Abstract
Objective
While the percentage of viable cells is a major determinant of graft performance during osteochondral allograft (OCA) transplantation, the baseline chondrocyte viability at the periphery of osteochondral plugs is defined at the time of harvest. In this laboratory study, we aimed to determine the optimal technique for OCA plug harvest by evaluating commercial standard techniques compared to sharp blade harvest technique.
Design
Osteochondral explants were harvested from bovine and human samples using 3 different techniques: (1) standard OATS manual punch device (Osteochondral Autograft Transplant System OATS; Arthrex, Naples, FL), (2) powered trephine device, and (3) fresh scalpel blade. Chondrocyte viability and the dead area at the periphery of the tissue were evaluated by LIVE/DEAD staining. Safranin-O and fast-green were performed for structural evaluation.
Results
For both bovine and human samples, the dead area at the periphery of the explant was significantly smaller after scalpel blade preparation compared to harvest with OATS (P < 0.001) and powered trephine devices (P < 0.001). In addition, while powered device had a smaller remaining dead area compared to the OATS device (P < 0.001), there was significantly greater tissue loss and peripheral contour change for plugs harvested with the powered trephine device.
Conclusion
Our study demonstrated that OCA plugs harvested with OATS and powered device lead to a significant mechanical injury at the periphery of the explants compared to a scalpel. We propose that the optimal technique for OCA harvest utilizes a combined approach incorporating a scalpel blade/circular scalpel to prepare the chondral surface and a powered trephine to prepare the osseous surface.
Introduction
Osteochondral allograft (OCA) transplantation is an effective strategy to treat cartilage defects, providing not only a mature, differentiated hyaline cartilage surface but also the supporting subchondral bone. 1 Traditionally, OCA transplantation has been performed using size-matched condyles, ensuring improved orthotopic congruence between the host and the donor, from which cylindrical OCAs, synonymously termed “cores,” “dowels,” or “plugs,” are harvested for transplantation. However, the availability of full condyle grafts remains a major limitation for OCA procedures here in the United States, potentially leading to increased costs and prolonged surgical delays as patients await appropriately size-matched grafts from a sparse supply. Such delay not only extends the period of disability or worsening of clinical symptoms suffered by patients, but may also portend toward progression of a patients’ lesion size over time. 2
In an effort to overcome this major limitation of graft availability here in the United States, the practice of harvesting multiple fresh “precut” allograft plugs from a single-donor condyle has emerged. In this way, not only are more precut plugs made available from a single donor, but these precut plugs can also be distributed and stocked by regional tissue banks/high-volume cartilage centers for more timely “off-the-shelf” accessibility. In short-term study, these fresh precut allograft plugs bear similar patient-reported clinical outcomes and MOCART scores when compared to traditionally harvested plugs taken from condyle allografts cut at the time of surgery with immediate implantation. 3 Precut plugs are manufactured and utilized for smaller cartilage lesions (<20 mm). Their smaller dimensions enhance the number of grafts that may ultimately be harvested from a single-donor condyle and are also believed to decrease the risk for orthotopic mismatching since their total cross-sectional surface area is small. 4 Two diameter sizes (10 mm and 16 mm) of precut plugs are available commercially currently; however, each of them is harvested using a different technique. The 10-mm plugs are harvested using a manual OATS punch device (Osteochondral Autograft Transplant System OATS; Arthrex, Naples, FL), while the 16-mm plugs are harvested with a powered trephine device (Osteochondral Resurfacing System; JRF, Centennial, CO). Despite promising initial short-term results, more extensive evaluation of graft survival for fresh precut allograft plugs versus traditional day-of-surgery harvested plugs is warranted to distinguish if late-term differences exist between these groups; or more specifically, if there are differences in long-term outcomes between the 10-mm precut and 16-mm precut plugs harvested by different techniques. It is important to note that the standard technique for the harvest from full fresh condylar grafts on the day-of-surgery more often utilizes the powered trephine device.
Chondrocytes are very fragile cells. Even minor traumatic mechanical insults may lead to significant chondral death and graft compromise. The process of OCA graft harvest is traumatic to donor chondrocytes, especially along their circumferential periphery where instrumentation, whether that be a manual punch or powered trephine, used to cut cylindrical plugs may interfere with chondrocyte viability.5 -7 During graft harvest, use of a powered trephine has been associated with large dead areas at the periphery of OCA plugs, with high chondrocyte death and disrupted cartilaginous matrix.8,9 The trauma suffered at the periphery of these plugs, and the associated chondrocyte death is particularly concerning as chondrocyte viability at time of implantation is a key determinant for overall graft survivorship in vivo.10,11 Furthermore, the peripheral chondrocytes and their surrounding matrix are responsible for ensuring a smooth transition and may influence the tissue integration along the cartilage-cartilage interface of donor and host tissues upon implantation.8,12
In analyzing outcomes among patients at our own institution, we compared graft survivorship at 2 years post-op among patients who underwent OCA transplantation by traditional, day-of-surgery OCA harvest from condylar grafts (151 patients) as well as those who underwent OCA transplantation using fresh precut “off-the-shelf” plugs (40 patients). Our unpublished analyses revealed a significantly lower graft survival rate at 2 years among patients who underwent OCA transplantation with 10-mm precut plugs using a punch device (8 patients graft failed out of 37 transplantations, 78.4% survival) compared to patients whose OCA was harvested from condylar grafts using a powered trephine at the time of the surgery (9 graft failure out of 111 transplantations, 91.9% survival) (P < 0.05). In addition, OCA survivorship at 2 years for the 16-mm precut plugs harvested using powered trephine represented a very small sample of total cases in our patient database, with no graft failures among the 3 cases of their use. Though these analyses are underpowered to be able to draw definitive conclusions on graft survivorship between those harvested by manual punch versus by powered trephine, these findings, nonetheless, raise the question as to which technique is in fact optimal for preserving chondrocyte viability and tissue architecture. We know that chondrocyte viability, a major determinant of graft performance in vivo, markedly decreases with prolonged OCA storage. 10 We also appreciate that the baseline status of chondrocyte viability at the periphery of harvested plugs is defined at the time of their harvest. Therefore, this controlled laboratory study aims to determine the optimal technique for OCA plug harvest by evaluating commercial standard techniques compared to sharp blade harvest technique, to identify which inflicts the least trauma or mechanical insult upon the OCA plugs, as measured by both histologic/structural quality and chondrocyte viability assays along the critical periphery of prepared OCA grafts.
