Abstract
Background:
Polycythemia vera (PV) is a myeloproliferative neoplasm driven by
Objectives:
To evaluate whether a ⩾50% reduction in the neutrophil-to-lymphocyte ratio (NLR) can serve as a surrogate marker of hematologic and molecular response during ropeginterferon therapy.
Design:
Secondary analysis of a multicenter, phase II, open-label trial in South Korea.
Methods:
Ninety-five patients with PV received ropeginterferon alfa-2b biweekly for 48 weeks. NLR and
Results:
NLR half reduction significantly predicted hematologic response (week 24 OR 6.42,
Conclusion:
NLR half reduction is a simple, cost-effective biomarker that may reflect molecular response and treatment efficacy in PV.
Plain Language Summary
Polycythemia vera (PV) is a rare blood cancer in which the bone marrow makes too many red blood cells. This disease is mainly caused by a change in a gene called JAK2, which leads to uncontrolled blood cell production and long-term inflammation. Doctors treat PV to reduce the risk of blood clots and to slow disease progression. One modern treatment, ropeginterferon alfa-2b, helps control blood counts and can reduce the JAK2 mutation level over time. Measuring the JAK2 mutation (“molecular monitoring”) is an important part of understanding how well the treatment works. However, this test requires special laboratory equipment and may not be available in all hospitals. In this study, we looked for an easier way to monitor treatment response using information from routine blood tests. We focused on the neutrophil-to-lymphocyte ratio (NLR), which reflects inflammation in the body and can be calculated from standard blood counts. We found that patients whose NLR dropped by at least 50% during treatment—what we called “NLR half reduction”—were more likely to achieve both good blood control and a decrease in JAK2 mutation levels. This means that NLR half reduction could serve as a simple, low-cost way to estimate whether the treatment is working, especially in places where molecular testing is not available. Our findings suggest that regular blood count results could give doctors useful information about disease activity and treatment success in patients with polycythemia vera.
Introduction
Myeloproliferative neoplasms (MPNs) are clonal hematologic disorders characterized by excessive production of mature blood cells and dysregulated inflammatory signaling by malignant clones.
1
Polycythemia vera (PV), a common MPN subtype, is primarily driven by activating
The neutrophil-to-lymphocyte ratio (NLR), derived from routine complete blood counts, serves as an accessible marker of systemic inflammation and has been explored across various malignancies and inflammatory states.
10
In PV, NLR is typically elevated due to panmyelosis with neutrophilia and relative lymphopenia. Prior studies have shown that NLR levels are higher in patients with MPNs than in healthy controls.11,12 We hypothesized that dynamic changes in NLR during treatment may reflect alterations in disease burden, particularly the
Methods
Study design and patients
We conducted a secondary analysis of a phase II, single-arm, open-label, investigator-initiated trial evaluating the efficacy of ropeginterferon alfa-2b in patients with PV across 16 centers in South Korea. 13 The study began in October 2021 and the core treatment period was completed in November 2023. Eligible patients were adults with a diagnosis of PV according to World Health Organization criteria who were candidates for interferon therapy; both hydroxyurea-naïve and previously treated patients were included. Patients with contraindications to interferon therapy, including uncontrolled autoimmune disease or significant psychiatric disorders, were excluded in accordance with the original trial protocol. Patients received ropeginterferon alfa-2b subcutaneously every 2 weeks, starting at 250 µg, escalating to 350 µg (week 3) and 500 µg (week 5), and maintained thereafter. A total of 95 patients who received at least one dose were included in the full analysis set.
Data collection and assessments
In total, 451 serial blood samples were analyzed, including complete blood counts and
Statistical analysis
Receiver operating characteristic (ROC) analysis was used to identify a 50% reduction in NLR as the optimal threshold to associate CHR and MR, termed “NLR half reduction.” Sensitivity, specificity, positive predictive value, and negative predictive value were calculated at each prespecified time point to evaluate the discriminative performance of NLR half reduction. Associations between NLR half reduction and clinical outcomes were evaluated using generalized estimating equations (GEE) with a logit link function to account for repeated measurements within individuals over time. Odds ratios (ORs) and 95% confidence intervals (CIs) were estimated from these logistic GEE models using an exchangeable working correlation structure.
