Abstract
Purpose
The present study investigated the effects of hyperbaric oxygen (HBO) on human chondrocyte proliferation and gene expression patterns.
Methods
Chondrocyte cultures were transferred to a HBO chamber and exposed to 100% oxygen for 7 consecutive days. Within groups, pressure was varied between 1 and 2 atm and duration of HBO administration was varied among 60, 90, and 120 minutes. Cell counts were performed using the WST-1 assay at 1, 3, 5, and 7 days after initiation of HBO treatment to obtain data to plot a growth curve. Gene expression of apoptosis markers PARP and caspase 3, as well as cartilage specific proteins collagen II and COMP, were detected by reverse transcription polymerase chain reaction.
Results
The experiments showed that in vitro administration of HBO inhibit chondrocyte growth. When applied compression was increased up to 2 atm, chondrocyte cell count was reduced by half at days 3 and 7 in association with an upregulation of the apoptosis markers PARP and caspase 3 as well as the cartilage specific proteins collagen II and COMP. No significant differences were monitored from varied duration of daily treatment.
Conclusion
Chondrocyte growth was inhibited in vitro by treatment of HBO. This inhibitory effect was even increased by elevating the applied pressure, while molecular testing showed reduced chondrocyte growth. Higher levels of HBO inhibited cell growth even more, but up-regulation of apoptosis specific markers and cartilage specific proteins were seen during administration of high oxygen levels. Thus, it has to be evaluated that there is a critical level of hypo-/hyperoxia required to stimulate or at least maintain chondrocyte cell proliferation.
Introduction
In the past decade, the intensity of competitive sports has taken on a whole new meaning with an increase of injuries to athletes. These sports injuries range from broken bones to ruptured ligaments, tendons, and cartilage trauma. 1 Different pathways have been held responsible for articular cartilage injury in athletes; one of the major causes2,3 is chronic repetitive loading of the articular cartilage, progressive articular cartilage degradation with accumulation of catabolic enzymes and cytokines, fragmentation of collagen and aggrecan, and resulting fissuring and progressive breakdown of the articular surface.
The healing of a sports injury follows a fairly constant pattern irrespective of the underlying cause. Three phases have been identified in this process: the inflammatory phase, the proliferative phase, and the remodeling phase. Oxygen has an important role in each of these phases. 2 In the inflammatory phase, the hypoxia-induced factor-1a, which promotes the glycolytic system, vascularization, and angiogenesis, has been shown to be important. In the proliferative phase, the oxygen supply to the injured area in the musculoskeletal tissue is gradually raised and is essential for the synthesis of extracellular matrix components such as fibronectin and proteoglycan. In the remodeling phase, tissue is slowly replaced over many hours using the oxygen supply provided by the blood vessel already built into the organization of the musculoskeletal system. If the damage is small, the tissue is recoverable with nearly perfect organization; however, if the extent of the damage is large, a scar (consisting mainly of collagen I) may replace tissue. 3
Collagen accounts for two-thirds of the dry weight of adult articular cartilage and confers on the tissue the capacity to withstand tensile and shear forces. Within articular cartilage, type II collagen is the dominant collagen, accounting for more than 90% of the total collagen.4,5 The nascent type II collagen fibril is a heteropolymer, with collagen IX molecules covalently linked to the surface and collagen XI forming the filamentous template of the fibril as a whole.
Type IX collagen is a fibril-associated collagen with interrupted triple helix that is proposed to stabilize the fibrillar and proteoglycan networks via lateral association with type II and type XI collagen, although only 1% to 5% of the total collagen of mature cartilage consist of this type of collagen. 5 Expression of types I and/or III and V collagen indicates phenotypic alteration to fibroblastic cells, and type X collagen is characteristic for hypertrophic chondrocyte.6,7
Successful repair of full-thickness defects in articular cartilage has been a difficult goal to achieve. Spontaneous repair often fails to completely fill the defect and the new tissue is composed of fibrocartilage rather than hyaline cartilage.8-13
Therefore, improvement in the quality of regenerating cartilage is necessary. Tissue engineering strategies, such as approaches using cellularized scaffolds, have been explored for cartilage replacement. The challenge, however, remains to be the production of a cartilaginous tissue incorporating functional chondrocytes, with the size and thickness big enough to be compatible with the replacement of articular defects.