Methods
Bovine humeral heads purchased from a local slaughterhouse (2-3 months old USDA-certified) and human femoral condyles allografts provided by Joint Restoration Foundation (JRF Ortho, Centennial, CO) were used in this study. Osteochondral (OC) explants were harvested from bovine and human samples under continuous cold irrigation with Dulbecco’s phosphate-buffered saline (DPBS; Gibco®, Life Technologies, Carlsbad, CA) using 3 different techniques: (1) a standard OATS manual punch device (Osteochondral Autograft Transplant System OATS; Arthrex), (2) a powered trephine device (Osteochondral Resurfacing System; JRF), and (3) a fresh scalpel blade (GEM, West Hempstead, NY). A total of 45 explants (15 per technique) from bovine samples and 18 explants (6 per technique) from human samples were obtained.
Harvesting Techniques
OATS punch (Osteochondral Autograft Transplant System OATS; Arthrex)
Briefly, the device was initially assembled with the screw-in core extruder knob and the collared pin per manufacturer’s published technique guide. Before harvesting the OC explant, care was taken to advance the collared pin 1 to 2 mm outside of the punch’s tip using the screw-in extruder knob. This step protects the cartilage from the sharp edge of the punch during its initial placement on the articular surface. Following, the punch was lowered to the surface of the intended harvest site of bovine shoulder or human femoral condyle, ensuring its perpendicularity to the cartilage surface. The screw-in core extruder knob was then removed from the back of the punch. This allows the edge of the punch to freely seat against the cartilage and then subchondral bone upon impaction. The punch was then impacted through cartilage and into subchondral bone to a depth of 10 mm using a mallet. Following impaction to this standardized depth, the T-handle tool was gently rotated 180° clockwise and then counterclockwise to release the explant from the deep bone. Importantly, due to the conical bevel at the tip of the impaction punch, the dowel is captured from the surrounding bone at the desired depth of impaction with these alternating rotations of the T-handle tool. The cylindrical plug is then withdrawn from the harvest bed, captured inside the punch device en bloc. The extruder knob was then threaded to release the harvested dowel from the punch device.
Powered trephine device (Osteochondral Resurfacing System; JRF)
Bovine shoulders and human femoral condyles were positioned and securely fixed in an Allograft OATS workstation jig before using powered devices. The powered trephine was centered along an axis perpendicular to the chondral surface using a collared central guide pin. Then a housing tool was placed over the indented graft site, perpendicularly to the cartilage surface. The rotary trephine was then assembled and connected to a powered drill and then passed within the housing tool until almost resting upon the graft’s cartilage surface. Subsequently, the trephine rotation was started and gently pressurizing the drill it was drilled at full speed across the length of the entire graft (articular cartilage and subchondral bone) to a depth of 10 mm, under constant cold irrigation. The OC plug was gently extracted from the surrounding bone with the assistance of a sagittal saw introduced perpendicularly through the condyle at 10 mm depth to the graft without contacting any of the freshly cut circumferential periphery of the cylindrical plug.
