Abstract
Objectives
During arthroscopic or open joint surgery, articular cartilage may be subjected to mechanical insults by accident or design. These may lead to chondrocyte death, cartilage breakdown and posttraumatic osteoarthritis. We have shown that increasing osmolarity of routinely used normal saline protected chondrocytes against injuries that may occur during orthopedic surgery. Often several liters of irrigation fluid are used during an orthopedic procedure, which is usually kept at room temperature, but is sometimes chilled. Here, we compared the effect of normal and hyperosmolar saline solution at different temperatures on chondrocyte viability following cartilage injury using
Design
Cartilage injury was induced in bovine osteochondral explants and the patellar groove of rats
Results
Hyperosmolar saline reduced scalpel-induced chondrocyte death in both bovine and rat cartilage by ~50% at all temperatures studied (4°C, 21°C, 37°C;
Conclusions
Increasing the osmolarity of normal saline and raising the temperature of the irrigation solutions to 37°C reduced chondrocyte death associated with scalpel-induced injury in both
Introduction
Articular cartilage has an extraordinary durability and capacity to adapt to the physiological loading patterns associated with normal joint activities. 1 However, exposing the tissue to unphysiological mechanical insults can result in chondrocyte death, potentially rendering articular cartilage more vulnerable to degeneration and development of posttraumatic osteoarthritis (PTOA). 2 Arthroscopic and open surgical interventions on articular cartilage may subject the tissue to various iatrogenic injuries resulting from cutting, probing, or drilling.3,4 Such injuries may be associated with loss of chondrocyte viability, cartilage damage, and poor lateral integration and therefore potentially affect the surgical outcome. 5 Minimizing chondrocyte death resulting from iatrogenic surgical injuries would thus be beneficial in reducing cartilage damage, promote the repair response, and improve integrative healing and patient outcome. 5
Various irrigation solutions are used in orthopedic surgical practice to improve visualization of the joint space and cartilage surface. During this process, the synovial fluid is replaced with an artificial solution that has a lower osmotic pressure (~250-300 mOsm) compared with that of normal synovial fluid (~400 mOsm).
6
Therefore, chondrocytes are exposed to a marked decrease in extracellular osmolarity during articular surgery, which is likely to affect their response to injury.
7
For example, lowering the osmolarity of the irrigation solution increased cell death caused by mechanical cartilage injury however raising osmolarity provided significant chondroprotection against mechanical trauma caused by drilling,
4
impact,
7
or scalpel cutting.
8
Using an
Temperature is an important factor that affects cell viability and metabolism13,14; however, the optimum temperature of the irrigation solution used in joint surgery has not yet been reported. Often a high volume (multiple liters) of irrigation fluid is used during an arthroscopic procedure, this is usually kept at room temperature, but is sometimes chilled as cold solutions have been proposed to reduce postoperative pain and inflammation after arthroscopic surgery.
15
However, no statistically or clinically significant effect was found in the first 4 postoperative days.
