Abstract
Background
Traumatic knee injury results in a 4- to 10-fold increased risk of post-traumatic osteoarthritis (PTOA). Currently, there are no successful interventions for preventing PTOA after knee injury. The aim of this study is to identify inflammatory proteins that are increased in serum and synovial fluid after acute knee injury, excluding intra-articular fractures.
Methods
A literature search was done according to the PRISMA guidelines. Articles reporting about inflammatory proteins after knee injury, except fractures, up to December 8, 2021 were collected. Inclusion criteria were as follows: patients younger than 45 years, no radiographic signs of knee osteoarthritis at baseline, and inflammatory protein measurement within 1 year after trauma. Risk of bias was assessed of the included studies. The level of evidence was determined by the Strength of Recommendation Taxonomy.
Results
Ten studies were included. All included studies used a healthy control group or the contralateral knee as healthy control. Strong evidence for interleukin 6 (IL-6) and limited evidence for CCL4 show elevated concentrations of these proteins in synovial fluid (SF) after acute knee injury; no upregulation in SF for IL-2, IL-10, CCL3, CCL5, CCL11, granulocyte colony-stimulating factor (G-CSF), and granulocyte-macrophage colony-stimulating factor (GM-CSF) was found. Limited evidence was found for no difference in serum concentration of IL-1β, IL-6, IL-10, CCL2, and tumor necrosis factor alpha (TNF-α) after knee injury.
Conclusion
Interleukin 6 and CCL4 are elevated in SF after acute knee injury. Included studies failed to demonstrate increased concentration of inflammatory proteins in SF samples taken 6 weeks after trauma. Future research should focus on SF inflammatory protein measurements taken less than 6 weeks after injury.
Keywords
Introduction
The knee is the most commonly injured joint with an estimated incidence of 2.29 per 1000 person years in the general population. 1 In the short term, knee injury results in decreased physical fitness and quality of life.2,3 In the long term, it is strongly associated with the development of post-traumatic osteoarthritis (PTOA) of the knee at an early age.4-6
The risk of knee OA after an anterior cruciate ligament (ACL) or meniscus injury, is reported to increase 4- to tenfold.5,7 PTOA can affect a population as young as 30 to 40 years of age.4,8 Thereby causing functional disability and lost productivity in a working-age population. 9 To date, there are no effective interventions for PTOA. Surgery, such as an ACL reconstruction or meniscus repair, has no protective effect against PTOA, while meniscectomy can even facilitate degeneration.4,10-14
Different types of knee injury can result in PTOA. Patients with intra-articular fractures of the knee have an estimated 23% to 44% chance of developing PTOA.15,16 Acute mechanical damage and chronic abnormal joint loading are thought to be the main contributors to cartilage breakdown after intra-articular fractures. 17 For soft tissue injuries of the knee such as ligament tears and meniscal injury, several factors have been suggested in the pathogenesis of PTOA. Possible contributing factors are concomitant cartilage defect or acute tissue damage at time of injury and secondary biomechanical changes as a consequence of the structural damage.16,18-20 Another suggested factor in the pathogenesis of PTOA is inflammation.16,19-21
Understanding the inflammatory response after knee injury is necessary to identify possible treatment strategies. Inflammatory biomarkers after trauma might predict the development of PTOA and subsequently become a target to prevent or treat PTOA development in an early stage. Because of the possible different pathways of PTOA after certain injuries, we included only knee injuries such as an ACL tear, posterior cruciate ligament (PCL) tear, meniscal tear, osteochondral fracture, or combined types and excluded knee fractures in this review. Osteochondral fractures are considered injuries where the cartilage is focally damaged with involvement of the direct underlying subchondral bone. 22 The bone damage does not exceed beyond the subchondral bone involvement in contrary to a knee fracture. Chondral fractures have focal cartilage damage without involvement of the subchondral bone. The aim of this systematic review is to identify inflammatory proteins that are increased in serum and synovial fluid after acute knee injury.
Methods
The reporting in this systematic review was conducted according to the PRISMA statement and published in a protocol in the PROSPERO database (CRD42020189896). 23
Data Sources and Searches
With help of a health science librarian with extensive experience in conducting literature search for systematic reviews a search of the literature was performed for relevant articles up to December 8, 2021. Search terms included inflammation, knee, anterior cruciate ligament, posterior cruciate ligament, meniscus, trauma, injury, prognosis, and biological marker. The full electronic search strategy for the Medline (OvidSP) database is presented in Appendix 1 (see Supplemental Material online). Similar search strategies were used in Embase, Web-of-Science, PubMed publisher, Cochrane, and Google Scholar. Additionally, the reference lists of all eligible studies were manually screened.
