Abstract
Objective
The medial meniscus extrusion (MME) is associated with increased stress on the knee joint, which leads to cartilage degeneration. To evaluate the etiology of knee osteoarthritis, it is extremely important to create animal models of the disease that more closely resemble actual clinical conditions in terms of symptomatology, molecular biology, and histology. This study aimed to create a clinically relevant model of MME in rats.
Design
Behavioral, molecular biological, and histological changes in the newly developed rat MME model were compared with those in sham and medial meniscus transection and medial collateral ligament transection (MMT) models to examine the characteristics of this model.
Results
In the MME rat model, behavioral evaluation shows abnormalities in gait compared with the other 2 groups, and molecular biological evaluation of the infrapatellar synovia of rats shows that gene expression of inflammatory cytokines, matrix-degrading enzymes, and pain-related nerve growth factor was increased compared with the sham group. Furthermore, histological evaluation reveals that cartilage degeneration was the most severe in the MME group.
Conclusions
The newly developed MME model reproduced the characteristic pathology of MME in clinical practice, such as severe pain, inflammation, and rapid progression of osteoarthritis. The MME model, which might more closely mimic human knee osteoarthritis (OA), could be a useful model for elucidating the pathophysiology and considering therapeutic management for knee OA.
Introduction
Knee osteoarthritis (OA) is a disease afflicting more than 27 million people in the United States and 250 million people worldwide,1,2 leading to decreased mobility owing to chronic pain and locomotor disorders causing frailty. 3 Knee OA is a result of degenerative changes centered on articular cartilage and the subchondral bone and occurs owing to synovial inflammation, meniscus degeneration, and progressive arthropathy.4-6 Although the detailed mechanism of knee OA has not yet been elucidated, meniscus degeneration is a significant mechanical factor. Meniscal fibers, which run in a circumferential pattern, form the hoop mechanism of the meniscus and play an important role in stress dispersion under load.7,8 The wedge-shaped meniscus is attached to the tibia by the anterior and posterior roots, which not only distributes the stress on the knee joint due to loading but also radially distributes the shear stress on the meniscus. 9 The meniscus degeneration and associated damage to the posterior horn of the medial meniscus can cause the meniscus to extrude from the medial articular surface of the tibia, disrupting the hoop mechanism necessary for load distribution and a force of transmission through the meniscus. 10 In recent years, it has become clear that the abnormal position of the medial meniscus, so-called medial meniscus extrusion (MME), is associated with increased stress on the knee joint,11,12 which leads to cartilage degeneration. 13 Knee pain was more frequently seen in knee OA patients with MME than in those without.14,15 Wang et al. 16 showed MME to be an independent predictor of bone marrow lesions which are linked to the progression of knee OA and pain. In addition, Chiba et al. have reported that more extruded medial meniscus showed a higher prevalence of both the onset and progression of knee OA in the epidemiological study based on longitudinal surveys conducted on the general population. 17 Biological factors also play a role in the development and progression of knee OA. As mechanical stress increases and cartilage degeneration occurs, wear fragments of type Ⅱ collagen induces an inflammatory response in the knee synovium. 18 The inflamed synovium increases the expression of pro-inflammatory cytokines such as interleukin (IL)-1β and tumor necrosis factor (TNF)-α, further amplifying the catabolic effects in the knee joint.19,20 Matrix degradation in knee OA may result from matrix metalloproteinase (MMP)-3 and a disintegrin and metalloproteinase with thrombospondin-1 domains (ADAMTS)-5, which degrade aggrecan, followed by increased activity of collagenases, in particular MMP-13, which is highly efficient at degrading type II collagen. 6 In addition, nerve growth factor (NGF), a pain signaling substance in peripheral nociceptors, plays an important role as a molecular biological factor involved in pain of knee OA. NGF production increases as cartilage damage in knee OA progresses, 21 and its production is thought to be related to inflammatory cytokines such as IL-1β and mechanical stress on articular cartilage. 22 Symptoms of knee OA caused by these changes are pain and functional impairment.23,24
Animal models of the disease are necessary, and many animal species (mice, rats, rabbits, and dogs) have been used to date.25-28 The advantage of using rats is that they have thicker cartilage than mice, which makes histological evaluation easier, and they are less expensive than rabbits. 27 In addition, various methods of inducing knee OA have been implemented, including intra-articular enzyme administration, impact loading, immobilization, and surgery.25,27,28 Commonly used induction methods include the monoiodoacetic acid-induced arthritis model (MIA injection model), 29 anterior cruciate ligament transection model (ACLT model), 30 and medial meniscus transection and medial collateral ligament (MCL) transection model (MMT model). 31 In the MIA model, intra-articular administration of MIA inhibits chondrocyte glycolytic energy metabolism and chondrocyte synthesis, leading to cartilage degeneration. The MMT model is used to mimic a post-traumatic OA degradation. The technique consists in cutting the meniscus into 2 parts, anterior and posterior, resulting in instability in the joint. This leads to atypical stress profiles that will damage the cartilage, an approach that better reflects the conditions for the appearance of secondary OA in humans. All these models cause arthritic changes such as cartilage degeneration and osteophyte formation, but the MIA model causes degeneration of the entire articular cartilage owing to drug administration. ACLT and MMT are both clinically relevant models of post-traumatic knee osteoarthritis, but they differ from the pathogenesis of knee OA caused by MME, which has attracted attention in recent years. As mentioned above, MME is a factor that greatly affects the onset and progression of knee osteoarthritis. Therefore, in recent years, there has been growing interest in performing arthroscopic centralization surgery to repair MME, 32 highlighting the significance of addressing MME. Establishing and investigating an animal model for MME can play a crucial role in elucidating the mechanisms of knee OA, developing new therapeutic approaches, and exploring methods to prevent cartilage degeneration.
