Abstract
Objective
To evaluate the efficacy and safety of radiotherapy plus immune checkpoint inhibitors versus radiotherapy plus chemotherapy in driver gene-negative patients with non–small cell lung cancer and brain metastases.
Methods
This single-center retrospective cohort study (Strengthening the Reporting of Observational Studies in Epidemiology–compliant) enrolled 60 consecutive driver gene-negative patients with non–small cell lung cancer and brain metastases (29 radiotherapy plus immune checkpoint inhibitors, 31 radiotherapy plus chemotherapy) treated between June 2018 and December 2023, with follow-up until July 2025. Survival, tumor response, and immune-related adverse events were analyzed using Kaplan–Meier methods, Cox models, and chi-square tests. The study was approved by the Institutional Review Board and used deidentified data.
Results
Radiotherapy plus immune checkpoint inhibitors significantly prolonged median overall survival (586 vs. 509 days, p = 0.0208) and progression-free survival (494 vs. 383 days, p = 0.0127) as well as improved objective response rate (34.48% vs. 19.35%, p = 0.0394) and disease control rate (75.86% vs. 51.61%, p = 0.0265) compared with radiotherapy plus chemotherapy. Favorable prognostic factors included age <60 years, Eastern Cooperative Oncology Group Performance Status <2, programmed death-ligand 1 tumor proportion score ≥50%, and absence of extracranial metastasis. Radiotherapy plus immune checkpoint inhibitors–related immune-related adverse events (24.14%) were mostly grades 1–2, with no grade ≥4 events.
Conclusions
Radiotherapy plus immune checkpoint inhibitors may confer survival benefits and favorable safety in driver gene-negative patients with non–small cell lung cancer and brain metastases. However, caution is warranted in interpreting these findings, which require validation in large-scale prospective studies.
Keywords
Introduction
Lung cancer remains the leading cause of cancer-related mortality worldwide, with persistently high incidence and mortality rates, ranking first among all malignancies. According to the latest global cancer statistics, approximately 2.2 million new lung cancer cases and 1.8 million deaths were reported in 2020, with a particularly heavy disease burden in Asia. Non–small cell lung cancer (NSCLC) accounts for more than 85% of all lung cancer cases and is characterized by high invasiveness and heterogeneity.1,2 At the time of diagnosis, approximately 50% of patients present with distant metastases. Brain metastases (BM) is one of the most common forms of distant metastasis in NSCLC, occurring in 30%–50% of cases. 3 These typically manifest as headaches (40%–50%), neurological deficits (30%–40%), and cognitive decline (20%–30%). Such complications significantly reduce both survival and quality of life. The prognosis for these patients is exceptionally poor, with a median overall survival (mOS) of only 1–2 months without treatment. Even with aggressive therapy, mOS extends to just 6–15 months.4,5
Advancements in molecular subtyping have facilitated breakthrough treatments for driver gene-positive NSCLC. For patients with EGFR mutations, occurring in approximately 40%–50% of Asian populations, and ALK rearrangements, found in approximately 3%–7%, tyrosine kinase inhibitors (TKIs) such as osimertinib 6 and alectinib 7 have significantly improved survival outcomes, even among patients with BM. In contrast, treatment options for patients with driver gene-negative NSCLC, particularly those with BM, remain limited and present a major clinical challenge. For these patients, conventional chemotherapy (CT) combined with radiotherapy (RT) results in an mOS of only 8.1–10.7 months. 8
Recent advancements in cancer immunotherapy have broadened therapeutic options. Immune checkpoint inhibitors (ICIs) targeting the programmed death (PD-1)/programmed death-ligand 1 (PD-L1) and CTLA-4 pathways can enhance survival by modulating the immunosuppressive tumor microenvironment and boosting antitumor immunity.9–12 Although PD-1/PD-L1 inhibitors show potential in treating patients with driver gene-negative NSCLC, 13 their effectiveness against BM remains controversial. Preclinical and clinical studies suggest that ICIs penetrate the blood–brain barrier and activate intracranial immune responses.14,15 However, some studies suggest that the immunosuppressive microenvironment of BM may limit their efficacy.16,17 The potential synergy between RT and immunotherapy, including abscopal effects, warrants further investigation.18–20 Notably, recent studies have indicated that the combination of ICIs and RT may improve intracranial tumor control by reversing local immunosuppression,21,22 although evidence specific to patients with driver gene-negative NSCLC and BM remains limited.
Despite recent advancements, comprehensive studies particularly targeting patients with driver gene-negative NSCLC and BM remain limited. Prior studies have largely focused on driver gene-positive cohorts or have not included molecular stratification. 4 This study aimed to conduct a thorough retrospective analysis of clinical characteristics and survival outcomes in this unique population, with particular focus on identifying prognostic factors and assessing the efficacy and safety of RT combined with immune checkpoint inhibitors (RT+ ICIs) versus RT combined with CT (RT + CT). Our findings may provide valuable evidence to guide clinical management and improve therapeutic strategies for this challenging patient population.
