Abstract
Objective:
Osteoarthritis (OA) is a degenerative disease that causes serious damage to joints, especially in elderly patients. The aim of study was to demonstrate the effectiveness of intraarticular therapies that are currently used or recently popularized in the treatment of OA.
Design:
The baseline values were determined by walking the rats on the CatWalk system. Afterwards, a monosodium iodoacetate (MIA)-induced knee OA model was created with intraarticular MIA, and the rats were walked again on the CatWalk system and post-OA values were recorded. At this stage, the rats were divided into 4 groups, and intraarticular astaxanthin, intraarticular corticosteroid, intraarticular hyaluronic acid, and intraarticular astaxanthin + hyaluronic acid were applied to the groups, respectively. The rats were walked once more and posttreatment values were obtained. Nine different dynamic gait parameters were used in the comparison.
Results:
Significant changes were measured in 6 of the 9 dynamic gait parameters after the MIA-induced knee OA model. While the best improvement was observed in run duration (P = 0.0022), stride length (P < 0.0001), and swing speed (P = 0.0355) in the astaxanthin group, the results closest to basal values in paw print length (P < 0.0001), paw print width (P = 0.0101), and paw print area (P = 0.0277) were seen in the astaxanthin + hyaluronic acid group.
Conclusion:
Astaxanthin gave better outcomes than corticosteroid and hyaluronic acid in both dynamic gait parameters and histological examinations. Intraarticular astaxanthin therapy can be a good alternative to corticosteroid and hyaluronic acid currently used in intraarticular therapy to treat OA.
Introduction
Knee osteoarthritis (OA) is a chronic joint disease involving degeneration in the articular cartilage, synovitis, remodeling of the subchondral bone, and atrophy of the joint muscles, especially in the elderly population. 1 Knee OA patients generally complain of increased pain in their knees with movement.
The primary goal of knee OA treatment is to reduce pain and increase functional capacity. Knee OA patients are treated with a wide range of methods from medication to surgery. 2 Knee arthroplasty is a safe and successful surgical method for severe knee OA. 3 However, both its high cost and its inability to be applied in patients with surgical contraindications have increased the importance of nonsurgical treatments. 4 Hyaluronic acid (HA) and corticosteroid (CS) are intraarticular (IA) injection therapies frequently used clinically.5,6 Some studies show that HA injection reduces pain and increases functional capacity in patients with knee OA.7,8 According to a systematic review, IA CS therapy is extremely successful in reducing knee pain. 9 Additionally, in recent years, in vivo animal studies have examined the effects of astaxanthin (AX) as IA injection therapy.10,11 Previous studies have indicated that AX inhibits expression of matrix metalloproteinases (MMPs) in human chondrocytes through p38 and ERK1/2 signaling pathway suppression; thus, it can be used in knee OA treatment due to its antioxidant and anti-inflammatory properties.12,13 In in vivo animal studies, there has been a need to compare the effectiveness of AX therapy with the HA and CS injections that are currently accepted and applied clinically in patients with knee OA.
In studies on the knee OA mechanism, many animal models have been developed for OA induction. Anterior cruciate ligament transection, medial meniscus destabilization, and medial meniscal tear are frequently used surgical methods for OA formation.14-16 Otherwise, monosodium iodoacetate (MIA) injection is chemical method used to induce OA.17,18 For the MIA injection model, it has been reported that joint damage progressively develops through inhibition of glycolysis and proteoglycan synthesis in chondrocytes, behaviors related to pronounced joint pain are displayed, and the mechanism of injury is very similar to that of OA.17-19 In addition, the pain and pain behavior of the MIA injection model in the knee joint has been demonstrated in several studies with a paw withdrawal mechanical threshold.20,21 The fact that it causes severe knee OA and mimics the pathogenesis of degenerative OA are the biggest factors in choosing the MIA injection model in the present study.