However, no animal model of MME-induced knee OA has been reported to date. The purpose of this study was to create a clinically relevant model of MME in rats.
Materials and Methods
The experimental protocol was approved by our institutional review board (Approval number 1017).
A total of 27 Sprague–Dawley male rats, each weighing approximately 250 g at the time of intervention, was used in the present study. During the experiment, rats were housed in pairs in 1 cage under a 12 h light/12 h dark cycle. They were given food pellets and water ad libitum.
Groups
Twenty-seven male rats were divided into 3 groups. In the MME group, rats underwent MME operation (n = 9) as mentioned below. In the sham group (n = 9), in which after transection of the MCL, a bone tunnel was made in the tibia and a thread passed through the margin of the meniscus, but no suturing was performed. The MMT model (n = 9), in which the medial meniscus was transected in addition to the MCL, 31 was used as comparative controls. Postoperatively, the rats walked freely in their cages.
Operation Method to Develop the MME Model
Rats were anesthetized in surgery with intraperitoneal administration of sodium pentobarbital (50 mg/kg), and the right knee was clipped and scrubbed in preparation for surgery. The medial side of the right knee joint was incised. Subsequent procedures were performed using a microscope. The medial collateral ligament was identified and transected, and the medial meniscus was identified in the deep layer. A 4-0 silk suture was passed through the middle segment of the medial meniscus on the side of the articular capsule using a surgical needle. Next, the suture was passed through the proximal medial tibial bone using a square needle to create a bone tunnel and then approximately half of the diameter of the meniscus was fixed in an extruded position. Finally, the skin was sutured with interrupted sutures using 4-0 nylon sutures (
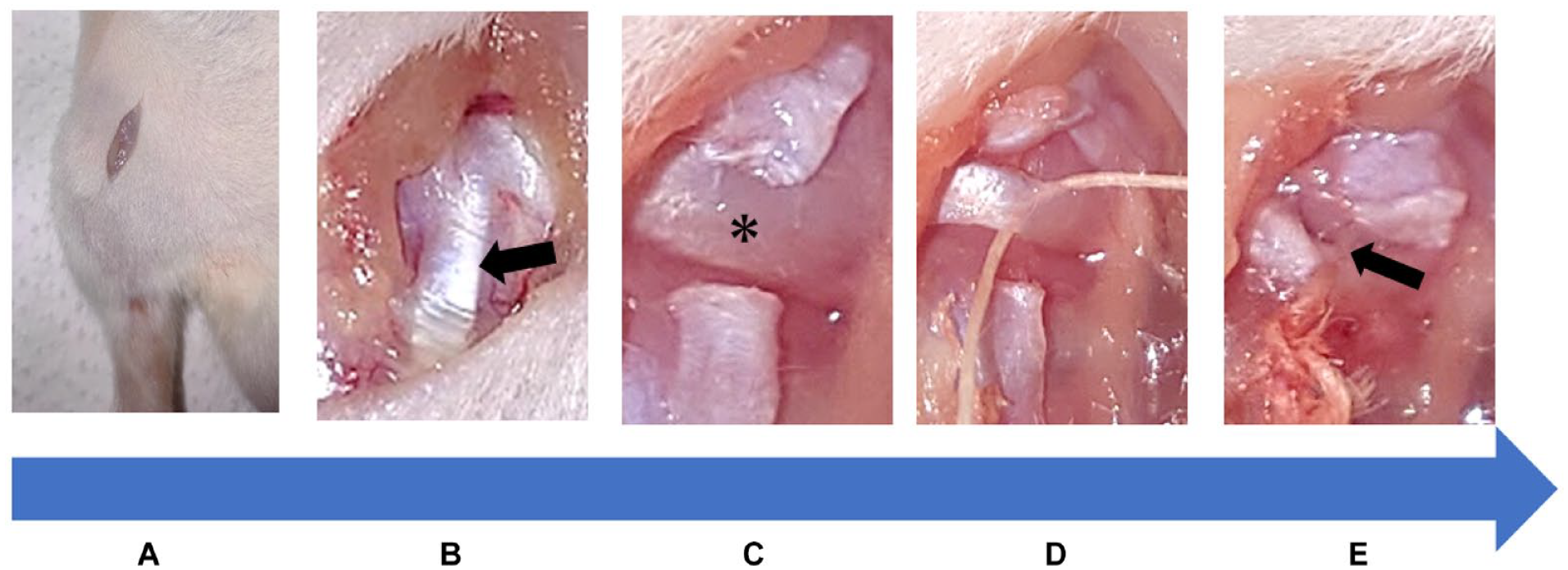
Medial meniscus extrusion model creation procedure. (
Study Design
Gait analysis under spontaneous walking of rats was performed every 2 weeks after the procedure until week 8 for symptomatic evaluation using the CatWalk system.33,34 In reference to previous reports,30,31 knee joint tissues of rats were harvested at 8 weeks for the following experiments. For the molecular biological evaluation of inflammatory cytokines and catabolic enzymes, gene expression analyses of the infrapatellar synovium at 8 weeks after the procedure were carried out using real-time polymerase chain reaction (PCR).