16
Moreover, an
The aim of this study was therefore to investigate the effect of a normal and hyperosmotic (potentially chondroprotective) saline solution at different temperatures on chondrocyte viability in carefully controlled
Materials and methods
In Vitro Cartilage Injury Model
Bovine osteochondral strips were harvested from the metacarpophalangeal joints of 3-year old cows ( Fig. 1A ) under aseptic conditions. These were then trimmed to produce rectangular explants of approx. 5 mm × 4 mm, 8 immediately placed in serum-free Dulbecco’s modified Eagle’s medium (DMEM, Invitrogen, Paisley, UK) and used within 30 minutes. The serum-free medium was used to provide more controlled environment for the cells and to eliminate the variance that may occur due to variability of serum composition. The osteochondral explants were immersed for 5 minutes in either normal saline (NaCl 300 mOsm, Baxter Healthcare Ltd, Berkshire, UK) or hyperosmolar saline solution (600 mOsm, 105 g sucrose addition to 1 L normal saline)8,9 and maintained at 4°C, 21°C, or 37°C. In this study, sucrose was the preferred osmolyte as it is impermeable, not metabolised by chondrocytes, relatively benign and thus one experimental variable. The addition of NaCl to raise osmolarity on the other hand can potentially alter a range of processes including the activity of sodium-dependent transport processes and membrane potential, and thus it would be difficult to separate out the effects of osmolarity from sodium-dependent effects. 9
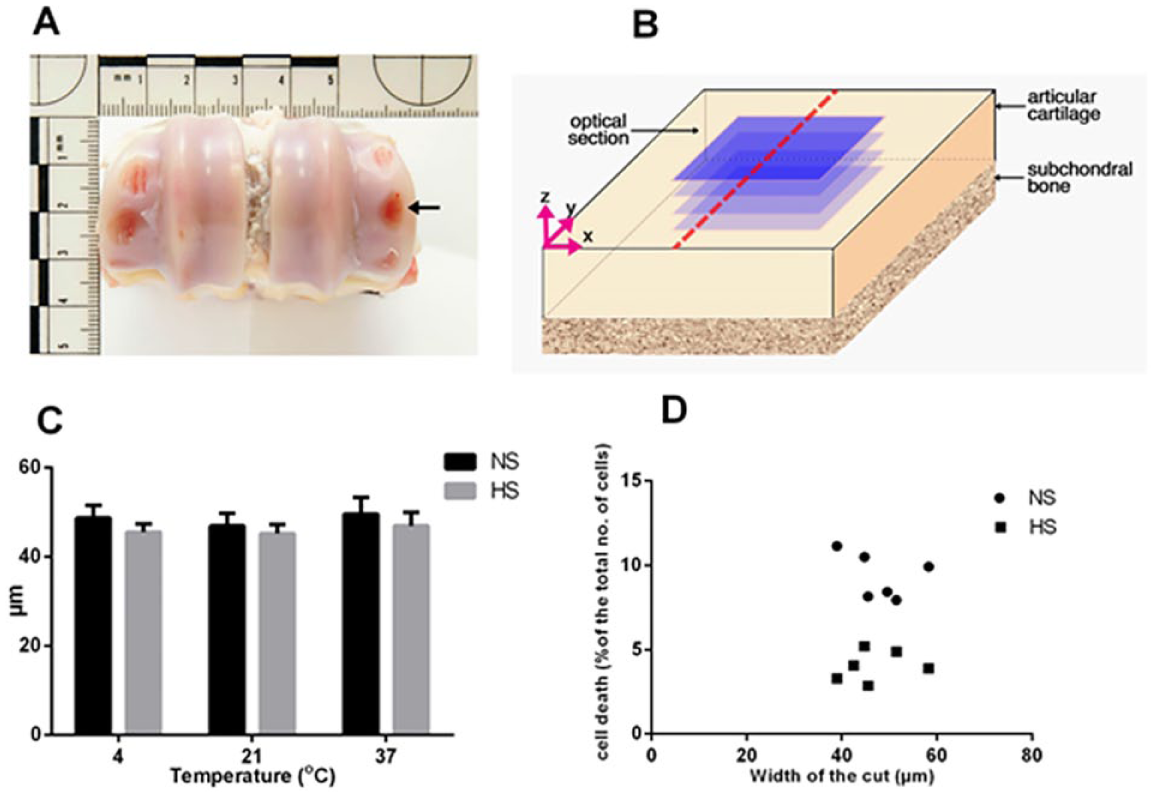
Generation and validation of an
The osmolarity of the solutions was measured by freezing point osmometer (Vitech Scientific Ltd, Horsham, UK). Explants were wounded with fresh No. 11 scalpel blades to produce a single partial thickness longitudinal cut in the middle of explant ( Fig. 1B ) and then returned to the designated solution for a further 5 minutes after injury before being assessed for cell viability. 8 Control uninjured explants were exposed to normal or hyperosmolar saline solution at 4°C, 21°C, or 37°C for 10 minutes before staining and fixation for microscopic analysis to determine the effect of raised osmolarity and temperature of the irrigation solution on chondrocyte viability.
In Vivo Scalpel Injury Model
Injury to the rat knee joint cartilage was induced as previously described.
