Abstract
Objective
To systematically review the available clinical evidence regarding the safety and efficacy of knee intraosseous injections for the treatment of bone marrow lesions in patients affected by knee osteoarthritis.
Design
A literature search was carried out on PubMed, Embase, and Google Scholar databases in January 2020. The following inclusion criteria were adopted: (1) studies of any level of evidence, dealing with subchondral injection of bone substitute materials and/or biologic agents; (2) studies with minimum 5 patients treated; and (3) studies with at least 6 months’ follow-up evaluation. All relevant data concerning clinical outcomes, adverse events, and rate of conversion to arthroplasty were extracted.
Results
A total of 12 studies were identified: 7 dealt with calcium phosphate administration, 3 with platelet-rich plasma, and 2 with bone marrow concentrate injection. Only 2 studies were randomized controlled trials, whereas 6 studies were prospective and the remaining 4 were retrospective. Studies included a total of 459 patients treated with intraosseous injections. Overall, only a few patients experienced adverse events and clinical improvement was documented in the majority of trial. The lack of any comparative evaluation versus subchondral drilling alone is the main limitation of the available evidence.
Conclusions
Knee intraosseous injections are a minimally invasive and safe procedure to address subchondral bone damage in osteoarthritic patients. They are able to provide beneficial effects at short-term evaluation. More high-quality evidence is needed to confirm their potential and to identify the best product to adopt in clinical practice.
Keywords
Introduction
A number of studies have highlighted that subchondral bone plays a key role in the initiation and progression of many knee joint pathologies. Bone marrow lesions (BMLs), detected at magnetic resonance imaging (MRI), are suggestive of the presence of subchondral bone damage, that can be linked to a number of different conditions:1,2 post-traumatic events, such as ligament injuries; primitive subchondral bone disorders, such as insufficiency fractures that could lead to spontaneous osteonecrosis of the knee (SONK); degenerative diseases, such as OA; inflammatory condition, like rheumatoid arthritis; metabolic disorders, for example, osteoporosis; neurologic impairment, such as in the case of algodystrophy; vascular impairment, leading to avascular osteoneocrosis (AVN). Differential diagnosis could be extremely complex and should rely both on accurate clinical evaluation and imaging, 3 sometimes requiring seriated MRI assessments over time to proper target the therapeutic approach. In most cases, BMLs are reversible but sometimes they can “chronicize” with the onset of irreversible conditions, like SONK, 4 characterized by collapse of subchondral bone and gradual flattening of the articular surface.
With regard to knee OA, it is one of the most prevalent chronic health conditions among adults, leading to pain and functional disability.5,6 BMLs are common findings in the context of knee OA, and there are pathogenetic hypothesis suggesting that OA starts as an alteration in the subchondral bone, which then leads to the destruction of the overlying articular cartilage. Typical histopathological findings include microcracks, microedemas, microbleeding within the subchondral region, as well as subchondral bone cysts.
7
The sclerotic subchondral bone mineralization is lower than in healthy joints, leading to a lower stiffness and mechanical resistance, as demonstrated through mechanical tests.8,9 Furthermore, as cartilage has no innervation, the highly innervated subchondral bone may be one of the primary sources of pain. Under a clinical point of view, the presence of BMLs correlates with pain and joint deterioration, and larger BMLs areas are associated to symptoms’ worsening over time.10-13 Knee OA patients with pattern of BMLs show also a poor prognosis and have an accelerated progression to end-stage disease, with a highly predictable need for total knee arthroplasty (TKA)14-16: A study by Scher
Although the incidence of OA is increasing and the issue of young and middle-aged active patients who prefer to postpone TKA is spreading, no fully effective conservative therapy has yet been established to restore the damaged cartilage. 18 Traditional injectable therapies have shown promising, but short-lasting results in patients affected by early or moderate OA.19-22 Biologic agents have also been introduced into clinical practice, and they are obviously attractive due to their potential to induce tissue healing and homeostatic regeneration without being deeply invasive.23,24 Since BMLs have shown to have a key role in the initiation and progression of the pathology, they represent a new possible target to be addressed for the treatment of OA.7,23,24
Intraosseous injections are a minimally invasive procedure performed to address chronic BMLs and improve subchondral bone quality without altering its physiological properties. They are carried out under fluoroscopic control to determine the injection site and the distribution of the injectable product in the subchondral region.12,25 Currently, there are different options to perform knee intraosseous injections:
(a) Calcium phosphate (CaP) bone substitute injection, also known as subchondroplasty, which exploits the good osteoconductive and osteoinductive properties of CaP ceramics to improve mineralization and promote local bone remodeling 26
(b) Platelet-rich plasma (PRP), which takes advantage of the several autologous growth factors (GFs) contained in platelets’ alpha-granules, which have been shown to promote osteogenic differentiation of resident mesenchymal stem cells (MSCs)23,24,27,28
(c) Bone marrow concentrate (BMC), which is a product rich in autologous MSCs harvested from the bone marrow. The intraosseous delivery of MSCs has been proven to be effective in bone fracture healing and in restoring the homeostasis of subchondral bone.29,30
The aim of the present article is to systematically review the available clinical evidence concerning the safety and efficacy of knee intraosseous injections. The primary outcome was the rate of conversion to arthroplasty, in order to understand whether this approach could play a role in delaying joint replacement.