35
Histological cartilage degeneration was evaluated by hematoxylin-eosin (HE) staining and Safranin-O staining using Osteoarthritis Research Society International (OARSI) scoring of knee joints from rats euthanized at 8 weeks
36
(
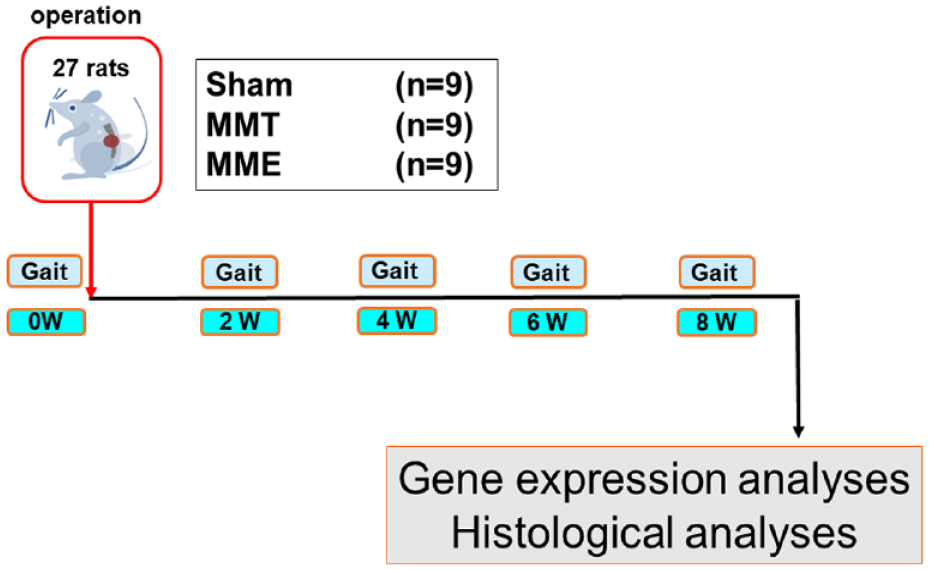
Experimental protocol. Gait analysis under spontaneous walking was performed every 2 weeks after the procedure until week 8 for symptomatic evaluation using the CatWalk system. The knee joint tissues of the rats were harvested at 8 weeks for subsequent experiments. For the molecular biological evaluation of inflammatory cytokines and catabolic enzymes, gene expression analyses of infrapatellar fat were performed using real-time PCR. Histological cartilage degeneration was evaluated by hematoxylin-eosin (HE) and Safranin-O staining using Osteoarthritis Research Society International (OARSI) scoring. PCR = polymerase chain reaction; MMT = medial meniscus transection; MME = medial meniscus extrusion.
CatWalk Analyses
Utilizing the CatWalk system (Noldus Information Technology, Wageningen, The Netherlands), a rat’s gait was analyzed under natural walking conditions. Rats were placed on a glass plate in a semi-darkened room and are free to walk along a straight corridor with a transparent floor (
Stand time (s). Stand time is the time that the paw was in contact with the glass plate in the stand phase.
Max contact area (mm 2 ). The maximum area is paw area contacted at the moment of maximal paw-glass plate contact.
Mean intensity. This parameter describes the mean pressure exerted by one individual paw on the walkway. Data analysis was performed with a threshold value of 40 (arbitrary units, possible range 0-225).
Swing speed (mm/s). Swing speed was calculated based on stride length and swing duration.
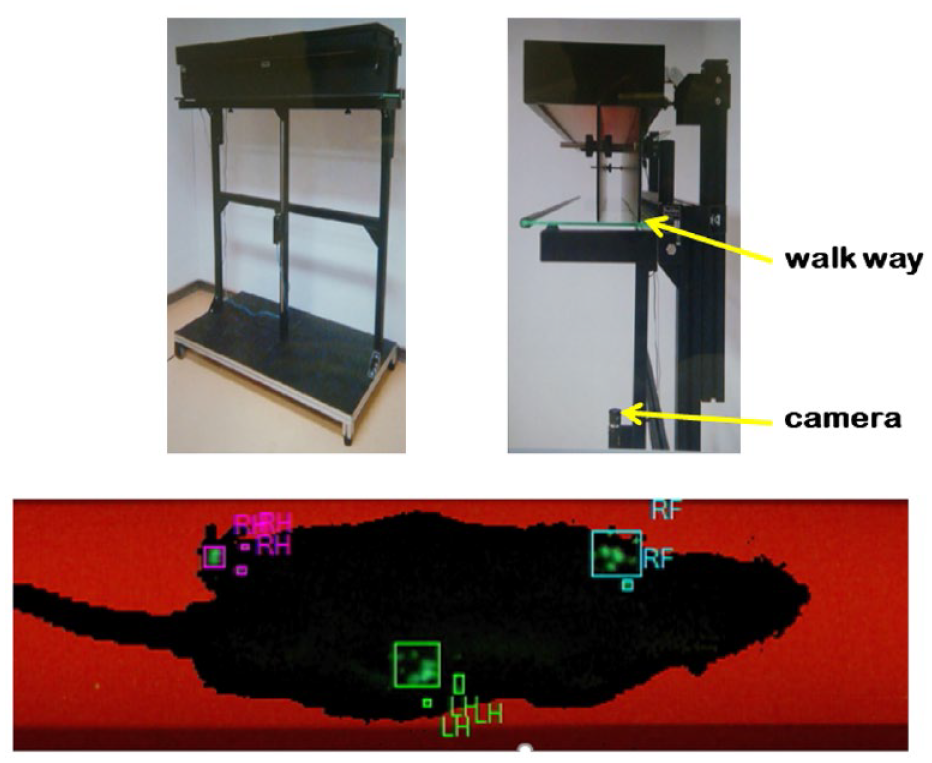
Gait analysis with the CatWalk system. The CatWalk system allows for detailed gait analysis. Rats were placed on a glass plate in a semi-darkened room and are free to walk along a straight corridor with a transparent floor. Fluorescent light is directed at the glass plate, and when the rat’s paw contacts the glass plate, the light is reflected downward, resulting in a clear bright footprint, which is recorded by a camera installed under the glass plate.