Materials and methods
Study patients
This retrospective study enrolled consecutive patients with driver gene-negative NSCLC and BM treated at our institution from June 2018 to December 2023. The study adhered to the principles of Declaration of Helsinki (1975), as revised in 2024, and was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 23 The following detailed clinical data were systematically collected from electronic health records: demographic variables (sex and age stratified as <60 or ≥60 years), performance status (PS) indicators (Karnofsky Performance Status (KPS) categorized as <90 or ≥90 and Eastern Cooperative Oncology Group Performance Status (ECOG PS) as <2 or ≥2), smoking history (yes, no, or unknown), tumor staging (T stage 1–4 and N stage 0–3 according to the 8th edition of the American Joint Committee on Cancer (AJCC) staging system), and BM characteristics (number of lesions, defining oligometastasis as 1–3 lesions and multiple metastases as ≥4 lesions; presence of large intracranial lesions ≥3 cm; and extracranial metastasis status). PD-L1 tumor proportion score (TPS) was assessed by immunohistochemistry using the Dako 22C3 antibody (stratified as <50%, ≥50%, or unknown). Treatment-related variables included RT modality, CT regimen, type and cycles of ICIs, and immune-related adverse events (irAEs) in patients treated with ICIs. Inclusion criteria were as follows: (a) histologically confirmed NSCLC, particularly adenocarcinoma or squamous cell carcinoma; (b) BM diagnosed via contrast-enhanced magnetic resonance imaging (MRI), the gold standard, with at least one measurable intracranial lesion according to the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1; (c) absence of driver gene alterations (EGFR/ALK/ROS1) identified through next-generation sequencing (NGS) or amplification-refractory mutation system polymerase chain reaction (ARMS-PCR); (d) receipt of RT + ICIs or RT + CT as first-line systemic therapy following BM diagnosis; and (e) availability of complete clinical and follow-up data. Exclusion criteria were as follows: (a) small cell lung cancer (SCLC) or unclassified lung cancer; (b) presence of driver gene alterations; (c) concurrent second primary malignancy; (d) severe comorbidities, including cardiac, hepatic, or renal dysfunction; (e) prior exposure to immunotherapy, brain RT, or CT; and (f) loss to follow-up within 3 months after treatment initiation. Clinical follow-up was conducted every 3 months during the first 2 years and every 6 months subsequently, with the last follow-up recorded in July 2025. For patients lost to follow-up, the most recent available survival status was used for censoring.
Patients were categorized into two groups based on the first-line systemic therapy regimen administered after the diagnosis of BM: the RT + ICIs group and the RT + CT group. Particularly, patients who received RT in combination with ICIs as first-line treatment were assigned to the RT + ICIs group, whereas those who received RT combined with CT were assigned to the RT + CT group. A total of 60 patients were included in the study, comprising 29 patients in the RT + ICIs group and 31 patients in the RT + CT group. Baseline clinical characteristics of both groups were collected and analyzed to ensure comparability.
Treatment plans
RT was tailored based on the number of BM lesions and the patient’s PS. For patients with multiple BM (≥4 lesions) or a PS of ≥2, whole-brain radiotherapy (WBRT) was administered at a dose of 36 Gy in 18 fractions, delivered at a rate of 5 fractions per week. In addition, the local tumor received a boost to 50–60 Gy in 18 fractions when appropriate. In contrast, patients with oligometastatic BM (1–3 lesions, diameter <3 cm) underwent stereotactic radiation therapy (SRT) at a dose of 6 Gy in 5 fractions. Neurosurgical resection was not performed because of the presence of multiple lesions or inadequate surgical candidacy. Dose modifications or temporary delays were implemented for patients with poor organ function or severe radiation-induced adverse events.
CT predominantly included platinum-based doublets, particularly cisplatin combined with pemetrexed for patients with adenocarcinoma and carboplatin combined with gemcitabine for patients with squamous cell carcinoma, with a planned regimen of 4–6 cycles every 3 weeks. Not all patients completed the full treatment course. Dose reductions, cycle prolongations, or early termination were applied based on individual tolerance, particularly in cases of grade ≥3 toxicity or poor PS. Patients received one of the following ICIs: camrelizumab, tislelizumab, sintilimab, pembrolizumab, durvalumab, or envafolimab, with administration tailored to individual needs. Common terminology criteria for adverse events (CTCAE) version 4.03 was used to assess irAEs. ICI treatment continued until disease progression, intolerable toxicity, or patient withdrawal. Dose adjustments or discontinuation were implemented based on the grade of irAEs.
Efficacy evaluation
Tumor response was assessed every 3 months using MRI and computed tomography (CT) scans according to RECIST version 1.1. Treatment responses were classified as complete response (CR), partial response (PR), stable disease (SD), or progressive disease (PD). The objective response rate (ORR) was defined as the percentage of patients achieving CR or PR, whereas the disease control rate (DCR) was defined as the percentage of patients achieving CR, PR, or SD.
Study endpoints
The primary endpoints of the study were intracranial progression-free survival (PFS) and overall survival (OS). PFS was defined as the duration from the diagnosis of BM and initiation of treatment up to intracranial progression or death from any cause, with the last follow-up data used for censored cases. OS was defined as the time from BM diagnosis and treatment initiation up to death from any cause, also using the last follow-up data for censored cases. Secondary endpoints included intracranial ORR, DCR, and treatment-related adverse events.
