Abstract
Purpose
This study aimed to verify the femoral head cartilage protective effect of labral reconstruction in a porcine model.
Methods
Twelve pigs (24 hips) were divided into 3 groups: labrum defect group, lateral meniscus (LM) allograft group, and LM allograft wrapped with acellular peritoneum matrix (LM-APM) group before undergoing bilateral hip surgery. The pigs were sacrificed at 12 and 24 weeks postoperatively, while the femoral head cartilage was retrieved and then subjected to imaging measurement, macroscopic observations, and biomechanical and histological assessment.
Results
Imaging measurement and macroscopic observations revealed that the defect area of the labrum was filled in LM and LM-APM allograft groups after 24 weeks, whereas the labrum defect remained at 24 weeks in the control group. The femoral head cartilage corresponding to the area of labral resection in the labral defect group had worse macroscopic Osteoarthritis Research Society International (OARSI) scores, uneven and discontinuous cartilage on hematoxylin and eosin (H&E) staining and Safranin O staining, decreased histopathology OARSI Osteoarthritis Cartilage Histopathology Assessment System (OOCHAS) scores, and decreased elastic modulus and hardness at 12 and 24 weeks after surgery compared with the meniscus allograft groups.
Conclusion
This study demonstrated that the LM allograft with or without APM for labral reconstruction had a chondroprotective effect on the femoral head in a porcine model.
Keywords
Introduction
Femoroacetabular impingement syndrome (FAIS) has been identified as a major cause of hip pain in young adult patients and a potential generator of secondary osteoarthritis (OA) in up to 50% of hip OA cases. 1 When FAIS symptoms are not alleviated by nonsurgical treatment, hip arthroscopy is employed as the mainstream surgical treatment. 2 Surgical correction involves the resection of impinging osseous structures as well as concurrent management of the associated chondral and labral pathology. 3 A circumferentially intact labrum is vital for the functionality of the hip joint and has been found to play a key role in preserving the hip fluid seal.4 -6 In addition, repairing the labrum results in a better anatomic correction and improved clinical outcomes compared with labral debridement. 3 Restoring its function during surgical intervention, therefore, remains an essential goal. 7 However, the hip labrum cannot always be repaired. Moreover, the resection of a nonrepairable acetabular labrum down to a bleeding bony surface does not stimulate the regrowth of tissue. 8 Given the disruption of the hip labral function that occurs with significant resection, labral allograft reconstruction has been used to restore the compromised function of a resected hip labrum. Hip labral reconstructions have been associated with early excellent clinical outcomes and low complication rates.5,6,9 Labral reconstruction has shown it can restore the suction seal of the normal labrum, reduce acetabular contact stress, reduce femoral head translation, and provide increased surface area to stabilize the femoral head. 10 Restoration of these functions could be chondroprotective. The importance of the femoral head cartilage is notable given chondral damage was considered an independent risk factor for poor clinical outcomes after hip arthroscopy, with even mild femoral head chondral damage being viewed as problematic. 11 One cadaveric study showed that the femoroacetabular articular cartilage surface friction increased after focal and complete labrectomy. 12 However, it is currently unknown whether labral reconstruction can protect femoral head cartilage.
A challenge with allograft hip labral reconstructions is the biologic incorporation of the allograft. Obtaining a sufficient number of cells is an important factor in improving the quality of labrum reconstruction. 13 The specific polar architecture of acellular peritoneum matrix (APM) is beneficial for cell retention, adhesion, and proliferation,13,14 because the loose arrangement of fibers are conducive to cell attachment, and the compact basement can prevent cell loss. 13 Consequently, using APM can contribute to the healing process of labrum reconstruction.
The purpose of this study was to evaluate the protective effect of labral reconstruction, with and without APM, in a porcine model. We hypothesized that labrum reconstruction with and without APM would have a protective effect on femoral head cartilage.
Methods
Ethical approval for this study was obtained from the Institutional Animal Care and Use Committee of Peking University Third Hospital (A2019013).