Gene Expression Analyses
Rat infrapatellar synovium was collected to evaluate gene expression of molecular biological factors according to a previous report 38 8 weeks after the procedure. Total ribonucleic acid (RNA) in infrapatellar synovium tissues was extracted using a tissue homogenizer (Polytron, Kinematica, Bohemia, NY) and Qiazol (Qiagen, Valencia, CA), with further purification by chloroform separation, followed by the RNeasy Lipid Tissue Mini Kit (Qiagen). The integrity of RNA pool was confirmed by electrophoresis. Double-stranded complementary deoxyribonucleic acid (cDNA) was synthesized by the reverse transcription of total RNA (1 μg) using PrimeScript RTase (Takara, Shiga. Japan). Real-time PCR using TB Green Premix Ex Taq II (Takara) was performed on a StepOne Plus (Thermo Fisher Scientific, Waltham, MA) with the following procedures: an initial denaturation step at 95°C for 30 seconds; 40 cycles at 95°C for 3 seconds; and 60°C for 30 seconds. The gene expression levels of IL-1β, TNFα, MMP-3, ADAMTS-5, MMP-13, and NGF were analyzed using pre-designed primers ( Table 1 ). Gene expression was determined by relative quantification (δδCT method 39 ) using the mitochondrial ribosomal protein of small subunit 18 (MRPS 18) as an internal control.
Primer Information.

IL = interleukin; TNF = tumor necrosis factor; MMP = matrix metalloproteinase; ADAMTS = a disintegrin and metalloproteinase with thrombospondin-1 domains; NGF = nerve growth factor; MRPS = mitochondrial ribosomal protein of the small subunit.
Histological Analyses
The right knee joint from rats sacrificed at 8 weeks was removed en bloc. After fixation in 10% formalin for 1 week, the knee joint tissues were decalcified using a 10% ethylenediaminetetraacetic acid solution and embedded in paraffin wax. Midcoronal sections (5 μm) of each right knee joint were stained with hematoxylin and eosin (HE) and Safranin-O. The histological sections at the medial articular surface of the tibial condyle were graded using the OARSI score. 36 The evaluation was performed in a blind manner by an orthopedic surgeon who specializes in treating knee joints and a pathologist. Since the intra- and inter-rater κ-coefficients were secured (the intra- and inter-rater κ-coefficients were 0.74 and 0.58, respectively), the numerical value of the orthopedic surgeon was used for evaluation.
Statistical Analysis
All values are presented as mean ± standard error. In the gait analyses, a 1- and 2-way analysis of variance (ANOVA) with Fisher’s post hoc least significant differences test was conducted to determine whether there were differences in the gait parameters measured between the groups at each measurement time point and over the entire experimental period. One-way ANOVA with Fisher’s post hoc least significant difference test was used for gene expression and histology analyses. Intra- and inter-rater reliability of the OA grade in OARSI scores in the histological analysis were evaluated by calculating the κ-coefficient. Statistical analysis was performed using The StatView 5.0 program (SAS Institute, Cary, NC). A P value of less than 0.05 was considered statistically significant.
Results
None of the rats exhibited stress reactions such as weight loss or decreased food intake.
CatWalk Analyses of the MME Model Compared With the Sham and MMT Group
There were no significant differences in CatWalk analyses of the contralateral side among the sham, MMT, and MME groups.
Stand time. In the ipsilateral side, the stand time was significantly decreased in the MME group, compared with the other 2 groups 2, 4, and 6 weeks after surgery (MME, 2 weeks: P < 0.01 vs. sham and MMT, 4 weeks: P < 0.01 vs. sham and MMT, 6 weeks: P < 0.01 vs. sham and MMT). At 8 weeks post-surgery, the stand time was significantly lower in the MME group than in the sham group (MME; P < 0.05 vs. sham).
Max contact area. The maximum contact area was significantly decreased in the MME group, compared with the other 2 groups at all observation periods (MME, 2 weeks: P < 0.01 vs. sham and MMT, 4 weeks: P < 0.01 vs. sham and MMT, 6 weeks: P < 0.01 vs. sham and MMT, 8 weeks: P < 0.05; vs. sham and p < 0.01 vs. MMT).
Mean intensity. The mean intensity was significantly lower in the MME group than in the other 2 groups at 2, 4, and 6 weeks after surgery (MME; 2 weeks: P < 0.01 vs. sham and P < 0.05 vs. MMT, 4 weeks: P < 0.05 vs. sham and MMT, 6 weeks: P < 0.01 vs. sham and MMT). At 8 weeks post-surgery, this was significantly lower in the MME group than in the sham group (MME; P < 0.05, vs. sham).