Study Selection
Two reviewers (M.N. and D.M.) independently assessed the studies for the eligibility criteria. Disagreements were solved by discussion and, if necessary, by a third author (M.R.). Additional citation tracking was performed by screening of the reference lists of the eligible studies.
Inclusion criteria for eligible studies were patients aged 45 years or younger at the time of biochemical collection or available data of an age-stratified <45-years group; acute knee injury (eg, ACL rupture, PCL rupture, meniscus tear, osteochondral fracture); inflammatory cytokines had to be measured within 1 year after injury; article had to be written in English, German, French, Spanish, or Dutch; and full text was available.
Exclusion criteria were history of knee trauma before current trauma; pre-existing radiological degenerative changes; knee fractures, except osteochondral fractures; no available control group, such as healthy controls or the contralateral knee; intra-articular anti-inflammatory treatment (eg, biologics, glucocorticoids); post-mortem studies; animal studies; not an original study (eg, reviews or editorials).
Data Extraction
One reviewer (MN) extracted the data of the included studies. Extracted patient characteristics were age, sex, body mass index (BMI), time between injury and sample collection and type of injury. Time between injury and protein measurement was classified as acute (0-6 weeks) and subacute (>6 weeks to 1 year). The included inflammatory proteins are shown in
Risk of Bias Assessment
To assess the potential risk of bias, 2 reviewers (M.N. and M.R.) independently assessed all included studies using the Cochrane Collaboration’s tool for assessing risk of bias of prognostic studies. Both reviewers discussed their findings and asked a third reviewer (DM) for consensus if necessary.
The checklist consists of 12 criteria, which were divided into 4 categories: selection bias, information bias, confounding, and statistical bias. We considered a study to have a high risk of bias in case of 1 negative score in any of the categories, moderate risk of bias in case of a question mark in any of the categories and low risk of bias in case of no negative scores or question marks.
Selection bias was assessed by 2 items, namely “did the study have a clearly described population composed of patients in the same part of the disease timeline” (1) and “was there a long enough follow-up” (2).
Information bias was assessed by 4 items, namely “were outcomes described explicitly and objectively” (3), “were measurement of outcomes measured in a valid and reliable manner” (4), “were outcome assessors blinded for prognostic factors” (5), and “was there a sufficient proportion (≥80%) of follow-up available” (6).
Confounding was assessed by 4 items namely, “were prognostic factors described explicitly and objectively” (7), “were measurements of prognostic factors measured in a valid and reliable manner” (8), “was measurement of prognostic factors measured the same way for all patients and during a comparable part of the disease period” (9), and “were prognostic factors measured in a sufficiently large part (≥90%) of the study population” (10).
Statistical bias was assessed by 2 items, namely “were all patients included in the final analysis” (11), and “was statistical analysis done correctly” (12).
Statistical Analysis
The clinical and methodological homogeneity of the included studies was checked to evaluate whether a meta-analysis could be performed. In case of heterogeneity a best evidence synthesis was performed. We divided extracted findings into 4 evidence levels: strong, limited, conflicting, and no evidence. 24 Therefore, we followed the Strength of Recommendation Taxonomy (SORT). 25 Strong evidence consisted from consistent findings (> 75%) from at least 2 studies with low or moderate risk of bias. Limited evidence consisted of 1 study with low or moderate risk of bias or consistent findings from studies with a high risk of bias. Conflicting evidence was defined as inconsistent findings (less than 75% consistency) in the studies. If none or only 1 study with a high risk of bias was available, we defined the outcomes as no evidence.
Results
Study Characteristics
A total of 7757 articles were identified of which finally 10 were included (Fig. 1). The 10 studies published data from 9 unique trials (Table 1). Struglics et al., 33 and Larsson et al., 31 both used data from the KANON trial. Baseline characteristics are found in Table 1. ACL injuries were evaluated in all studies. Six of the reviewed studies only assessed patients with ACL injuries.26-29,32, 33 One study evaluated patients with ACL tears, meniscal tears, cartilage injury or ACL, and meniscal injury and 1 study analyzed patients with ACL tears with or without concomitant cartilage injury.27,30 Two studies included a heterogenous group of knee injuries combining ACL injuries, meniscal injuries and other injuries as a group.34,35 Of the investigated studies 8 evaluated synovial fluid samples and 5 evaluated serum samples (Table 2). An overview of the inflammatory biomarkers per study can be found in Table 2.