9
Briefly, male Sprague-Dawley rats (8 weeks old) were anesthetized using 3% isoflurane. The patella was dislocated laterally after medial parapatellar arthrotomy to expose the patellar groove. The articular cartilage was then wounded along the groove by a single pass of a fresh No. 11 scalpel blade held in a standard holder, and applied under its own weight. Joints were irrigated for 5 minutes before, and 5 minutes after the induction of the cartilage injury by normal saline (300 mOsm) or hyperosmolar saline solution (600 mOsm) at 4°C, 21°C, or 37°C to allow the
Cell Viability Assay
Cartilage Imaging by Confocal Laser Scanning Microscopy
Fluorescently labeled and fixed osteochondral explants and rat knee joints were anchored to the base of a Petri dish with Blu-Tack (Bostik Ltd, Leicester, UK), immersed in PBS with the articular surface facing upward. Consecutive series of axial optical sections of the labeled
Live and dead cells were identified in the green and red channels, respectively, by thresholding voxel (volumetric pixel) intensity. A histogram of measured values for all objects in each channel was used to set the percentage threshold for the intensity (upper limit =100%; lower limit >5%) and the percentage cell death (PCD = 100 × number of dead cells/number of dead and live cells) calculated in the ROI. The width of the injury was measured by LSM imaging software (Carl Zeiss Ltd) at 100-µm intervals along the
Statistical Analysis
Data analyses were performed using GraphPad Prism (version 6.0, GraphPad Software, Inc., San Diego, CA, USA). All data were presented as mean ± standard error of mean (SEM) where
Results
Assessment of Chondrocyte Viability Following Exposure to Varying Osmolarities and Temperatures
Control uninjured bovine osteochondral explants and sham-operated rat knee joints were irrigated with either normal or hyperosmolar saline solution at 4°C, 21°C, or 37°C to evaluate the effect of increasing osmolarity and temperature on cell viability. The PCD in articular cartilage was negligible (<1%) under all conditions in both