Data and statistical analysis
Survival analysis was conducted using the Kaplan–Meier method, with between-group comparisons evaluated using log-rank tests. Cox proportional hazards regression models were used to identify prognostic factors, incorporating variables with p <0.05 in univariate analysis into the multivariate models. All statistical analyses were performed using Statistical Package for Social Sciences (SPSS) version 23.0 and GraphPad Prism version 8.0, with a two-sided significance level of p <0.05. This analytical framework ensured a robust evaluation of treatment outcomes and preserved scientific validity for this patient population.
Results
Patient baseline characteristics
Between June 2018 and December 2023, a total of 158 patients with lung cancer and BM were initially screened, including 75 cases of SCLC and 83 cases of NSCLC. After excluding 23 patients with driver gene mutations (EGFR/ALK/ROS1), 60 patients with driver gene-negative NSCLC and BM were included in the final analysis. These patients were categorized into two groups based on their first-line treatment regimens: 29 patients in the RT + ICIs group and 31 patients in the RT + CT group. In the overall cohort (n = 60), sex distribution comprised 33 males (55.0%) and 27 females (45.0%), with 31 patients (51.7%) being of a specific demographic. The median age was 62 years (range: 38–78). In terms of performance status, 34 patients (56.7%) had a KPS score ≥90, whereas 42 patients (70.0%) had an ECOG PS score of <2. A smoking history was reported in 30 patients (50.0%), whereas 30 patients (50.0%) had no smoking history or an unknown status. According to the 8th edition of the AJCC tumor staging system, 34 patients (56.7%) were classified as having T1–T2 stage, whereas 26 patients (43.3%) were categorized as having T3–T4 stage. In terms of nodal involvement, 22 patients (36.7%) presented with N0–N1 stage, whereas 38 patients (63.3%) had N2–N3 stage. Regarding BM characteristics, 43 patients (71.7%) had oligometastatic disease, defined as 1–3 lesions, and 17 patients (28.3%) had multiple metastases, defined as ≥4 lesions. Furthermore, 40 patients (66.7%) did not have large intracranial metastatic tumors (≥3 cm), 12 patients (20.0%) had tumors measuring 3–4 cm, 7 patients (11.7%) had tumors measuring 4–5 cm, and 1 patient (1.7%) had tumors exceeding 5 cm. For PD-L1 TPS, 29 patients (48.3%) had TPS <50%, 25 patients (41.7%) had TPS ≥50%, and 6 patients (10.0%) had unknown status. Additionally, 26 patients (43.3%) had no extracranial metastasis, whereas 34 patients (56.7%) had concurrent extracranial metastases. A comparison of baseline clinical characteristics between the two groups showed no statistically significant differences across all indicators (all p > 0.05), indicating good comparability. Detailed baseline clinical characteristics are presented in Table 1.
Baseline clinical characteristics of driver gene-negative patients with NSCLC and brain metastases in our cohort (n = 60).

KPS: Karnofsky Performance Status; PD-L1 TPS: programmed death-ligand 1 tumor proportion score; PS: performance status; RT + CT: radiotherapy plus chemotherapy; RT + ICIs: radiotherapy plus immune checkpoint inhibitors; NSCLC: non–small cell lung cancer.
Survival outcomes
The Kaplan–Meier survival analysis showed an mOS of 533 days for the entire cohort, with a 1-year survival rate of 85%. The median progression-free survival (mPFS) was 436 days, with a 1-year PFS rate of 61.67% (Figure 1). In the RT + ICIs group, the mOS was 586 days, whereas in the RT + CT group it was 509 days (p = 0.0208*, hazard ratio (HR) = 1.891, 95% confidence interval (CI): 1.057–3.383; Figure 2(a)). The median PFS in the RT + ICIs group was 494 days, compared with 383 days in the RT + CT group (p = 0.0127*, HR = 2.146, 95% CI: 1.154–3.989; Figure 2(b)). The 1-year OS rates were 83.8% for the RT + CT group and 93.1% for the RT + ICIs group, whereas the 1-year PFS rates were 54.8% and 70%, respectively.

Kaplan–Meier curves of OS and PFS in the entire cohort. (a) OS curve (1y-OS: 85%); (b) PFS curve (1y-PFS: 61.67%).

Comparison of OS and PFS between RT + CT and RT + ICIs groups. (a) OS curve (1y-OS: 83.8% vs 93.1%, P = 0.0208); (b) PFS curve (1y-PFS: 54.8% vs 70%, P = 0.0127). Log-rank test, *P < 0.05.
Univariate and multivariate analysis
Univariate analysis showed significant associations between PFS and several factors, including age ≥60 years (Figure 3(b); p < 0.001***), ECOG PS (PS) ≥2 (Figure 3(d); p < 0.001***), PD-L1 TPS <50% (Figure 3(e); p < 0.001***), and the presence of multiple metastatic lesions (Figure 3(f); p = 0.0124*). In contrast, sex (Figure 3(a); p = 0.493), KPS <90 (Figure 3(c); p = 0.1161), and the presence of large metastatic tumors (Figure 3(g); p = 0.062) were not significantly associated with PFS (all p > 0.05; Table 2). For OS, significant factors included age ≥60 years (Figure 4(b); p < 0.001***), ECOG PS ≥2 (Figure 4(d); p < 0.001***), PD-L1 TPS <50% (Figure 4(e); p < 0.001***), multiple metastatic lesions (Figure 4(f); p = 0.0417*), the presence of large intracranial metastases (Figure 4(g); p = 0.0437*), and the presence of extracranial metastases (Figure 4(h); p = 0.0072**). In contrast, sex (Figure 4(a); p = 0.7089) and KPS <90 (Figure 4(c); p = 0.2412) did not show statistically significant correlations with OS (all p > 0.05; Table 3).