Fresh scalpel blade (GEM)
To evaluate the use of a straight sharp blade as opposed to the circular and beveled sharp tip of the Arthrex OATS punch, OC explants were initially harvested using the OATS device utilizing the same technique as outlined above. Subsequently A 0.23-mm-thick single-edge straight scalpel blade was then used to cut the borders of the cylindrical explants perpendicularly into square grafts. This process created a cubic sample, of which all exposed peripheral margins were prepared by a straight fresh scalpel blade only (
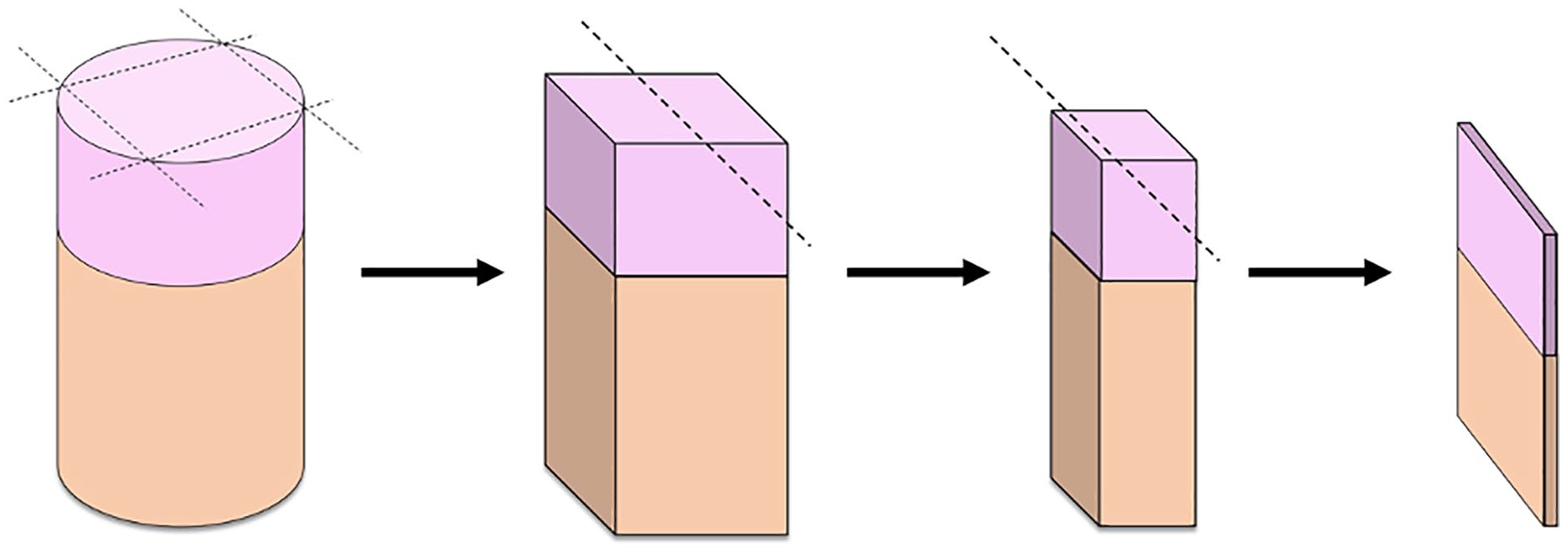
Illustration of OC explants cut perpendicularly with fresh straight scalpel from cylindrical to cubic graft shapes. Dotted lines represent the planes cut by scalpel blade. OC = osteochondral.
Old, Dull OATS versus New, Sharp OATS Punch
To evaluate whether the sharpness of the tip of the OATS device affects the histologic/structural quality and chondrocyte viability at the periphery of explanted plugs, a subset of grafts were harvested using an older, OATS punch with a duller conical bevel tip after prior use upon more than 20 bovine shoulders (old OATS punch), while a second subset of grafts were harvested using a brand-new, sharp OATS punch debuted for these experiment (new OATS punch). Explanted plugs in each subset were harvested using their respective old, dull or new, sharp OATS device utilizing the same technique as outlined above.
Graft Processing after Harvest
After OC explant harvest, all explants were rinsed with DPBS. To assess chondrocyte viability, the explants were each cut in the half lengthwise from the surface to the bottom, and a 0.5-mm-thick wafer of articular cartilage and underlying bone representative of the mid-sagittal graft were obtained using standard microtome technique (
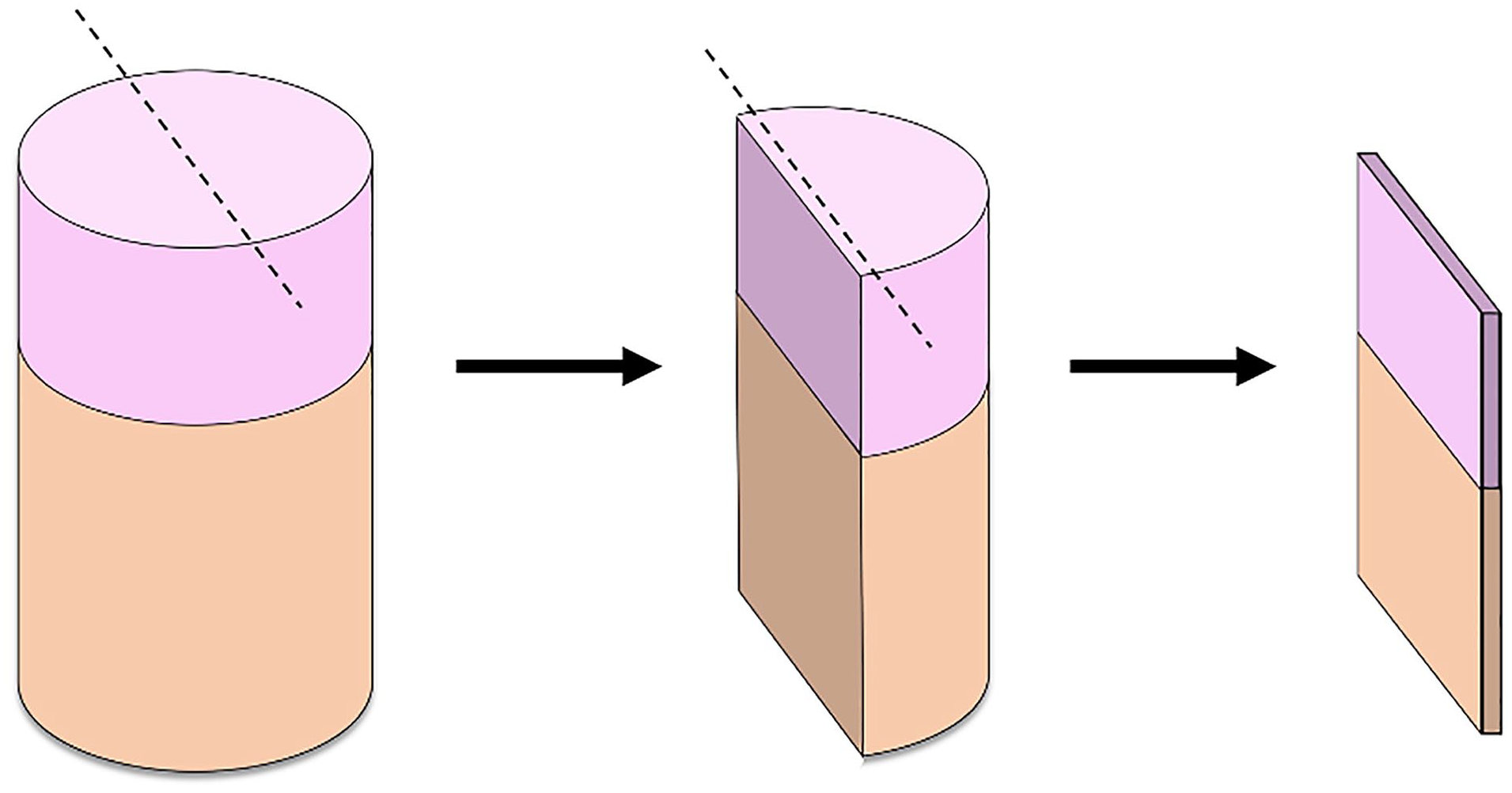
Illustration of OC explants cut lengthwise to render the 0.5-mm mid-sagittal wafer of articular cartilage and subchondral bone used for chondrocyte viability and microscopic structural evaluation. Dotted lines represent the planes cut by standard microtome technique. OC = osteochondral.