Surgical Procedures
Twelve 6-month-old skeletally mature male Bama mini experimental pigs weighing 25 to 30 kg were subjected to bilateral labral surgery. The hips (24 in total; 8 hips per group) were randomly divided into 3 groups: the labrum defect group (control group), lateral meniscus (LM) allograft group, and LM allograft wrapped with APM (LM-APM) group. LM from sacrificed Bama mini experimental pigs used in other research was used as the graft source. Notably, the grafts had been stored for 2 to 4 months at −80°C. Before the start of the experiment, the LM was trimmed to form a size of 1.5 cm in length and 4 mm in width, and the transverse section was trimmed to a triangular shape (
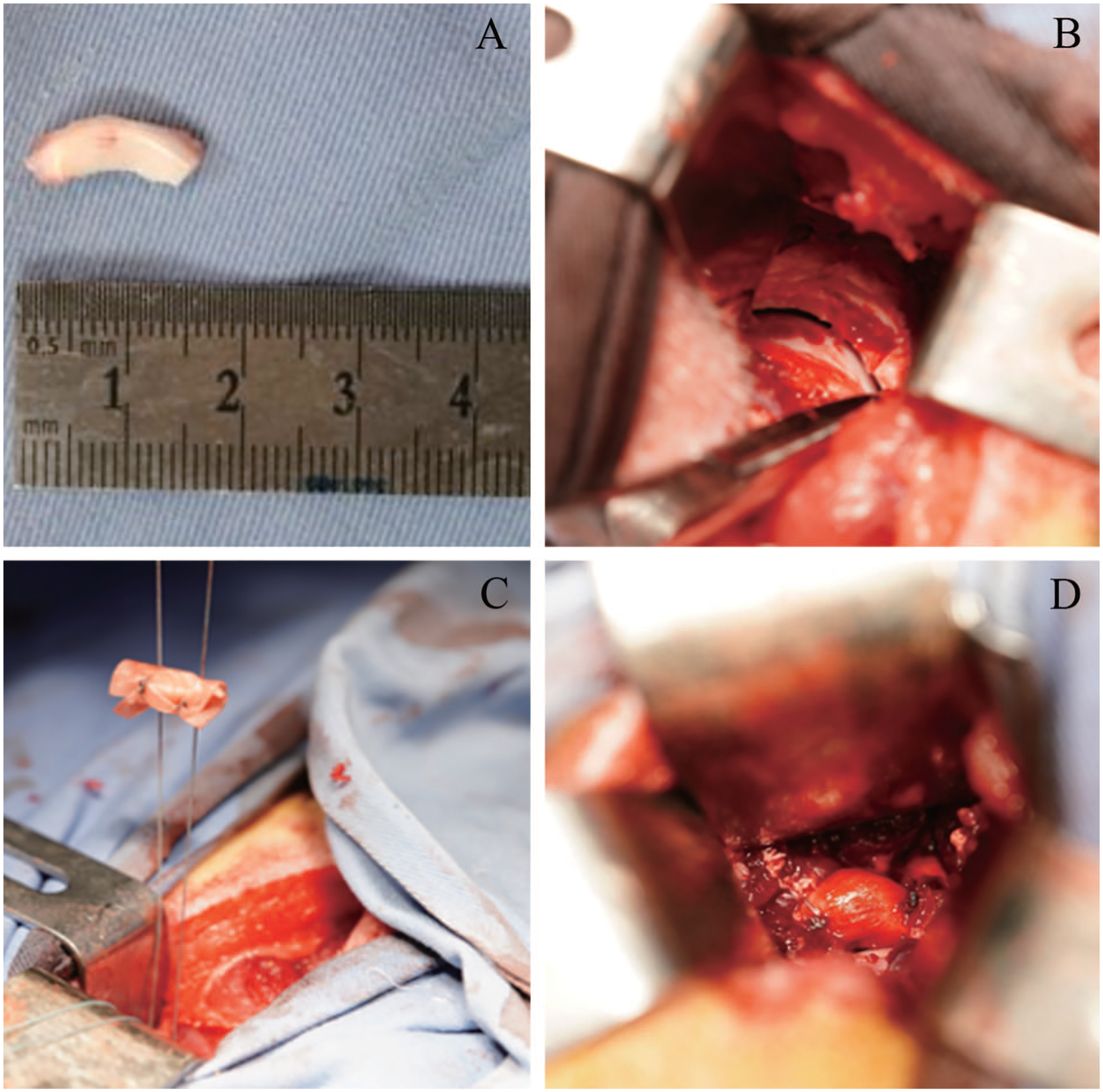
Preparation process of the lateral meniscus allograft and the surgical procedure. (
Briefly, pigs were anesthetized intravenously with propofol (20 drops/min) during the operation. Iodine and alcohol were used to disinfect the skin. The skin, subcutaneous superficial fascia, and aponeurotic fascia were incised with an approximate 10 cm arc incision posterosuperior to the greater trochanter. Next, blunt dissection of the space between the gluteus medius and gluteus maximus was performed, followed by anterior retraction of the gluteus medius, thus exposing the hip joint capsule. After incising the capsule, the labrum was exposed. A 1.5-cm-long section in the anterior dorsal labrum was then sharply resected to the acetabulum (
The pigs were then allowed to recover without weightbearing restriction or immobilization postoperatively. Antibiotic prophylaxis (penicillin, intramuscular injection; 500 mg twice daily) was continued for 5 days. After 12 or 24 weeks postoperatively, animals were sacrificed using pentobarbital sodium.