Hyperbaric oxygen (HBO) therapy is defined as the administration of 100% oxygen at atmospheric pressures greater than sea level. It thus increases dissolved oxygen content to above physiologic levels and has been available and advocated for many years as a therapeutic modality for many clinical conditions such as carbon monoxide poisoning decompression sickness, arterial gas embolism, osteomyelitis, wound healing (diabetic foot ulcers), malunion and nonunion fractures, acute traumatic ischemia, and muscle damage.14-24. Although the exact mechanisms remain unclear, the presence of oxygen is known to speed the process of wound healing.25,26 It may be due to its role the production of collagen matrix and subsequent angiogenesis,12,19 the presence and beneficial effects of reactive oxygen species (ROS),27-29 or other unknown reasons. Little research has been done on the effect of HBO on the musculoskeletal tissue thus far: Positive effects on chondrocyte and fibroblast were shown by Yuan et al.30-32 and Chan et al., 33 while Seol et al. 34 demonstrated prevention of chondrocyte death and cartilage degradation by hypoxia and Anderson et al. 35 showed promotion of the articular chondrocyte phenotype in highly chondrogenic cells. Hsieh et al. 36 demonstrated that HBO significantly promoted osteoblast proliferation and cell cycle progression, increased mRNA expression of fibroblast growth factor as well as protein expression levels. Studies have shown close associations between HBO, IL-1b, nitric oxide (NO), and caspase. HBO treatment increases tissue/microvascular partial pressure of oxygen, 37 decrease IL-1b secretion from monocytes 38 and degenerated disc cells, 39 suppress apoptosis of degenerated disc cells, 40 reduce caspase 3 activities in rat brain, 41 and as earlier mentioned already suppress inducible nitric oxide synthase (iNOS) expression and apoptosis of chondrocytes in rabbit cartilage defects 31 and cerebral endothelial cells. 42
Concurrently, negative effects of the administration of HBO have been shown,43-46 which might be due to the toxic effects of hyperoxia from ROS caused by the mechanically induced effects of high atmospheric pressure. There seem to be a delicate balance between having enough and too much oxygen and atmospheric pressure when promoting cell growth. 47 Articular chondrocytes are specialized cells that can survive in an avascular environment and obtain oxygen via the diffusion pathway. The normal synovium is highly vascularized to satisfy the metabolic demands of the avascular cartilage; however, in the case of joint disease, the oxygen gradient could be disturbed. Cell death in osteoarthritic cartilage possesses certain features of apoptosis or programmed cell death. Apoptosis of chondrocytes can be induced by localized hypoxia and NO production and is mediated by a cascade of aspartate-specific cysteine proteases or caspases through an extrinsic and intrinsic pathway. Caspase 3, both in extrinsic and intrinsic pathways, is responsible for the nuclear changes in apoptosis.48-50 Increased caspase expression has been correlated with reduced cell density in human osteoarthritic cartilage. 51 It can be assessed by measuring parameters like DNA degradation, disintegration of lipid bilayer, mitochondrial activity, or immunohistochemical expression. Caspase activity and caspase processing was shown to be specific in the detection of chondrocyte apopotosis. 52 Poly-ADP-ribose polymerase 1 (PARP-1) applies as an indicator for DNA single strand breakage or its repair mechanisms. 53
Oxidant conditioning protects the cartilage from mechanically induced damage. Suppression of NO production after chondral trauma, or during the early stage of osteoarthritis (OA), may inhibit the initiation or progression of OA.
Materials and Methods
Cellplating
Human chondrocytes that were partly purchased (Provitro, Berlin) and partly harvested from one donor were seeded in monolayer triple-flasks at a density of 104 cells/cm2 in Dubecco’s modified Eagle medium/HamF12-medium, containing 10% fetal calf serum (PAA, Pasching, Austria), 1% amino acids, 1% glutamine, 25 µg/mL ascorbic acid, 2500 µg/mL amphotericin B (Biochrome, Germany), and 50 U/mL penicillin/streptomycin (Sigma, Germany), while cells from different origin were cultivated separately. Cell viability was confirmed with trypan blue dye exclusion. Chondrocytes were then diluted in growth medium at densities appropriate for each assay. For the proliferation assay, the chondrocytes were seeded in 96-well plates (Nunc, Germany) at a density of 2.5 × 103 cells/well. For the gene expression analysis, the chondrocytes were seeded in 10 cm petri dishes (Nunc, Germany) at a density of 104 cells/dish.