Methods
A systematic review was performed on 3 medical electronic databases (PubMed, Embase, and Google Scholar) by 2 independent authors (FO and AP) on January 28, 2020. To achieve the maximum search strategy sensitivity we combined keywords with Boolean operators “OR” or “AND” for the literature terms “calcium phosphate,” “tricalcium,” “bone substitute,” “bony substitute,” “PRP,” “ACP,” “platelet-rich,” “Accufill,” “platelet derived,” “platelet concentrate,” “growth factor,” “stem cells,” “mesenchymal,” “adipose,” “BMC,” “bone marrow concentrate,” “SVF,” with the terms “subchondroplasty,” “subchondral injection,” “subchondral,” “intraosseous,” “bone cartilage interface,” and “interface.”
A total of 13,536 potential articles were identified through our database search.
The following inclusion criteria were adopted for study selection: clinical trials of any level of evidence, written in English, and reporting clinical results following knee intraosseous injections of bone substitutes or biologic agents (PRP or MSCs products), with a minimum number of 5 patients treated and a minimum follow-up period of 6 months. We excluded all articles coming from non-peer-reviewed journals, surveys, editorials, special topics, conference presentations, narrative reviews, articles where the access to the full text was blocked and case reports or mini case-series (<5 patients).
After title and abstract screening, 82 studies were assessed for eligibility. Full text was retrieved and after a deep and careful review 74 articles were excluded because not related to the knee joint, presented less than 5 cases or with an insufficient period of follow-up.
All the references of the retrieved articles were further reviewed for identification of potentially relevant studies and reassessed using the same inclusion and exclusion criteria stated above: 4 additional studies were included from the references. A PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flowchart of the selection and screening process is provided in Figure 1 .
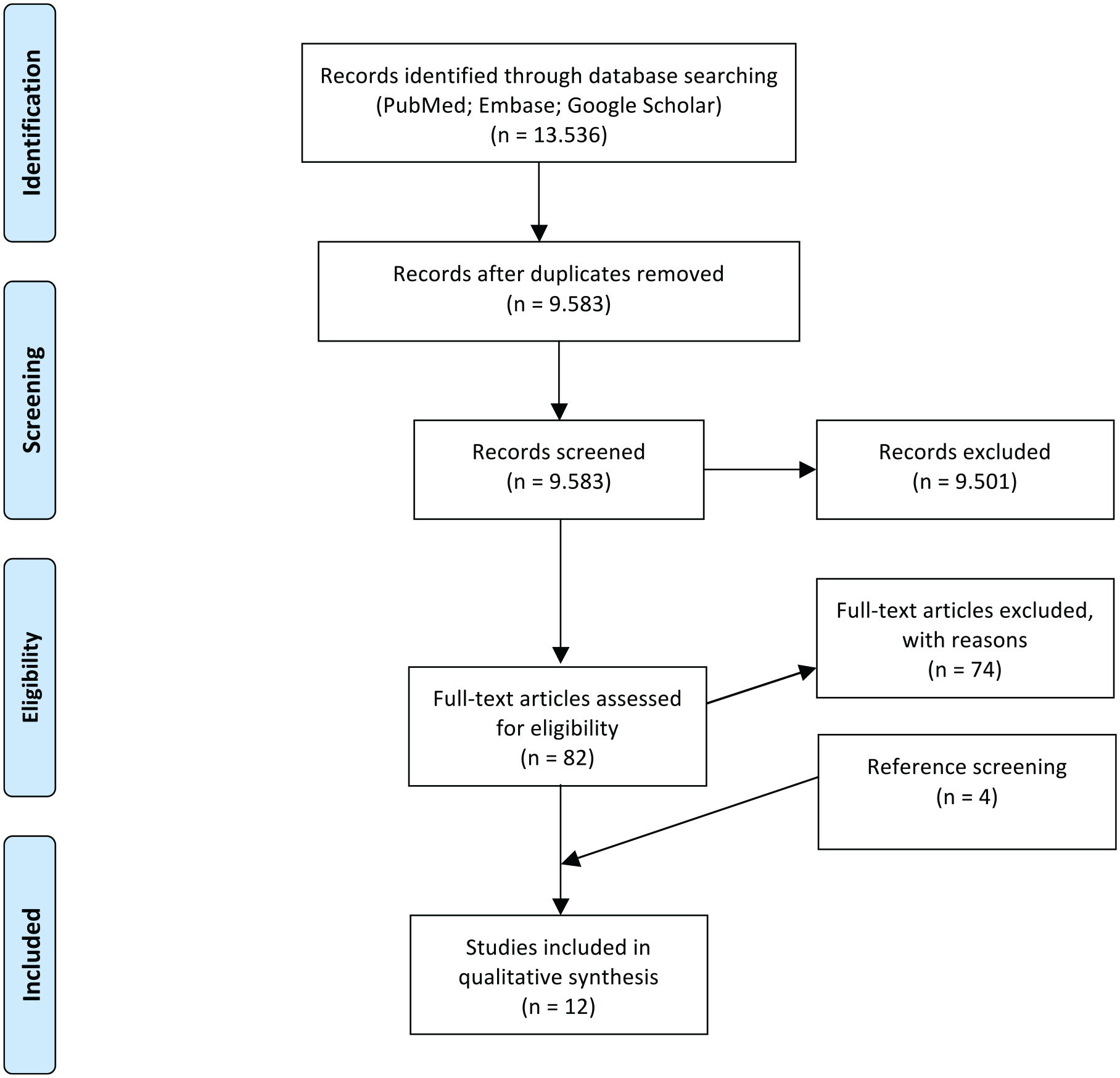
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flowchart showing the studies’ selection process.