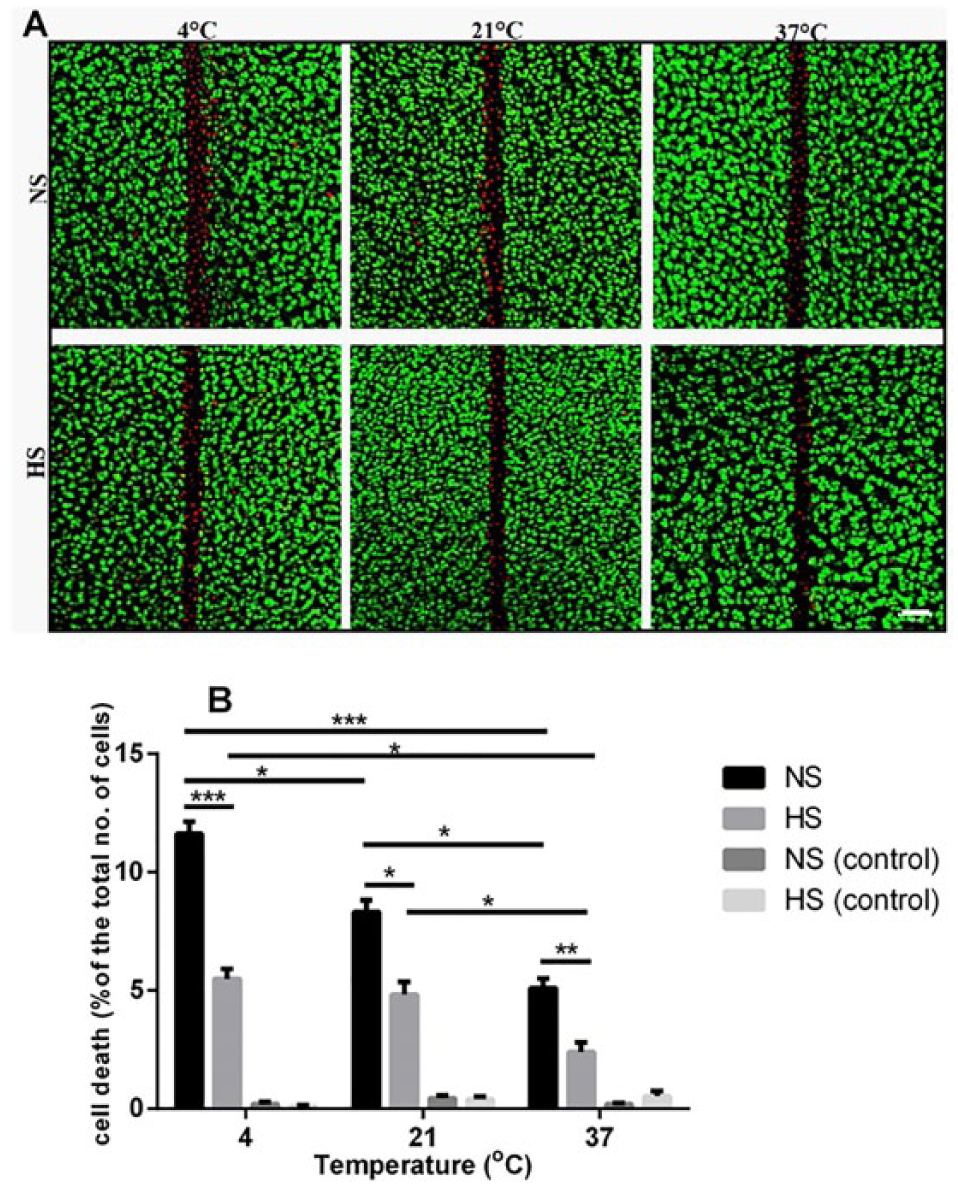
Effect of the hyperosmolar saline solution on chondrocyte death in injured osteochondral explants at different temperatures
Development of an In Vitro Cartilage Injury Model
Cartilage injury in osteochondral bovine explants was induced by fresh scalpel blades to produce a single longitudinal partial thickness cut in the middle of explant (
Fig. 1A
and
B
). To assess the reproducibility of the cut across all the experimental conditions, the mean width was calculated from 10 measurements taken at 100-μm intervals. The width of the defect was 47.1 ± 6.5 µm (mean ± SD;
Hyperosmolarity and Physiological Temperature Reduce Chondrocyte Death due to Scalpel-Induced Injury in Bovine Cartilage
Axial CLSM projection images showed a band of cell death at/around the edges of scalpel-induced injury in bovine osteochondral explants (
Fig. 2A
). Exposing cartilage explants to hyperosmolar saline solution significantly (
The Chondroprotective Effect of Hyperosmolar Saline Solution and Increased Temperature in an In Vivo Model of Cartilage Injury
The PCD in the specified ROI used for the
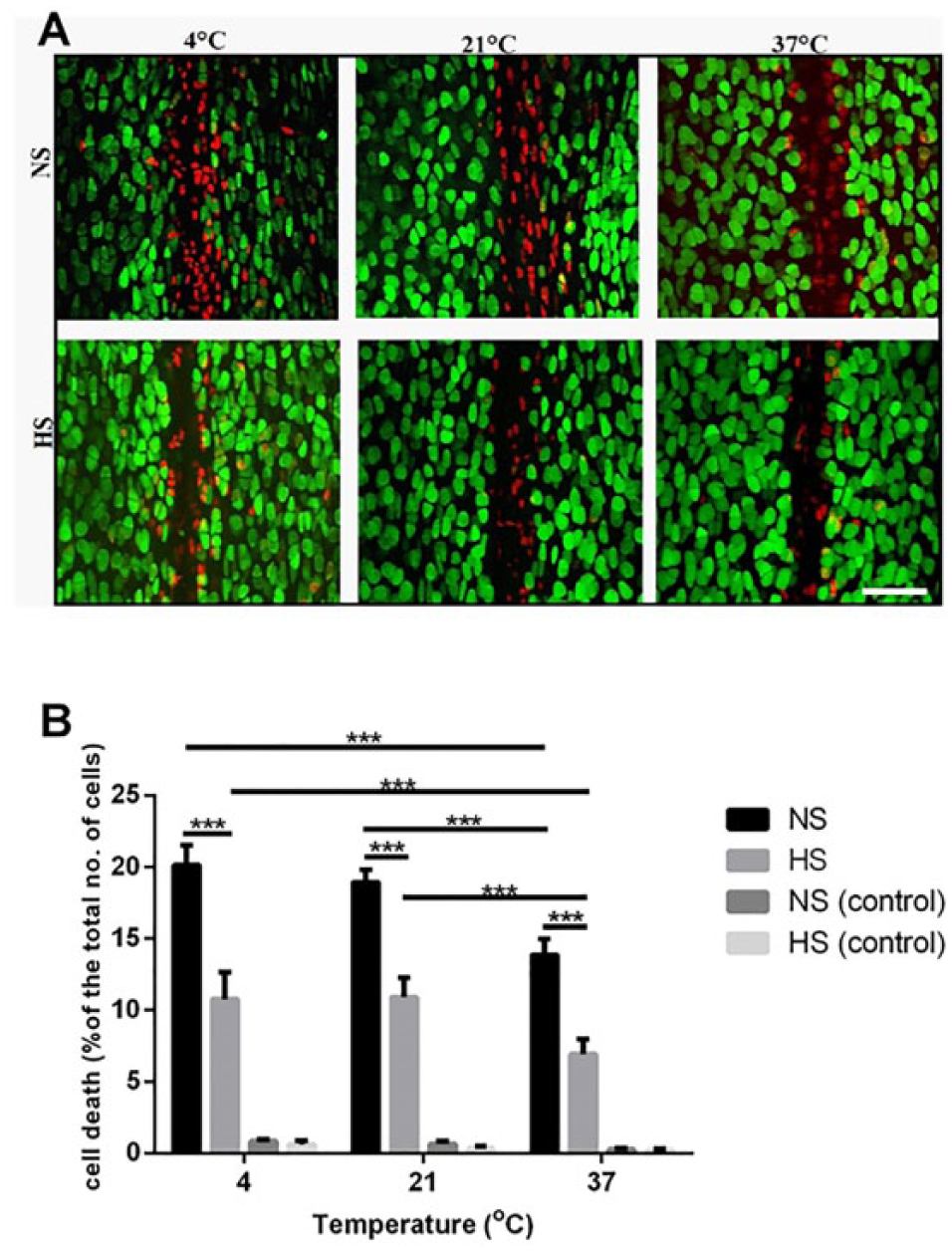
The chondroprotective effect of hyperosmotic saline solution and increased irrigation temperature in an
Raising the temperature of the hyperosmolar saline solution to 37°C also significantly decreased the PCD in injured joints compared with this solution maintained at either 4°C (
Discussion
An isotonic NaCl saline solution at room temperature is commonly used for joint irrigation during orthopedic and arthroscopic procedures to provide a clear bloodless surgical field. However, this is likely to alter the temperature and extracellular osmolarity of
In the
Irrigating healthy uninjured articular cartilage with either normal or hyperosmolar saline solution at 4°C, 21°C, or 37°C did not affect cell viability in either
