Abstract
Objective
This proof-of-concept study investigated an improved cell-based injection therapy combining mesenchymal stem cells (MSCs) and meniscus cells (MCs) to support superior meniscus allograft repopulation and early revival compared to injecting MSCs alone.
Design
In this controlled laboratory study, frozen meniscus allograft samples were injected vertically with a cell suspension containing different ratios of MSCs and MCs or control (lactated ringers) and cultured for 28 days. Samples were analyzed weekly for cell viability, migration, and metabolism using histological and biochemical assays. Tissue medium was analyzed for matrix metalloproteinase (MMP) expression using zymography.
Results
Cellular repopulation of frozen allografts injected with different cell suspensions was validated by immunohistochemistry. Significant higher DNA content was evidenced in grafts treated with suspensions of MCs or MC:MSC (1:4 ratio). Cell metabolic activity was significantly different between all treated groups and control group after 1 week. Allografts injected with MCs showed significantly more cell proliferation than injections with MSCs. MMP2 activity was detected in medium of all grafts cellularized with MCs with or without MSCs. Scanning electron microscopy (SEM) analysis showed resolution of the needle puncture, but not in the control group. Cell labeling of MCs upon injection of mixed MC:MSC suspensions revealed a gradual increase in the cell ratio.
Conclusions
The findings of this study establish that injection of MCs with or without MSCs enhances the cellularity of meniscus allograft to support early graft revival and remodeling.
Introduction
When the meniscus is damaged and symptomatic, repair or substitution of the tissue is a priority to improve symptoms and possibly prevent deterioration of the underlying cartilage. 1 Meniscus allograft transplantation (MAT) represents an established treatment option for symptomatic patients without advanced osteoarthritis to restore biomechanical functioning of the joint. 2 Several preservation methods exist for meniscus allografts. Fresh meniscus allografts, which contain viable cells, are not often used, because they are associated with high costs and limited availability.3,4 Therefore, frozen allografts have become the most prevalent due to lower costs, logistical benefits, and extended storage times that increase availability. 3 However, preservation by freezing has drawbacks, including the loss of cell viability (due to cell membrane fragility) within a tissue characterized by limited native cellularity. The lack of viable cells within the allograft may adversely affect graft performance and is associated with the common complications of shrinkage, extrusion, and tearing of the graft. 5
Graft repopulation by spontaneous host cell influx may promote tissue incorporation, graft survival, and clinical performance, but this putative mechanism does not translate to frozen MAT.6,7 Several groups have demonstrated limited cellular repopulation of frozen allografts, with influx of viable cells limited to superficial layers.8-10 These studies emphasize that the high-density collagen fiber network of the meniscus poses a formidable barrier for cell migration. However, the presence of viable cells, and subsequent extracellular matrix (ECM) remodeling, could improve cellular repopulation. The importance of initial meniscus allograft viability to graft outcome and durability remains unclear. However, in the field of cartilage restoration surgery, chondrocyte viability within a graft upon transplantation is associated with superior performance and clinical outcomes.11,12 Therefore, a viable cell population contributing to early remodeling and biological incorporation of meniscus grafts may facilitate favorable outcomes, patient rehabilitation, and return to sport and activities of daily living.2,13
There is a large clinical need for an improved meniscus allograft that maintains the logistical benefits of frozen allografts while improving graft properties and performance. A cell-based treatment of the frozen allograft, pre-transplantation may facilitate graft viability, early remodeling, and biological incorporation. In a previous proof-of-concept study, we examined the feasibility of injecting mesenchymal stromal/stem cell (MSC) suspensions in meniscus allografts and established cell survival up to 28 days of incubation. 14 Treating a frozen meniscus allograft with this cell-based injection therapy would maintain high availability of allografts and include viable cells working to maintain meniscus ultrastructure and subsequent biomechanical properties.
Previous in vitro studies and a human clinical trial for cartilage defects have firmly established the principle of improving the regenerative capacity of allografts with cell-based injections that combine native cells with mesenchymal stromal cells (MSCs).15,16 In the current study, we addressed the hypothesis that cellular treatment of the allograft with a cell suspension containing native meniscus cells (MCs) with or without MSCs will lead to superior graft revival and early remodeling in comparison to treatment with MSCs alone.