MRI Measurement
The hips were harvested after the pigs were sacrificed. The muscle around each hip joint was carefully dissected and partially retained, reserving the joint capsule. Two hips were randomly selected at each time point and then subjected to MRI measurement using a 3.0T Siemens scanner (MAGNETOM Verio Tim System, Erlangen, Germany) with a 12-channel-RF coil. The graft was identified by the junction area of bone defect and anchor, after which proton density-weighted images were captured to assess the condition of the femoral head cartilage. Five sequences were included in the protocol for morphological analysis. The total acquisition time was approximately 35 minutes. All MRI results were reported by the same senior radiologist (Y.Y.C.).
Macroscopic Morphological and Histological Analysis
The hip joints were carefully dissected, after which the implanted tissues were harvested for histological assessment. The gross morphology of the articular cartilage surface of the femoral head and integration with the acetabulum of implants were evaluated and photographed at 12 and 24 weeks. A semiquantitative scoring system for the femoral head cartilage was used by 3 researchers who were blinded to the nature of the experiment and the treatment allocation using the OARSI guidelines. 15 All researchers received instructions prior to morphologic and histologic assessments to maintain consistency. The scores from 3 observers were collected to calculate the mean value of the final score. If a score differed substantially from the other 2 scores, the sample was reassessed, and a consensus score was made. Next, the hip samples were then fixed in 10% neutral buffered formalin and then decalcified in 10% ethylenediaminetetraacetic acid (EDTA) for 20 days. All specimens were then dehydrated in alcohol and embedded in paraffin. Histopathological analysis to evaluate the integration of the meniscal allograft with the surrounding tissue were performed by sectioning the samples in 5-µm-thick sections and then subjected to hematoxylin and eosin (H&E) staining and Safranin O/fast green staining. The severity of the OA on the hip samples was determined using the OARSI Osteoarthritis Cartilage Histopathology Assessment System (OOCHAS). 16
Nanoindentation Evaluation of Cartilage
The biomechanical features of cartilage, including microscopic geomorphology, elastic modulus, and hardness, were assessed using nanoindentation. Cartilage samples (n = 4 per group) were harvested from the femoral head of animals in each experimental group. For blank controls, 4 normal hips were also used from sacrificed Bama mini experimental pigs taken from other studies. Circumfluent phosphate-buffered saline (PBS) solution was used to maintain hydration during the tests. A TriboIndenter (Hysitron Inc., Minneapolis, MN, USA) with a conospherical diamond probe tip with 400-mm radial curvature was applied to evaluate the samples. Each indent site was tested using a trapezoidal load function that included loading (10 seconds), hold (2 seconds), and unloading (10 seconds). The maximum depth of indentation was 500 nm. A micro-scanning apparatus was used to capture the microscopic geomorphology of the indentation zones.
Statistical Analyses
All statistical analyses were performed using SPSS 26.0 statistical software (IBM Corp.). The statistical review of the present study was performed by a biomedical statistician (L.N.). Data were recorded as mean ± standard error. One-way analysis of variance (ANOVA; homogeneity of variance, 3 groups) was employed to analyze normally distributed data (OOCHAS scores, elastic modulus, and hardness). While, non-parametric tests were used to analyze non-normally distributed data (macroscopic OARSI scores). P < 0.05 was considered statistically significant.
Results
General Postoperative Course
During the experimental period, all pigs were in good general condition, and there were no postoperative complications, such as wound infection or death. Normal walking and standing activities returned within 1 to 2 weeks. No signs of joint effusion were detected when the animals were sacrificed at 12- and 24-week time points.