Chondrocyte Viability
Chondrocyte viability and the dead (acellular) area at the periphery of the tissue sections were evaluated by the LIVE/DEAD staining (Calcein-AM and ethidium homodimer) as previously published.
13
In brief, 0.5-mm-thick wafers of cartilage with subchondral bone were incubated in calcein-AM 5 µg/ml and ethidium homodimer 10 µg/ml (LIVE/DEAD Cell Viability Kits; Life Technologies) dissolved in DPBS at room temperature for 40 minutes. Subsequently, the wafers were rinsed in DPBS 3 times for 10 minutes. Explants were evaluated with an inverted fluorescent microscope at exposure of 150 to 200 nm (live) and 600 to 700 nm (dead). Three depths along the peripheral region of interest (pROI) were surveyed along the full depth of each explant’s margin (300 × 300 pixels) ensuring the full span from articular surface through underlying subchondral bone was viewed. Area was measured in the selected pROI for each sample using ImageJ software (version 1.47q; NIH, Bethesda, MD).
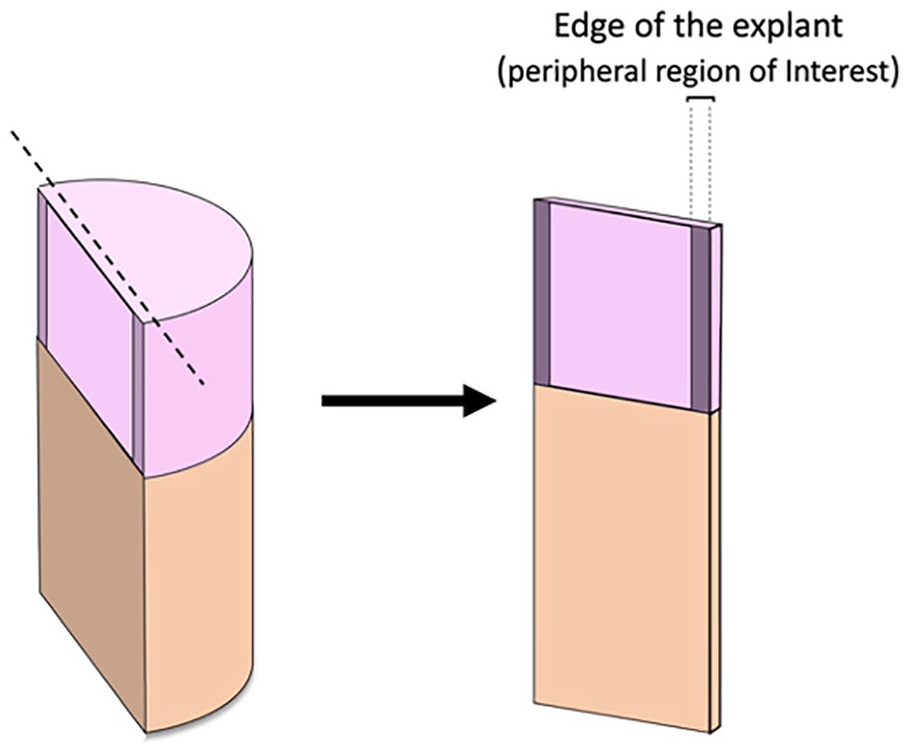
Image demonstrating the analyzed peripheral region of interest (pROI) at the explant’s margin.
Structural Evaluation
Following fixation, explant samples were decalcified using 10% ethylenediaminetetraacetic acid (EDTA). Samples were then processed, embedded in paraffin, and cut into 7-µm-thick sections. Sections were deparaffinized in xylene and rehydrated in a series of graded alcohol, and Safranin-O and fast-green (Fisher Scientific) and hematoxylin-eosin staining was performed.
Statistical Analysis
Mean ± standard deviation was determined in each group. One-way analysis of variance (ANOVA) with post hoc Bonferroni test was used to determine differences among the groups in the area of dead cells at the periphery of the OC explant. Student’s t test was used to compare differences between the new and the old OATS punch. SPSS version 28.0 (SPSS Inc, Chicago, IL) was used for statistical analysis. Statistical significance was set at P < 0.05.