Methods
Donor Grafts, Cell Isolation, and Culture Conditions
Low passage human MSCs were acquired through the Immune Progenitor and Cell Therapy (IMPACT) laboratory at Mayo Clinic (Rochester, MN). Cells from a single female adult donor (age: 42 years) were obtained during liposuction. For all experiments, MSCs were expanded in T175 flasks with advanced MEM (Gibco, Thermofisher Scientific, Waltham, MA) supplemented with commercial stabilized human platelet lysate product (PLTMax, Mill Creek Life Sciences, Rochester, MN), 1% Penicillin/Streptomycin (Pen/Strep, Gibco, Thermofisher Scientific, Waltham, MA),
Cell Injections and Culture
Co-culture experiments
For all experiments, early passage MCs and MSCs were suspended in lactated ringers containing cell ratios 100% MSC, 80:20% MSC:MC, or 100% MCs. Frozen allografts were brought to room temperature and cut into equal pieces of approximately 1 cm2. All samples were treated with vertical injections of 50 µL cell solution (0.7 × 10E6 cells) of one of the cell ratios each accompanied by a mock-injected control sample using a 20-G needle (
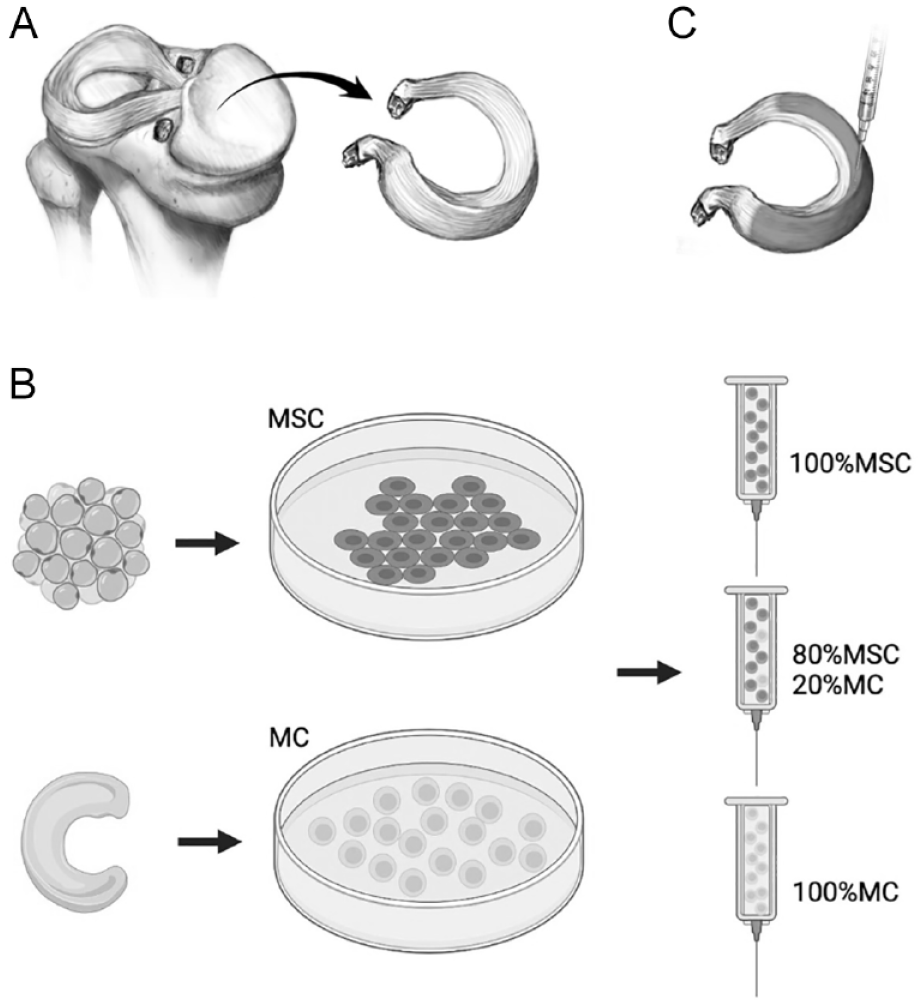
(
Co-culture cell labeling
Cell-cell interactions and presence of injected MCs and MSCs at different time points were studied through cell labeling. MSCs were labeled with a red fluorescent membrane dye (PKH26-red, Sigma-Aldrich) pre-injection according to the manufacturer’s protocol and MCs were labeled with mCherry through lentivirus infection. To study cell-cell interactions, labeled MSCs (80%) were suspended with plain MCs (20%) and samples were stained for connexin-43 (Gap Junction Protein Alpha 1; GJA1). Cell residence over 28 days of culture was investigated using labeled MCs and plain MSCs. At days 0, 4, 7, 14, and 28, samples (n = 24) were collected, embedded in Cryomatrix Embedding Resin (Thermo Fisher Scientific, Waltham, MA), processed with liquid nitrogen snap-freezing, and sliced using a cryostat (Leica, CM3050S-3-1-1) at 10 µm thickness.
Histology, Immunohistochemistry, and Microscopic Imaging
Histology and confocal immunofluorescence microscopy
Samples were dehydrated and clarified using stepwise dehydration, embedded in paraffin, sectioned at 10 µm using a microtome, and stained with 0.125% Safranin-O (Sigma-Aldrich, counterstained with Weigert’s hematoxylin (Sigma-Aldrich), and 0.02% fast green (Sigma-Aldrich)). Connexin 43 (GJA1) expression was determined by confocal IF microscopy. Sections were blocked using 5% bovine serum albumin (BSA) in PBS for 1 hour followed by an overnight incubation (4°C) with a primary antibody against connexin 43 (rabbit anti-connexin 43, ab66151, 1/50; Abcam) followed by secondary antibody Alexa Fluor 647 (A-21245, Thermo Fisher Scientific) and nuclear counterstain Hoechst 33342 (Thermo Fisher Scientific).
Cell viability and distribution was analyzed using LIVE/DEAD assay for mammalian cells consisting of calcein AM and ethidium homodimer-1 (Thermo Fisher Scientific) as described earlier. 14 For the co-culture cell labeling experiments, samples were submerged with a working solution of Calcein-AM only in PBS to differentiate living cells (total number of living cells in green) from labeled cells (MCs or MSCs—green and red). All (immuno)fluorescence imaging was performed by confocal microscopy using standard filters for blue green and red channels.