Gait analysis systems are used to evaluate both static and dynamic functions of the lower limb in animal experimental models. 22 The CatWalk system (CatWalk, Noldus Company, Wageningen, the Netherlands) is a video-based automated gait analysis system designed to evaluate paw print and gait changes in rats. 23 It automatically records and analyzes many parameters, such as paw print area, run duration, stride length, and swing speed. The CatWalk system presents gait changes in animal pain models by quickly and objectively providing a wide range of static and dynamic gait-related parameters. Changes in these parameters are crucial in detecting pain behavior in animal models. In a study conducted by Gabriel et al. on rats, it was shown that the CatWalk system automatically and completely provides data on pain-related gait changes in a carrageenan-induced acute pain model. 24 It has been also a guide in evaluating both the changes caused by OA in rats and the efficacy of analgesic drugs in OA treatment.25-27
The aim of the present study was to evaluate the efficacy and success of different IA agents both alone and in combination by analyzing and comparing the data obtained from the CatWalk system in MIA-induced knee OA models.
Materials and Methods
Animals
The research was approved by the Research Ethics Committee for Animal Experiments of our institution. Forty-four male, mean 19.6 ± 3.4 weeks old (range 16-24), conventional Wistar rats weighing approximately mean 304.1 ± 27.5 g (range 272-331) were included in the study. Throughout the 8-week study period, 2 rats were euthanized to measure the baseline score and 2 rats were euthanized to determine the post-OA score. Therefore, the study was completed with 40 rats. During the study, the rats were kept in wire cages. For hygiene reasons, the cages were cleaned every 2 days. The rats were kept at room temperature (25 °C) in 60% humidity and on a 12-hour light/12-hour dark cycle simulating daylight, and 100% air exchange was performed 12 times in 1 hour. A pellet diet was given along with refined tap water. The water was supplied in autoclavable Makrolon bottles. Food was restricted 24 hours before and drink was restricted 6 hours before the gait analysis and injection applications. Before the study, the rats were walked on the platform of the device 3 times a day in a dark room to allow them to get used to the gait analysis device. If they walked to the end of the walking platform, they were rewarded with chocolate milk and cheese. At the end of the study, all rats were euthanized by cervical dislocation.
MIA-Induced Knee OA Animal Model
The animals were anesthetized with 100 mg/kg intramuscular sodium ketamine hydrochloride (Alfamine, Egevet, 100 mg/mL) and 20 mg/kg intramuscular xylazine (Alfazyne, Egevet, 2%) through a 30 G needle. Under general anesthesia, all rats were injected with 1 mg of MIA (Sigma-Aldrich Inc., St. Louis, MO) in 30 μL of saline into the left knee with a 27 G needle along the infrapatellar ligament with the knee joint flexed at 90°.28,29 Four weeks were allowed for OA formation after the injection.
Application of IA Injection Therapies
Similar to the MIA injection, the animals were anesthetized with 100 mg/kg intramuscular sodium ketamine hydrochloride (Alfamine, Egevet, 100 mg/mL) and 20 mg/kg intramuscular xylazine (Alfazyne, Egevet, 2%) through a 30 G needle. Forty rats were divided into 4 groups with 10 rats in each. Optimal doses of injection therapies to be administered to rats by a veterinarian were determined before. Under general anesthesia, each injection consisted of 0.3 mL in the left knee of the rats with OA; AX (150 μM/mL) 11 was injected in the first group, CS (2 mg/mL) 30 in the second group, HA (50 μg/mL) 31 in the third group, and AX + HA combination in the fourth group via a 27 G needle along the infrapatellar ligament with the knee joint flexed at 90° in accordance with the literature. After the procedure, 4 weeks were allowed for the efficacy of the therapy.