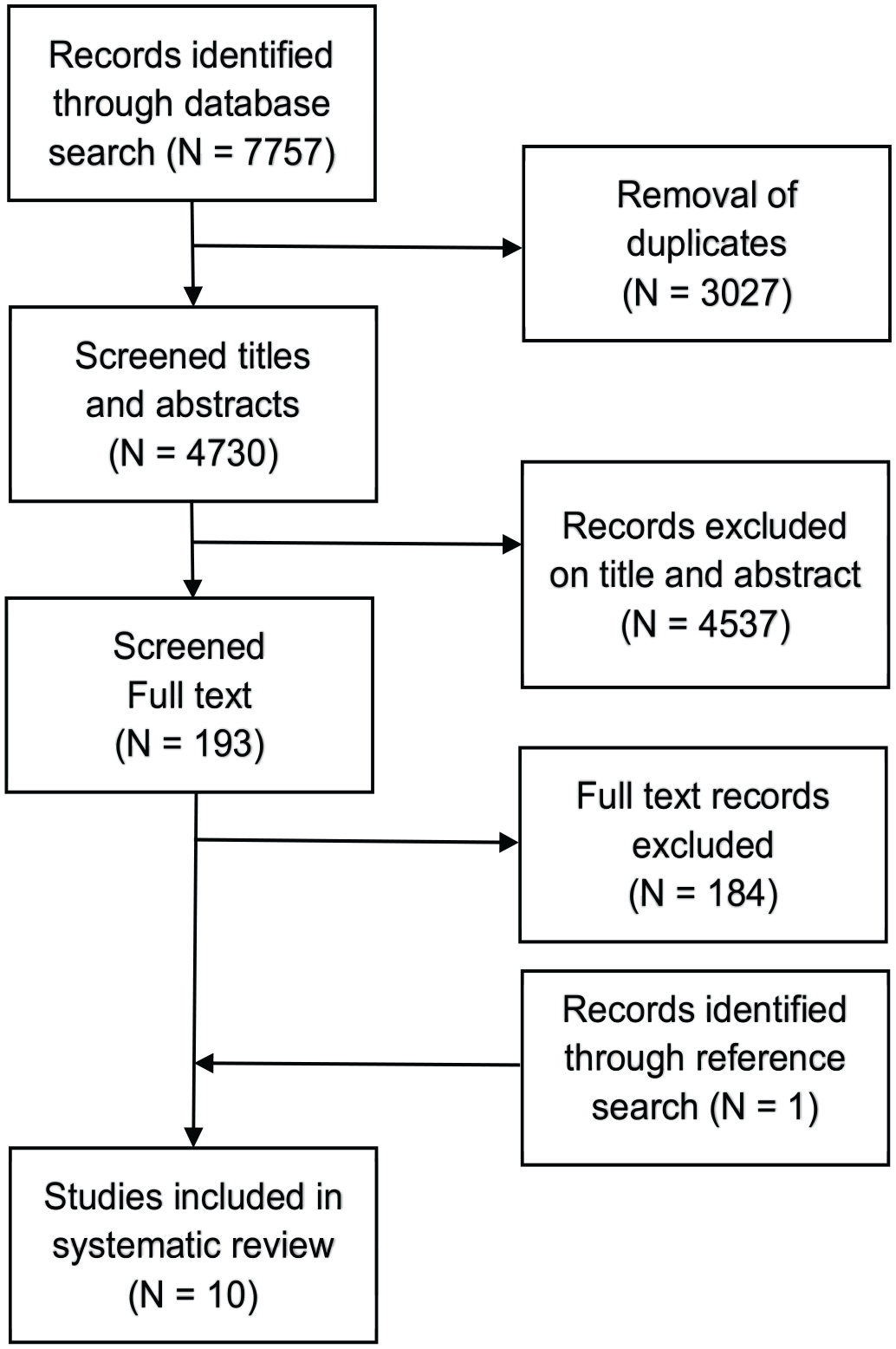
Flow chart of included and excluded studies.
Characteristics of the Included Studies.