HBO Administration
Chondrocyte cultures within the 96-well plates were transferred to an HBO chamber (Haux, Karlsbad, Germany) which was sealed, flushed with 100% oxygen and pressurized to the appropriate level as measured by an attached barometer ( Fig. 1 ). To avoid compression damage to cell membranes, pressure was built up by 0.1 atm per second. Treatment groups were exposed to 100% oxygen at 1.0 and 2.0 atm daily for 60, 90, or 120 minutes for 7 consecutive days, while control plates were exposed to ambient room air under sterile conditions for the duration of the experiment. Subsequently, decompression was accomplished in intervals of 0.1 atm per second and both the experimental groups and control groups were transferred in the incubator without delay.

Hyperbaric oxygen (HBO) chamber (Haux, Karlsbad, Germany): Treatment groups were exposed to 100% oxygen at 1.0 and 2.0 atm daily for 60, 90, or 120 minutes for 7 consecutive days, while control plates were exposed to ambient room air under sterile conditions for the duration of the experiment.
Chondrocyte Proliferation
Cell counts were performed using the WST-1 assay (Cell Proliferation Reagent WST-1, Roche, Germany) at 1, 3, 5, and 7 days after initiation of HBO treatment to obtain data to plot a growth curve. The WST-1 assay is a colorimetric assay used for the quantification of cell proliferation and cell viability based on the cleavage of the tetrazolium salt WST-1 by mitochondrial dehydrogenases in viable cells. The WST-1 agent is converted to orange formazan by the mitochondria of active cells. Hence, it can be used to analyse the activity of cells. The level of orange product increases when cell activity increases and can be quantified using a spectrophotometer at a wavelength of 460 nm.
Gene Expression Analysis of the Chondrocytes
In a LightCycler (Roche Applied Science, Mannheim, Germany), cyclophilin B, cartilage-specific proteins like collagen II and COMP, and additionally apoptosis specific proteins caspase-3 and PARP-1, were used as primers (Search-LC Heidelberg, Germany) and total RNA was directly isolated from chondrocytes HBO administration on day 1, 4, and 7 of HBO administration using the RNeasy mini Kit (Qiagen, Hilden, Germany). For cDNA systhesis, 1 µg of total RNA was reverse-transcribed using the Transcriptor High Fidelity cDNA Synthesis Kit (Roche Applied Science). Amplification reactions were performed using a LightCycler FastStart DNA Master SYBR Green I Kit (Roche applied Science). One microliter of the 1/10 diluted sample was used for the quantitative 2-step polymerase chain reaction (PCR) using the following protocol (10-minute step at 95°C, following by 40 cycles of 60 seconds at 58°C and 16 seconds at 72°C). Assays were performed in duplicate and relative quantification of the target gene expression was generated normalizing to cyclophilin B.
Statistical Analysis
Statistical analysis was performed using the software program GraphPad (GraphPad Software Inc, San Diego, CA). All data were analyzed by using Wilcoxon matched pairs test, a nonparametric t test with paired samples with significance level set at P < 0.05.
Results
Chondrocytes cultures were maintained in the in vitro cell culture model successfully for the duration of the experiment, while the control cells kept at ambient room air demonstrated roughly linear growth.
At day 1, all cells (HBO-treated and controls) showed a relative decrease in proliferation even though after 5 days the nontreated and the 2-atm-group doubled in number.
From day 3 on, all HBO-treated cells demonstrated significantly decreased proliferation compared with controls ( Fig. 2 ), which then remained static from day 5 of the experiment.