Gait Analysis
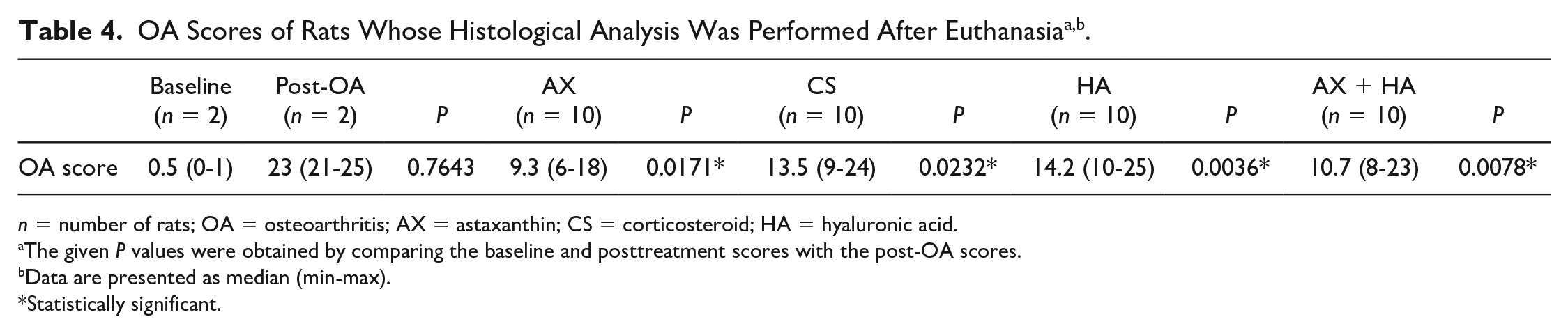
The gait of the rats was analyzed using the CatWalk system ( Fig. 1a and b ). This system is described in detail in Hamers et al. 23 Briefly, rats are placed on a glass plate in a dark room and are allowed to walk freely. A light beam from a fluorescent lamp is propagated across the entire glass plate. The light beam is fully reflected through the glass plate. When a paw touches the glass plate, the light beam is reflected downwards. As a result, a sharp and bright image of the paw print is formed. The entire walk is recorded with a video camera. All data are collected and analyzed in detail using the CatWalk software. 32 In our research, evaluations were made from the data obtained: run duration, run average speed, mean stance, maximum contact intensity, paw print length, paw print width, paw print area, stride length, and swing speed.
(
Forty-four rats were primarily run on the CatWalk platform, and the data obtained were recorded as baseline values. Later, 2 rats were euthanized for histological examination. At this stage, MIA was injected into the left knee of 42 rats, and after a 4-week waiting period, an MIA-induced knee OA model was created. Two rats were euthanized for histological examination again. Then the rats were run again on the CatWalk platform and post-OA data were recorded. After walking, 40 rats were divided into 4 groups with 10 rats in each group, and each group underwent the previously mentioned treatments. Four weeks were allowed for the treatment to take effect. Subsequently, the rats were run on the CatWalk platform for a third time to obtain posttreatment data. After this step, all rats were euthanized for histological analysis.
Histological Analysis
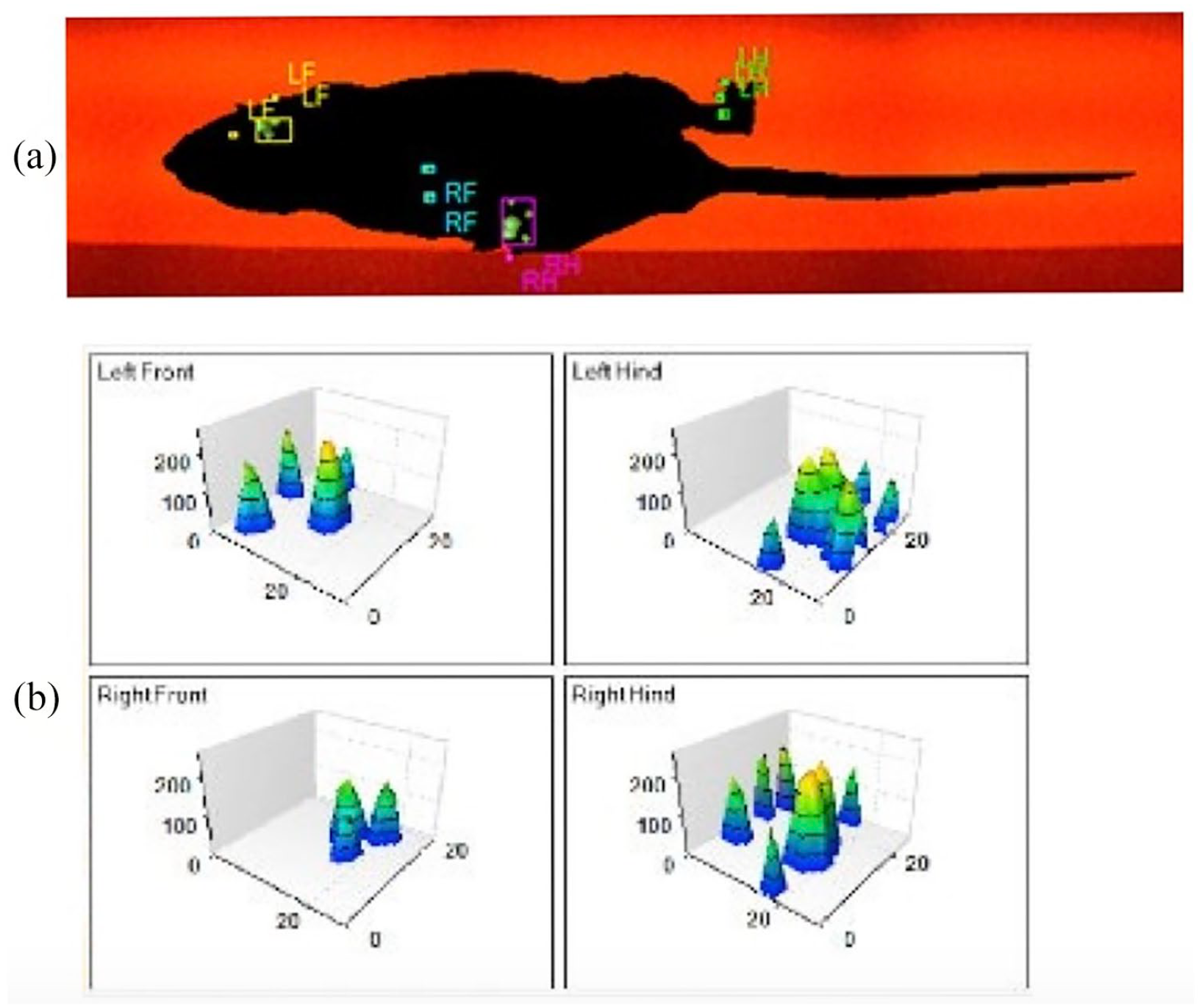