All data were extracted and reviewed from article texts, tables, and figures by 2 independent investigators (FO and AP). Discrepancies between the 2 reviewers were resolved by discussion and consensus. The final results were reviewed by the senior investigators (BDM and EK). The main outcome of the present systematic review was the rate of conversion to arthroplasty during follow-up evaluation. Demographics of patients, results in terms of pain evaluation and patients’ subjective reported outcomes were also collected.
Risk of bias and quality assessment of the included articles was done following the Coleman methodology score modified by Kon
Results
Application Methods and Quality Assessment of the Available Literature
According to the aforementioned inclusion and exclusion criteria, we identified a total of 12 studies. Seven of the included articles involved the use of bone substitutes (i.e. subchondroplasty), 3 studies used the injection of PRP and other 2 involved injection of BMC. Studies quality was assessed by Coleman methodology modified by Kon
Quality Assessment of the Included Studies by Coleman Methodology Score Modified by Kon
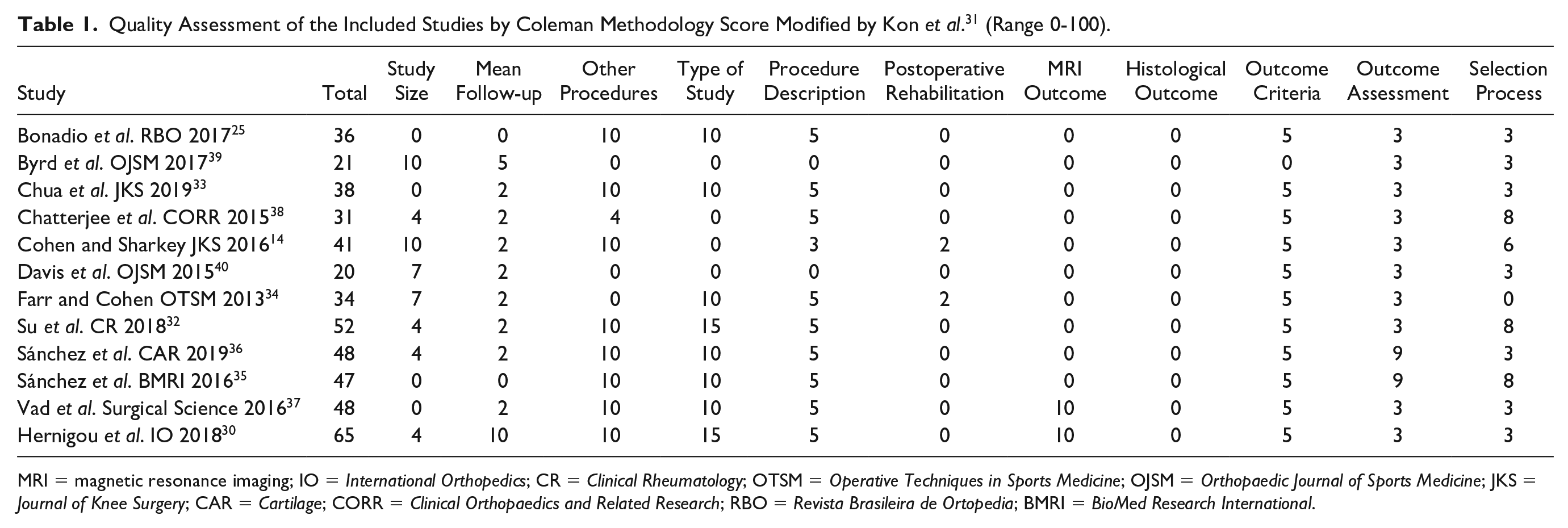
MRI = magnetic resonance imaging; IO =
Patients’ Demographics
The studies included a total of 459 patients, with a mean age of 58 years (range 28-68 years) and an average follow-up of 23 months (range 6-144 months) as shown in
Table 2
. Most of the studies had a mean follow-up around 14 months with the exception of Bonadio
Patients’ Demographics and Relevant Characteristics of the Included Studies.
