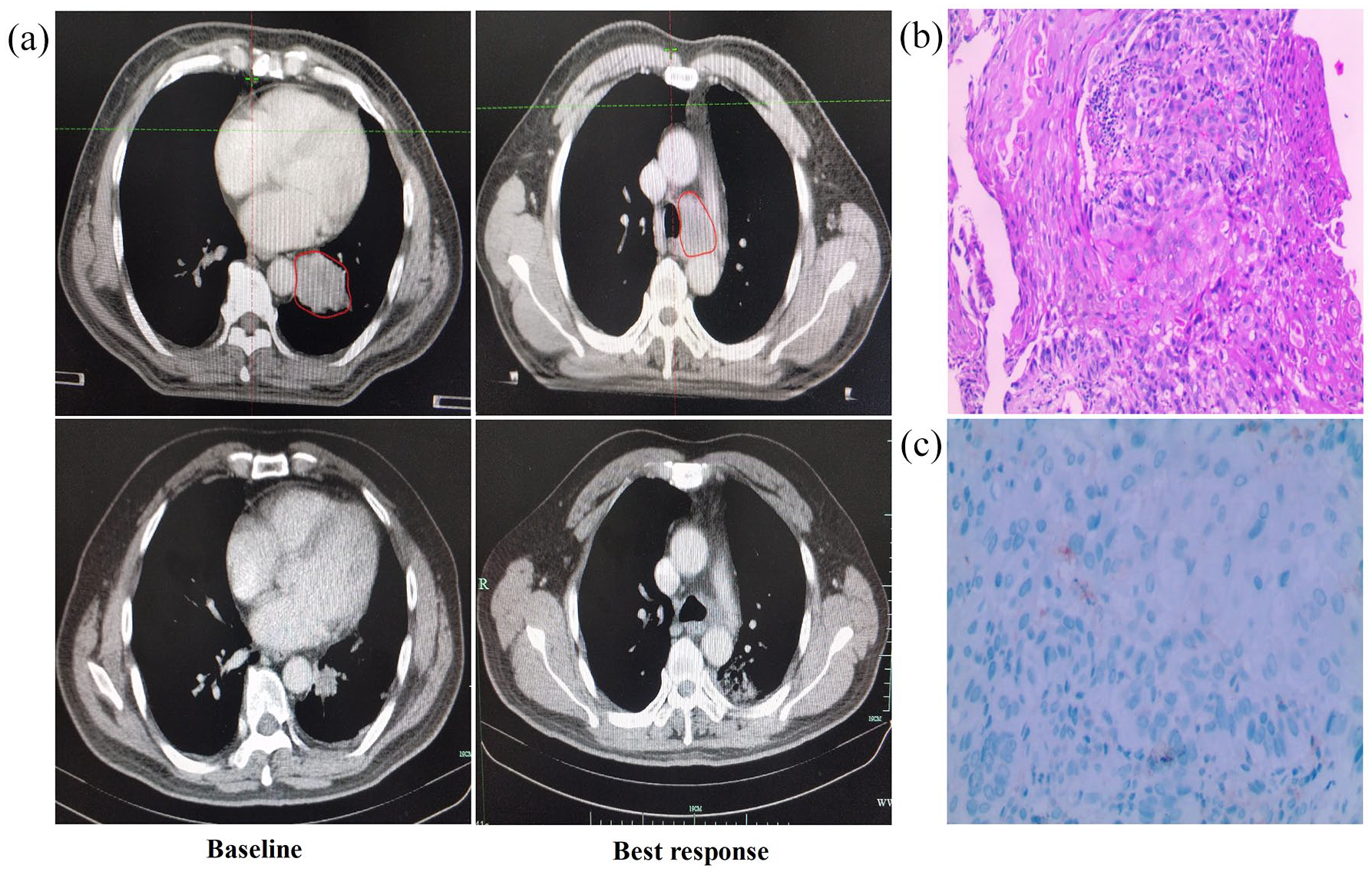
(Patient 1) (a) Contrast-enhanced chest CT prior and best response to treatment. (b) Hematoxylin and eosin-stained section of lymph node biopsy specimen. (c) PD-L1 protein expression assessed by immunohistochemistry.