Scanning electron microscopy
On day 28, samples were collected and processed for scanning electron microscopy (SEM). Samples (n = 6) were transferred to Trumps fixative at 4°C, washed in phosphate buffer, rinsed in water, and dehydrated through a graded series of ethanols. Subsequently, samples were critical point dried using carbon dioxide, mounted on an aluminum stub, and sputter-coated with gold-palladium. Finally, samples were imaged in a Hitachi S-4700 cold field emission scanning electron microscope (Hitachi High Technologies America, Inc., IL) at 5 kV (meniscus) accelerating voltage. The preparation and the imaging of the SEM samples were performed by the Microscopy and Cell analysis core laboratory of the Mayo Clinic.
Biochemical Analysis
DMMB and PicoGreen DsDNA assay
On days 0, 4, 7, 14, and 28, the PicoGreen dsDNA assay (Invitrogen) and DMMB assay (Chondrex, Woodinville, WA) were performed according to the manufacturer’s protocol after overnight digestions at 60°C in 1.5 mL papain enzyme solution consisting of 5 mM
MTS assay
Treated grafts were harvested at days 0, 1, 2, 3, and 7 and subjected to (3-(4,5-dimethylthiazol-2yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium) (MTS) assays (Promega Corporation, WI) to analyze cell metabolic activity. The assays were carried out according to the manufacturer’s protocol. For each treated sample, a positive control (living meniscus tissue) and negative control (mock-injected allograft sample) was included for reference. MTS assays were analyzed using a SpectraMax Plus Plate Reader (Molecular Devices, Sunnyvale, CA) at an absorbance wavelength of 490 nm.
MMP zymography
A detergent-compatible (DC) protein assay (BioRad, Hercules, CA) was conducted on the conditioned medium using a BSA ladder supplemented with media without platelet lysate to determine protein concentration. A volume of conditioned media associated with 10 µg of protein was added to 5 µL Tris-Glycine SDS Sample Buffer (2x) with sufficient deionized