Outcomes were assessed at prespecified time points (weeks 12, 24, 36, and 48), and separate multivariable models were constructed for each outcome (CHR and MR) at each time point. Variables included in the multivariable models were selected a priori from baseline demographic and clinical characteristics summarized in Table S1, including age, sex, hydroxyurea treatment status (naïve vs pretreated), and alcohol consumption. Statistical analyses were conducted using R software (version 4.2.2; R Foundation for Statistical Computing, Vienna, Austria). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 14
Results
Patient characteristics
A total of 95 patients with PV were included in the full analysis set, with 451 serial blood test results collected during the 48-week treatment period. The median age was 58 years (range, 26–81), and 54% were male. Forty-eight percent had high-risk PV, 54.7% were hydroxyurea-naïve, and the median disease duration was 907 days. The median baseline NLR was 4.87 (range, 1.17–32.34; Table S1).
Correlation between NLR and JAK2 allele burden
We assessed the correlation between

ROC curves for CHR and MR prediction based on NLR reduction. (a) ROC curve for predicting CHR across all assessment time points. The optimal NLR reduction cut-off was −0.51, with an AUC of 0.671 (95% CI: 0.614–0.728,
Association of NLR half reduction with treatment response
In multivariate analysis, NLR half reduction was not independently associated with CHR at week 12; instead, alcohol consumption (OR 2.37,
For molecular response, NLR half reduction was a strong and consistent predictor across all time points: week 12 (OR 7.49,
Multivariate regression analysis for predictors of CHR and MR.
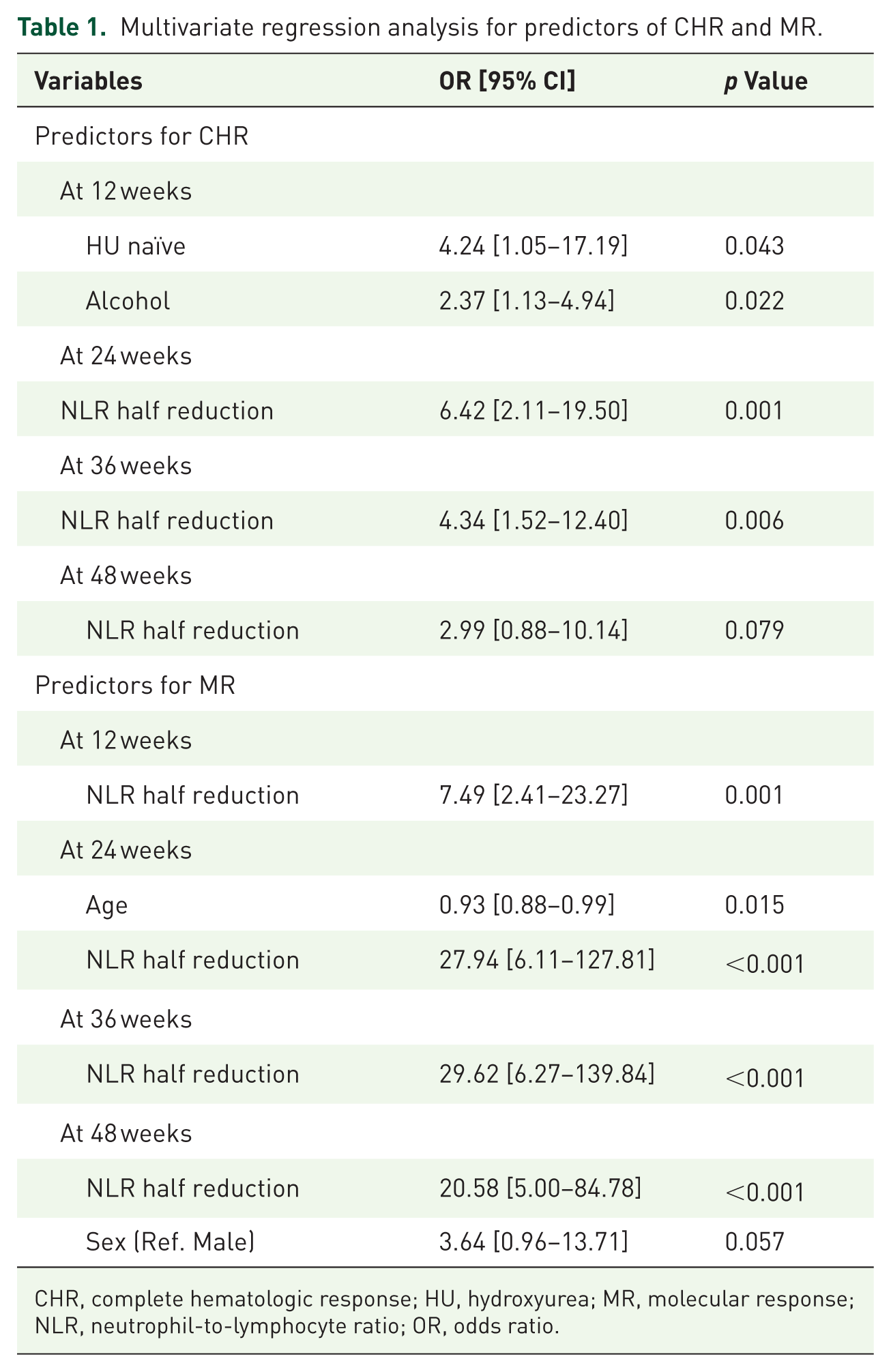
CHR, complete hematologic response; HU, hydroxyurea; MR, molecular response; NLR, neutrophil-to-lymphocyte ratio; OR, odds ratio.
Sensitivity and specificity analyses based on ROC curves demonstrated that NLR half reduction showed moderate to good discriminative performance for molecular response, particularly at weeks 24–48, whereas its accuracy for predicting CHR was modest across time points (Figures S3 and S4).
Association of NLR half reduction with clinical events
During the 48-week core study period, three thrombotic events and one case of myelofibrotic transformation were observed. Because these events did not consistently coincide with scheduled study visits, NLR half reduction status at the most recent regular visit preceding the event was used for analysis. Event rates were lower among patients who had achieved NLR half reduction compared with those who had not (1.7% vs 9.1%). NLR half reduction was associated with a lower odds of thrombotic or myelofibrotic events (OR 0.17, 95% CI: 0.003–2.18), although this association did not reach statistical significance due to the small number of events (Fisher’s exact test,