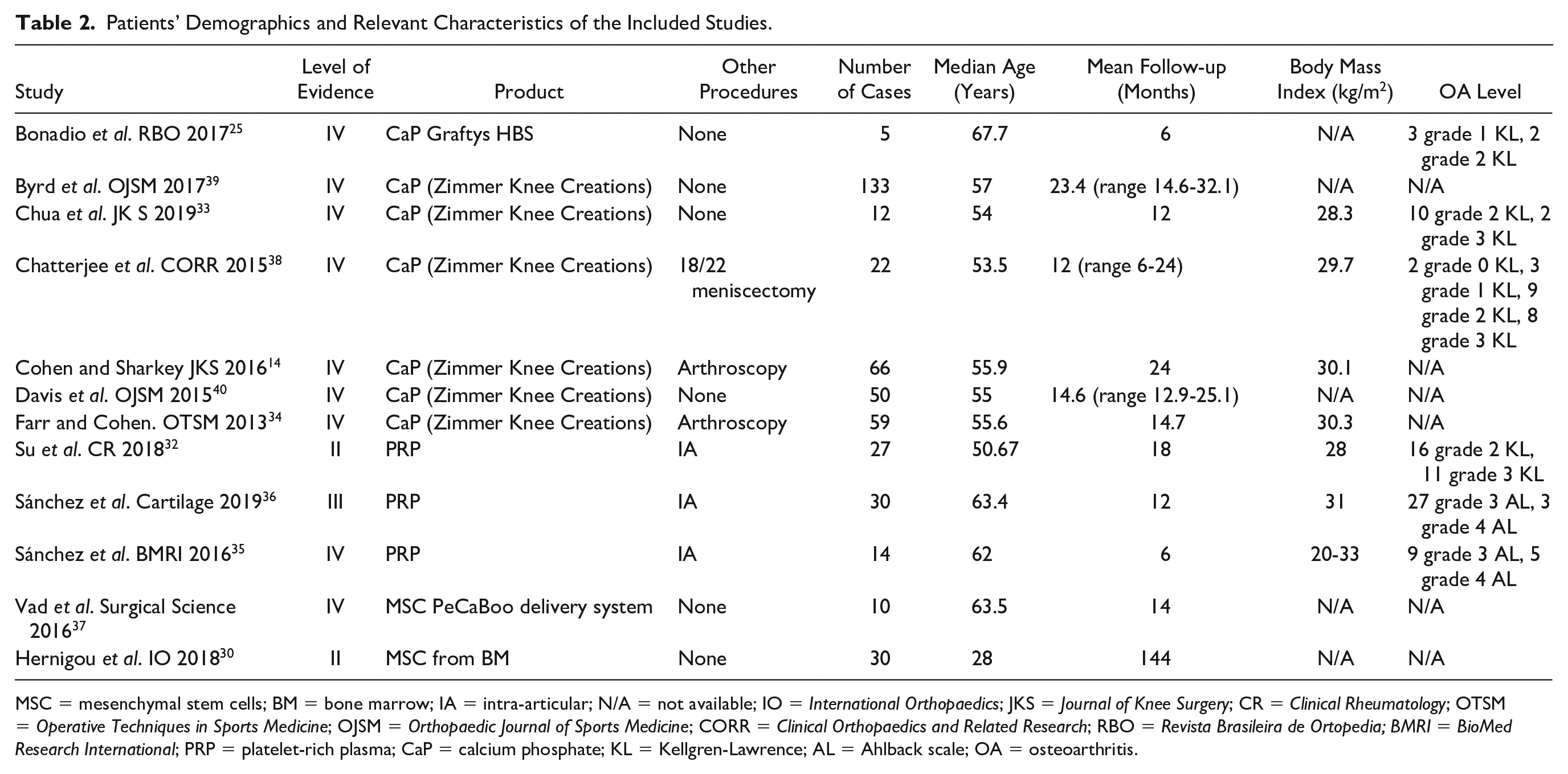
MSC = mesenchymal stem cells; BM = bone marrow; IA = intra-articular; N/A = not available; IO =
Concerning the study design, 2 studies were randomized controlled trials (RCTs),30,32 6 studies were prospective,25,33-37 whereas the remaining 4 were retrospective case series.14,38-40
Bone Substitutes
Six studies ( Table 3 ) investigated the outcome of CaP bone substitute Subchondroplasty (SCP; Zimmer Knee Creations, USA), whereas 1 study adopted the Graftys HBS (Graftys, France). 25 Three studies were prospective case series25,33,34 whereas 4 had a retrospective design.14,38-40
Clinical Outcomes of Studies Dealing with Calcium Phosphate Bone Substitutes.