MRI Measurement
In the control labral defect group, the signal density of the femoral head articular cartilage was uneven, and there was contrast infiltration into the cartilage tissue, suggesting that the labral resection procedure induced degenerative changes to the femoral chondral surfaces. Cartilage degeneration and damage were more pronounced at 24 weeks (
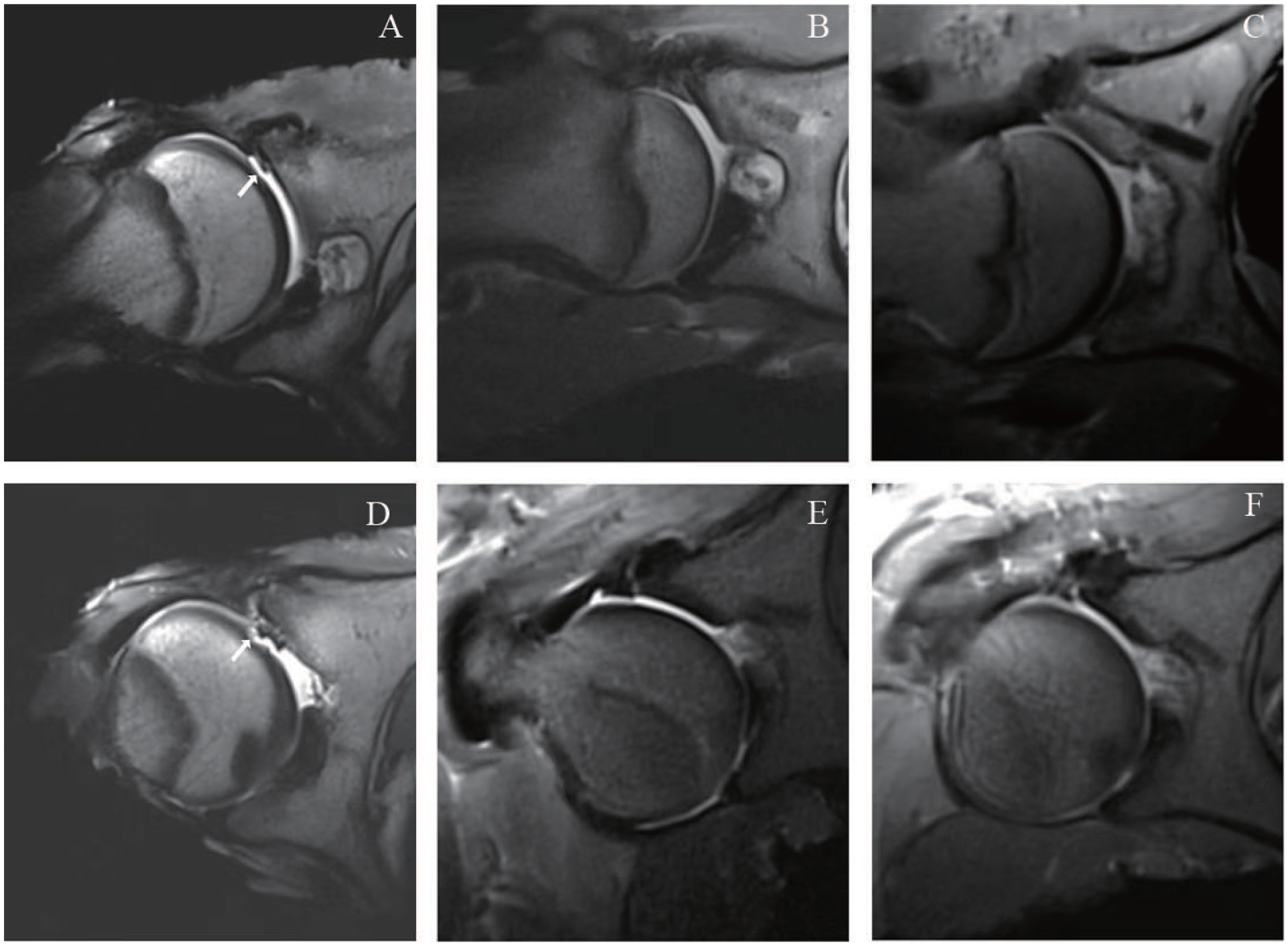
Cartilage features of femoral head observed using MRI at 12 and 24 weeks postoperatively (arrows represent articular cartilage; n = 3 for each group). (
Macroscopic Evaluation of Grafts and Cartilage
Macroscopic Evaluation of Grafts
At 12 weeks, the macroscopic analysis indicated that the continuity of the labrum was interrupted in the labrum defect group and that the defect was covered by a small amount of soft tissue. In LM allograft group, the graft was found to be closely attached to the bone bed of the acetabulum. In LM-APM allograft group, no APM scaffold structure was observed, the grafts showed complete healing at both ends of the labrum and were closely attached to the acetabulum. At 24 weeks, no clear regrowth of the labrum was observed in any specimen in the control group. All grafts in LM and LM-APM allograft group exhibited complete healing and filled the defect area of the labrum. In addition, the implants connected with the acetabular cartilage at 12 and 24 weeks after surgery and formed smooth articular surfaces in LM and LM-APM allograft groups, effectively deepening the acetabular volume.