Swing speed. The swing speed was significantly decreased in the MME group, compared with the other 2 groups at all observation periods (MME, 2 weeks: P < 0.01 vs. sham and MMT, 4 weeks: P < 0.01 vs. sham and MMT, 6 weeks: P < 0.01 vs. sham and MMT, 8 weeks: P < 0.01 vs. sham and MMT). In addition, the MME group was significantly lower than the other 2 groups in all parameters throughout the experimental period (MME; P < 0.01, vs. sham and MMT) (
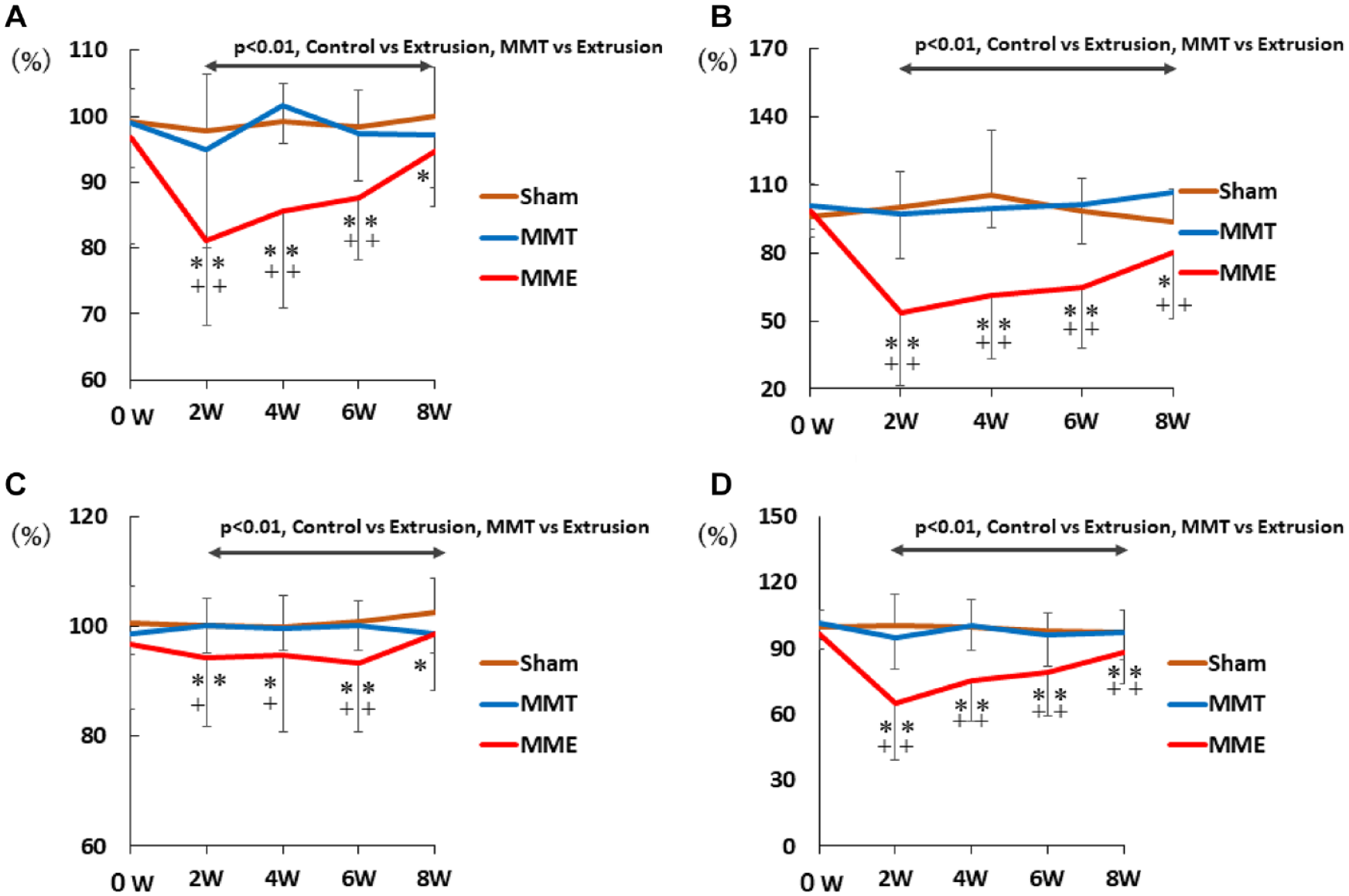
Gait analysis with the CatWalk system. The MME group was significantly lower than the other 2 groups in all parameters throughout the experimental period (MME; P < 0.01, vs. sham and MMT). The 2-way ANOVA with Fisher’s post hoc least significant differences test was used. (
Gene Expression Analyses of the MME Model
Pro-inflammatory genes
IL-1β gene expression was significantly higher in the MMT and MME groups than in the sham group (Sham; 1.00 ± 0.14, MMT; 2.10 ± 0.41, MME; 2.38 ± 0.19, MMT; P < 0.05, MME; P < 0.01, vs. sham). The TNF-α levels in the MME group were significantly lower than those in the MMT group (Sham; 1.00 ± 0.17, MMT; 1.33 ± 0.35, MME; 0.55 ± 0.07, MMT; P < 0.05, vs. MME) (
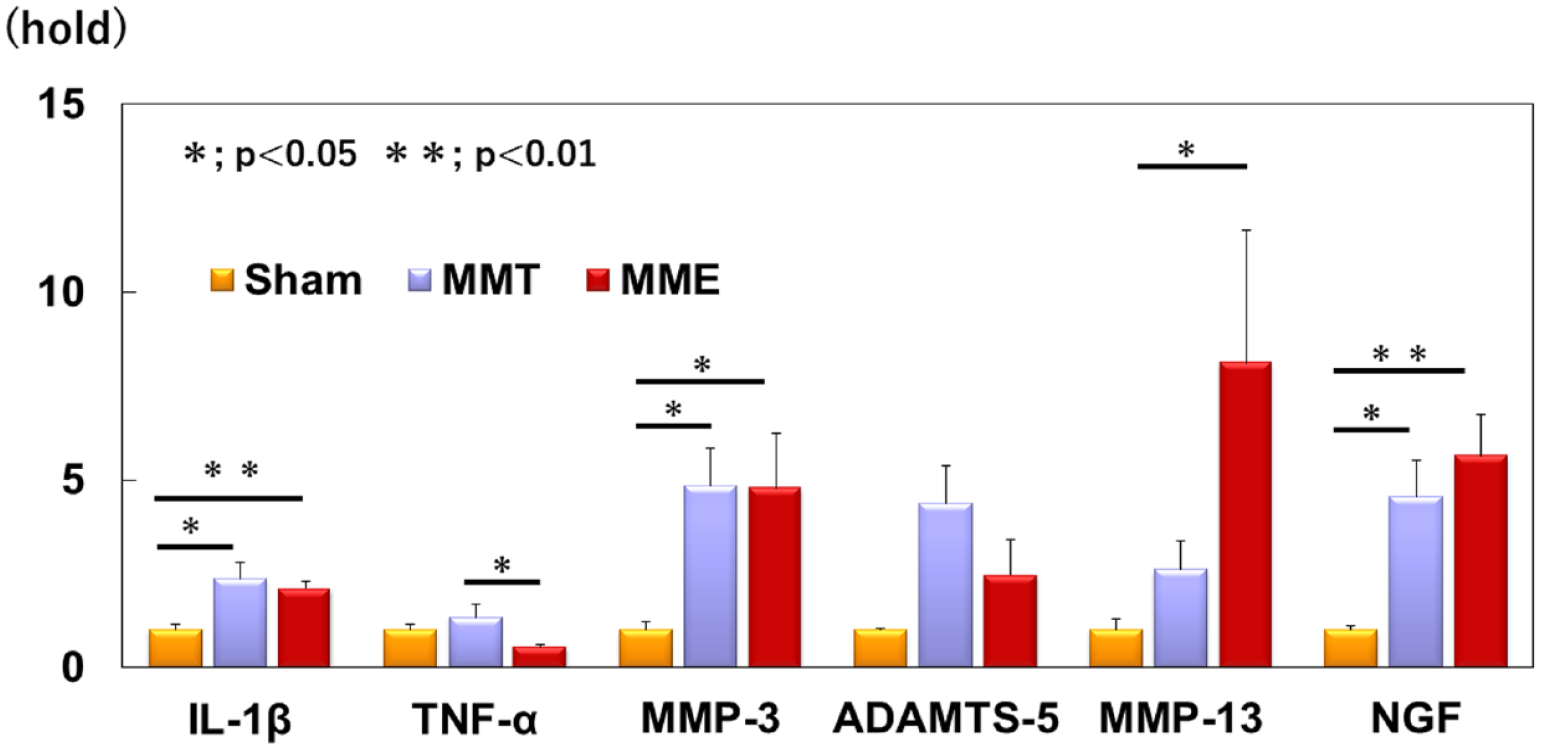
Gene expression analyses. Real-time polymerase chain reaction (PCR) results for gene expressions of pro-inflammatory genes: interleukin-1β (IL-1β), tumor necrosis factor-α (TNF-α), matrix-degrading enzymes: matrix metalloproteinase (MMP)-3, a disintegrin and metalloproteinase with thrombospondin-1 domains (ADAMTS)-5 and MMP-13 and pain-related molecules: nerve