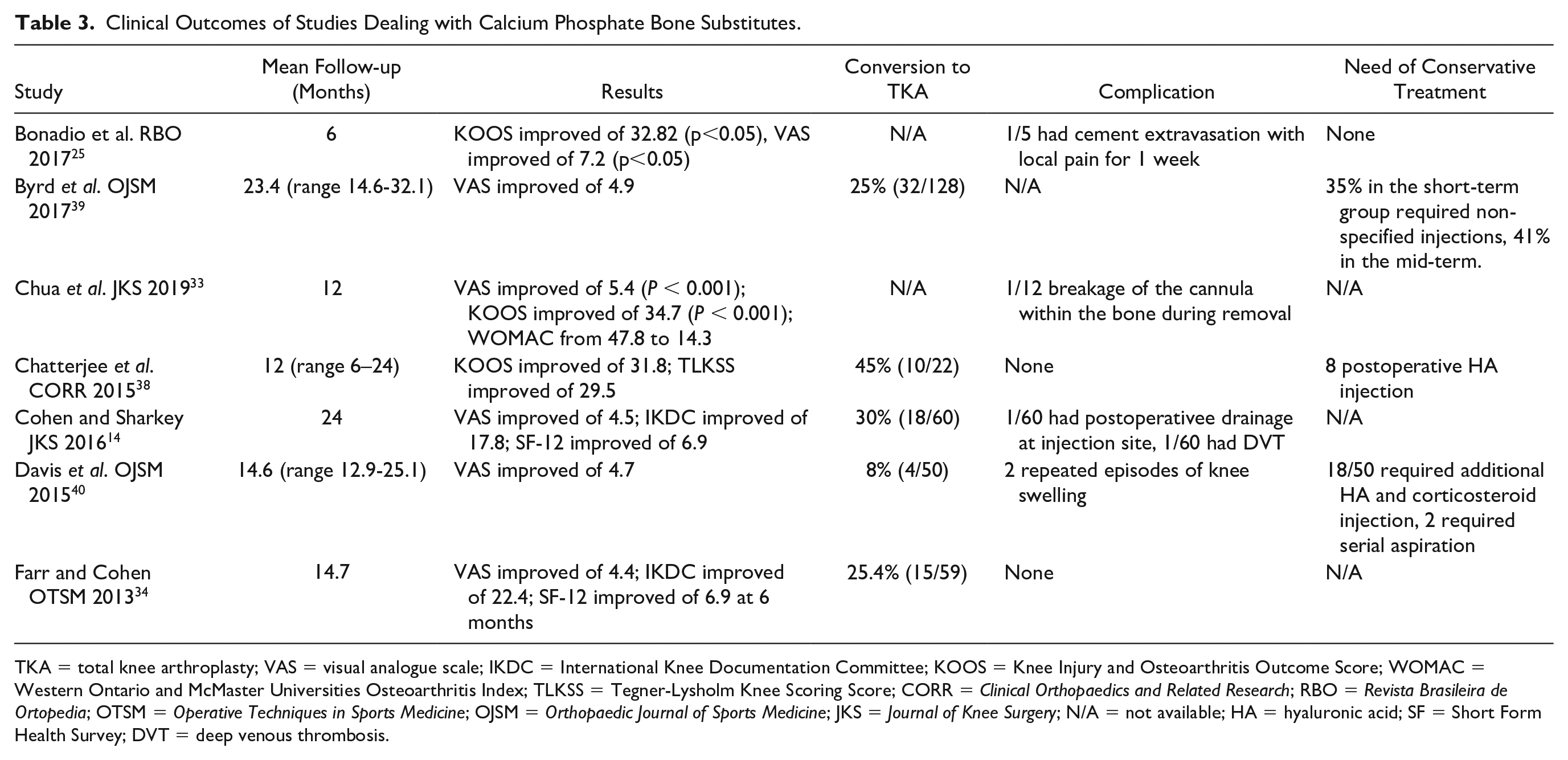
TKA = total knee arthroplasty; VAS = visual analogue scale; IKDC = International Knee Documentation Committee; KOOS = Knee Injury and Osteoarthritis Outcome Score; WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index; TLKSS = Tegner-Lysholm Knee Scoring Score; CORR =
Arthroscopic control to assess the eventual extravasation of CaP into the joint was performed in 4 cases,14,33,34,38 whereas Bonadio
Clinical Findings
A statistically significant improvement in pain and function of the treated knees was reported in all but one trial, as detailed in Table 3 . One article showed poor results, with 10 patients out of 22 (45%) requiring conversion to arthroplasty within 1 year.
Looking at complications, only 6 patients out of 347 experienced adverse events. In one case, there was extra articular cement extravasation, which did not require further surgical treatment and was managed just by analgesic drugs in the postoperative phases. Cohen and Sharkey
14
reported 1 case of postoperative drainage, which required surgical irrigation debridement, and 1 episode of deep venous thrombosis. Davis
With regard to the need of conservative retreatment, Byrd
Looking at the TKA conversion rate, it ranged from 8% at mean 14 months’ follow-up 37 to 45% at mean 12 months’ follow-up. 38 Three studies14,34,39 showed similar percentage of conversion to arthroplasty (25%-30%) at comparable time intervals (approximately 24 months after treatment). In 2 studies, no data on TKA conversion rate are available.25,33
Platelet-Rich Plasma
Only 3 studies included in this review analyzed the results of knee intraosseous leukocyte-poor PRP injections.32,35,36 Sánchez
Su
Clinical Findings
Both the comparative trial by Sánchez
Clinical Outcomes of Studies Dealing with PRP Application.