water to reach a total volume of 10 µL. Samples were electrophoresed into a 10% acrylamide gelatin gel (BioRad) at 125 V for 60 minutes. Zymography gels were developed and treated with renaturation buffer, prior to staining with Colloidal Blue staining kit according to the manufacturer’s protocol (Invitrogen). Stained matrix metalloproteinase (MMP) bands were then visualized using a ChemiDoc Touch Imaging System (BioRad).
Statistical Analysis
Data were analyzed using GraphPad Prism version 9 (GraphPad Software, San Diego, CA) and presented as the mean ± standard deviation (SD). DsDNA content was compared between mock-injected (control) group and all other groups using Welch ANOVA with Dunnett’s T3 correction post hoc for multiple comparisons because variances were not equal. Similarly, cell metabolic activity was compared between the mock-injected control group and different treatment groups using Welch ANOVA with Dunnett’s T3 correction. Differences were considered statistically significant when P < 0.05 and indicated by a single asterisk (*). Cell labeling and (immunofluorescent) histological results are provided as a qualitative illustration of the findings and are therefore a descriptive assessment of the data.
Results
Primary meniscal cells, containing both fibroblast-like cells and more spherical fibrochondrocytes, were characterized by a panel of RT-qPCR and appear to express fibroblast markers (COL1A1 and MMP13) and not cartilage markers (COL2A1, ACAN, and COL10A1) (Suppl. Fig. S1).
Allografts Treated With Various Cell Ratios
Small allograft specimen treated with monocultures of MSCs (group 1) and MCs (group 3) and combined injections of 80% MSC and 20% MCs (group 2) were compared with vehicle-injected allograft specimen (lactated ringers; group 4) with n = 15 samples per group.
Cellular repopulation is enhanced in treated allografts