Results
Measurements of Dead Cell Area
Bovine samples
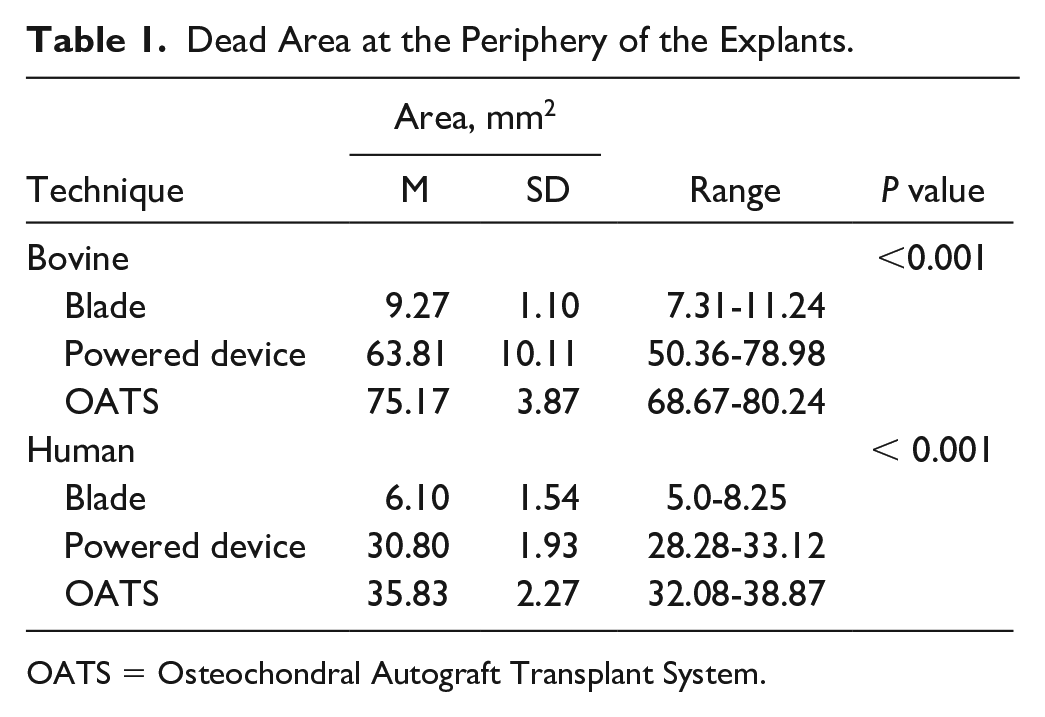
The dead area at the periphery of the explant was significantly smaller after scalpel blade preparation compared to harvest with OATS (P < 0.001) and powered trephine devices (P < 0.001). And though samples harvested with the powered trephine device had a smaller remaining dead area compared to the dead area of samples harvested using the OATS device (P < 0.001), there was significantly greater tissue loss and peripheral contour change for plugs harvested with the powered trephine device. Said differently, though fewer dead cells remained along the periphery of plugs harvested with the powered device as compared to the OATS device, the overall total area of remaining peripheral tissues was less among samples harvested by powered device; demonstrating that the powered device inflicted the greatest structural damage and peripheral cartilage tissue loss among all the harvest techniques.
Table 1
and
Dead Area at the Periphery of the Explants.
OATS = Osteochondral Autograft Transplant System.
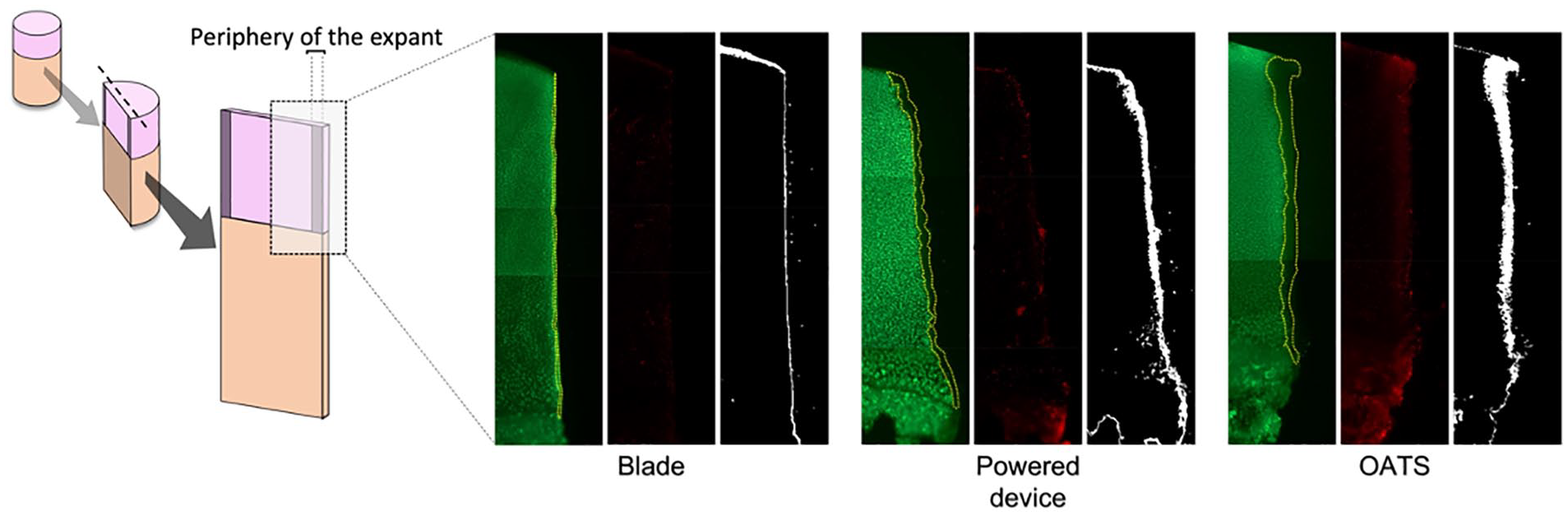
Images showing the dead area at the periphery of the explants—outlined in yellow—using the 3 different harvest techniques. Scale (white bar) represents 150 μm.