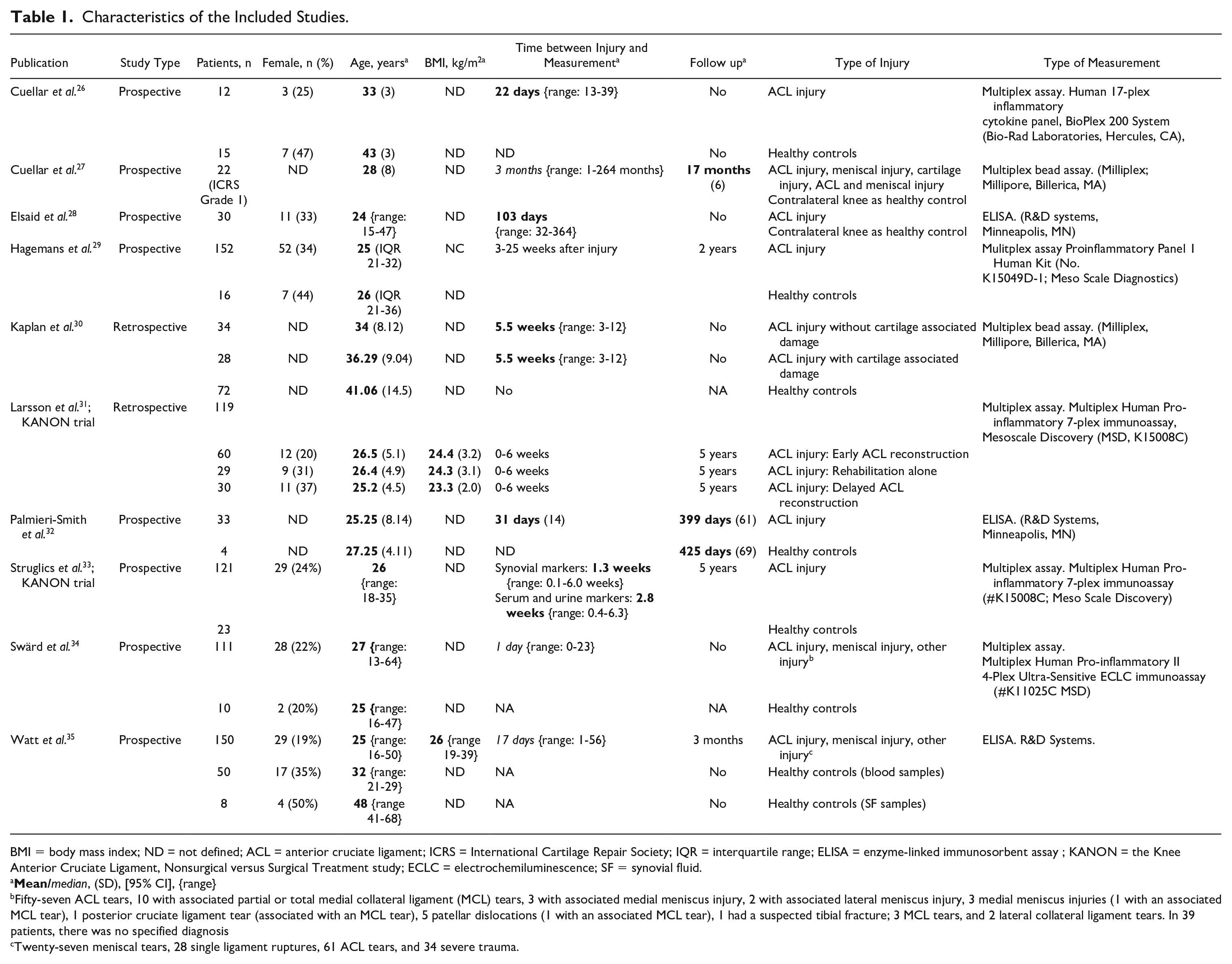
BMI = body mass index; ND = not defined; ACL = anterior cruciate ligament; ICRS = International Cartilage Repair Society; IQR = interquartile range; ELISA = enzyme-linked immunosorbent assay ; KANON = the Knee Anterior Cruciate Ligament, Nonsurgical versus Surgical Treatment study; ECLC = electrochemiluminescence; SF = synovial fluid.