Macroscopic Evaluation of Femoral Head Cartilage and OARSI Score
Macroscopic evaluation of the hip samples revealed significant cartilage degeneration and damage were observed in the labrum defect group. There were discontinuity, fibrillation, severe erosion, crevasse, and delamination on the cartilage surfaces of the femoral heads, especially in the zone where the anterior (cranial) dorsal labrum was removed, which suggested that degenerative changes were induced by labral deficiency. Furthermore, cartilage degeneration and damage were more pronounced at 24 weeks than at 12 weeks (
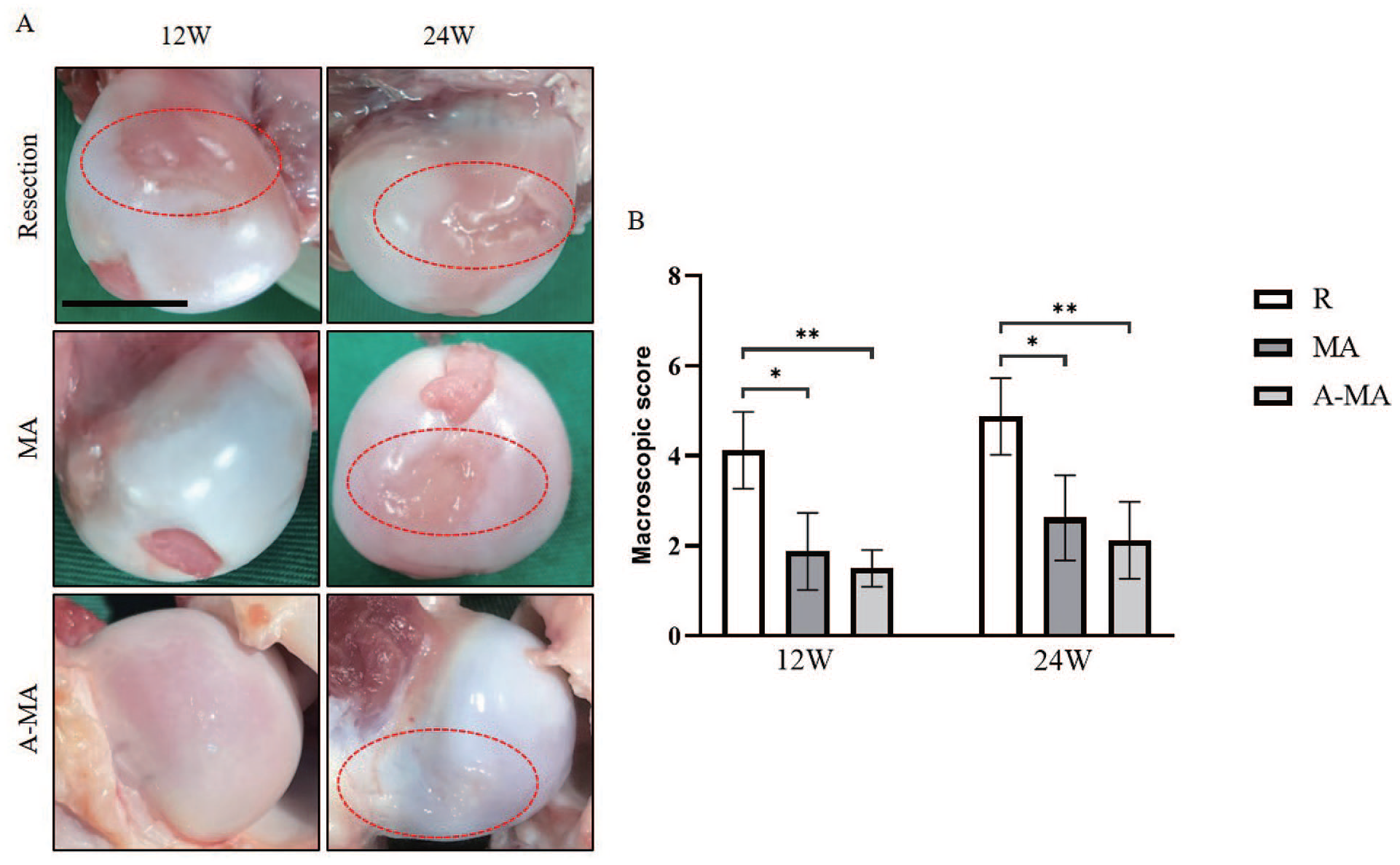
Macroscopic appearance
At 12 weeks, OARSI score was 3.3 ± 0.8 in the labrum defect group, 1.5 ± 0.4 in LM allograft group, and 1.8 ± 0.6 in LM-APM allograft group. At 24 weeks, the score was 5.4 ± 1.0 in the labrum defect group, 3.1 ± 1.1 in LM allograft group, and 3.1 ± 0.2 in LM-APM allograft group (
Histological Evaluation