PFS curves from univariate analysis in driver gene-negative NSCLC with brain metastases. (a) Gender: P = 0.493; (b) Age: P < 0.001***; (c) KPS: P = 0.1161; (d) PS: P < 0.001***; (e) PD-L1 TPS: P < 0.001***; (f) Metastatic lesions: P = 0.0144*; (g) Large metastatic tumor: P = 0.062; (h) Extracranial metastasis: P = 0.0124*. Log-rank test.
Univariate and multivariate analyses of progression-free survival (PFS) in driver gene-negative patients with NSCLC and brain metastases. *p < 0.05.

*p <0.05, indicating a statistical significant difference.
CI: confidence interval; HR: hazard ratio; KPS: Karnofsky Performance Status; PD-L1 TPS: programmed death-ligand 1 tumor proportion score; PS: performance status; NSCLC: non–small cell lung cancer.

OS curves from univariate analysis in driver gene-negative NSCLC with brain metastases. (a) Gender: P = 0.7089; (b) Age: P < 0.001***; (c) KPS: P = 0.2412; (d) PS: P < 0.001***; (e) PD-L1 TPS: P < 0.001***; (f) Metastatic lesions: P = 0.0417*; (g) Large metastatic tumor: P = 0.0437*; (h) Extracranial metastasis: P = 0.0072**. Log-rank test.
Univariate and multivariate analyses of overall survival (OS) in driver gene-negative patients with NSCLC and brain metastases.*p <0.05.
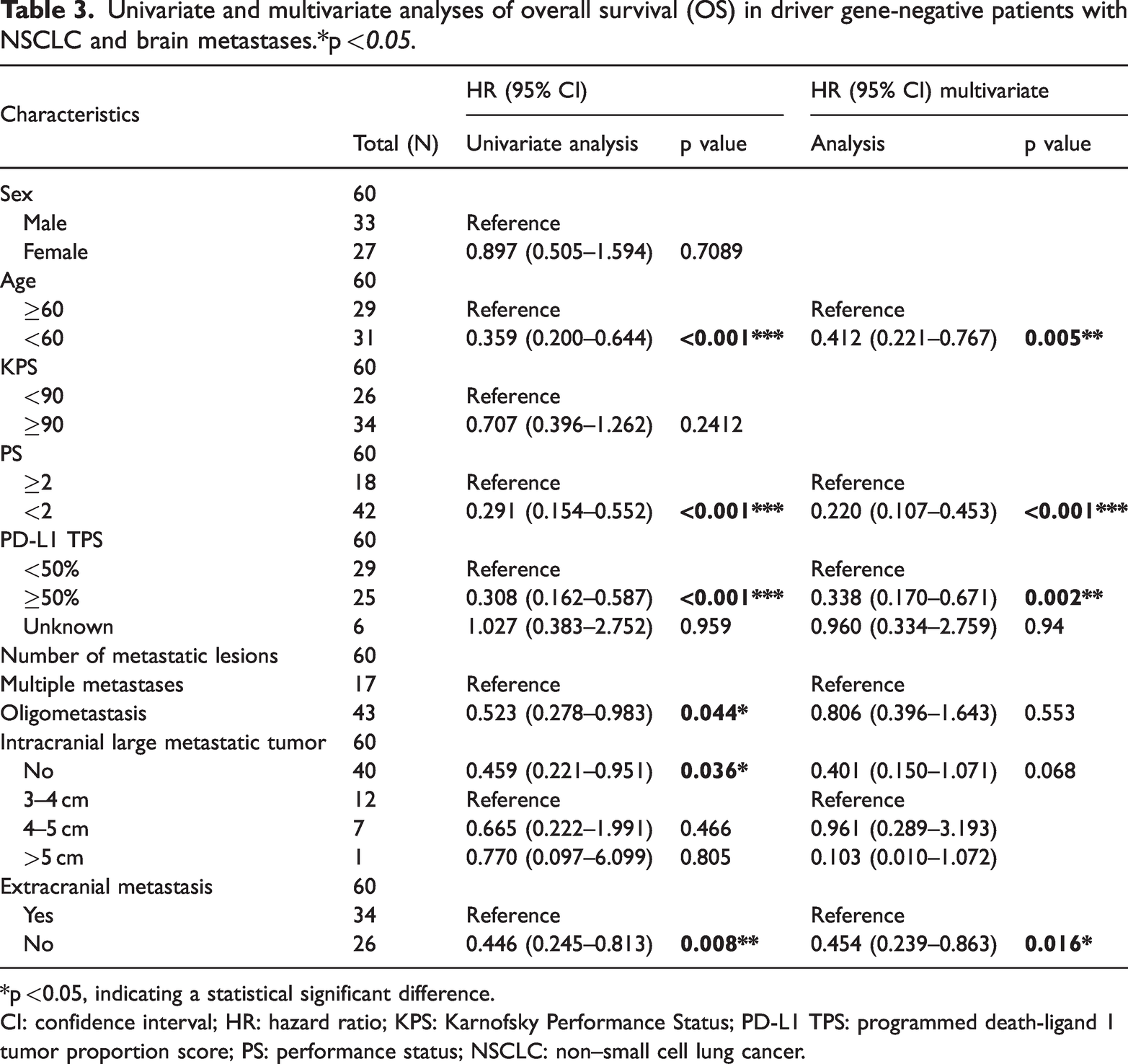
*p <0.05, indicating a statistical significant difference.