Discussion
Our findings demonstrate that a ⩾50% reduction in the NLR during ropeginterferon alfa-2b therapy is significantly associated with both hematologic and molecular responses in patients with PV. In particular, the consistent association with MR across all time points supports NLR half reduction as a dynamic biomarker of treatment efficacy.
While hematologic response mitigates immediate clinical risks such as thrombosis, molecular response—defined by reductions in
NLR offers a practical and accessible alternative. Easily derived from complete blood counts, it provides a real-time, cost-effective, and universally available tool. Its utility may be particularly high in resource-limited environments or for treatment monitoring between allele burden assessments. Moreover, recent evidence suggests that patients with high baseline NLR may exhibit greater sensitivity to interferon therapy. 15 Another prospective study also demonstrated that NLR ⩾5 was independently associated with increased mortality and a more proliferative PV phenotype, reinforcing its potential as a clinical biomarker. 16 Our data extend these observations by showing that treatment-induced reductions in NLR—not just baseline values—are predictive of molecular response. Notably, NLR half reduction was not associated with CHR at the early 12-week time point, at which alcohol consumption and treatment-related factors were more strongly associated with hematologic response. This finding suggests that early hematologic responses may reflect patient- or treatment-related characteristics, whereas NLR kinetics become more informative for sustained hematologic and molecular responses over time. Collectively, these findings suggest that NLR may serve not only as a prognostic marker but also as a biomarker associated with response to interferon therapy, potentially informing individualized treatment strategies. In exploratory ROC analyses, NLR half reduction showed moderate discriminative performance for molecular response, particularly at later time points, whereas its performance for CHR was more modest. These findings support the role of NLR half reduction primarily as an associated biomarker of molecular response rather than as a standalone predictive tool.