Histological and immunofluorescence (IF) analyses allowed qualitative assessment of cell presence within meniscus allograft samples. While injected cells are initially clustered together around the injection canal, histological analysis at day 7 revealed several separated cells surrounded by loose matrix indicative of dynamic matrix processes resulting in cell migration in alignment with collagen fibers (
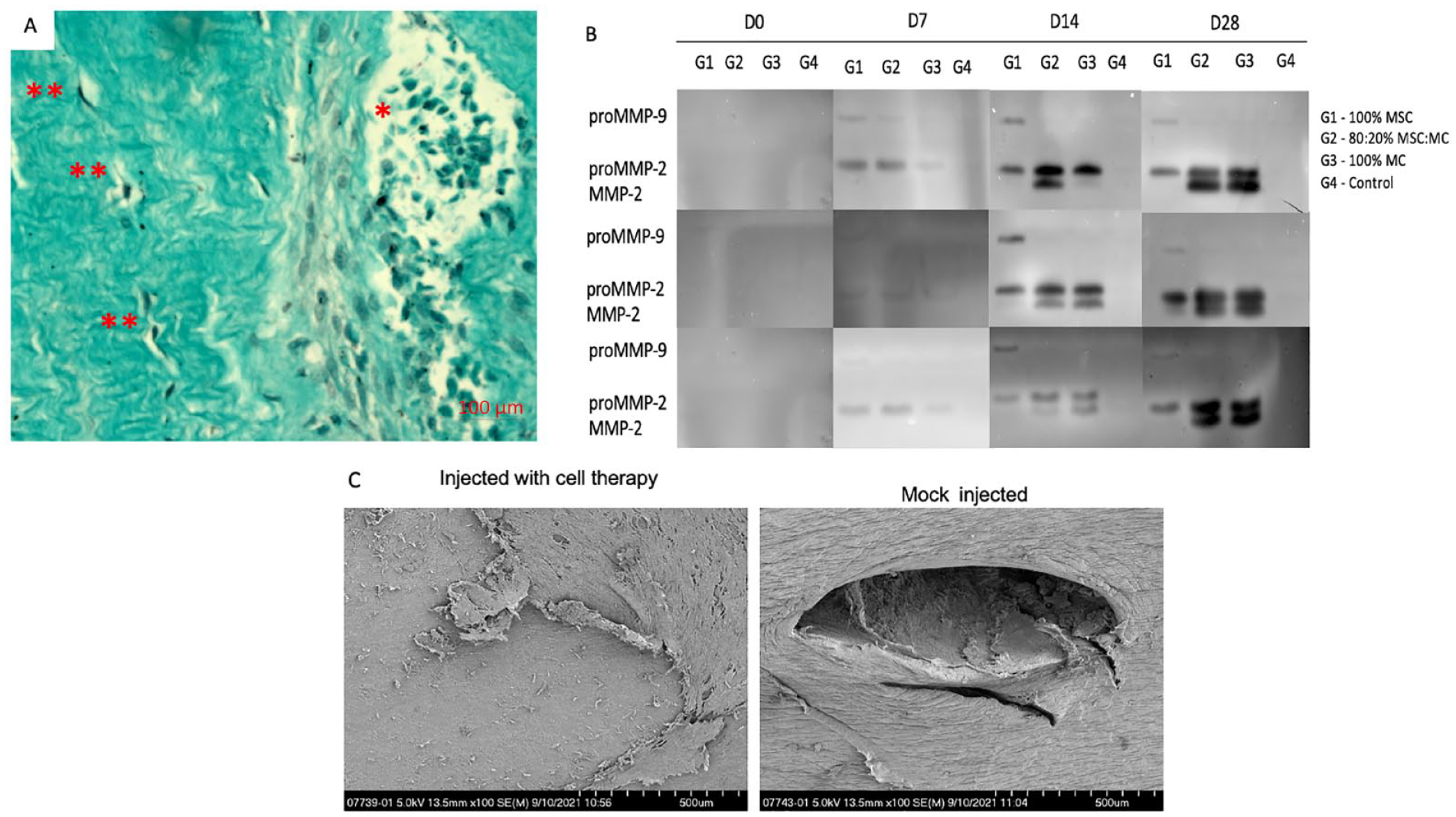
Matrix synthesis, deposition, and remodeling. (
(
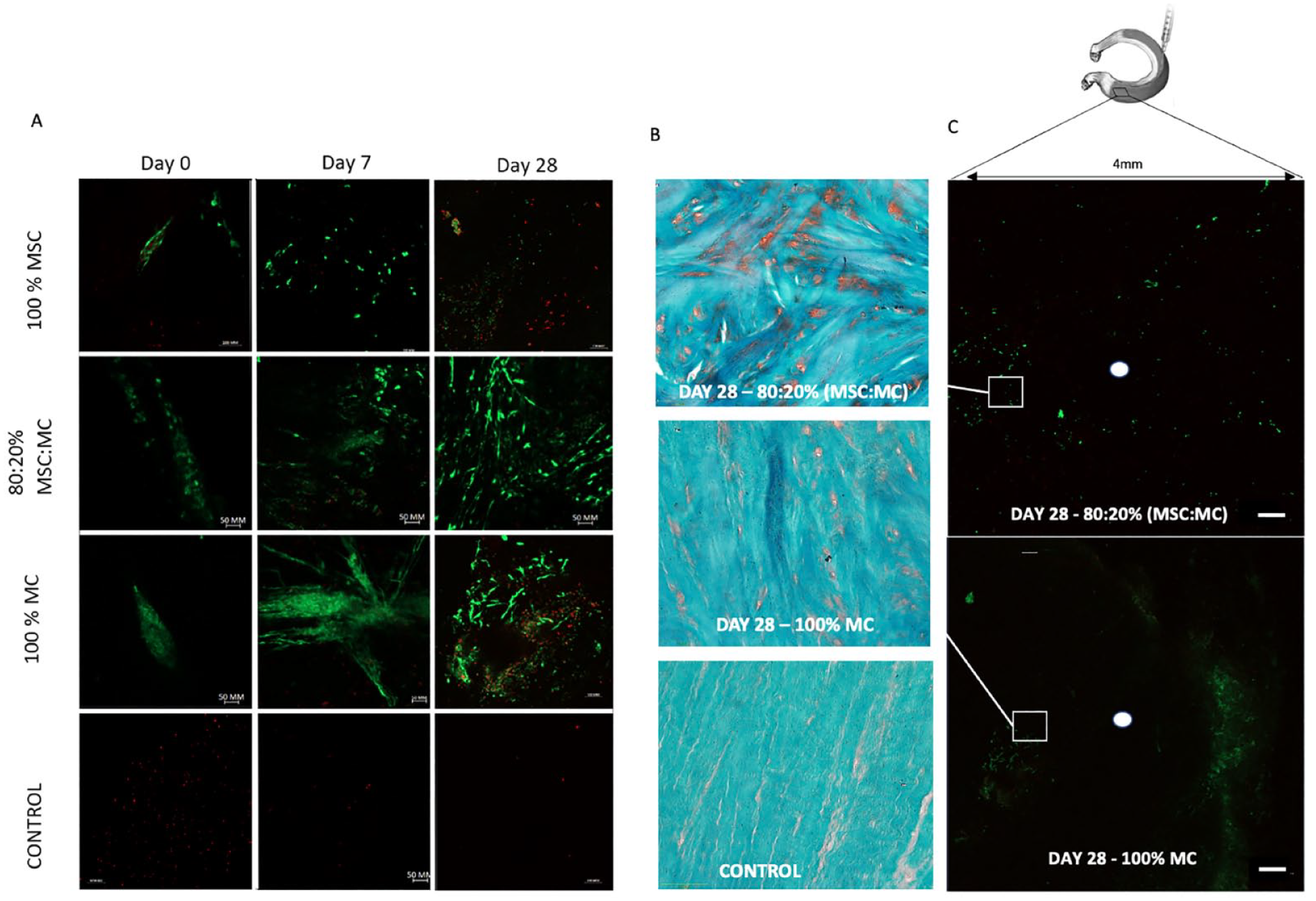
All treatment groups showed viable cells up to day 28, except for mock-injected samples where live cells were absent. MCs adapt an elongated fibroblast-like morphology after 4 days of residing in the graft, while other fibroblast-like spindle morphologies became evident for MSCs in grafts treated with either MSCs or MSC:MC mixtures (80%:20%) (
(
Quantification of DNA within all meniscus allografts corroborated the retention of injected MCs in the meniscal allograft. After 2 weeks of culture and beyond, allografts treated with monocultures of native meniscal cells and co-cultures of MCs and MSCs show a significant higher presence of DNA in the allografts compared to their respective control samples (Day 14: 5.44 ± 0.31, P = 0.0043, and 4.35 ± 0.43, P = 0.0054, vs. 1.34 ± 0.60. Day 28: 5.16 ± 0.69, P = 0.0058, and 4.92 ± 0.73, P = 0.0075 vs. 0.57 ± 0.46, respectively). Moreover, grafts treated with MSCs show no significant difference in DNA content with mock-injected control samples (
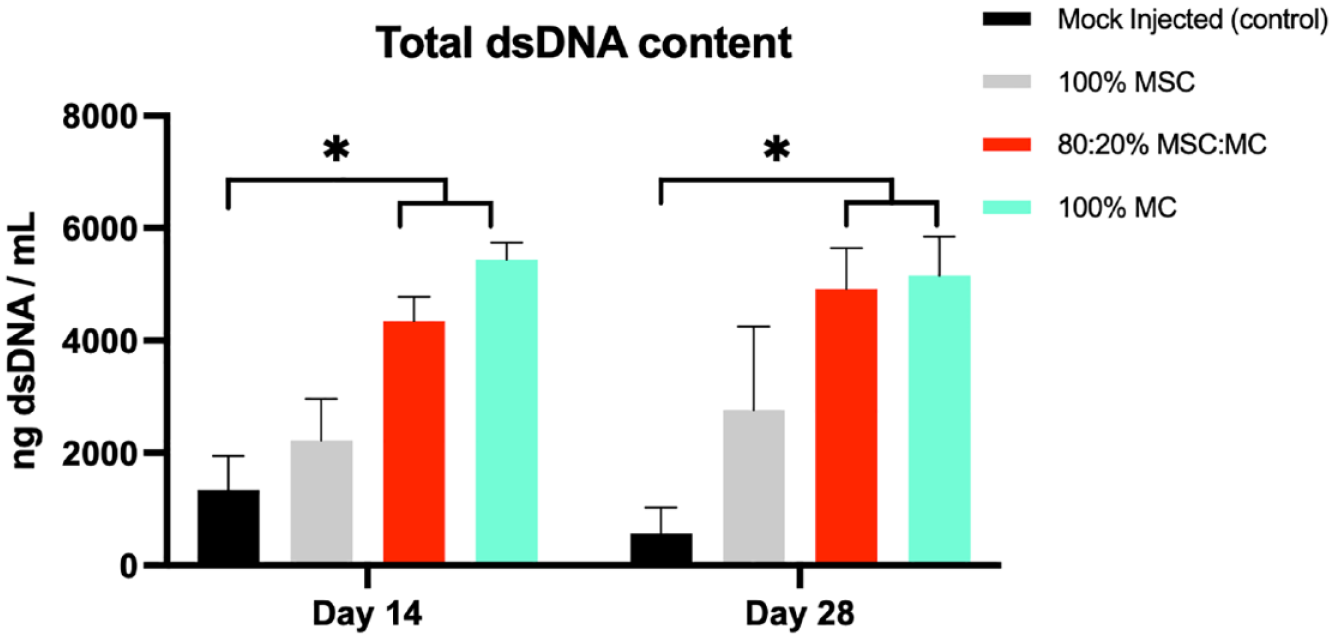
PicoGreen dsDNA assay analysis of allografts treated with different groups after 14 and 28 days of culture. Groups treated with 80:20% MSC:MCs and 100% MCs have a significant higher total dsDNA content in comparison to mock-injected (control) samples. (80:20% MSC:MCs. Day 14: P = 0.0054, Day 28: P = 0.0075 and 100% MCs. Day 14: P = 0.0043, Day 28: P = 0.0058). Grafts treated with monocultures of MSCs were not significantly different from control group. (Day 14: P = 0.4020, Day 28: P = 0.2691). MSC = mesenchymal stem cells; MC = meniscus cells. *P < 0.05.