Histological analysis was carried out to determine both the destruction caused by MIA and the regenerative effects of the injection therapies on the articular cartilage (Fig. 2a-f). Before OA induction, after OA induction, and 4 weeks after the treatment phase, the rats were euthanized by cervical dislocation, and the affected left knee joints were dissected, fixed in 10% phosphate-buffered formalin for 24 hours, and decalcified in 5% formic acid for 2 weeks. Sections (thickness, 7 μm) were stained with toluidine blue to assess both glycosaminoglycans loss after MIA injection and articular cartilage regeneration after injection therapies. All sections were observed under a Carl Zeiss Axiolab 5 microscope (Carl Zeiss, Deisenhofen, Germany). All stained sections were histologically evaluated and were scored on a scale of 0 to 25 by double-blind observation according to the rat OA scoring system shown in Table 1 . 33
Microscopic view of a cross-section of a toluidine-stained knee joint of a rat presented at all stages. (
The Rat Osteoarthritis Scoring System Used in the Research (0-25).

Statistical Analysis
The data are expressed as mean ± standard deviation (SD) or median (minimum-maximum) values. First, the suitability of the data to normal distribution was evaluated with the Kolmogorov-Smirnov test, and it was seen that all data were compatible with normal distribution. A paired sample t-test was used for comparisons between baseline and post-OA data. One-way analysis of variance (ANOVA) was used followed by Bonferroni’s post hoc test in which the 4 posttreatment groups were statistically compared with each other. The Wilcoxon rank-sum test was used to compare OA scores. The analyses were carried out using IBM SPSS Statistics for Windows, version 21 (IBM Corp., Armonk, NY). A value of P < 0.05 was accepted as significant in all statistical tests.