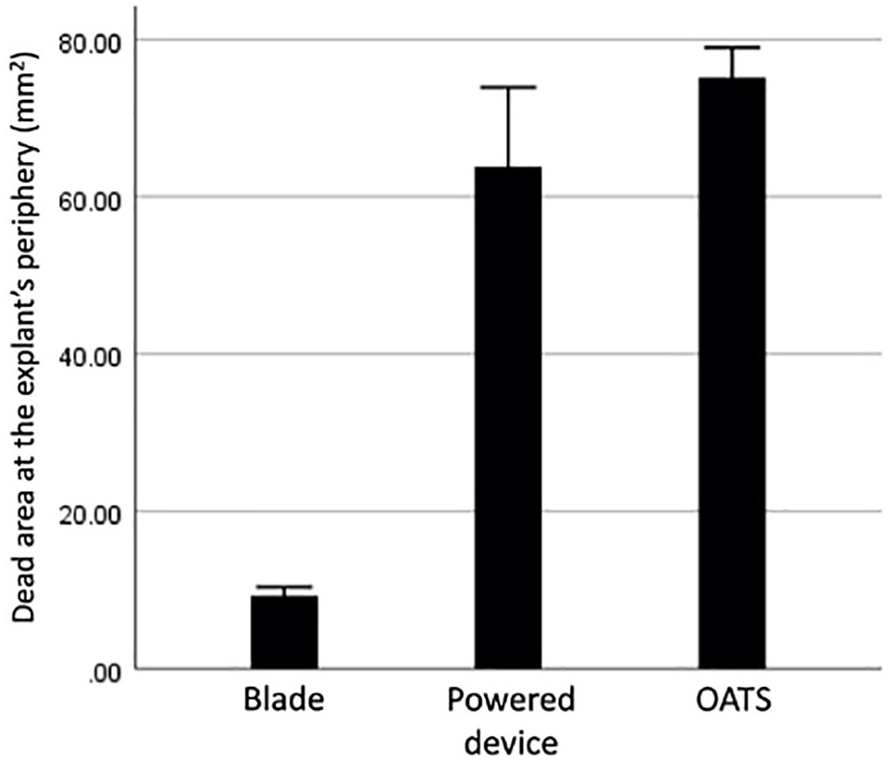
Measurement of the dead area at the periphery of the explants in bovine samples. OATS = Osteochondral Autograft Transplant System.
Human samples
We observed similar changes after harvest in human explants. The dead area was minimal under microscopic evaluation for scalpel blade preparation, which was significantly smaller compared to the other groups (OATS punch, P < 0.001; powered trephine device, P < 0.001). In addition, although the powered device again presented a smaller remaining area of death when compared to the manual OATS punch, the difference did not reach statistical significance. (P = 0.065 and
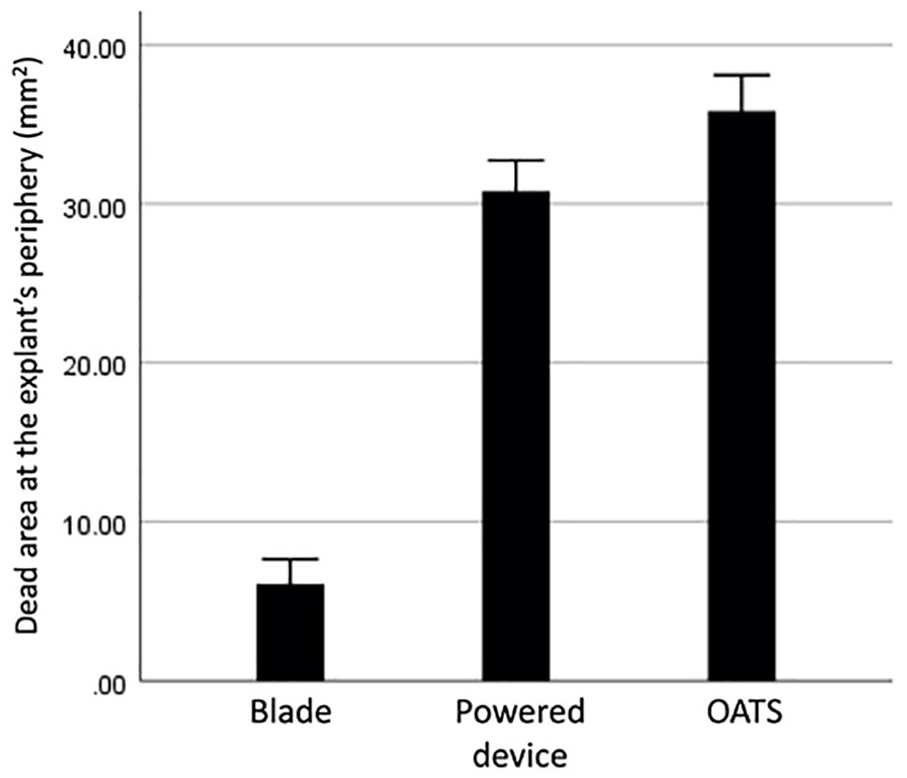
Measurement of the dead area at the periphery of the explants in human samples. OATS = Osteochondral Autograft Transplant System.