Cell viability and proliferation was validated using MTS assay with both quantification (
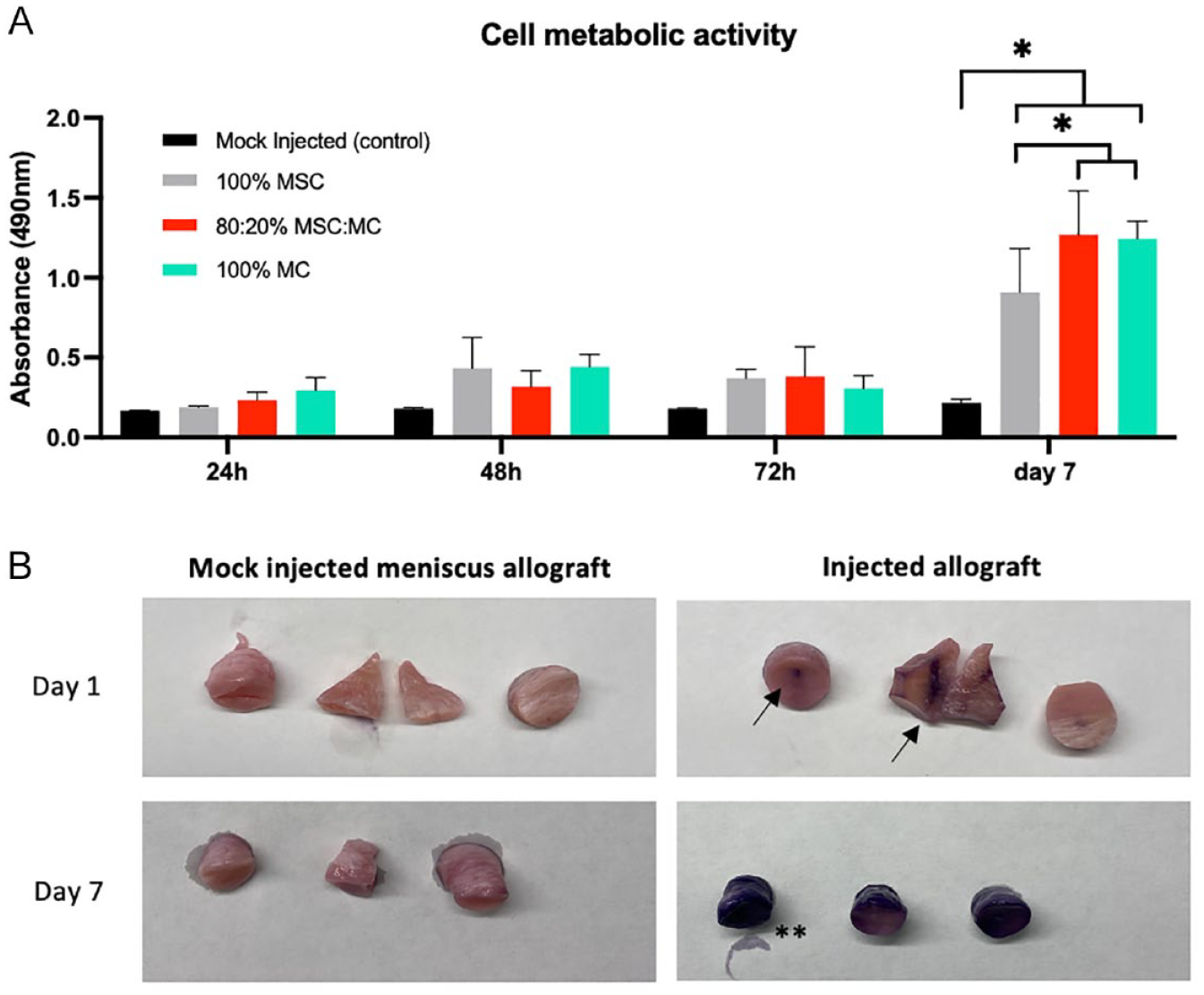
Cell metabolic activity using 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium (MTS) proliferation assay. (
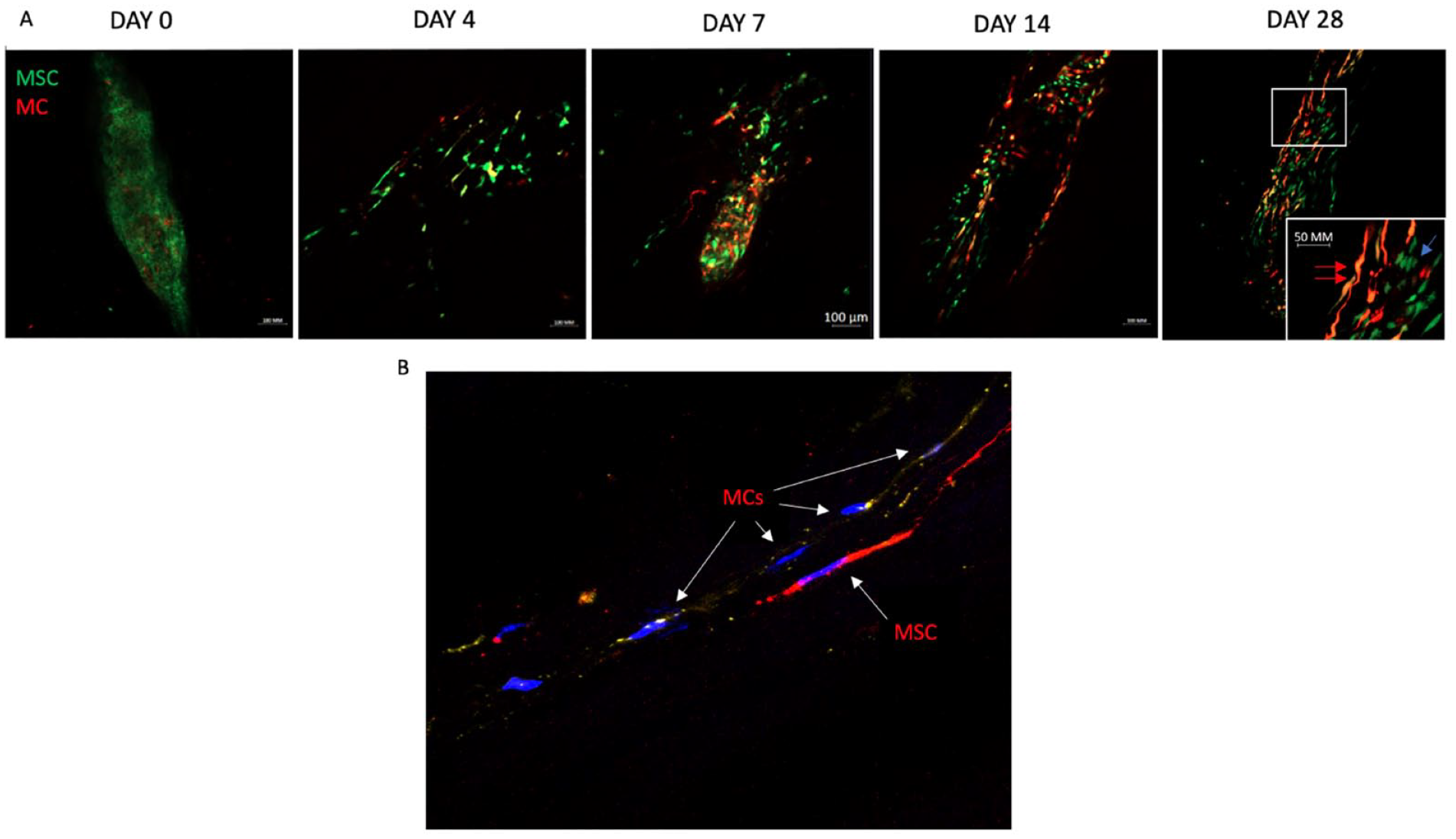
Cell retention and cell-cell interaction
Both MSCs and MCs in monoculture were labeled successfully with cell membrane fluorescent dye PKH26 and mCherry, respectively. Labeled cells retained their fluorescent dye after 28 days in culture. Comparing allograft samples treated with plain MSCs and labeled MCs (group 2) on different time points revealed a gradual decrease in the MSC:MC ratio (
Matrix synthesis, deposition, and remodeling in cellularized meniscus allografts
The presence of viable MCs in the allograft may permit interactions with and remodeling of the meniscal ECM. Therefore, we examined whether cell-injected allografts express enzymes capable of modifying the matrix. Competency for matrix degradation and remodeling was measured by gelatin gel zymography using media that was conditioned by incubation of allografts. Allografts do not exhibit zymography bands on day 0 while proMMP2 bands are evident on day 7 for all treatment groups with levels of these bands increasing significantly at later time points, especially for grafts treated with 100% native MCs and the 80%:20% MSC:MC mixtures. These same groups (group 2 and 3) showed a band on day 14 for MMP2, and this MMP detection increased on day 28. Grafts treated only with MSCs showed a band for proMMP9 on day 7 and day 14 which became weaker on day 28. No bands were detected for media derived from mock-injected samples (
After 28 days of culture, SEM demonstrated remnants of the injection hole in mock-injected allografts (
Discussion
This study pioneers the recellularization of essentially acellular frozen meniscal allografts by injection of MSCs and/or native MCs, inspired by our earlier proof-of-concept study in which meniscus allografts were injected with MSCs. 14 In this study, we firmly establish that it is feasible to repopulate a frozen meniscus allograft using a cell-based injection therapy with native MCs, potentially using MSCs as an auxiliary cell type. Moreover, the findings of this study validate our hypothesis that injecting MCs or a combination of MCs with MSCs show superior results in terms of early graft revival and remodeling of decellularized meniscus allografts.