Case 2
A 59-year-old male never-smoking initially presented in June 2018 with several months of persistent cough. CT revealed a 4.5 × 3.3 cm right lung mass (Figure 2(a)) as well as mediastinal lymphadenopathy. There was no positive finding on the brain MRI. Biopsies of the lung showed lung squamous cell carcinoma (Figure 2(b)). No mutations were detected for EGFR or ALK. The PD-L1 expression (TPS) was negative (Figure 2(c)). His disease was characterized as stage IIIA (T2N2M0). The patient was unsuitable for surgery after a careful multidisciplinary discussion. The patient had an ECOG score of 1 and adequate organ and pulmonary function. The patient initially refused chemotherapy. Thus, he received the combination of local radiation with pembrolizumab. One month after the combination therapy, the patient was deemed to show a stable disease (SD) on August 2018 (Figure 2(a)). He experienced grade 2 radiation pneumonitis. After administration of oral steroids, the patient’s symptoms improved significantly and no treatment interruption happened. This SD was maintained in less than 4 months, and the patient developed a new liver metastasis. Treatment consisted of radiofrequency ablation of the liver metastasis, followed by 6 cycles of nab-paclitaxel (400 mg/m2, each 3-week cycle). After completing treatment in March 2019, the patient developed 24 months disease stabilization and is alive at his last visit without local recurrence or any other metastasis (March 2021).

(Patient 2) (a) Contrast-enhanced chest CT prior and best response to treatment. (b) Hematoxylin and eosin-stained section of lung lesion biopsy specimen. (c) PD-L1 protein expression assessed by immunohistochemistry.
Case 3
A 59-year-old male former smoker initially presented in August 2018 with 1 month of progressive dyspnea, and persistent cough. CT revealed a 5.5 × 3.6 cm right hilar mediastinum mass (Figure 3(a)). There was no positive finding on the brain MRI. Bronchoscopy biopsy of the lung showed lung squamous cell carcinoma (Figure 3(b)). No mutations were detected for EGFR or ALK. The PD-L1 expression (TPS) was 30% (Figure 3(c)). His disease was characterized as stage IIIB (T3N2M0). The patient was unsuitable for surgery after a careful multidisciplinary discussion. The patient had an ECOG score of 1 and adequate organ and pulmonary function. The patient initially refused chemotherapy. The patient received the combination of local radiation with pembrolizumab. One month after the combination therapy, the patient was deemed to show a PR on October 2018. There was a 80% local tumor reduction based on contrast-enhanced CT analysis (Figure 3(a)). The treatment were tolerated without serious adverse effects. The patient experienced grade 1 fatigue. The patient continued to be in stabilization for 29 months up to the last follow-up without local recurrence or metastasis (March 2021).
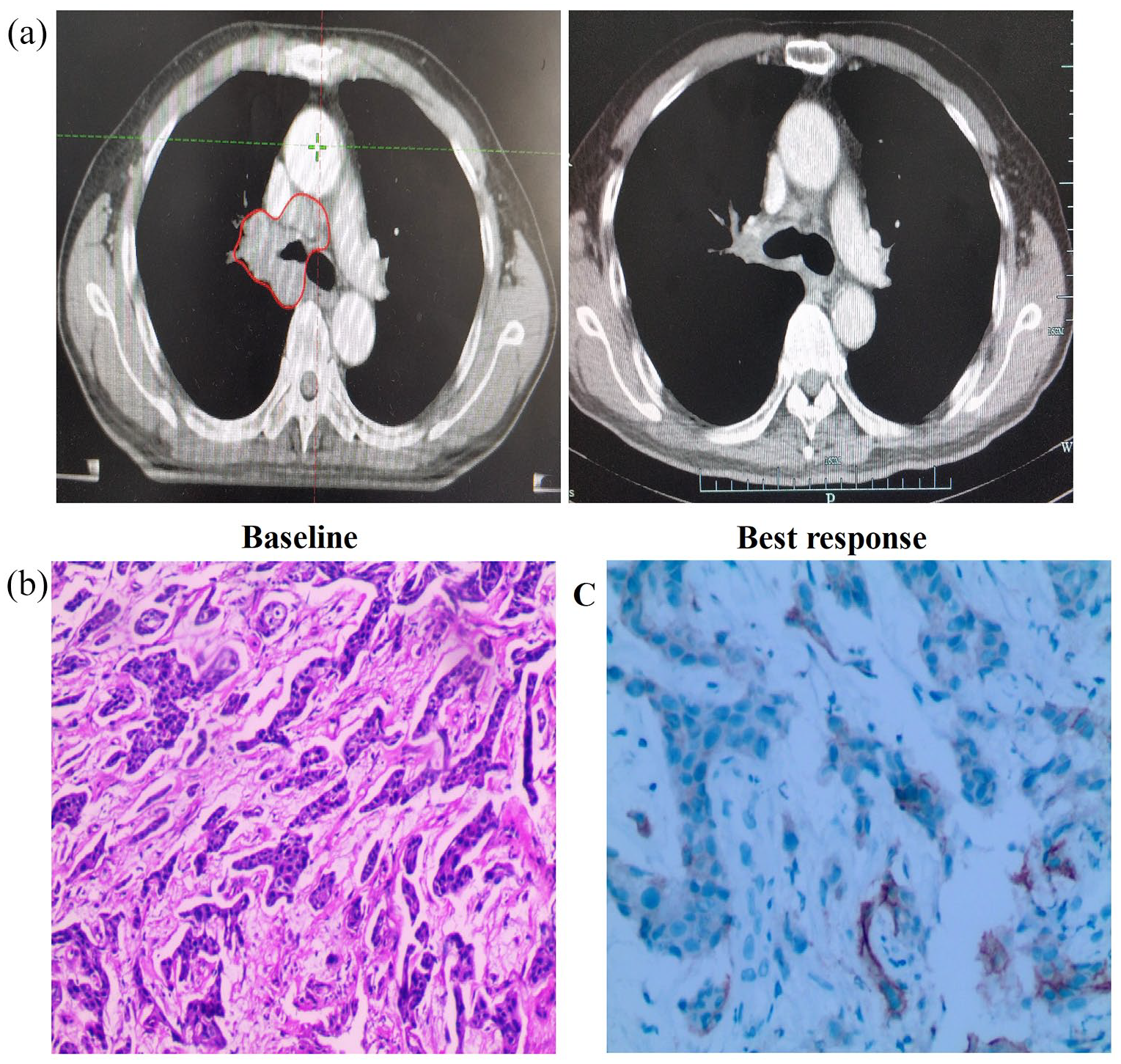
(Patient 3) (a) Contrast-enhanced chest CT prior and best response to treatment. (b) Hematoxylin and eosin-stained section of lung lesion biopsy specimen. (c) PD-L1 protein expression assessed by immunohistochemistry.
Discussion