Old OATS versus New OATS Punch
The size of the dead cell area at the periphery of the graft was similar between samples harvested using the old, dull OATS punch (37.13 ± 1.74 mm2) or the new, sharp OATS punch (35.83 ± 2.27 mm2) (P = 0.292).
Measurement of Dead Cell Pattern and Tissue Contour
Samples harvested with the powered trephine demonstrated irregular margins with the greatest amount of tissue loss at the periphery of harvested grafts when compared to the other groups. Conversely, the straight scalpel blade generated more perpendicular, straight, regular margins with the least amount of tissue loss. Last, the OATS punch generated conical-shaped, regular margins, and the powered device generated conical-shaped, ragged, and irregular margins. Focusing on the pattern of chondrocyte death along these peripheral margins, the scalpel blade margins were narrow and straight with minimal cell death, while the OATS punch generated a funnel-shaped pattern of chondrocyte death in which the dead area at the superficial zone was wider than the dead area at the deep zone (
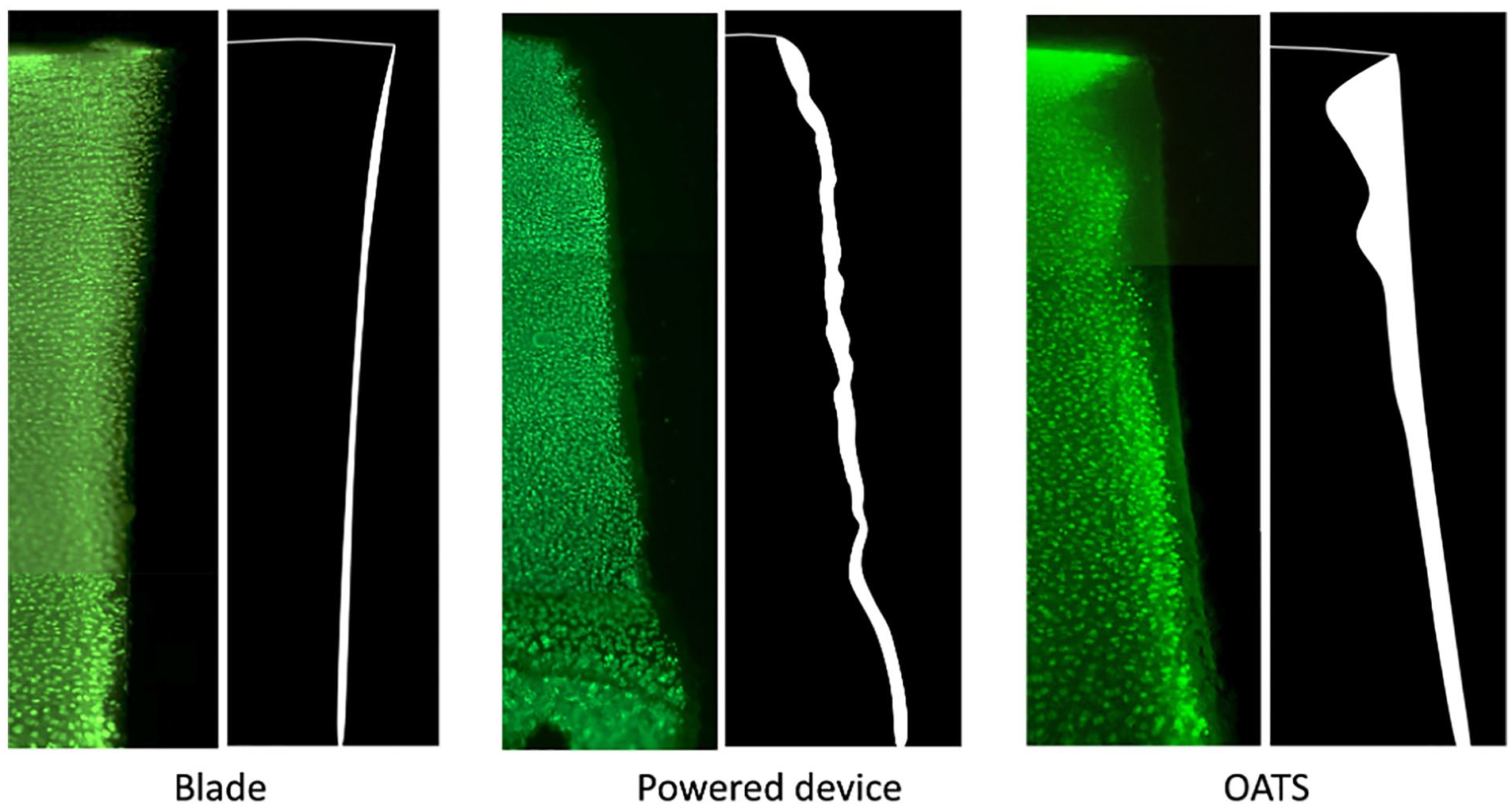
Imaging representing the tissue contour and dead area at the chondral periphery of the harvested explants. Note the irregular edge of samples harvested using the powered device. In addition, OATS punch generates a funnel-shaped pattern of cell death, with a wider pattern of cell death at the superficial zone and a narrower pattern of death at the deep zone. OATS = Osteochondral Autograft Transplant System.