Confidence in our findings is based on a combination of quantitative biochemical data on cell metabolism, dsDNA content, and dynamic matrix remodeling as well as (immuno)histological data illustrating migration and repopulation patterns up to 28 days of culture.
First, cell proliferation after 7 days of culture was significantly higher in allografts treated with a cellular suspension containing native MCs (mono or combination therapy) in comparison to grafts treated with monocultures of MSCs. Likewise, dsDNA content after 14 days and 28 days of culture was significantly higher in grafts treated with native MCs in comparison to mock-injected allografts while this was not the case for grafts treated with MSCs only. Moreover, supernatant media from grafts treated with native MCs contained increasing levels of proMMP2 and MMP2 expression, starting at day 7 and most pronounced at day 28. This indicates comparable dynamic matrix remodeling in grafts treated with native MCs in mono- or combination therapy. Grafts treated only with MSCs lack MMP2 expression, an activated MMP, at all time points, while proMMP9 bands, an indicator for inactive MMPs, remain present at all time points, indicating that MSCs-ECM interactions did not result in activation of matrix remodeling MMPs. 22 Confocal IF microscopy, fluorescence microscopy, and histological data using live/dead stain and cell labeling corroborate these findings by showing that cellular migration patterns were more apparent at earlier time points (day 7) and more extensive at day 28 in groups treated with native MCs (single cell type infusion or combination cell therapy). One interpretation of this finding is that MCs are programmed to function in dense collagen networks and adapt to a fibroblast-like phenotype when residing in such environment. The latter would translate to earlier migration and generally more cellular repopulation in comparison to grafts treated with MSCs only.