CI: confidence interval; HR: hazard ratio; KPS: Karnofsky Performance Status; PD-L1 TPS: programmed death-ligand 1 tumor proportion score; PS: performance status; NSCLC: non–small cell lung cancer.
Multivariate Cox proportional hazards regression analysis was performed to adjust for potential confounding factors. The results showed that age (HR = 0.373, 95% CI: 0.182–0.763, p = 0.007** for PFS; HR = 0.412, 95% CI: 0.221–0.767, p =0.005** for OS), ECOG PS (HR = 0.393, 95% CI: 0.187–0.827, p = 0.014* for PFS; HR = 0.220, 95% CI: 0.107–0.453, p < 0.001*** for OS), PD-L1 TPS <50% (HR = 0.289, 95% CI: 0.131–0.635, p = 0.002** for PFS; HR = 0.412, 95% CI: 0.170–0.671, p = 0.002** for OS), and extracranial metastasis (HR = 0.467, 95% CI: 0.231–0.943, p = 0.034* for PFS; HR =0.454, 95% CI: 0.239–0.863, p = 0.016* for OS) were independent adverse prognostic factors for both PFS and OS. Notably, the presence of large metastatic tumors was not identified as an independent prognostic factor for OS (p > 0.05), and the number of metastatic lesions did not demonstrate independent prognostic value for either OS or PFS (p > 0.05 for both). Complete statistical details of the univariate and multivariate analyses are provided in Tables 2 and 3.
Treatment administration and adverse events
RT regimens were categorized based on BM burden and ECOG PS. Patients with multiple BM (≥4 lesions) or ECOG PS ≥2 were treated with WBRT (36 Gy in 18 fractions, 5 fractions/week) with a local boost (50–60 Gy in 18 fractions), totaling 17 patients. In contrast, the remaining 43 patients with oligometastatic BM (1–3 lesions, diameter <3 cm) received SRT (6 Gy in five fractions). Neurosurgical resection was not performed because of either multiple lesions or poor surgical candidacy, and five patients experienced dose adjustments or temporary treatment pauses due to organ dysfunction or severe radiation-induced adverse events.
All 31 patients in the RT + CT group received platinum-based doublet CT tailored to pathological subtype. Specifically, 29 patients (93.5%) with adenocarcinoma received cisplatin plus pemetrexed, whereas 2 patients (6.5%) with squamous cell carcinoma received carboplatin plus gemcitabine. Among them, 24 patients (77.4%) completed 4–6 cycles every 3 weeks, whereas 7 patients (22.6%) required dose reductions, cycle extensions, or early discontinuation due to grade ≥3 toxicity (n = 4) or deteriorating PS (n = 3).
The 29 patients in the RT + ICIs group received personalized ICI monotherapy, including camrelizumab (n = 4, 13.8%), tislelizumab (n = 11, 37.9%), sintilimab (n = 5, 17.2%), pembrolizumab (n = 5, 17.2%), durvalumab (n = 3, 10.3%), and envafolimab (n = 1, 3.4%). The irAEs were assessed according to CTCAE version 4.03 guidelines, and detailed data are presented in Table 4. Among the 29 patients in the RT + ICIs group, 7 (24.1%) developed irAEs, including 3 cases of rash, 2 cases of immune-mediated colitis, 1 case of mild hypothyroidism, and 1 case of reactive cutaneous capillary proliferation. Symptomatic treatment effectively managed all irAEs, resulting in complete resolution during follow-up, with no residual complications. None of the irAEs resulted in fatalities.
Immune checkpoint inhibitors and immune-related adverse events (irAEs) of RT + ICIs group.