Many patients with stage III NSCLC are ineligible for concurrent CRT, medically unsuitable or refuse to chemotherapy. The prognosis of these patients received RT alone was poor. Immune checkpoint blockade is an exciting treatment option for advanced NSCLC. Nivolumab improved OS compared with chemotherapy in metastatic squamous and non-squamous NSCLC as second-line treatment (CheckMate017, CheckMate057). 15 Pembrolizumab and atezolizumab also demonstrated prolonged OS compared with chemotherapy as second-line therapy (Keynote-010, OAK trial).16,17 As first-line treatments, pembrolizumab monotherapy significantly improved OS among patients with PD-L1 ⩾ 50% (Keynote-024). 9 Combined with platinum-based doublet chemotherapy, pembrolizumab significantly prolonged OS in non-squamous NSCLC (Keynote-189) and squamous NSCLC (Keynote-407).18,19 In the consolidation setting, the update data from the PACIFIC trial in 2021 American Society of Clinical Oncology (ASCO) showed 5-year OS and PFS rate of the patients who received durvalumab were 42.9% and 33.1%, respectively. The results demonstrated that the durvalumab as consolidation therapy after CRT has durable OS and PFS benefits.
It has been demonstrated that immune checkpoint inhibitors and radiation had a synergistic effect.20,21 The combination has shown encouraging safety and efficacy results in metastatic solid tumors. Immunotherapy plus radiotherapy could improve the prognosis of NSCLC. In the phase I Keynote-001 trial, any prior radiation followed by pembrolizumab significantly improved OS in patients with advanced NSCLC compared with those receiving pembrolizumab alone. 22 In a randomized phase II study, advanced NSCLC patients treated with stereotactic body radiotherapy and sequential pembrolizumab showed favorable survival compared with pembrolizumab alone. 23 The median OS was 38.8 months and 2-year OS rate was 63.7% for unresectable stage III NSCLC patients received nivolumab and standard CRT in the NICOLAS single-arm phase II trial. 24
Our case series demonstrated the effect of curative radiation and anti-PD-1 immunotherapy in patients with stage III unresectable NSCLC. Limited by the short follow-up time and small sample size, we still found the potential effect of this treatment. All patients had no local-regional recurrence. The case series only received 5 cycles of pembrolizumab treatment, but the responses maybe long-lasting. The obvious regression of the tumor in immunotherapy could be translated into the long-term survival of the patient. Meanwhile, the pembrolizumab can be safely added to radiotherapy without an increase in the toxicity profiles. No patient experienced ⩾ grade 3 adverse event (AE), and no patient discontinued treatment because of an AE. During follow-up, one patient developed liver metastasis. The data about distant control adding immunotherapy to radiotherapy alone in a curative setting are currently lacking. We can get some hints from the existing research. The durvalumab arm had a lower frequency of brain metastasis than the placebo arm (6.3% vs 11.8%) in the PACIFIC trial. Durvalumab as consolidation therapy after CRT could retardate the local recurrence and distant metastasis. In a mouse model of breast cancer, the combination of local radiotherapy with cytotoxic lymphocyte antigen 4 (CTLA-4) blockade significantly prolonged survival time and inhibited of lung metastases formation. 25 Integration of immune checkpoint inhibitors with radiotherapy may have the potential to improve local and systemic immune profiles reducing the risk of distant metastases.