Results
The baseline and post-OA values obtained in the CatWalk system for the rats are shown in Table 2 . Nine parameters obtained from the CatWalk system were evaluated, and all of these parameters are data for the affected knees of the rats. When compared with the baseline value, there was a significant increase in run duration (6.63 ± 0.48 to 8.79 ± 0.52, P = 0.0017) after MIA-induced OA. On the other hand, run average speed (18.80 ± 1.08 to 14.43 ± 1.37, P < 0.0001), paw print length (1.76 ± 0.11 to 1.21 ± 0.11, P = 0.0025), paw print width (1.47 ± 0.08 to 1.36 ± 0.09, P = 0.0029), paw print area (2.58 ± 0.18 to 1.94 ± 0.15, P = 0.0107), and swing speed (123.14 ± 3.98 to 99.03 ± 4.26, P = 0.0026) decreased significantly. The changes in mean stance, maximum contact intensity, and stride length were not significant.
Baseline and Post-OA Data Obtained in the CatWalk Gait Analysis System of Left Knees of Rats a .
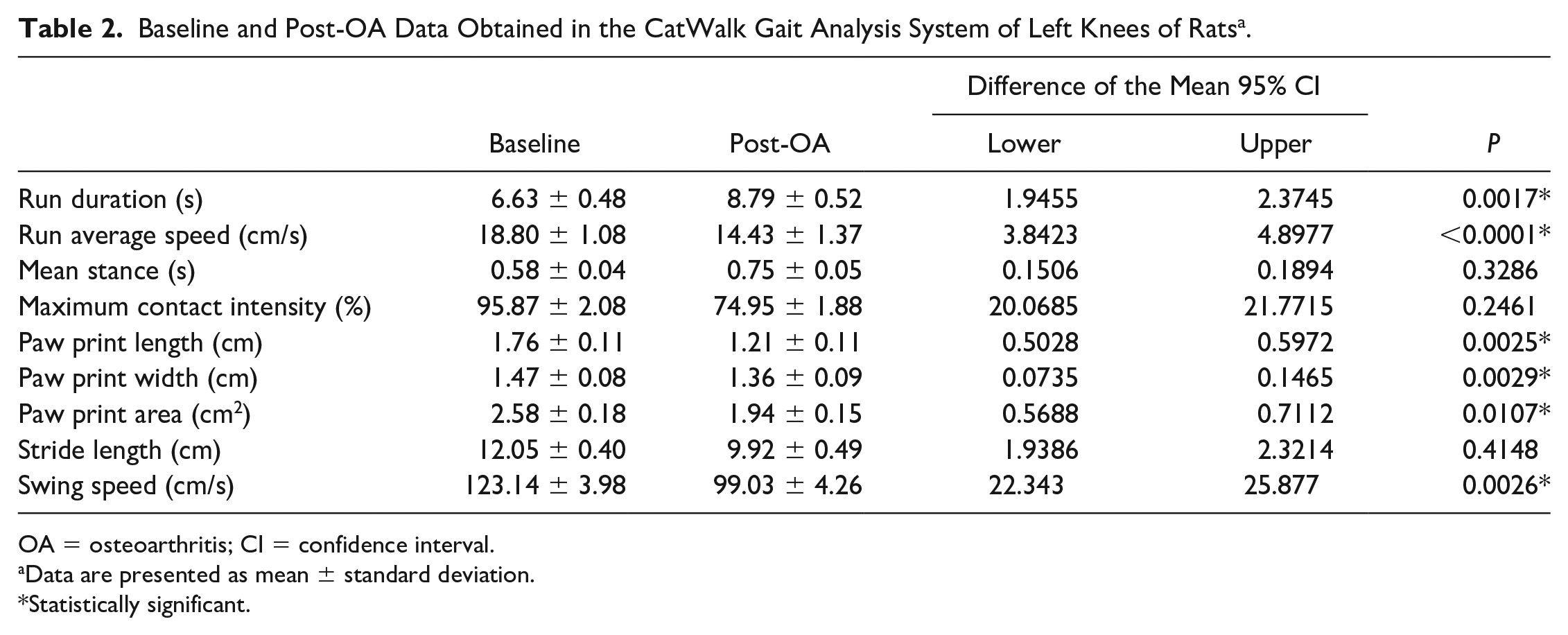
OA = osteoarthritis; CI = confidence interval.
Data are presented as mean ± standard deviation.
Statistically significant.