It is important to note that although this study provided proof of principle that increasing the osmolarity and temperature of normal saline reduced chondrocyte death associated with scalpel-induced injury, it has certain limitations. We emphasise that the results obtained from our experimental model/procedures cannot be regarded as translational to those occurring during orthopedic surgery and additionally only a single outcome measure, that is, that of chondrocyte viability, was assessed. The cartilage was only briefly exposed to the different irrigation solutions to elucidate the immediate influence of varying osmolarity and temperatures on the extent of cell death following cartilage injury. Previous studies have established that chondrocytes sense and respond within 5 minutes.
18
Thus, it is in the early phase following the addition of the chondroprotective solution possibly as a result of chondrocyte shrinkage7,18 that influences the extent of chondrocyte death following mechanical injury. Research using an
There have been relatively few studies on the effect of longer term exposure of intact articular cartilage to irrigation solutions of differing osmolarity and temperature, and the influence on other cartilage structural and biological properties, for example, gene expression, synthetic and catabolic activities remain to be evaluated. However, in a porcine osteochondral explant model, exposure to saline irrigation solution at room temperature had a detrimental effect on chondrocyte metabolism and RNA synthesis. 14 In addition, a recent report using a canine shoulder arthroscopy model 20 demonstrated no deleterious effect on chondrocyte viability or cartilage hydration after 2hrs of irrigation with a hyperosmolar saline (600 mOsm by raised NaCl). Clearly, further work on the influence of long-term exposure of chondroprotective solutions on cartilage properties, extracellular matrix metabolism, and chondrocyte viability is warranted.