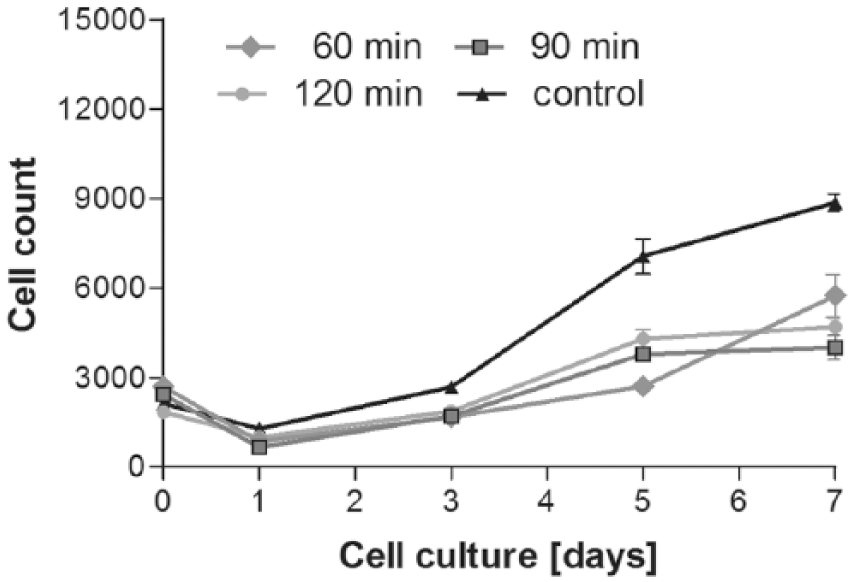
Impact of treatment time with hyperbaric oxygen (HBO; 60 vs. 90 vs. 120 minutes) on chondrocyte cell count. At day 1, all cells (HBO-treated and controls) showed a relative decrease in proliferation. Values are expressed with * showing significant difference (P < 0.05).
While in the 3-atm group chondrocyte population and proliferation were decreased from the beginning and further halved at days 3 and 7, no significant differences were monitored among groups with varied duration of HBO administration (60, 90, and 120 minutes) ( Fig. 3 ).
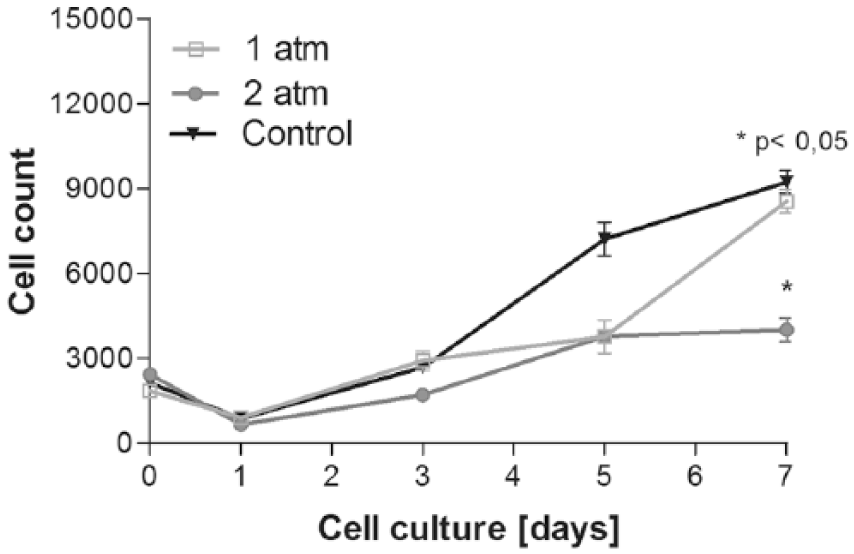
Impact of treatment pressure (1 vs. 2 atm) within the chamber on chondrocyte cell count. Values are expressed with * showing significant difference (P < 0.05).
Cartilage consists mainly of collagen II, IX, und XI. As up to 95% of collagen is the dominant type II we decided to identify the gene expression pattern of collagen II representatively for evaluating the influence of hyperbaric oxygenation on the synthesis of collagen in human chondrocytes. Even before administering pressure, collagen synthesis of collagen II was measured to confirm the chondrocytes as cells of hyaline joint cartilage ( Fig. 4 ). However, collagen type II was only isolated in the donor-harvested human chondrocytes while even in Light Cycler PCR merely no collagen type II was detected in the industrial purchased chondrocytes ( Fig. 5 ).