In an exploratory post hoc analysis of clinical events, patients who achieved NLR half reduction showed a lower incidence of thrombotic or myelofibrotic events compared with those who did not. Although these findings suggest a potential association between NLR dynamics and disease-related complications, they should be interpreted with caution, given the small number of events and the lack of NLR measurements at the exact time of event occurrence.
Limitations
This study has several limitations. It was a secondary analysis of a single-arm, non-randomized trial, and causal inference cannot be established due to its observational design. The present analysis was also limited to the 48-week core treatment period of the trial; therefore, more comprehensive evaluations of long-term clinical outcomes and hematologic toxicity will be more appropriately addressed upon completion of the ongoing extended follow-up study. The small number of thrombotic and myelofibrotic events and the lack of NLR measurements at the exact time of event occurrence limited the ability to draw definitive conclusions regarding the association between NLR reduction and clinical events. In addition, we did not perform stratified analyses according to baseline
Conclusion
Nonetheless, the consistent association between NLR half reduction and MR suggests that NLR may serve as a simple and cost-effective surrogate marker in clinical practice where molecular testing is not always feasible. Future prospective and randomized studies are warranted to validate the role of NLR kinetics as a surrogate marker and to define its place in the therapeutic monitoring of PV.
Supplemental Material
sj-docx-1-tah-10.1177_20406207261431904 – Supplemental material for Neutrophil-to-lymphocyte ratio half reduction as a surrogate of molecular response in polycythemia vera treated with ropeginterferon alfa-2b
Supplemental material, sj-docx-1-tah-10.1177_20406207261431904 for Neutrophil-to-lymphocyte ratio half reduction as a surrogate of molecular response in polycythemia vera treated with ropeginterferon alfa-2b by Seug Yun Yoon, Sung-Soo Yoon, Deok-Hwan Yang, Gyeong-Won Lee, Sang Kyun Sohn, Ho-Jin Shin, Sung Hwa Bae, Chul Won Choi, Eun-Ji Choi, June-Won Cheong, Soo-Mee Bang, Joon Seong Park, Yong Park, Young Hoon Park and Sung-Eun Lee in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-2-tah-10.1177_20406207261431904 – Supplemental material for Neutrophil-to-lymphocyte ratio half reduction as a surrogate of molecular response in polycythemia vera treated with ropeginterferon alfa-2b
Supplemental material, sj-docx-2-tah-10.1177_20406207261431904 for Neutrophil-to-lymphocyte ratio half reduction as a surrogate of molecular response in polycythemia vera treated with ropeginterferon alfa-2b by Seug Yun Yoon, Sung-Soo Yoon, Deok-Hwan Yang, Gyeong-Won Lee, Sang Kyun Sohn, Ho-Jin Shin, Sung Hwa Bae, Chul Won Choi, Eun-Ji Choi, June-Won Cheong, Soo-Mee Bang, Joon Seong Park, Yong Park, Young Hoon Park and Sung-Eun Lee in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-3-tah-10.1177_20406207261431904 – Supplemental material for Neutrophil-to-lymphocyte ratio half reduction as a surrogate of molecular response in polycythemia vera treated with ropeginterferon alfa-2b