H&E staining revealed that in the labrum defect group, the cartilaginous surfaces of the femoral head were uneven and discontinuous, with complex vertical and horizontal cracks that progressed to the subchondral bone. Microfractures and fibrous tissue repair were simultaneously visible, the tidemark was disrupted, and the number of chondrocytes was relatively low (
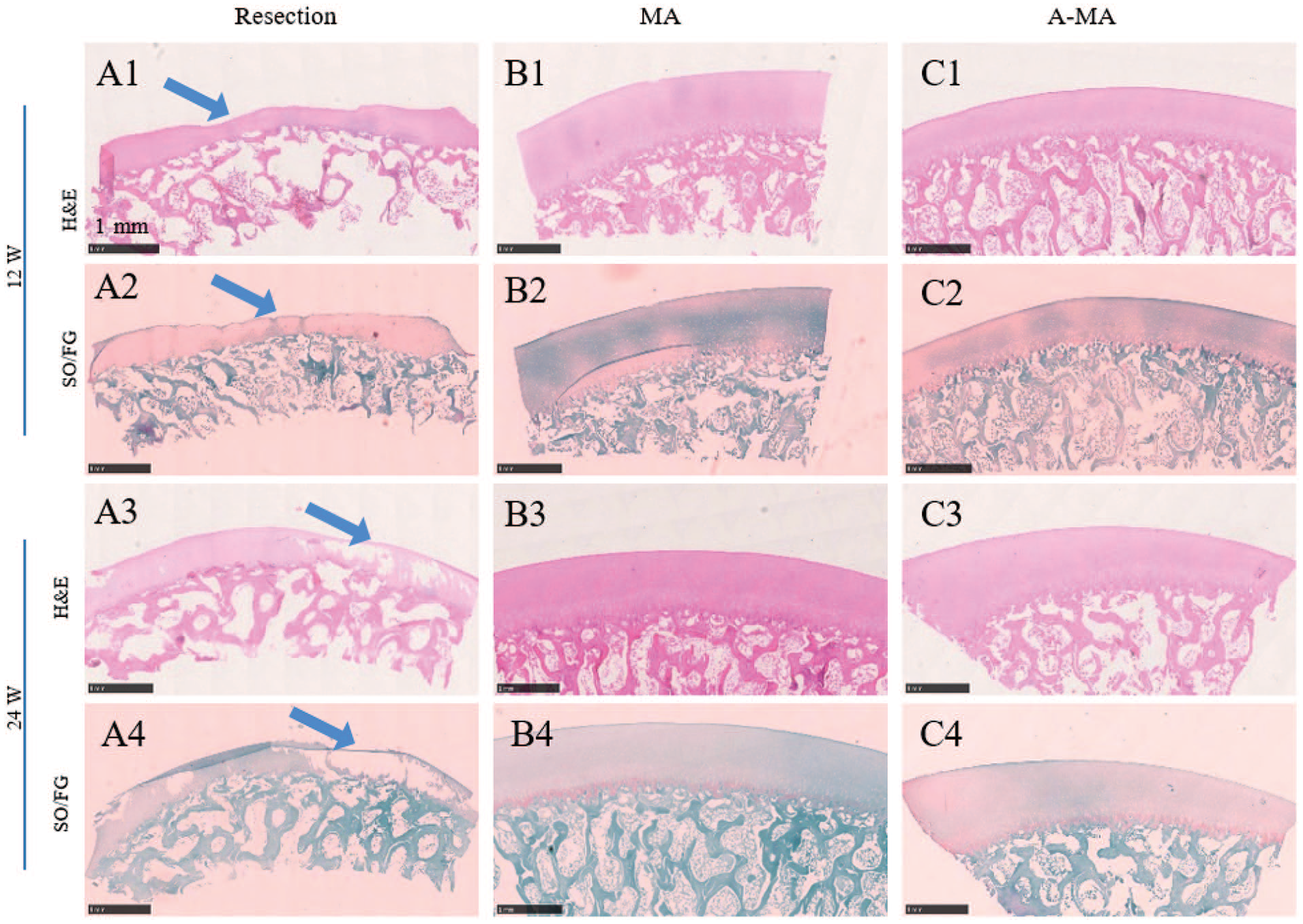
Histological analysis on the femoral head cartilage, including H&E and SO/FO staining. H&E = hematoxylin and eosin. SO/FO = Safranin O/fast green. MA = lateral meniscus allograft group; A-MA = lateral meniscus allograft wrapped with acellular peritoneum matrix group.
Safranin O staining revealed that the cartilage surface was not flat in the labrum defect group at 12 weeks (
At 12 weeks, OOCHAS score for the labrum defect group was 10.1 ± 3.7, significantly higher than for allograft groups (1.9 ± 0.7 and 2.3 ± 1.1, respectively) indicative of greater cartilage degeneration in the control group. At 24 weeks, OOCHAS score for the labrum defect group was 10.0 ± 4.0, which was also higher than that for the allograft groups (1.9 ± 0.7 and 1.4 ± 0.4, respectively) indicative of greater cartilage degeneration in the control group.