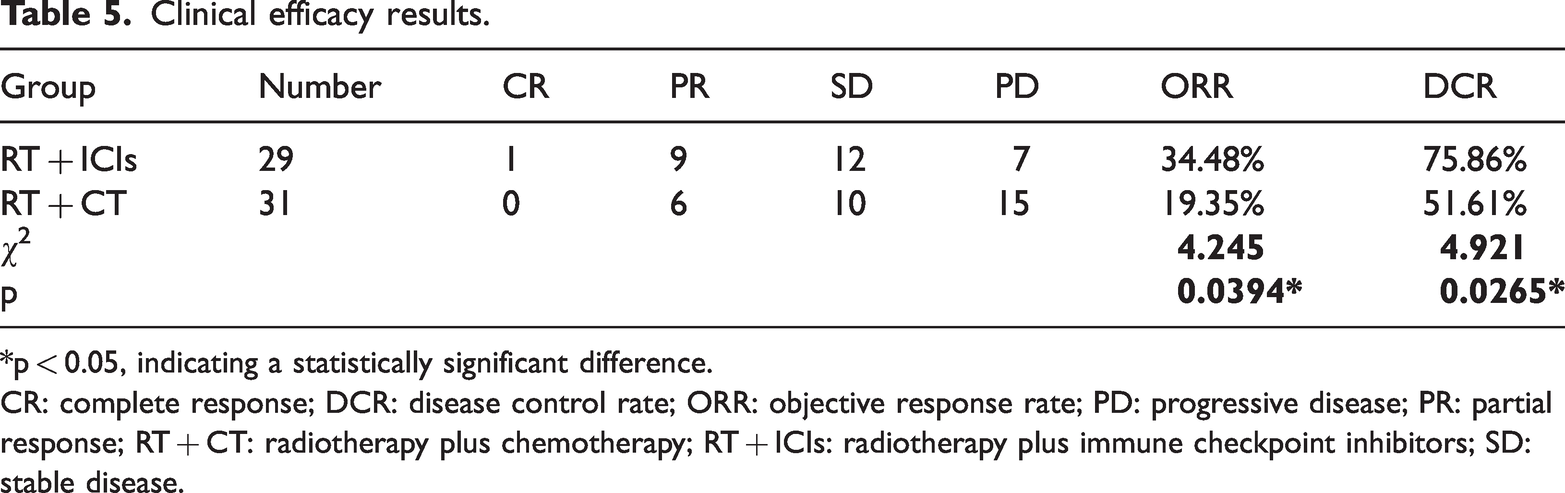
RT + ICIs: radiotherapy plus immune checkpoint inhibitors; CTCAE: Common Terminology Criteria for Adverse Events.
Efficacy evaluation
Treatment responses were assessed according to RECIST version 1.1. The RT + ICIs cohort included 29 patients, whereas the RT + CT cohort included 31 patients. Specifically, 1 patient (3.45%) in the RT + ICIs group achieved CR, whereas no CR was observed in the RT + CT group. PR was documented in 9 patients (31.03%) in the RT + ICIs group versus 6 patients (19.35%) in the RT + CT group. SD was observed in 12 patients (41.38%) in the RT + ICIs group and 10 patients (32.26%) in the RT + CT group, whereas PD occurred in 7 patients (24.14%) in the RT + ICIs group and 15 patients (48.39%) in the RT + CT group. Accordingly, the ORR was significantly higher in the RT + ICIs group than in the RT + CT group (34.48% vs. 19.35%; χ2 = 4.245, p = 0.0394*). In addition, DCR was significantly higher in the RT + ICIs group (75.86% vs. 51.61%; χ2 = 4.921, p = 0.0265*), as summarized in Table 5.
Clinical efficacy results.

*p < 0.05, indicating a statistically significant difference.
CR: complete response; DCR: disease control rate; ORR: objective response rate; PD: progressive disease; PR: partial response; RT + CT: radiotherapy plus chemotherapy; RT + ICIs: radiotherapy plus immune checkpoint inhibitors; SD: stable disease.
Discussion
NSCLC is the predominant histological subtype, accounting for approximately 85% of all lung cancer cases. Patients with driver gene-negative NSCLC and BM have a poor prognosis due to the limited effectiveness of conventional treatments such as CT and RT, and many patients are not suitable for surgical resection of BM at diagnosis. 3 Immunotherapy has emerged as a groundbreaking treatment approach in recent years and has demonstrated significant efficacy in patients with NSCLC, especially patients with tumors lacking driver gene alterations.24,25 This retrospective study focused on patients with driver gene-negative NSCLC complicated by BM to identify prognostic factors influencing survival in this specific group. In addition, it evaluated the efficacy of RT + ICI therapy and the safety of ICIs in this patient cohort, providing important insights for clinical decision-making.
The therapeutic potential of ICIs in patients with NSCLC and BM has been extensively investigated. In a phase II trial, Goldberg et al. 14 demonstrated that pembrolizumab can improve both systemic and intracranial objective response rates, indicating its ability to effectively manage systemic and intracranial lesions simultaneously. Our analysis of patients with driver gene-negative NSCLC and BM showed that RT combined with immunotherapy resulted in higher intracranial ORR and DCR than the RT + CT regimen. This finding highlights the effectiveness of immunotherapy in targeting intracranial lesions in this patient population. In a pivotal randomized controlled trial (RCT), Reck et al. 13 confirmed that in patients with advanced NSCLC and PD-L1 expression ≥50% and without EGFR/ALK alterations, anti-PD-1 therapy outperformed platinum-based CT, significantly prolonging PFS (10.3 vs. 6.0 months; HR = 0.50) and OS (30.0 vs. 14.2 months; HR = 0.63), with a lower incidence of adverse events. A meta-analysis by de Alencar et al. 26 further supported these findings, indicating that ICIs may produce synergistic therapeutic effects between systemic and intracranial disease sites, providing survival benefits without significant neurotoxicity. The IMpower110 RCT reported by Herbst et al. 27 demonstrated that atezolizumab prolonged mOS by 7.1 months compared with CT (20.2 vs. 13.1 months; HR = 0.59; p = 0.01) in patients with advanced NSCLC without EGFR/ALK mutations. Atezolizumab also showed a more favorable safety profile (incidence of grade ≥3 adverse events: 30.1% vs. 52.5%) and a higher intracranial ORR in the BM subgroup (35.7% vs. 20.0%). In our study, multivariate analysis identified PD-L1 expression ≥50% as an independent adverse prognostic factor for OS and PFS in this patient cohort. Consistent with the aforementioned results, we concluded that the efficacy of RT + ICIs group surpassed that of the RT + CT group following RT. In addition, the incidence of irAEs in the RT + ICIs group was less than one-third, and no grade ≥3 irAEs were reported, further supporting the favorable safety profile of immunotherapy. This single-center retrospective study has inherent limitations, our findings, although not directly comparable with those of the aforementioned trials, are consistent with their overall conclusions.