Histologic Evaluation of Structural Damage and Tissue Loss
Safranin-O staining showed that samples harvested with OATS and powered device presented higher evidence of mechanical injury at the edge of the explants in comparison with scalpel blade samples. In addition, a ragged aspect was more evident in the powered device samples when compared to the other techniques, which was in line with our observation during chondrocyte viability testing (

Safranin-O staining showing the structure of the periphery of harvested explants. White bar represents 100 μm scale. OATS = Osteochondral Autograft Transplant System.
Discussion
This controlled laboratory study aimed to determine the optimal technique for OCA plug harvest comparing the use of commercially available manual punch, powered trephine, or a straight scalpel as assessed by chondrocyte viability and microscopic tissue damage along the critical periphery of grafts prepared using each technique. The findings of this study are in accord with those of many others.5,8,9,12,14 Use of powered trephine demonstrates the greatest amount of tissue damage at the periphery of prepared plugs compared to other techniques (
Our findings are also consistent with those of previous studies that characterize a middle-tier harvest profile with the use of manual punch.8,9,14 In an ovine model comparing use of powered trephine to manual punch in harvesting OCA plugs from the trochlea, Evans et al. found significantly improved chondrocyte viability and decreased microscopic tissue damage in the manual punch groups compared to powered trephine groups, and further caution against the use of powered trephines entirely.
9
Similar to our own findings, Evans et al. and Huntley et al. appreciated a moderate degree of chondrocyte death and tissue damage with use of manual punch devices among the 3 techniques examined in this study (
The use of an open system technique as opposed to an enclosed punch or trephine technique avoids many of the proposed factors believed to contribute to mechanical injury at the periphery of OCA plugs during their harvest. In an open system, as is the case when using the straight and narrow profile edge of a sharp scalpel blade, the compressive/compaction injury associated with a beveled manual punch is avoided. The potentially caustic axial compression applied to the cartilage surface of plugs necessary to extrude them from within enclosed punch and trephine systems is also avoided. Furthermore, use of a straight scalpel blade only to the depth of the articular cartilage alone not only avoids both thermal injury and rotatory shear associated with powered trephine use, but also decreases vertical shear injury by minimizing the time spent with frictional contact against the inner cutting surface of a manual punch or powered trephine especially when additional force is necessary to breach the subchondral bone under traditional techniques.
For these reasons, the results of our study are expectedly consistent with those of previous work concluding that the use of a straight sharp scalpel blade bears the least harm to peripheral chondrocytes, with the least amount of tissue loss during OCA plug harvest
Optimizing the technique for OCA harvest to preserve chondrocyte viability and tissue integrity is vital to the success of OCA implantation whether OCA grafts are precut prior to surgery or prepared intra-operatively. Furthermore, given the scarcity of full condyle donor grafts, it is important that we continue to enhance the successful use of precut OCA plugs as they represent a more affordable and accessible graft option. 4 Based on the findings of our current study, and in light of concordant prior work by others, the optimal technique for OCA harvest may utilize a combined approach incorporating both straight or circular scalpel and powered trephine devices. It has been demonstrated by others, and further supported by this study, that scalpel use confers the least trauma to peripheral cartilage during OCA harvest compared to use of manual punch or powered trephine.8,12 However, a thin straight scalpel blade trades the benefits of its narrow, straight, and open profile with sharp cutting edge for a decreased stiffness and material strength, making it suboptimal for breaching the subchondral plate and deeper cancellous bone. For this reason, a powered trephine is necessary to prepare the deeper bone. However, it remains an important consideration to ensure that peripheral chondrocytes are shielded from the various mechanical, shearing, and thermal injuries associated with powered trephine use. Therefore, we propose that the optimal technique for OCA harvest incorporate same-axis antegrade and retrograde approaches to prepare each of the osteochondral tissue types to the level of the subchondral plate—specifically, retrograde scalpel blade or a circular scalpel use for preparation of the chondral surface, and antegrade powered trephine use for preparation of the boney surface. This study suggests there is opportunity for improvement upon the current harvesting technique standards, and further studies are warranted to assess the clinical success of OCAs harvested by the optimal technique proposed herein.
Conclusion
This controlled laboratory study demonstrated that the OATS punch and the powered device led to a significant mechanical injury at the edge of the explants when compared to a scalpel incision. In particular, grafts after OATS harvest demonstrated the largest dead chondrocyte area at the periphery of the graft, while the utilization of a powered device resulted in substantial tissue damage. Consequently, we propose that the optimal technique for OCA harvest utilizes a combined approach incorporating retrograde scalpel blade or a circular scalpel preparation of the chondral surface and antegrade powered trephine use to prepare the underlying boney plug.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Our project received the JRF Ortho Investigator Initiated Grant, Co-Investigator ($15,000 total direct cost).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: G.M. has received consulting fees from JRF and two grants from JRF Ortho. E.M.F. has received support for education from Kairos Surgical. A.H.G. has received royalties from Organogenesis; honoraria from Vericel, JRF Ortho, and Fidia Pharma; consulting fees from JRF Ortho, Vericel, Smith & Nephew, and Aastrom Biosciences; speaking fees from Aastrom Biosciences, LifeNet Health, Pacira Pharmaceuticals Inc., and Linvatec; consulting fees from Genzyme, Sanofi, and JRF Ortho; travel fees from LifeNet Health; and hospitality payments from Fidia Pharma and LifeNet Health. AOSSM checks author disclosures. C.L. has received honoraria from Arthrosurface, Vericel, and JRF Ortho; consulting fees from Zimmer Biomet, Aastrom Biosciences, Sanofi-Aventis, Vericel, Flexion Therapeutics, JRF, and Samumed; and nonconsulting fees from Aesculap and Arthrosurface.