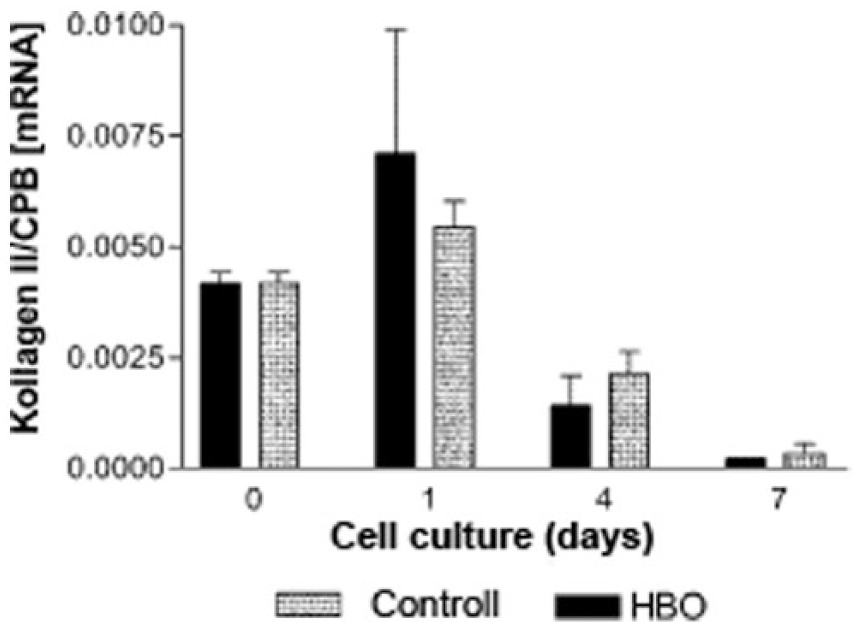
Differences between hyperbaric oxygen (HBO) group (300 atm/90 min) versus control in gene expression of collagen II. Values are expressed with * showing significant difference (P < 0.05).
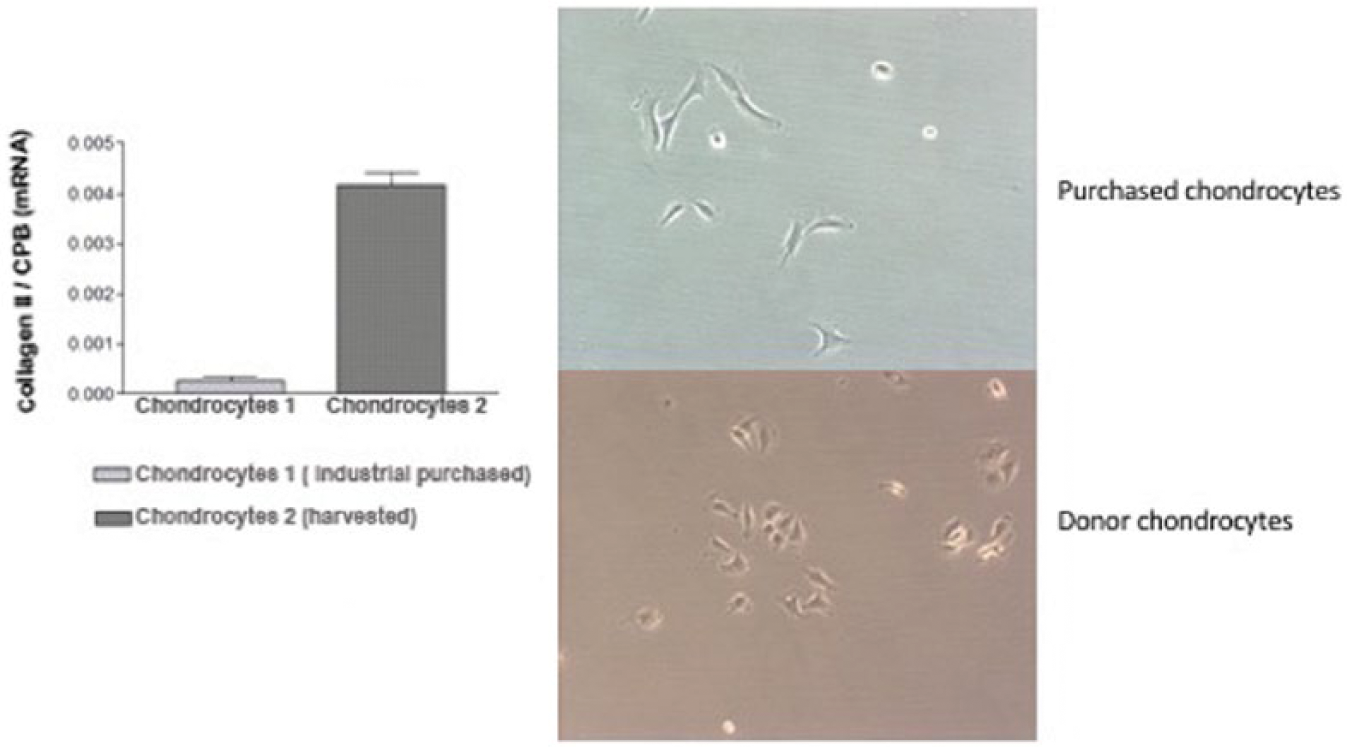
Differences in collagen II expression in harvested and purchased chondrocytes. Collagen II expression in patient harvested chondrocytes was enhanced.