Fifty-seven ACL tears, 10 with associated partial or total medial collateral ligament (MCL) tears, 3 with associated medial meniscus injury, 2 with associated lateral meniscus injury, 3 medial meniscus injuries (1 with an associated MCL tear), 1 posterior cruciate ligament tear (associated with an MCL tear), 5 patellar dislocations (1 with an associated MCL tear), 1 had a suspected tibial fracture; 3 MCL tears, and 2 lateral collateral ligament tears. In 39 patients, there was no specified diagnosis
Twenty-seven meniscal tears, 28 single ligament ruptures, 61 ACL tears, and 34 severe trauma.
Overview of the Used Inflammatory Proteins Per Study.
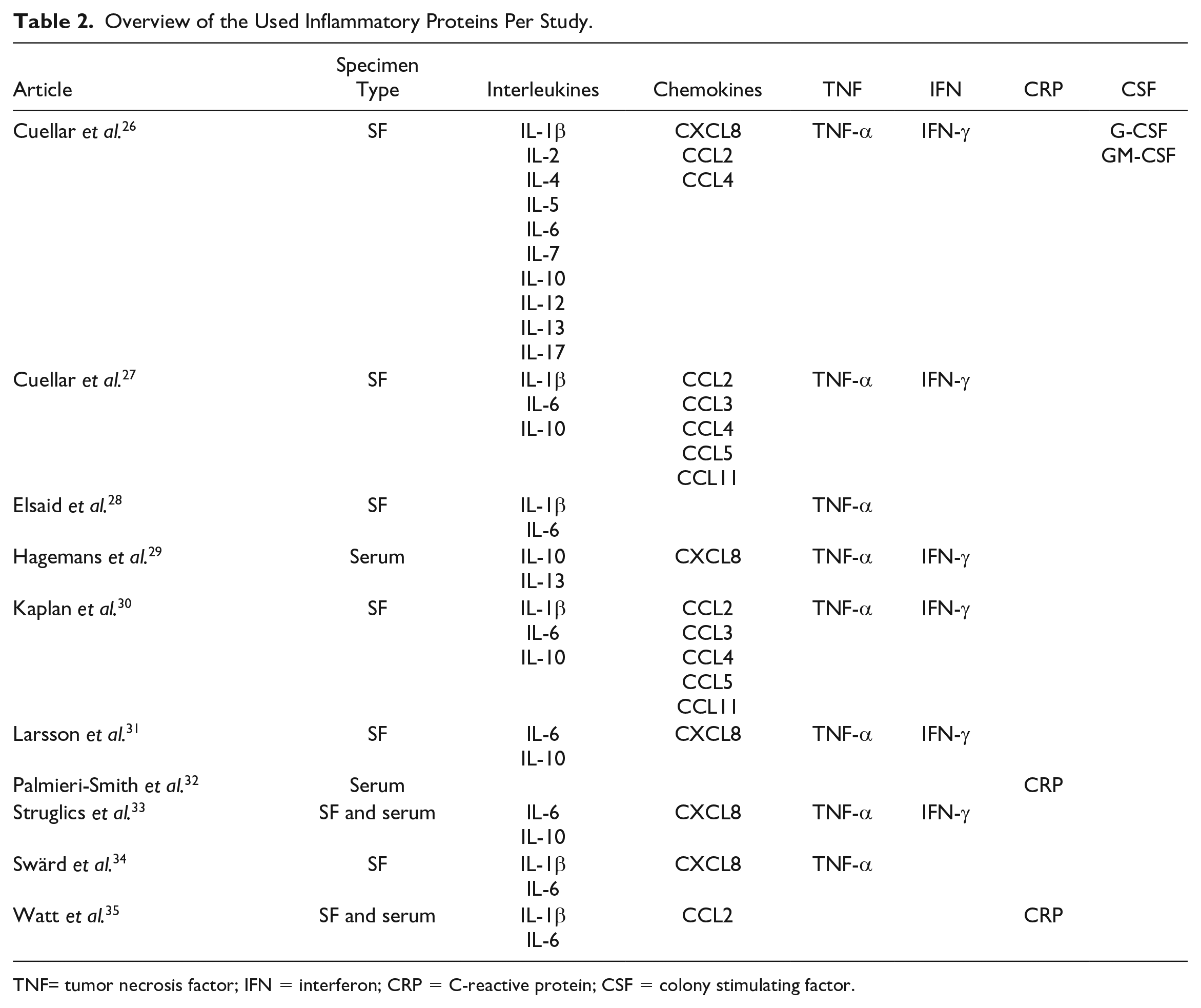
TNF= tumor necrosis factor; IFN = interferon; CRP = C-reactive protein; CSF = colony stimulating factor.