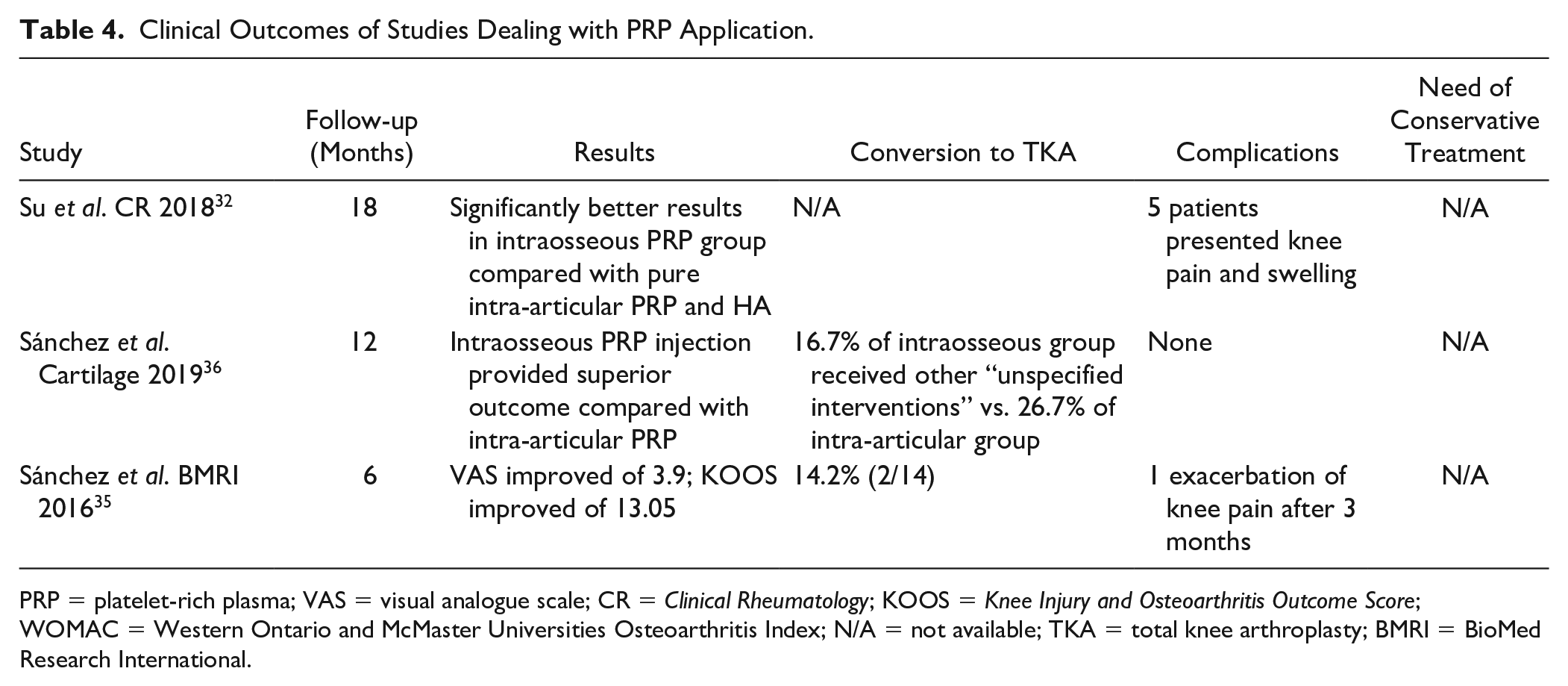
PRP = platelet-rich plasma; VAS = visual analogue scale; CR =
Considering the 3 studies, 5 minor adverse events (knee pain and swelling) were seen by Su
The rate of conversion to TKA has been mentioned by Sánchez
Bone Marrow Concentrate
Only 2 trials investigated the subchondral injection of BMC: one of them is a prospective study, 37 whereas the other is an RCT comparing the outcomes of BMC injection versus TKA on the contralateral knee, in patients with previous long-term, high-dose corticosteroid therapy resulting in bilateral knee OA associated to osteonecrosis. 30
The therapeutic protocols were different: Vad
Clinical and MRI Findings
With regard to the clinical outcomes, both studies reported improvement in patients’ reported scores. In particular, Hernigou
Clinical Outcomes of Studies Dealing with BMC Application.