The data obtained for each group after the therapeutic injections are presented in Table 3 . All the data obtained were for the left knees and the same parameters were examined. The greatest improvements in run duration (7.12 ± 1.07, P = 0.0022), stride length (11.06 ± 1.05, P < 0.0001), and swing speed (115.01 ± 15.76, P = 0.0355), that is, the closest to baseline values, were seen in the AX group and they were significant. In paw print length (1.59 ± 0.30, P = 0.0005), paw print width (1.42 ± 0.16, P = 0.0101), and paw print area (2.22 ± 0.25, P = 0.0277), the most significant improvements were detected in the AX + HA group. Mean stance (0.61 ± 0.06, P < 0.0001) was closest to the baseline value in the CS group. Changes in run average speed and maximum contact intensity were not significant.
Data Collected after Injection Therapy from the CatWalk Gait Analysis System of Left Knees of the Rats a .
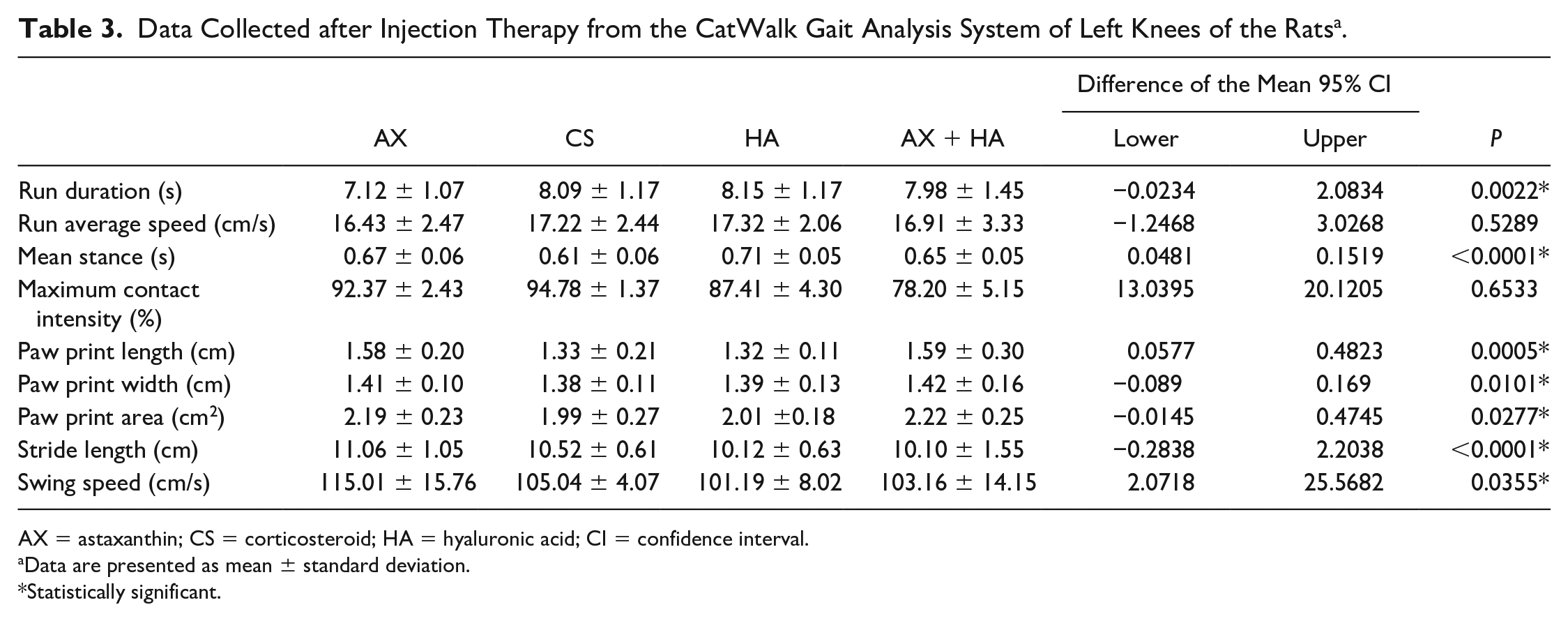
AX = astaxanthin; CS = corticosteroid; HA = hyaluronic acid; CI = confidence interval.
Data are presented as mean ± standard deviation.
Statistically significant.