Risk of Bias Assessment
Table 3 shows the risk of bias for the included studies. One study has a low risk of bias, 26 and 2 studies a moderate risk of bias.29,34
Risk of Bias Assessment.
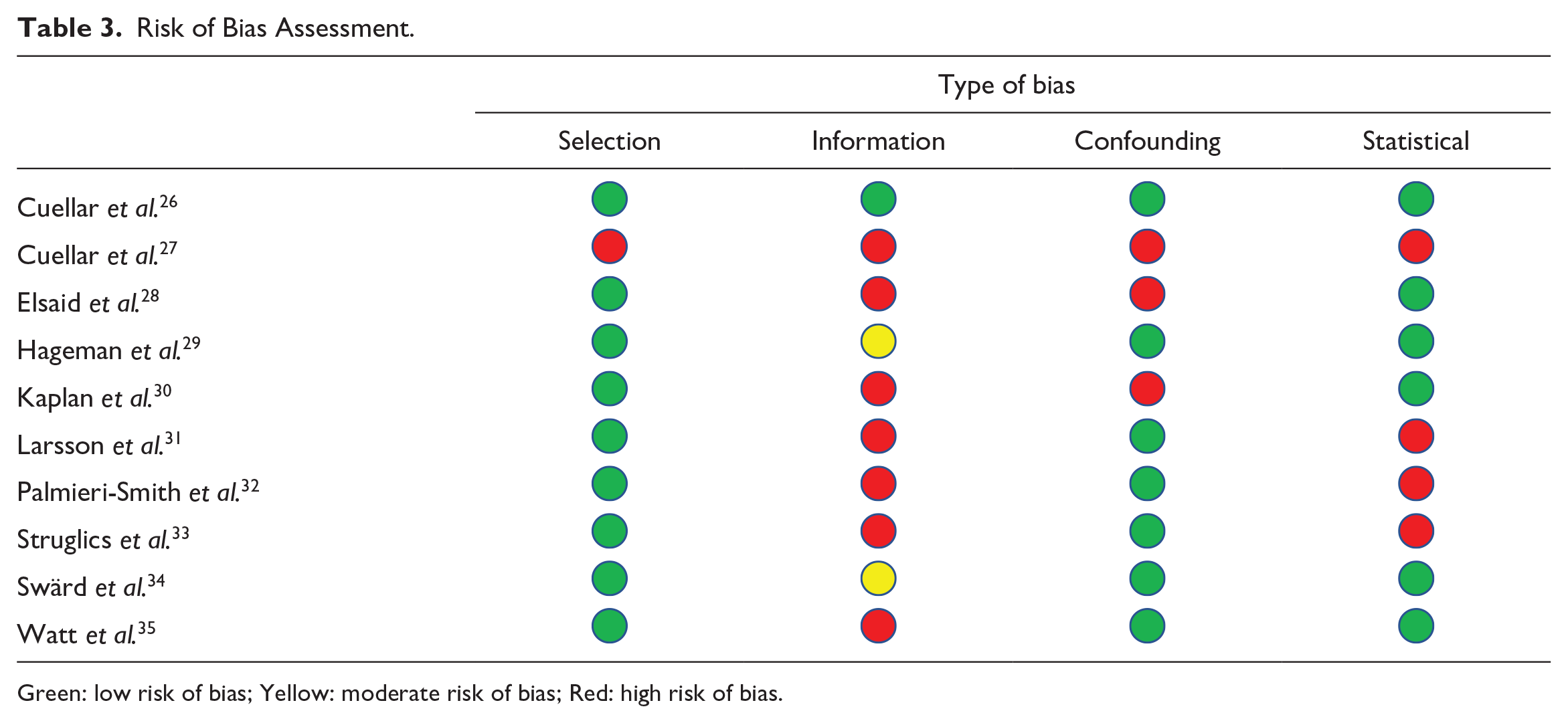
Green: low risk of bias; yellow: moderate risk of bias; red: high risk of bias.