Nanoindentation Evaluation of Cartilage
Using micro-scanning, we observed that the cartilage surface in the labrum defect group was much rougher and more uneven than that in the normal joints. In contrast, the cartilage surfaces in the allograft groups were smooth and homogeneous, similar to those in the normal joints. Moreover, the elastic modulus and hardness of the cartilage were markedly decreased in the labrum defect group compared with those in the normal cartilage. These properties were effectively restored in the 2 allograft groups, illustrating the recovery of compression properties after labrum reconstruction (Fig. 5).
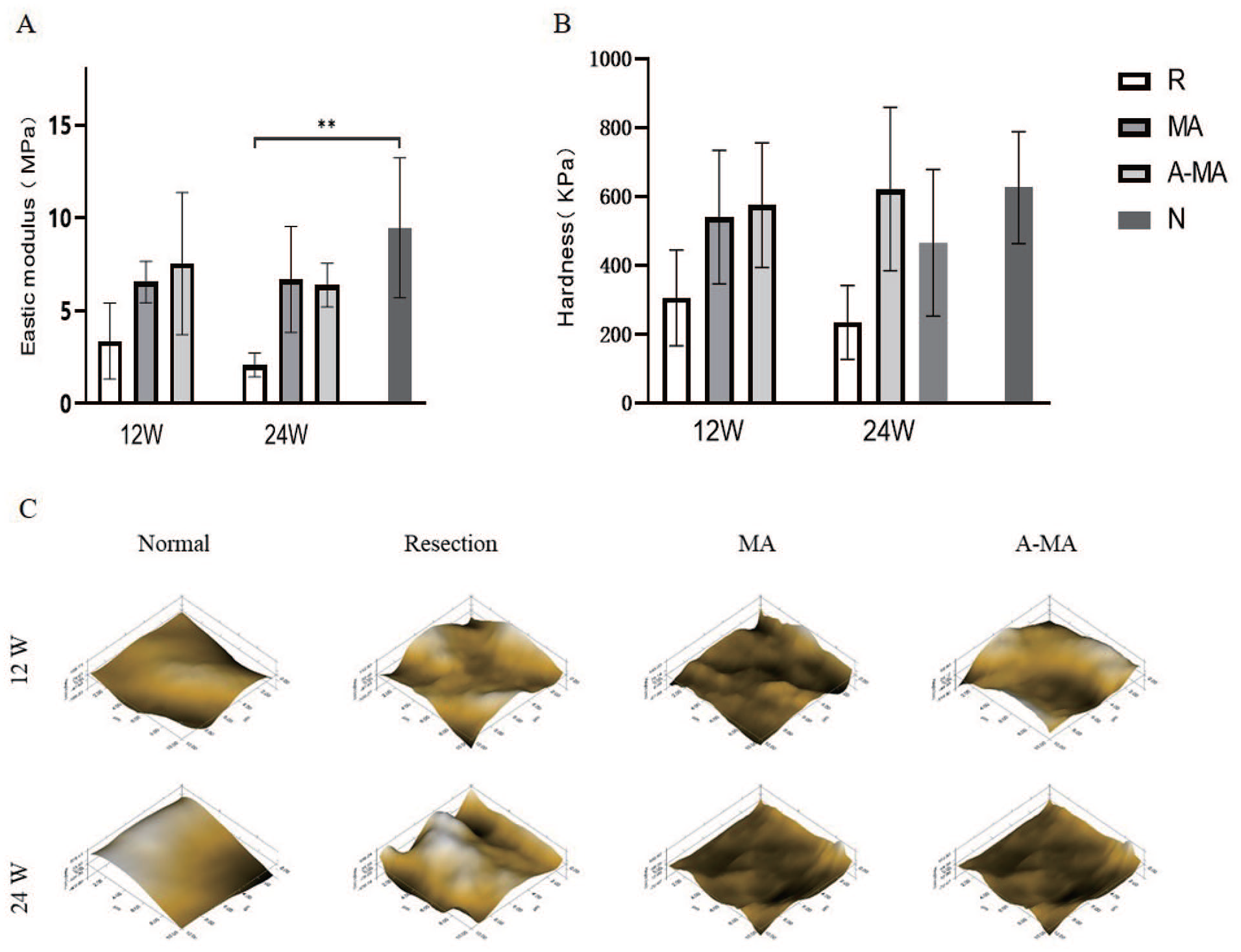
Elastic modulus (
Discussion
The main finding of this study was that LM and LM-APM allograft labrum reconstructions had a markedly chondroprotective effect on femoral head cartilage compared with the labrum defect group in a porcine model. The femoral head cartilage of the labral defect group had worse macroscopic OARSI scores, uneven and discontinuous cartilage on H&E staining and Safranin O staining, decreased OOCHAS scores, and decreased elastic modulus and hardness at 12 and 24 weeks after surgery compared with both meniscus allograft groups. These findings support prior research showing promising clinical outcomes for labral allograft reconstruction.17-19
LM allograft, which was used for labrum reconstruction in the present study, has several potential advantages. First, the native labrum and LM share many similarities in composition. The collagen content of the labrum and the LM is predominantly type I collagen. 20 Second, given that the structure of the two is also similar, the shape of the labrum and LM can be well matched. Notably, the labrum is circular, whereas the LM is O-shaped; consequently, the meniscus allograft is a better match for the labrum than the tendon allograft.