Cartilage oligomeric matrix protein (COMP) is a glycoprotein of extracellular matrix and is used as an early marker for chondrocyte proliferation. While COMP synthesis was enhanced after the first HBO treatment, gene expression diminished within the following days in both groups (treatment/control) ( Fig. 6 ).
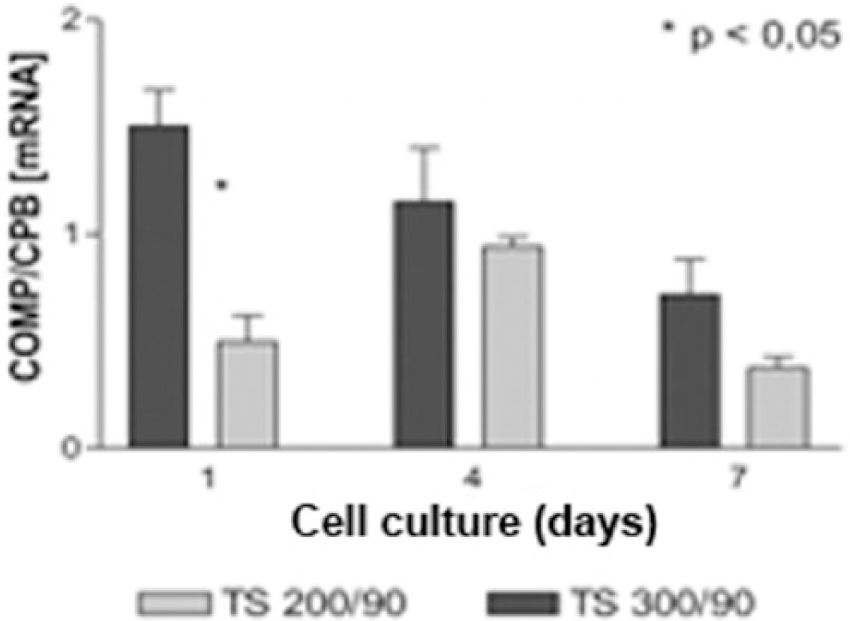
Influence of pressure variation (1 vs. 2 atm) on gene expression of cartilage oligomeric matrix protein (COMP).
PARP-1 and caspase 3 are markers for apoptosis. To detect any noxious effect of HBO on chondrocytes, expressions of these markers were measured by Light-Cycler PCR. Upregulation of the apoptosis markers was detected in the 2 atm–90 minute treatment group ( Figs 7 and 8 ).
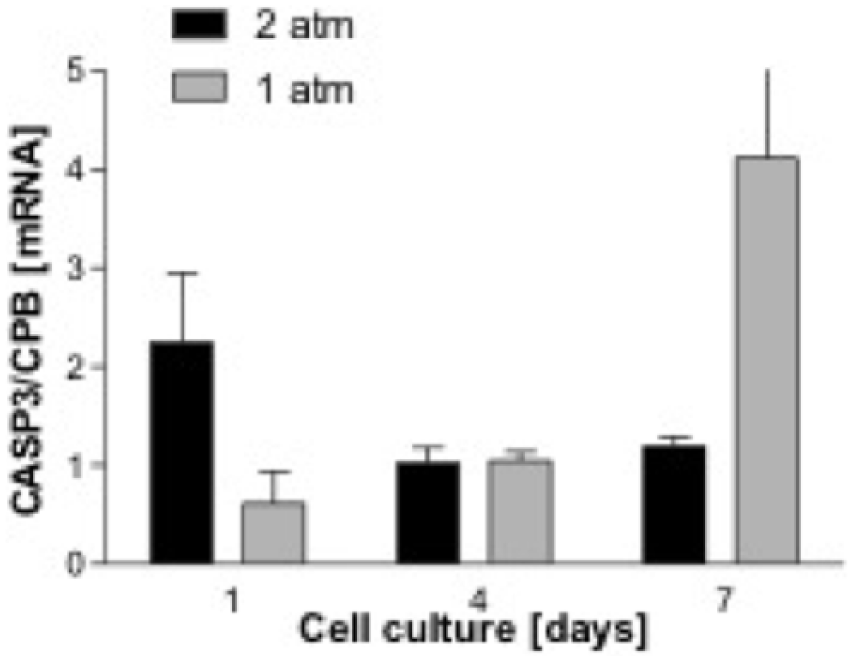
Impact of treatment pressure (1 vs. 2 atm) on gene expression of caspase 3 (CASP3).