growth factor (NGF) at 8 weeks after the procedure. Data are expressed as the mean ± standard error (n = 9). The 1-way ANOVA with Fisher’s post hoc least significant differences test was used. *P < 0.05. **P < 0.01. ANOVA = analysis of variance.
Matrix-degrading enzymes
Gene expression of MMP-3 was significantly higher in the MMT and MME groups than in the sham group (Sham; 1.00 ± 0.22, MMT; 4.85 ± 0.99, MME; 4.78 ± 1.46, MMT; P < 0.05, MME; P < 0.05, vs. sham). ADAMTS-5 levels were significantly higher in the MMT group than in the sham group (Sham; 1.00 ± 0.05, MMT; 4.37 ± 0.99, MME; 2.48 ± 0.92, MMT; P < 0.05 vs. sham). Although gene expression of ADAMTS-5 was similarly upregulated in the MME group, there were no significant differences between the MMT and sham groups. MMP-13 expression was higher in the MME group than in the sham group (Sham; 1.00 ± 0.31, MMT; 2.61 ± 0.76, MME; 8.11 ± 3.54, MME; P < 0.05, vs. sham) (
Pain-related molecules
In the MMT and MME groups, gene expression of NGF was significantly upregulated compared with the sham group (Sham; 1.00 ± 0.12, MMT; 4.55 ± 0.96, MME; 5.64 ± 1.09, MMT; P < 0.05, MME; P < 0.01 vs. sham) (
Histological Analyses
The medial articular surface of the tibia showed grade 1 to 2 changes, mainly superficial fibrillation and partial degradation of the matrix in the sham group. In the sham group, almost no arthropathic changes occurred, whereas in the MMT group, complex cracks and large cavities owing to intercellular detachment were observed in relatively deep areas, indicating mainly grade 3 to 4 cartilage degeneration.