Calculated according to the rat OA scoring system presented in Table 1 , OA scores obtained after the histological examination are shown in Table 4 . There was an enormous increase in the score after MIA-induced OA to the baseline score but it was not significant. The greatest improvement in OA score after injection treatments was seen in the AX group (9.3, P = 0.0171), followed by the AX + HA (10.7, P = 0.0078), CS (13.5, P = 0.0232), and HA (14.2, P = 0.0036) groups.
n = number of rats; OA = osteoarthritis; AX = astaxanthin; CS = corticosteroid; HA = hyaluronic acid.
The given P values were obtained by comparing the baseline and posttreatment scores with the post-OA scores.
Data are presented as median (min-max).
Statistically significant.
Discussion
Many techniques have been described in the literature to create a knee OA model in animal studies. Myers et al. reported that instability in the knee joint after anterior cruciate ligament transection caused damage to the articular cartilage, resulting in OA. 34 Marijnissen et al. described the “Groove” model that predisposes to OA by causing articular cartilage damage. 35 Bendele reported that rupture of the medial meniscus caused rapid degeneration of the joint fracture in rats and subsequent OA. 16 Another study used the complete Freund’s adjuvant-induced OA model. 36 In MIA-induced knee OA, chondrocyte death occurs as a result of glycolysis inhibition and avascular cartilage is formed. The MIA-induced OA model has been found to be superior to other methods in evaluating knee pain. 37 In addition, one of the most important features of the MIA-induced knee OA model is that the pathological changes in the articular cartilage are very similar to those of degenerative OA. 38 Therefore, MIA-induced knee OA rat model is frequently used in studies.26,37,39 In our study, MIA was also chosen for OA induction because it was an important criterion for us to have effects similar to those of degenerative OA. Due to these properties, MIA is frequently used in rat knee OA models during the investigation of various IA therapies used in the treatment of OA.30,40,41 The success of MIA in OA induction was demonstrated in the present study both by changes in digital gait parameters in the CatWalk system and by histological examinations.
The CatWalk gait analysis system works by recording each step of rats on a walking platform and automatically classifying the data generated by those steps. There are studies in the literature regarding the use of the CatWalk system in the evaluation of rat OA models.24,27,28,42 Ferland et al. showed a significant difference in dynamic gait parameters between injured knees and contralateral knees in the MIA-induced knee OA rat model. 26 Rats begin to limp post-OA in their knees and show a reluctance to put weight on the affected limb secondary to pain. This results in a decrease in swing speed and an increase in run duration in the affected limb. 26 In a CatWalk analysis by Parvathy and Masocha, after Freund's adjuvant-induced knee OA rat model, decreases in swing speed and paw print area and an increase in run duration were observed post-OA. 36 In another study, after MIA-induced knee OA, swing speed and print area were significantly decreased. 37 In our research, a total of 9 parameters including swing speed, run duration, and paw print area were analyzed with the CatWalk system, and a significant change was observed in 6 of these parameters post-OA. The CatWalk gait analysis system is practical, convenient, and reliable for detecting post-OA changes in the rat OA model.
Lifestyle changes, physical therapy, and nonsteroidal anti-inflammatory drugs are inadequate, especially in patients with knee OA who have contraindications for knee arthroplasty and who do not agree to surgical treatment; therefore, IA injection treatments are becoming increasingly important.43,44 One of these is HA, which acts by increasing proteoglycan synthesis, decreasing soluble pro-inflammatory mediators and degradative enzymes activity, and regulating the behavior of immune cells. 45 It also protects joint chondrocytes against oxygen-induced free radical damage by showing antioxidant properties. 46 In studies conducted, IA HA injection therapy was applied as a treatment in rat knee OA models and positive results were reported.11,47,48 Another important IA treatment agent is CS, which inhibits pro-inflammatory cytokine secretion and angiogenesis, and it relieves synovitis and pain seriously with a strong anti-inflammatory effect. 49 Studies have shown that IA CS treatment is used in rat OA models, and there is significant relief of pain.50,51 In our study, IA CS was applied to the second group and IA HA was applied to the third group, and according to the post-OA, run duration, mean stance, paw print length, paw print width, paw print area, stride length, and swing speed recovered significantly. In addition, amelioration was observed in the scores obtained after histological analysis compared to the post-OA scores. Although IA HA and CS give good outcomes in knee OA treatment, they have some disadvantages. The important drawback of IA HA is the limited duration of stay in the joint and rapid removal from the body. 44 The negative feature of CS is that it can cause muscle weakness and hormone imbalance in the case of long-term use. 52 For these reasons, although they are used in IA therapies, they are not the gold standard.