Beyond efficacy and prognostic biomarkers, the timing of RT + ICIs and PD-L1 expression status are key determinants of outcomes in patients with NSCLC and BM. As reported by Qian et al., 21 concurrent administration of RT and ICIs (RT within 90 days of ICI initiation) confers superior therapeutic effects. Meanwhile, a study from the Dana-Farber Brigham Cancer Center identified PD-L1 TPS ≥50% as a favorable prognostic factor, whereas also noting that high PD-L1 expression is associated with an increased risk of radiation necrosis, emphasizing the need for long-term toxicity surveillance in this patient population. 22 In our single-center retrospective study with a relatively small sample size, several limitations should be acknowledged. Due to the constraints of retrospective design, limited sample size, and considerations related to patients’ physical status and economic capacity, RT combined with ICIs or CT was generally administered concurrently within 1 month of hospitalization. Patients receiving sequential treatment were not included for comparison. This represents an inherent limitation, and future prospective studies with larger cohorts should incorporate both concurrent and sequential treatment arms to clarify the optimal timing of the RT–ICI combination. Additionally, our study did not evaluate the association between high PD-L1 expression and the risk of radiation necrosis, nor did it include long-term toxicity surveillance. This limitation restricts our ability to fully assess the safety profile of ICIs in this population. Future studies should therefore incorporate systematic long-term follow-up and toxicity monitoring protocols to capture late-onset adverse events such as radiation necrosis.
Our study enrolled 60 patients with driver gene-negative NSCLC and BM, and the preliminary exploratory findings generally align with the core conclusions of the aforementioned large-scale studies. The RT + ICIs group appeared to demonstrate potential clinical benefits compared with the RT + CT group, with an mOS of 586 versus 509 days (p = 0.0208*, HR = 1.891, 95% CI: 1.057–3.383), an mPFS of 494 versus 383 days (p = 0.0127*, HR = 2.146, 95% CI: 1.154–3.989), an ORR of 34.48% versus 19.35% (p = 0.0394*), and a DCR of 75.86% versus 51.61% (p = 0.0265*). These findings provide exploratory evidence suggesting potential advantages of the RT + ICIs regimen over the conventional RT + CT regimen in this patient population. However, the small sample size (n = 60) limits the generalizability of these findings, and they should therefore be interpreted cautiously. Multivariate Cox proportional hazards regression analysis further suggested that PD-L1 TPS ≥50% may serve as a potential independent prognostic factor for both OS (HR = 0.338, 95% CI: 0.170–0.671, p = 0.002**) and PFS (HR = 0.289, 95% CI: 0.131–0.635, p = 0.002**). These findings provide preliminary support for the potential role of PD-L1 TPS as a biomarker to identify patients who may benefit from RT + ICIs and to guide personalized treatment decisions.
RT regimens in this study were stratified by BM burden: SRT was administered at 6 Gy in 5 fractions for patients with oligometastatic lesions (1–3 lesions) and lesion diameter <3 cm, whereas WBRT was delivered at 36 Gy in 18 fractions combined with local boost irradiation at 50–60 Gy in 18 fractions for patients with multiple metastases (≥4 lesions) or poor ECOG PS (≥2). This stratified strategy achieved a balance between therapeutic efficacy and safety; however, a notable limitation of this study is the lack of further investigation into whether such individualized RT modalities exert differential impacts on survival prognosis and therapeutic efficacy. Future prospective studies with larger sample sizes are warranted to further elucidate the effects of different RT modalities combined with ICIs on the survival prognosis of this patient population.
This study also tentatively identified potential independent adverse prognostic factors that have not been extensively addressed in previous studies: age ≥60 years (OS: HR = 0.412, 95% CI: 0.221–0.767, p = 0.005**; PFS: HR = 0.373, 95% CI: 0.182–0.763, p = 0.007**), ECOG PS ≥2 (OS: HR = 0.220, 95% CI: 0.107–0.453, p < 0.001***; PFS: HR = 0.393, 95% CI: 0.187–0.827, p = 0.014*), and extracranial metastasis (OS: HR = 0.454, 95% CI: 0.239–0.863, p = 0.016*; PFS: HR = 0.467, 95% CI: 0.231–0.943, p = 0.034*). These preliminary observations may tentatively enrich the prognostic assessment system for patients with driver gene-negative NSCLC and BM, highlighting the potential need to integrate patients’ baseline characteristics (age, PS, and extracranial disease burden) into individualized treatment planning. For patients aged ≥60 years, with ECOG PS ≥2, or with extracranial metastasis, intensified supportive care and close monitoring may be warranted to maximize potential treatment benefits, although these recommendations remain exploratory pending validation in larger cohorts.