Meng et al. 13 used APM scaffold to repair cartilage lesions, demonstrating that APM scaffold can recruit attachment of bone marrow-derived mesenchymal stem cells (BM-MSCs) and fill the cartilage defects in the femoral trochlear of rabbits. Moreover, Tsai et al. 14 used APM to construct human skin equivalents in vitro, and showed that it had excellent skin cell attachment and proliferation for human dermal fibroblasts (HDF) and immortalized human keratinocytes. Meanwhile, APM was used in this study to promote the healing process of labrum reconstruction, and it was found that both LM allograft and LM-APM allograft can fully fill the labral defect in a porcine model. The condition of femoral head cartilage was similar in both groups. The results showed that LM alone can fully fill the labral defect and protect the femoral head cartilage.
Many clinical studies have explored the causal relationship between a labral deficiency and cartilage damage. Philippon et al., 21 for instance, reported that patients with severely damaged labral tissue often have poor cartilage on the acetabulum edge, while Mayes et al. 22 found labral tears were associated with cartilage defects in professional ballet dancers. The animal models used in the present study confirmed these clinical findings. The labrum provides mechanical support by effectively deepening the acetabular socket. Therefore, once the labrum is damaged or deficient, potential for the mechanical support becomes compromised, predisposing the hip joint to femoral head instability within the acetabulum.
Another possible reason for the chondroprotective effect of labral reconstruction on femoral head cartilage is that it retains the sealing function. The acetabular labrum provides a sealing effect, which creates both hydrostatic pressures to slow cartilage consolidation during hip joint motion and a “suction effect” to resist the distraction of the hip joint. Furthermore, Philippon et al. 23 found that labral reconstruction effectively restored intra-articular fluid pressurization to levels similar to the intact state via the hip fluid seal, whereas labral resection decreased intra-articular fluid pressurization. Similarly, Ferguson et al.24,25 reported that hips with damaged labrum are consistent with reduced fluid pressure and increased solid–solid contact stress between articular cartilage surfaces in vitro investigation and finite element model. Finally, Song et al. 12 identified that resistance to rotation at the articular cartilage surface was significantly increased following focal and complete labral resection in a cadaver, which indicates that articular cartilage friction in an intact hip was significantly increased. Retention and/or restoration of the functionality of the hip labrum should therefore be a prime consideration in surgical treatment in FAIS patients. The chondroprotective effect that labrum reconstruction has may delay the progression of OA and help to avoid or at least postpone total hip arthroplasty.
While our study has several strengths including providing preclinical evidence on the importance of hip labral reconstruction and the length of follow-up in our animal model (6 months), we should acknowledge that the study has some limitations as well. One limitation is our study did not evaluate other tissue allografts that can be used to reconstruct the hip labrum including iliotibial band or hamstring tendons. Notably, we did not include these groups because previous studies had suggested that those allograft may have poorer mechanical properties and are ineffective at healing the interface of graft-labrum compared with native labrum. 26 A second limitation is our study’s sample size in each group was relatively small. Despite the small sample size, we were able to show some meaningful differences between labral defect versus labral reconstruction in this porcine model. Finally, although we were able to provide structural and biomechanical properties in our preclinical study, certain biologic parameters including inflammatory cytokines analyses of the tissues and synovial fluid were not analyzed.
Conclusion
This study demonstrated that the LM allograft with or without APM for labral reconstruction had a chondroprotective effect on the femoral head in a porcine model.
Footnotes
Author Contributions
H.H. and J.W. have given substantial contributions to the conception and the design of the manuscript; F.Y. and M.M. to acquisition, analysis, and interpretation of the data. All authors have participated to drafting the manuscript, X.Z. and Z.H. revised it critically. All authors read and approved the final version of the manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (Grant Nos. 81902205, 82072403); Beijing Natural Science Foundation Haidian Original Innovation Joint Fund Frontier Project (L212052); and Clinical Medicine Plus X—Young Scholars Project, Peking University, the Fundamental Research Funds for the Central Universities (PKU2022LCXQ005, PKU2022LCXQ015).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The design of the present study adhered to the tenets of the Declaration of Helsinki and the protocol was reviewed and approved by the Ethics Committee of Peking University Third Hospital.