In recent studies, it is a matter of discussion whether AX can be used in IA injections. In the study by Chen et al., AX was shown to reduce the expression of MMP-1, MMP-3, and MMP-13 as well as the phosphorylation of 2 mitogen-activated proteins kinases in IL-1β-stimulated human chondrocytes. 13 AX also blocks the degradation of 1κB-α. 13 In another study, it was reported that AX provides cartilage homeostasis through modulation of Nrf2 signaling. 12 In vivo studies found that AX suppresses inflammation by reducing pro-inflammatory cytokines such as TNF-α, IL-1β, IL-6, and IFN-γ due to its powerful antioxidant effect, and it can be used in the treatment of OA in the early stage.10,11,13,53 AX can be applied alone or in combination with other molecules. In a recent study by Park et al., the combination of AX and HA was applied to the MIA-induced rat OA model, and they found pain relief, minimization of articular cartilage damage, and improvement in functional status. 54 In our study, IA AX injection was applied to the first group and IA AX+HA was applied to the fourth group. Considering the significant parameters, the closest data to baseline values in run duration, stride length, and swing speed were obtained in the AX group and in paw print length, paw print width, and paw print area were obtained in the AX + HA group. It was observed that there was greater improvement in these 2 groups compared to the HA and CS groups. In addition, histological analysis also found better results in OA scores in these 2 groups compared to IA HA and CS. Volunteers were selected and safety tests were performed on AX, low dose (3.85 mg AX) was given to one group and high dose (19.25 mg AX) was given to the other group for 29 consecutive days, and no disease or intoxication was observed in either group. 55 According to a systematic review of clinical studies, no serious adverse effect were reported in patients using oral and topical AX. 56 Although there is not much data in the literature on the IA form of AX, its reliability in oral and topical forms puts it one step ahead in terms of possible side effects. 54
The study has some limitations. First of all, rats have to undergo a certain amount of training to get used to the CatWalk device and walk on it without error. Second, in the study, the posttreatment groups were compared with each other and there was no control group. Finally, although there were 10 rats in each treatment group, more significant data can be obtained with a higher number of subjects.
Conclusion
AX, which is a powerful antioxidant and is still in a more experimental phase, provided both functional and histological improvement in knee OA rat models and gave satisfactory results. IA AX therapy can be a good alternative to IA HA and CS, which are currently used in the clinic in the treatment of OA. In order to better determine the efficacy and safety of IA AX, experiments with larger samples and clinical studies in humans are needed.
Supplemental Material
sj-pdf-1-car-10.1177_19476035211046042 – Supplemental material for Evaluation of Different Intraarticular Injection Therapies with Gait Analysis in a Rat Osteoarthritis Model
Supplemental material, sj-pdf-1-car-10.1177_19476035211046042 for Evaluation of Different Intraarticular Injection Therapies with Gait Analysis in a Rat Osteoarthritis Model by Ceyhun Çağlar, Halil Kara, Okan Ateş and Mahmut Uğurlu in CARTILAGE
Supplemental Material
sj-pdf-2-car-10.1177_19476035211046042 – Supplemental material for Evaluation of Different Intraarticular Injection Therapies with Gait Analysis in a Rat Osteoarthritis Model
Supplemental material, sj-pdf-2-car-10.1177_19476035211046042 for Evaluation of Different Intraarticular Injection Therapies with Gait Analysis in a Rat Osteoarthritis Model by Ceyhun Çağlar, Halil Kara, Okan Ateş and Mahmut Uğurlu in CARTILAGE
Footnotes
The reported study was conducted in Ankara City Hospital.
Acknowledgments and Funding
We would like to thank Prof. Dr. Siyami Karahan for histological analysis and microscopic views. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Ankara Yıldırım Beyazıt University Scientific Research Projects Coordination Unit.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The Research Ethics Committee for Animal Experiments at our institution approved of the study.
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.