In terms of safety, preliminary data from this study showed that the incidence of irAEs in the RT + ICIs group was 24.1% (7/29), including 3 cases of rash, 2 cases of immune-mediated colitis, 1 case of mild hypothyroidism, and 1 case of reactive cutaneous capillary proliferation. All irAEs achieved complete resolution after symptomatic treatment without residual sequelae or treatment-related deaths, which tentatively aligns with the safety profile reported in previous studies, 27 offering preliminary evidence suggesting that ICIs may be well tolerated in this patient population. The incidence of severe irAEs (grades 4–5) was 0, a finding that may be attributed to individualized ICI selection and close toxicity monitoring; however, this association remains speculative given the small sample size. Collectively, these observations provide preliminary, tentative support for the potential clinical applicability of RT + ICIs, but they are exploratory in nature and require validation in larger cohorts.
This study has several inherent limitations that should be objectively acknowledged. First, as a single-center retrospective cohort study, it strictly adhered to the STROBE guidelines and the Declaration of Helsinki (1975, revised 2024), with Institutional Review Board approval and deidentified patient data. However, nonrandom enrollment is an inherent characteristic of retrospective studies, as patient allocation relies on clinical treatment decisions rather than predefined randomization protocols, selection bias is inevitably introduced. Even with baseline balance analysis and multivariate adjustments, residual confounding from unmeasured variables remains a pervasive challenge in retrospective designs and cannot be fully eliminated, regardless of statistical adjustments. Second, the relatively small sample size (n = 60) is a common limitation of single-center retrospective studies, which directly results in insufficient statistical power for subgroup analyses (e.g. only two patients with squamous cell carcinoma were included in the RT + CT group). This inherent limitation may undermine the stability of multivariate Cox regression results, and the findings of this study remain preliminary and exploratory. They require validation in larger-scale, multicenter cohorts to confirm their reliability. Third, the completeness and granularity of clinical data collection are inherently limited in retrospective research. PD-L1 expression data collection was inadequate in this study, with only binary stratification (TPS <50% vs. ≥50%) and 10.0% (6/60) of patients having unknown PD-L1 expression status. This inherent data limitation, stemming from the retrospective study design, prevented refined analysis of prognostic value across distinct PD-L1 expression gradients, thereby hindering the precise identification of potential beneficiaries of ICI therapy. Fourth, due to inherent limitations in clinical data archiving and follow-up management, this study was unable to comprehensively assess late-onset toxicities such as radiation necrosis. Additionally, the impact of RT–ICI combination timing on therapeutic efficacy was not incorporated into the systematic analysis. This is because this key treatment parameter is often not systematically documented in retrospective clinical records, another consequence of the inherent limitations of retrospective study designs. Furthermore, the retrospective nature inherently precludes independent verification of certain study data, which may exert a slight impact on the reproducibility of the research methodology, a pervasive challenge in nonprospective study designs. Future studies should adopt prospective designs by expanding the sample size to overcome the inherent limitations of retrospective analyses, improving the granularity of biomarker data collection, standardizing the documentation of local treatment regimens and RT–ICI combination timing, and extending the follow-up period, thereby enhancing the reliability and clinical translatability of research findings.
Footnotes
Acknowledgments
We sincerely thank the Institutional Ethics Committee of The Second Hospital of Hebei Medical University for their guidance and approval. We also appreciate all medical staff and researchers in the Department of Radiotherapy and the Central Laboratory of the hospital for their efforts in data collection, collation, and follow-up.
Author contributions
Zizhou Zhang contributed to conceptualization, data curation, investigation, and writing of the original draft. Jiayuan Li was responsible for data curation and formal statistical analysis. Huandi Zhou participated in methodology design and manuscript review. Liubing Hou assisted with clinical data verification and follow-up information collation. Yu Wang conducted literature retrieval and reference formatting. Mengting Zhang supported result interpretation and figure preparation. Yufeng Zhang participated in the optimization of statistical methods. Xiaoying Xue supervised the entire study, secured institutional ethics approval, and finalized the manuscript for submission. All authors have read and agreed to the published version of the manuscript.
Data availability statement
The clinical data used in this retrospective study are subject to patient confidentiality and are not publicly available. Requests for data access may be directed to the corresponding author.
Declaration of conflicting interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as potential conflicts of interest.
Funding
The project is supported by the Hebei Province Medical Applicable Technology Tracking Project (Grant No: GZ20260028) and the Second Hospital of Hebei Medical University, Hospital-Enterprise Cooperation Special Project (Grant No: 2HY202552).
Ethical approval
The study protocol was reviewed and approved by the Institutional Ethics Committee of The Second Hospital of Hebei Medical University (Approval No. 2025-R725) in accordance with the International Council for Harmonisation Good Clinical Practice (ICH-GCP) guidelines, relevant Chinese regulations, the Declaration of Helsinki, and national regulations for the ethical review of biomedical research involving human subjects. Written informed consent was obtained from all participants.