A preclinical study demonstrated that hypofractionated radiation could induce more anti-tumor immunity compared to daily fractionated low-dose radiation. 26 Compared with conventional daily low-dose fractionated radiotherapy, hypofractionated radiotherapy significantly inhibited tumor growth through inhibiting vascular endothelial growth factor (VEGF) expression and reducing the recruitment of myeloid-derived suppressor cells (MDSCs) into tumors. 27 However, Klug et al 28 reported conventional daily low-dose per-fraction radiation could normalize aberrant vasculature and recruit more cytotoxic T cells into the tumor microenvironment than hypofractionated radiation in xenotransplant mouse tumor models. Low-dose radiation could improve anti-tumor immune response by promoting the polarization of tumor-associated macrophages (TAMs) from M2 to nitric oxide synthase (NOS)+ M1. 29 A retrospective study showed that higher doses of radiation to the immune system were associated with tumor progression and death after the definitive treatment of stage III NSCLC. 30 In addition, a systematic review of melanoma patients treated with radiotherapy and ipilimumab demonstrated that multiple low fraction radiation regimens were associated with a more favorable response. 31 Current data from our experience showed conventionally fractionated radiotherapy also has good synergistic effect with concurrent anti-PD-1 antibody. The optimal dose fractioned schedule in the immunoradiation remains controversial, and the researches in this area should take into account biological features of tumor cells and the immune state of each individual patient.
Immunohistochemistry assay for PD-L1 expression remains the current standard for identifying patients with NSCLC who are more likely to benefit from immunotherapy. 32 Many prospective trials have demonstrated a correlation between the level of tissue PD-L1 expression and clinical efficacy.18,19 However, not all patients with high PD-L1 expression respond to therapy, and some patients with low or no expression of PD-L1 also achieve benefit. 33 In this case series, the Case #1 patient with no expression of PD-L1 still demonstrate a durable clinical benefit from the treatment of pembrolizumab plus radiation. Currently, PD-L1 still remains a controversial biomarker with some limitations: differences in testing platforms, the use of different cutoff points for different immunotherapy agents, and the heterogenous expression within tumors. Compared with monotherapy, it is more difficult to identify which patients may derive the benefit from combined immunotherapy. 34 It has been reported that four different types of tumor microenvironment exist based on tumor-infiltrating lymphocytes (TIL) and PD-L1 expression. 35 Type I (PD-L1+TIL+) and Type IV (PD-L1-TIL+) considered as ‘hot tumors’ are sensitive to anti-PD-1 or anti-PD-L1 monotherapy or combination therapy. For type II (PD-L1-TIL-) and type III cancers (PD-L1+TIL-) without TIL in the tumor, anti-PD-1 or anti-PD-L1 therapies would be ineffective. Radiotherapy has the potential to induce immunogenic cell death and liberate neoantigens resulting T-cell responses in combination with anti-PD-1 or anti-PD-L1 in this type of tumor. The combination evaluation TIL and PD-L1 expression would be a theoretically reliable biomarker.
There are several ongoing clinical trials determining safety, feasibility, and efficacy of immunoradiotherapy in a curative setting. In the trial TRADE-hypo, elderly and frail patients with unresectable stage III NSCLC unfit for chemotherapy undergo combined durvalumab with either conventionally fractionated (CON-group) or hypofractionated (HYPO-group) thoracic radiotherapy. 36 In the SPIRAL-RT trial, durvalumab as consolidation therapy following radiation monotherapy in patients with stage III NSCLC ineligible for chemoradiotherapy. 37 Furthermore, the DART trial will enroll patients with locally advanced stage II-III NSCLC unfit for concurrent CRT and administer standard RT with concurrent and consolidation durvalumab. The role of concurrent or sequential immunotherapy with radiotherapy in patients unfit for systemic chemotherapy needs further research.
Conclusion
We provided the case series of combination of curative radiation with pembrolizumab for unresectable stage III NSCLC patients who refuse chemotherapy. Although there might be different immune backgrounds and biomarkers of immune response in the three patients, we successfully treated them with radiation and the PD-1 inhibitor pembrolizumab. All patients achieved long-term local control with acceptable tolerance. The advent of concurrent immunoradiotherapy may bring a valid alternative treatment of stage III NSCLC patients who refuse or are ineligible for chemotherapy.
Footnotes
Author contributions
ZJ wrote the manuscript; RJZ contributed immunohistochemical analyses and the pathohistological pictures. ZJ and NZ revised the manuscript. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This retrospective study was approved by the Medical Ethics Committee of Zhejiang hospital (2021-CA-10).
Consent for publication
The patients provided written informed consent for the publication of their information.