Furthermore, MC-MSC interactions were analyzed through cell labeling experiments and positive immunofluorescent staining for connexin 43 (GJA1), indicating information exchange through gap junctions (
Overall, our findings demonstrate that native MCs in monotherapy or combined with MSCs represent a viable strategy for recellularization of frozen meniscus allografts to improve their surgical utility in joint repair. We propose that the effectiveness of MCs may be attributable to the apparent intrinsic ability of these cells to support meniscal repair, as was evident in the repair of a needle-sized injury. While MCs are an ideal cell type, translating this cellular injection therapy to the clinic requires a sufficient number of cells to be obtained from recycled meniscus tissue, which is often limited within patients with symptomatic meniscus deficiency. This limitation could potentially be overcome using a combined cell injection therapy of MSCs and MCs, because the combination of MSC and MCs reduces the number of necessary MCs, in agreement with similar strategies developed in previous studies for native-like meniscus tissue production and cartilage defect filling.15,16,27
Several studies have reported on decellularization and recellularization strategies for meniscus repair or replacement.28-31 One prior study used a similar approach as our study by seeding cells into a meniscus allograft using a manual needle injection technique. 32 This study by Maier and colleagues showed that injected MCs into chemically decellularized allografts remain viable for more than 4 weeks as a viable cellularized meniscus construct. 32 Similar to our findings, viable cells were detected through all areas of complete scaffold cross-sections, while seeded fibrochondrocytes showed a typical configuration of elongated fibroblast-like cells. 32 Our study shows results comparable to this earlier study, while extending the analysis by inclusion of data on cell behavior and ECM production. Another study examined cell suspensions of human primary chondrocytes that were seeded on the surface of decellularized meniscus scaffolds. These allograft constructs were cultivated up to 28 days and exhibited ECM deposition of newly synthesized proteins. These findings correspond to our SEM observations and gelatin gel zymography indicative for dynamic matrix remodeling and ECM production. 33
The current study identified several limitations that require resolution in the future to translate this injection therapy to clinical applications. This work represents an in vitro study which does not allow us to conclude that this therapy should work in a complex postsurgical in vivo environment. Further validation, such as mechanical testing and introduction of a synovial fluid-like environment, still needs to be performed to support the clinical application of this therapy. However, our group conducted a robotic mechanical study on injected meniscus grafts and did not show a higher rate of tears in these grafts compared with the control group, suggesting that the meniscus tensile properties were not altered due to the injections (unpublished observations). Another study investigated the mechanical properties of a needle punched meniscus (28G, 1-mm2 spacing) and demonstrated no altered static and dynamic compressive material properties relative to their control meniscus samples while their material property testing results were consistent with results reported in other studies. 9 Needle passage has negligible effects on the molecular and cellular properties of MSCs and is not predicted to affect MCs. 34 SEM of injected allografts also did not show remnants of needle punctures among all treatment groups indicating ex vivo remodeling and repair of the defects. Furthermore, the MC populations used in this study are derived from patients with osteoarthritis which may influence cell behavior and phenotypes when applied to a healthy meniscus allograft repopulation and regeneration model. Finally, we did not examine specimen near the root of the meniscus, because vertical injections are technically challenging due to limited height of the specimen. Therefore, we excluded these specimens.
In conclusion, this study demonstrates the feasibility of recellularizing a frozen meniscus allograft with our cell-based injection therapy using MSCs and native MCs, either as a single cell or combination cell therapy, as reflected by microscopic evidence of cellular repopulation and matrix remodeling. Our findings provide proof of concept that enhancements in the cellular composition and other important biological attributes of meniscal allografts can be attained by introducing native MCs, either independently or in combination with allogeneic MSCs. These results encourage continuing research on this promising injection therapy to improve MAT surgery.
Supplemental Material
sj-pptx-1-car-10.1177_19476035231224802 – Supplemental material for Cellular Enhancement of Frozen Meniscus Allograft Combining Native Meniscus and Mesenchymal Stromal Cell Injections
Supplemental material, sj-pptx-1-car-10.1177_19476035231224802 for Cellular Enhancement of Frozen Meniscus Allograft Combining Native Meniscus and Mesenchymal Stromal Cell Injections by Caroline Struijk, Katherine L. Lydon, Martin Husen, Peter Verdonk, Jozef Michielsen, Andre J. van Wijnen, Aaron J. Krych and Daniel B.F. Saris in CARTILAGE
Footnotes
Author Contributions
C.S. performed experiments, data collection, and manuscript writing. K.L.L. performed zymography analysis. C.S., K.L.L., and D.B.F.S. contributed to the study conception and design. All authors commented previous versions of the manuscript. All authors have read and approved the final submitted manuscript.
Acknowledgments and Funding
C.S. performed this work while being a recipient of a Fellowship of the Belgian American Educational Foundation. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The contributions of A.J.v.W. were supported by NIH R01 AR049069.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Written informed consent was obtained and all collection procedures were performed based on Institutional Review Board (IRB)-approved protocols.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