It is likely that unless care is taken, normal joint temperature will be reduced during articular surgery as a result of irrigation solution temperature, the length of the surgical procedure and exposure to the ambient conditions of the operating theatre. For example, it has been demonstrated that intra-articular temperature decreases from ~35°C at the beginning of anterior cruciate ligament (ACL) reconstruction or menisectomy surgery to ~25°C toward the end of the procedure after about 1.5hrs. 21 These changes might have detrimental effects on the structural, physiological and biomechanical properties of articular cartilage, and potentially increase the sensitivity of chondrocytes to mechanical injury. In addition, exposure of articular cartilage to cold saline significantly altered the ultrastructure of the articular surface. 22 During thermal chondroplasty, the temperature of the lavage solution influenced chondrocyte viability and surface contouring. 22 The use of cool lavage solution (22°C) led to a significant increase in the depth of chondrocyte death and roughening of articular surface compared to a 37°C solution. 22 Thus, the use of warmed, rather than cool, irrigation solutions has the potential to limit these deleterious effects if used for joint lavage.
The mechanism underlying the chondroprotective effect of warmed hyperosmolar saline against mechanical injury is unclear. This could be due to changes in matrix mechanics and/or direct effects on chondrocytes or chondrocyte-matrix interactions rendering them less sensitive to scalpel-induced injury. Previous work has demonstrated that raised temperature increased the dynamic and equilibrium stiffness of cartilage 23 and therefore at 37°C the extracellular matrix at the injury edge could be stiffer and more resistant to disruption when injured by the scalpel. Although raising osmolarity might have a similar effect, the injury width was identical for scalpel-induced trauma with both saline and hyperosmotic saline irrigation over the temperature range studied ( Fig. 1C ). It is probable that a more direct action of temperature and osmolarity on chondrocytes or chondrocyte-matrix interactions are more important than their effects on bulk properties of cartilage. Changes to osmolarity affect the sensitivity and viability of chondrocytes following exposure to injurious stimuli.7-9 Thus, raising the osmolarity surrounding cartilage will decrease chondrocyte volume, 18 membrane transport activity 24 and lead to cytoskeletal reorganization. 25 Changes to chondrocyte volume can initiate intracellular signaling cascades, including regulation of gene expression, 25 metabolic activity, 19 and calcium concentration, 26 with the latter potentially interacting with other pathways such as those that generate reactive oxygen species (ROS), which control chondrocyte viability. 27 Reducing temperature also has a variety of effects directly on cells, including increased membrane viscosity and stability 28 decreased permeability 28 and partial disassembly of spindle microtubules and cortical microfilaments.29,30 It is notable that the chondrocyte death at the site of matrix disruption reported here ( Figs. 2 and 3 ) is similar to that observed following impact injury where cell death was localized around the cartilage cracks and not present in impacted areas without cracks.7,31 Indirect effects of reduced temperature and osmolarity on cell viability on the interactions between matrix proteins, chondrocyte integrins, and cytoplasmic elements may also be crucial as they are known to be essential for chondrocyte survival. 32
In the short-term experiments reported here, warmed hyperosmolar saline was chondroprotective following mechanical injury. However, the long-term protection of cartilage integrity, stimulation of wound healing and the quality of the repair tissue remains to be investigated. Inhibiting chondrocyte death at the wound edge reduces matrix loss and enhances cartilage integration.
33
We have previously shown that hyperosmotic lavage at room temperature resulted in a more developed repair tissue
This study provided evidence that in our animal models, increasing the osmolarity and temperature of the saline irrigation solution significantly reduced the extent of chondrocyte death associated with scalpel-induced injury. Furthermore, both the saline irrigation solution and the hyperosmolar solution had no detectable effects on the viability of uninjured cartilage. The use of this relatively simple and cheap hyperosmotic irrigation solution at 37°C may provide an effective strategy for conferring chondroprotection against mechanical injury both
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Arthritis Research (UK) grant number 19665.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was obtained from the Local Ethics Committee and Animals (Scientific Procedures) Act 1986 UK Home Office (PPL 60/4052).
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