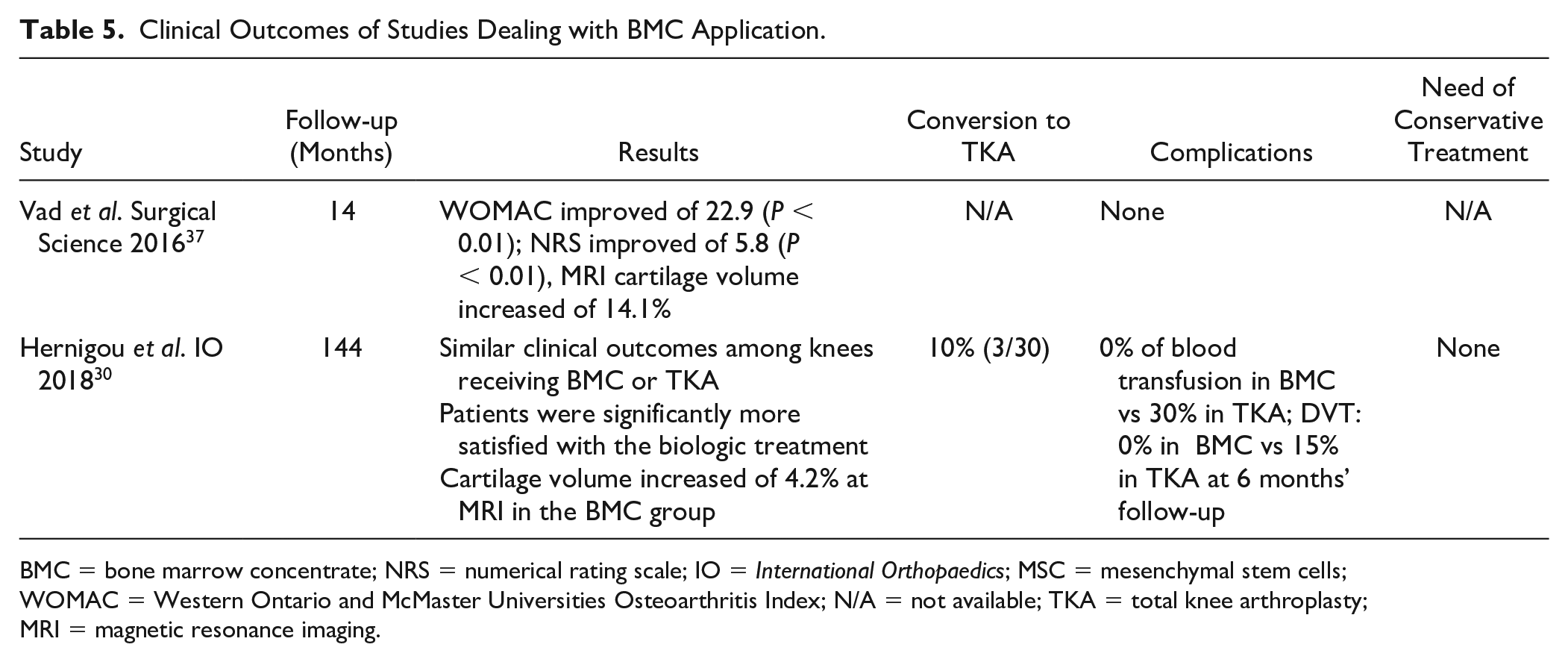
BMC = bone marrow concentrate; NRS = numerical rating scale; IO =
Even in terms of MRI outcomes, results were encouraging: Vad
Vad
Looking at conversion rate, 3 knees that received BMC in the study by Hernigou
Discussion
The main finding of the present systematic review is the lack of high-quality clinical evidence concerning the efficacy of knee intraosseous injection, due to the paucity of RCTs and the modest overall methodology, often related to the limited number of patients included, their heterogeneity in terms of OA grade (from initial to severe OA), and the short-term evaluation, as testified by the low average scores assessed through the Coleman methodology modified by Kon
Knee OA is a multifaceted disease, which involves all the intra- and extra-articular structures, thus representing a challenge in terms of treatment due to the impossibility of targeting all the tissues involved. Actually, under the name of OA there are different pathological entities characterized by different clinical presentations, ranging from inflammatory patterns to more mechanical features.41,42 There are several etiopathogenetic pathways currently investigated,43,44 and especially in recent years there has been a peculiar recognition of the role of subchondral bone: In fact it has been shown that patients presenting the so-called BMLs at MRI are usually more symptomatic and tend to have worse prognosis.16,17 Therefore, intraosseous injections were introduced with the aim of treating the pathologic subchondral bone tissue. The biologic rationale was to stimulate subchondral bone remodeling and healing in order to reduce symptoms and postpone more aggressive surgical procedures. The primary role of the subchondral treatment is therefore to delay joint replacement, especially in patients too young or in those affected by severe comorbidities that could increase the risk of postoperative complications.45,46
There are 2 different options for knee subchondral treatment: one is the “subchondroplasty” approach, consisting in the use of CaP bone substitutes, whereas the other is the “biologic” solution, which consists in the use of platelet-derived growth factors or concentrated MSCs, both of which demonstrated potential in promoting healing of different musculoskeletal tissues.47,48 The goal is to normalize the subchondral bone environment, although with very different mechanisms: in fact, the use of CaP ceramics, beyond promoting bone mineralization over time, is able to provide also immediate mechanical support in the subchondral region due to the physicochemical properties of these biomimetic ceramics: this could reduce micro-motion in the subchondral trabeculae thus accounting for a reduction in pain perceived by patients. 34 Differently, stimulation through growth factors or cell-based approaches activates different healing pathways in the subchondral bone, without immediate effects on the mechanical stability of the subchondral tissue. 23
The use of CaP is currently more represented in literature (7 studies vs 3 for PRP vs 2 for BMC), but no comparative analysis has yet been performed, and further randomized trials are necessary to understand whether a specific substance is able to provide better or longer lasting results. Furthermore, no clear indication can be drawn on the amount of substance that must be injected in the pathologic subchondral area, and whether there is any correlation with the size of BMLs observed at MRI.