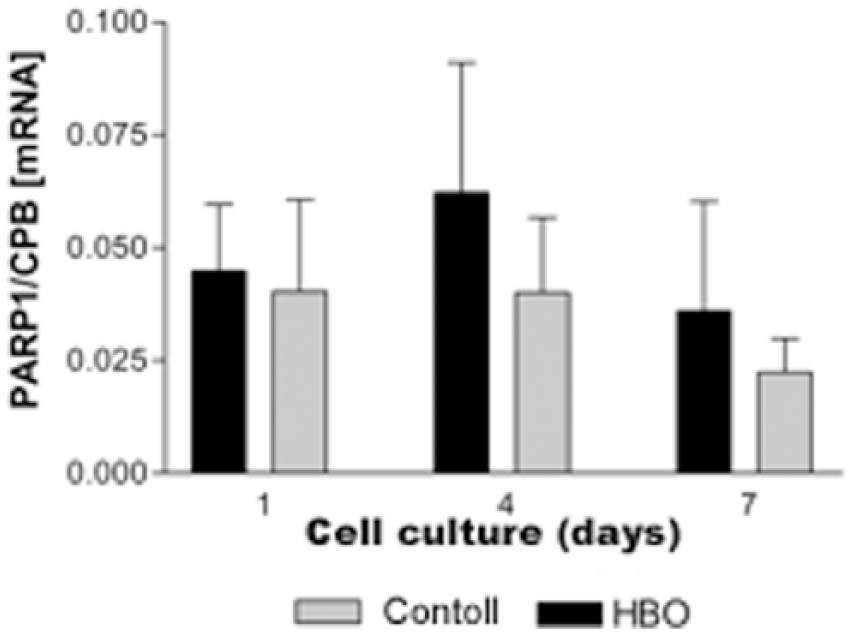
Influence of hyperbaric oxygen (HBO) on gene expression of poly-ADP-ribose polymerase 1 (PARP-1).
Discussion
For chondrocytes, adequate oxygen levels are known to be required for hydroxylation processes involved in collagen release. As articular cartilage is stratified into zones by characteristic changes in cellular, matrix and nutritive components, the different zones are exposed to levels of nutrients due to the avascular nature. Especially the oxygen partial pressure declines from 7% to 10% at the superficial layer to less than 1% near the subchondral bone. 54 A further decrease in synovial fluid oxygen tension and environmental changes in traumatic injury or degenerative diseases of articular cartilage have been reported. While hyperoxic conditions appear to enhance collagen and glycosaminoglycan synthesis in fibroblasts, 55 the effect on chondrogenesis remains unclear. Previous studies showed that HBO treatment increased oxygen tension in rabbit synovial fluid and suppressed the iNOS expression and apoptosis of chondrocytes.
As mentioned earlier, until now only Yuan et al., 30 Chen et al., 10 and Ueng et al. 56 have analyzed the effects of HBO on chondrocytes in an in vivo animal model. Whereas the Yuan group could show an increase of cell accumulation, upregulated mRNA expression and enhancement of cartilage repair, in our study, chondrocyte cell proliferation in response to HBO was inhibited after repeated daily treatments before remaining static after 5 days of treatment at 2 atm. These differences might be due to different treatment and analysis regimes as cell proliferation in our study was evaluated immediately (30 minutes) after administering HBO treatment, whereas Yuan et al. 30 determined proliferation after a 48-hour interval and in the case of Chen et al., 10 2, 4, 6, and 12 weeks after surgery. Potentially this time period is sufficient to compensate for potential toxic effects of HBO or constitute protection mechanisms toward ROS and oxidative stress. Ueng et al. 56 demonstrated that HBO treatment prevents NO-induced apoptosis in articular cartilage injury via enhancement of the expression of heat shock protein. Furthermore, beneficial effects of HBO treatment were evident in their rabbit cartilage repair model.