Supplemental material, sj-docx-3-tah-10.1177_20406207261431904 for Neutrophil-to-lymphocyte ratio half reduction as a surrogate of molecular response in polycythemia vera treated with ropeginterferon alfa-2b by Seug Yun Yoon, Sung-Soo Yoon, Deok-Hwan Yang, Gyeong-Won Lee, Sang Kyun Sohn, Ho-Jin Shin, Sung Hwa Bae, Chul Won Choi, Eun-Ji Choi, June-Won Cheong, Soo-Mee Bang, Joon Seong Park, Yong Park, Young Hoon Park and Sung-Eun Lee in Therapeutic Advances in Hematology
Supplemental Material
sj-tiff-4-tah-10.1177_20406207261431904 – Supplemental material for Neutrophil-to-lymphocyte ratio half reduction as a surrogate of molecular response in polycythemia vera treated with ropeginterferon alfa-2b
Supplemental material, sj-tiff-4-tah-10.1177_20406207261431904 for Neutrophil-to-lymphocyte ratio half reduction as a surrogate of molecular response in polycythemia vera treated with ropeginterferon alfa-2b by Seug Yun Yoon, Sung-Soo Yoon, Deok-Hwan Yang, Gyeong-Won Lee, Sang Kyun Sohn, Ho-Jin Shin, Sung Hwa Bae, Chul Won Choi, Eun-Ji Choi, June-Won Cheong, Soo-Mee Bang, Joon Seong Park, Yong Park, Young Hoon Park and Sung-Eun Lee in Therapeutic Advances in Hematology
Supplemental Material
sj-tiff-5-tah-10.1177_20406207261431904 – Supplemental material for Neutrophil-to-lymphocyte ratio half reduction as a surrogate of molecular response in polycythemia vera treated with ropeginterferon alfa-2b
Supplemental material, sj-tiff-5-tah-10.1177_20406207261431904 for Neutrophil-to-lymphocyte ratio half reduction as a surrogate of molecular response in polycythemia vera treated with ropeginterferon alfa-2b by Seug Yun Yoon, Sung-Soo Yoon, Deok-Hwan Yang, Gyeong-Won Lee, Sang Kyun Sohn, Ho-Jin Shin, Sung Hwa Bae, Chul Won Choi, Eun-Ji Choi, June-Won Cheong, Soo-Mee Bang, Joon Seong Park, Yong Park, Young Hoon Park and Sung-Eun Lee in Therapeutic Advances in Hematology
Supplemental Material
sj-tiff-6-tah-10.1177_20406207261431904 – Supplemental material for Neutrophil-to-lymphocyte ratio half reduction as a surrogate of molecular response in polycythemia vera treated with ropeginterferon alfa-2b
Supplemental material, sj-tiff-6-tah-10.1177_20406207261431904 for Neutrophil-to-lymphocyte ratio half reduction as a surrogate of molecular response in polycythemia vera treated with ropeginterferon alfa-2b by Seug Yun Yoon, Sung-Soo Yoon, Deok-Hwan Yang, Gyeong-Won Lee, Sang Kyun Sohn, Ho-Jin Shin, Sung Hwa Bae, Chul Won Choi, Eun-Ji Choi, June-Won Cheong, Soo-Mee Bang, Joon Seong Park, Yong Park, Young Hoon Park and Sung-Eun Lee in Therapeutic Advances in Hematology
Supplemental Material
sj-tiff-7-tah-10.1177_20406207261431904 – Supplemental material for Neutrophil-to-lymphocyte ratio half reduction as a surrogate of molecular response in polycythemia vera treated with ropeginterferon alfa-2b
Supplemental material, sj-tiff-7-tah-10.1177_20406207261431904 for Neutrophil-to-lymphocyte ratio half reduction as a surrogate of molecular response in polycythemia vera treated with ropeginterferon alfa-2b by Seug Yun Yoon, Sung-Soo Yoon, Deok-Hwan Yang, Gyeong-Won Lee, Sang Kyun Sohn, Ho-Jin Shin, Sung Hwa Bae, Chul Won Choi, Eun-Ji Choi, June-Won Cheong, Soo-Mee Bang, Joon Seong Park, Yong Park, Young Hoon Park and Sung-Eun Lee in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