In the MME group, the most severe degeneration with extensive loss of cartilage matrix and replacement with fibrous cartilage tissue was observed. In some rats, cartilage was almost completely lost, and the transition to bone tissue was irregular. Although grade 4 changes were predominant, grade 6 rats with the most severe cartilage degeneration were observed only in the MME model (
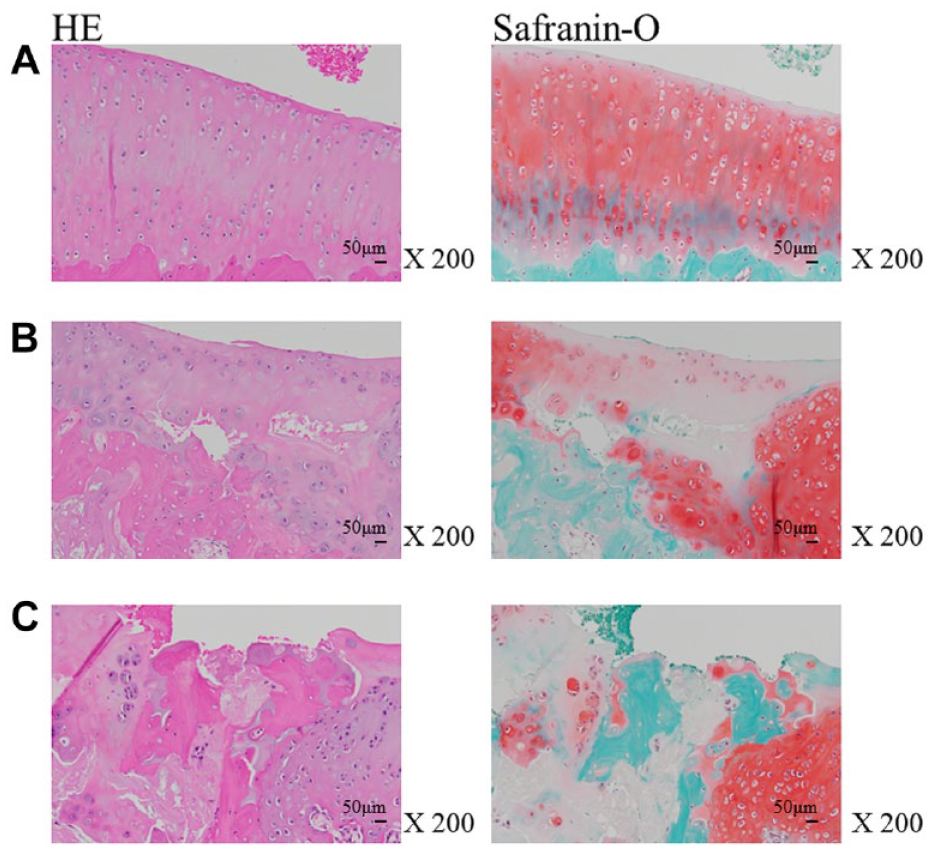
Histological images of the rat knee joint. Histological cartilage degeneration was evaluated by hematoxylin-eosin (HE) staining and Safranin-O staining. (
The OA grade in OARSI score of both MMT and MME was significantly higher than that of the sham (Sham; 1.25 ± 0.15, MMT; 3.33 ± 0.37, MME; 3.75 ± 0.42, MMT; P < 0.01, MME; P < 0.01 vs. sham) (
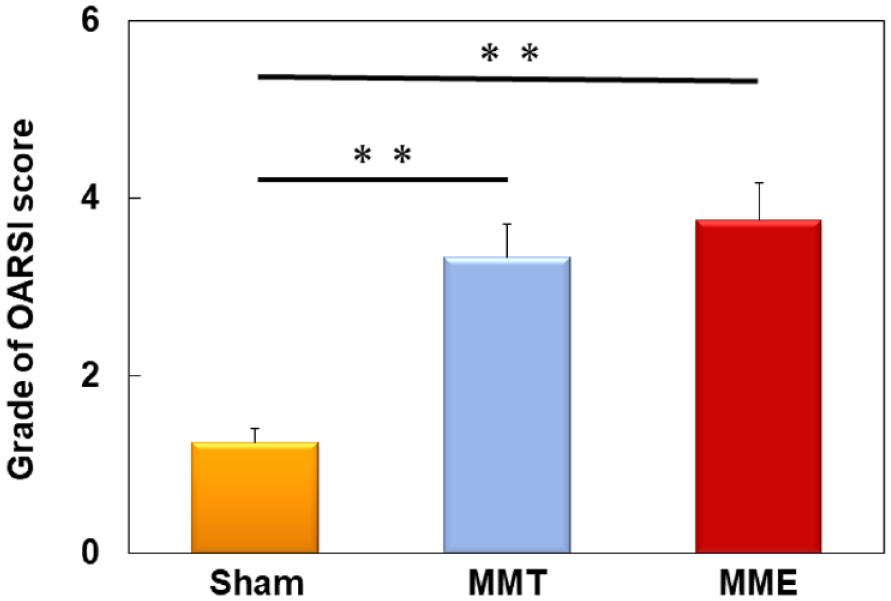
Histological analyses of the OA rat model. The Osteoarthritis Research Society International (OARSI) grades of both the MMT and MME models were significantly higher than that of the sham group. Data are expressed as the mean ± standard error (n = 9). The 1-way ANOVA with Fisher’s post hoc least significant differences test was used. **P < 0.01. OA = osteoarthritis; ANOVA = analysis of variance; MMT = medial meniscus transection; MME = medial meniscus extrusion.
Discussion
Gait Patterns in Knee OA
Sun et al. compared the gait of knee OA patients with that of healthy participants and reported that lower limb velocity was decreased in knee OA. 40 Kara et al. 33 also reported that gait parameters such as swing speed, print length, print width, and contact intensity obtained by CatWalk system are decreased in the rat OA model induced by MIA injection. The gait abnormality observed in our developed model is similar to the results of gait analysis in the patients of knee OA and MIA rat OA model. In this experiment, although the MMT model did not exhibit gait abnormalities as assessed by gait analysis using CatWalk, the MME model showed evidence of gait abnormalities: stand time, contact area, swing speed, and intensity. Vrinten et al. indicated that the decrease in intensity and stand time were involved in mechanical allodynia. 41 It is difficult to judge whether the gait abnormality in the MME model represents pain itself or escape behavior to avoid pain, but in any case, it is considered to be pain-related behavior.