Both Ysart and Mason 57 and Speit et al. 46 demonstrated a time-dependent response of cells to HBO. Not only fibroblast cell proliferation in response to HBO was initially inhibited but then recovered after repeated daily treatments, but also lymphocytes of HBO-treated patients showed evidence of DNA damage only after the first treatment, while subsequent treatments failed to result in DNA damage. Both groups attribute this finding to an upregulated expression of the antioxidant enzymes after initial HBO treatment even though this has not been proven terminally.
While higher levels of HBO inhibited cell growth even more, it should be evaluated that there is a critical level of hypo-/hyperoxia required to stimulate or at least maintain chondrocyte cell proliferation. Furthermore, the question arises if the positive effects shown in all in vivo studies are due to direct chondrocyte activation or might be triggered by additional involvement of cells participating in wound healing. This especially needs further consideration as all defects treated extended into the subchondral, vascularised bone, which might lead to recruitment of mesenchymal stem cells following treatment with HBO. In addition to increasing neovascularization, hyperbaric oxygenation may directly affect growth factors necessary for mesenchymal stem cell migration or subsequent regenerative processes. Unfortunately, the effects of HBO on tissue regeneration are difficult to determine because they do not occur at the same oxygen concentrations. While glycosaminoglycan synthesis in bovine chondrocytes reaches its maximum at 21% O2, proteoglycan aggregation peaks at 3% O2. 58 In addition, chondrogenesis by periosteum is maximal at O2 concentrations of 12% to 15%, while inhibition of cartilage formation and collagen synthesis occurs at very high (>90%) and very low (<5%) oxygen levels. 59 As little as we yet know about optimal oxygen concentrations for regenerating cartilage under hyperbaric conditions, it has been estimated that HBO at 2 to 2.4 atm increases oxygen concentrations in bone and vascularity of wound tissue and fibroblast proliferation.47,60,61 Our findings suggest that hyperbaric conditions of 1 to 2 atm might be the most favorable for enhancement of cell count in chondrocytes, which is verified by the work of Schulze et al., 62 who showed that fibroblasts exposed to HBO at a pressure of 1.5 bar exhibited the highest cell number. Articular chondrocytes live in a physiologic environment of 1% to 5% (8-40 mm Hg) oxygen, and bone marrow resides in approximately 7% oxygen (50 mm Hg).63-66
Shapiro’s studies on chicks queries that chondrocytes in endochondral growth cartilage are not hypoxic, 13 while the current consensus still postulates the opposite. Anderson et al. 35 showed that lowering oxygen from atmospheric level (hyperoxia) to the physiological level (physioxia) of articular promotes the articular chondrocyte phenotype in highly chondrogenic cells.
This is undermined by detection of upregulation of apoptosis specific markers and cartilage specific proteins during administration of high oxygen levels. The decrease in chondrocyte growth during high levels of HBO we saw in our study may be due to enhanced apoptosis, which can be induced by various pathways. On one hand, cell membrane receptors (CD95/Fas, tumor necrosis factor receptor [TNFR]) may receive signals, which are transmitted by signal transducers like caspase3 to the nucleus; on the other hand, direct modulation might cause conversation of transcription- and translation activities activating protein synthesis leading to changes in the cell membrane, DNA degeneration or necrosis. This is undermined by our findings that chondrocyte cell count dipped within the first days but showed recovery from day 5 on. Li et al. 67 tried to evaluate the necessary oxygen level in a combined experimental-computerized approach. Their findings support the hypothesis that a threshold oxygen tension and pressure needs to be determined to better develop new cartilage matrix. As many different modulatory effects where already shown during HBO treatment, further experiments must evaluate the course of chondrocyte growth and the necessary level of pressure and treatment time and develop better treatment options in sports-related and degenerative cartilage damage.
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was obtained from Ethical Comission of LMU Munich (115-13).
Informed Consent
Verbal informed consent was obtained from all subjects before the study.
Trial Registration
Not applicable.