Under a clinical point of view, the first relevant aspect coming from the analysis of literature is the overall safety of knee subchondral injections, which has been confirmed by all the trials available. Beyond the risks linked to surgical procedures in general, the most common complications are knee swelling and postoperative pain, which can be easily managed. The only real concern is the intra-articular leakage of the product when using CaP bone substitutes, which is more likely to happen in case of osteoporotic patients or when the cannula is placed too close to the articular surface. Overall, beneficial effects on symptoms and functional recovery have been documented at short term, with the majority of trials having performed evaluation in the range of 12 to 24 months after treatment. With regard to the survival rate, 1 trial on subchondroplasty
38
reported frankly disappointing results, with 45% of patients converted to TKA at short term, whereas the majority of the other subchondroplasty studies revealed a failure rate in the range of 25% to 30% up to 2 years, even if a significant amount of patients needing occasional retreatment (intra-articular injections of HA or corticosteroids) has been reported.39,40 Given the minimally invasive nature of knee intraosseous injections, a yearly failure rate of 10% to 15% could be still acceptable, provided that patients are clearly informed on the outcomes to expect following such treatment. At present moment, the only long-term evaluation has been performed by Hernigou
Beyond differences in the mechanism of action, there are also “practical” differences which might have an impact on surgical choices: in fact, CaP bone substitutes are an “on-the shelf” products always available in the operating room, whereas autologous biologic products requires to be harvested, immediately prepared and then applied with inherent longer surgical time. Moreover, the use of biologic products, in particular MSCs, is often strictly regulated by health authorities, 49 and this led to the development of “minimal manipulation” strategies, 50 which allows immediate processing of MSCs in the operating room.
Looking at surgical technique, knee intraosseous injections are usually performed under fluoroscopic control, which could be a limiting factor since the optimal placement of the cannula within the BML is not guaranteed through fluoroscopy: the exact assessment of the extension and location of the lesion is done only by MRI and, therefore, X-ray guidance can results in a wrong positioning of the injecting device, especially in relatively small BMLs. To this purpose, novel software have been developed that are able to use preoperative MRI to calculate the optimal placement of the cannula, which can be controlled by intraoperative computed tomography.51,52 Alternatively, patient-specific MRI-based pointing devices could be realized to allow correct placement. 53 The use of these technologies is still not routine and should be evaluated against the obvious increase in costs.
Concerning the association of knee arthroscopy, it is recommended only when using CaP bone substitutes (subchondroplasty), to assess eventual intra-articular extravasation of the product, which could cause persistent joint inflammation. Arthroscopic approach also allows concurrent treatments such as cartilage procedures, articular lavage, or meniscal treatment, which could play a role in determining the outcome. On the other hand, arthroscopy may have a negative impact on the joint environment and increase the risk of infection: therefore, arthroscopy is not suggested when using biologic products, also because PRP and BMC have the advantage of being injected both intraosseously and intra-articularly, so arthroscopic fluid could dilute them and reduce their potential. On this specific aspect, it is relevant to note that 2 studies on PRP proved that subchondral injections significantly improve the outcomes compared with pure intra-articular delivery, thus confirming the necessity of addressing specifically the subchondral bone damage.32,36,54
All the aforementioned encouraging findings should be always carefully considered in the light of the many limitations of the current literature, in particular the paucity of studies available, mostly case series, the small number of patients included (just 4 trials had more than 50 patients), the short follow-up, and the lack of serial MRI evaluations to understand the healing potential associated to subchondral treatment. Anyway, the most relevant flaw emerged is the lack of randomized trials comparing knee intraosseous injections with subchondral “decompression” alone. Mechanical stimulation through a single or multiple subchondral perforations has been originally employed for the treatment of femoral head osteonecrosis with the aim of reducing intraosseous pressure, restore normal vascular flow and ultimately promote subchondral bone remodeling.55,56 Therefore, knee subchondral perforation, which always takes place before injecting any substance, has a beneficial effect
Beyond the limitations of the literature, we need also to consider some potential barriers to the spread of subchondral treatment in routine practice: in particular, the high costs of the products and the surgical kits necessary to perform the procedure, also considering the lack of studies investigating the cost-effectiveness of this novel surgical approach. This is particularly true when it comes to biologic products, a field where there is a large inter-product variability and standardization is far from being reached.57-59
To conclude, knee intraosseous injections can be regarded as a minimally invasive and safe procedure to address subchondral bone damage, with the goal of delaying joint replacement or offering a salvage option to those not eligible for arthroplasty. Current evidence does not allow to identify the superiority of a specific product over the others; looking at the data on the rate of conversion to arthroplasty (25%-30% on average up to two years’ evaluation), patient counselling is fundamental to avoid unrealistic expectations after subchondral procedures, and further studies are needed to answer fundamental questions: the first one is whether subchondral injections (of any substance) in the knee provide better outcome than subchondral decompression alone. Then, it should be elucidated if any difference exists among the various products available in terms of durability of results and, last, the best therapeutic protocols to apply.
Footnotes
Authors’ Note
This investigation was performed at the Humanitas Clinical and Research Center, IRCCS, Milan, Italy.
Acknowledgment and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