Gene Expression in the Synovial Membrane of Knee OA
Synovitis occurs when mononuclear cells infiltrate the synovium and trigger the production of inflammatory mediators such as IL-1β, TNF-α, and chemokines. In this study, the gene expression of the inflammatory cytokine IL-1β was elevated in both the MMT and MME groups. Regarding MMP-3 and ADAMTS-5, which are involved in proteoglycan degradation,20,42 the gene expression levels of MMP-3 were higher in the MMT and MME models than those in the sham group, whereas MMP-3 gene expression was only increased in the MME model. The expression of MMP-13, which is associated with the degradation of type II collagen, 42 was elevated only in the MME group. Degradation of the extracellular matrix in the knee articular cartilage begins with proteoglycan degeneration in the early stages, followed by collagen destruction. We speculate that, in this study, since at the time of observation, cartilage degeneration was more advanced in the MME model, the expression of MMP-13 increased only in the MME model. NGF is known to be upregulated in chondrocytes and synovial fibroblasts of OA patients and is involved in the pathogenesis.21,43 In the present study, the gene expression of NGF was most elevated in MME models, and the elevation might result from inflammatory responses and mechanical stress.
Histology of Cartilage Degeneration of the Knee Joint
Bendle reported that MMT surgery causes histological arthropathic changes on cartilage. 44 In the present study, although the MMT model also showed cartilage degeneration 8 weeks after surgery, more severe morphological disintegration of the cartilage in the knee was observed in the MME model.
In the state of MME, there is an increased stress on the knee joint cartilage.11,12 Patients with MME demonstrated significant volume loss in the medial tibial cartilage during a 2-year follow-up. 45 Furthermore, a group of patients with medial meniscus posterior root tears, one of the causes of MME, were conservatively followed up for 5 years. The Kellgren-Lawrence (KL) grade, which averaged 1.5 at baseline, progressed to an average of 2.4 after 4.3 years of observation, and at the final follow-up, 78% of the patients exhibited a KL grade of 2 or higher. Moreover, 31% of the patients underwent total knee arthroplasty. 46 In the current study, the observed severe histological degeneration of articular cartilage in the MME model suggests a substantial increase in load on the articular cartilage in this animal model as well.
Clinical Relevance
In clinical practice, knee OA is characterized by pain, inflammation,14,15 and the progression of cartilage degeneration. 47 Miyagi et al. described that the medial meniscus manipulation model resembles the anatomical pathology of knee OA in human, but the MIA model is more effective in the evaluation of knee pain because it is likely to cause pain. 48 The MME is known to cause severe knee pain in clinical practice. 13 On MRI, it is associated with bone marrow lesion, 16 which are linked to the progression of knee OA and pain. In addition, it is a significant factor in the development of spontaneous osteonecrosis of the knee. 49 The MME model used in this study successfully induced rapid extrusion, leading to pain-related behaviors in walking analysis and showing the most advanced joint changes in histological evaluation. This aligns with the clinical characteristics of MME, which accelerate the symptoms and progression of knee OA. The MME is a major factor in the development of knee OA, and this model, which replicates the process, is valuable for evaluating the mechanisms of MME-induced knee OA and devising and validating pharmacological or novel surgical interventions for the MME.
Limitations
This study has several limitations. First, the animal model used was insufficient for clinical relevance, since rats are small animals whose knee joints are obviously different from those of humans. Second, the early evaluation is also important. This time, in order to reduce the effect of pain due to surgical invasion itself, the behavioral experiment was conducted from the second week after the operation, and gene expression and tissue evaluation are performed at the 8 weeks after the surgery. Although gene expression changes such as inflammatory cytokines and histological changes observed in knee OA were clearly seen in the MME model, a further time course evaluation of the cytokines and histological changes observed in this study would be needed. In future, precise examination of these changes over time might provide a deeper understanding of gait abnormalities, which are thought to be pain-related behaviors.
Conclusion
We demonstrated for the first time that MME resulted in progression of knee OA in the rat. Gait abnormalities inferred to be pain-related behaviors, increased gene expression of inflammatory cytokines and catabolic genes in the synovium, and histological evidence of articular cartilage degeneration were clearly shown in our developed MME model. The MME model, which might more closely mimic human knee OA, could be a useful model for elucidating the pathophysiology of knee OA and considering therapeutic management for knee OA.
Supplemental Material
sj-docx-1-car-10.1177_19476035231205680 – Supplemental material for Development of a Novel Rat Knee Osteoarthritis Model Induced by Medial Meniscus Extrusion
Supplemental material, sj-docx-1-car-10.1177_19476035231205680 for Development of a Novel Rat Knee Osteoarthritis Model Induced by Medial Meniscus Extrusion by Daisuke Fukui, Daisuke Nishiyama, Manabu Yamanaka, Hidenobu Tamai, Naoko Nishio, Mamoru Kawakami and Hiroshi Yamada in CARTILAGE
Footnotes
Acknowledgments and Funding
We sincerely appreciate the support and financial assistance of Eiji Sugimoto, founder of the Nissho Highway Co. We would like to thank Editage (![]() ) for English language editing. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by JSPS KAKENHI (Grant Number 20K18038).
) for English language editing. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by JSPS KAKENHI (Grant Number 20K18038).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by our institutional review board (Approval number 1017).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