Synovial Fluid Biomarkers
Several studies investigated the levels of IL-1β, IL-6, IL-10, CXCL8, CCL2, CCL4, TNF-α, and IFN-γ in synovial fluid after an acute knee injury, using a control group. Tables 4 and 5 and
Upregulation after trauma
Strong evidence was found for increased concentrations of SF biomarker IL-6 (Table 4) in acute injuries of the knee (0-6 weeks after trauma).26,27,30,31,33-35 Limited evidence was found for increased concentrations of SF biomarkers CCL4 (Table 5) in acute injuries of the knee.26,27,30 For IL-6, the available evidence for the subacute phase is conflicting showing no significant difference after 6 weeks between injured knees and controls in the KANON trial and a significant difference at 3 months in the study by Cuellar et al. (see Table 4).26,27,30,31,33-35 Regarding subacute measurements not enough data are available for CCL4.
Synovial Fluid IL-6 Outcomes.
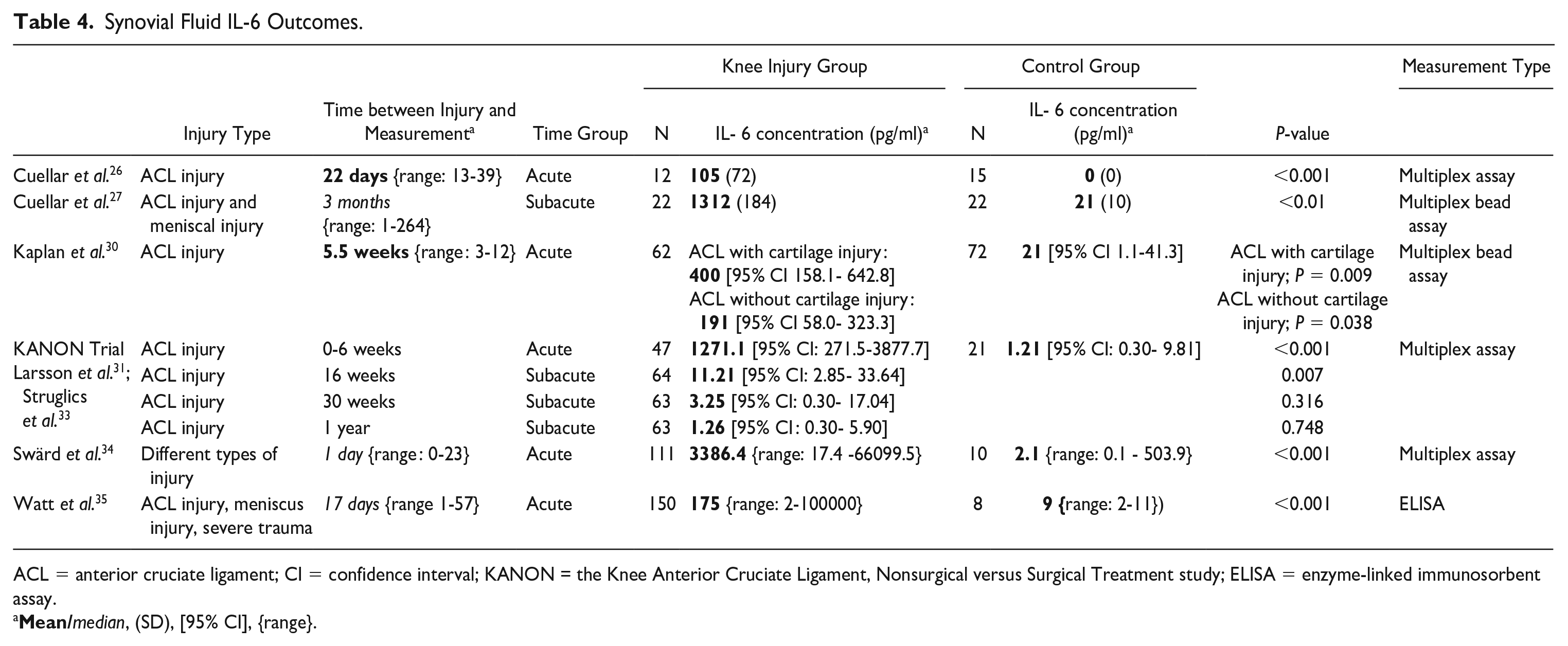
ACL = anterior cruciate ligament; CI = confidence interval; KANON = the Knee Anterior Cruciate Ligament, Nonsurgical versus Surgical Treatment study; ELISA = enzyme-linked immunosorbent assay.
Synovial Fluid CCL4 Outcomes.
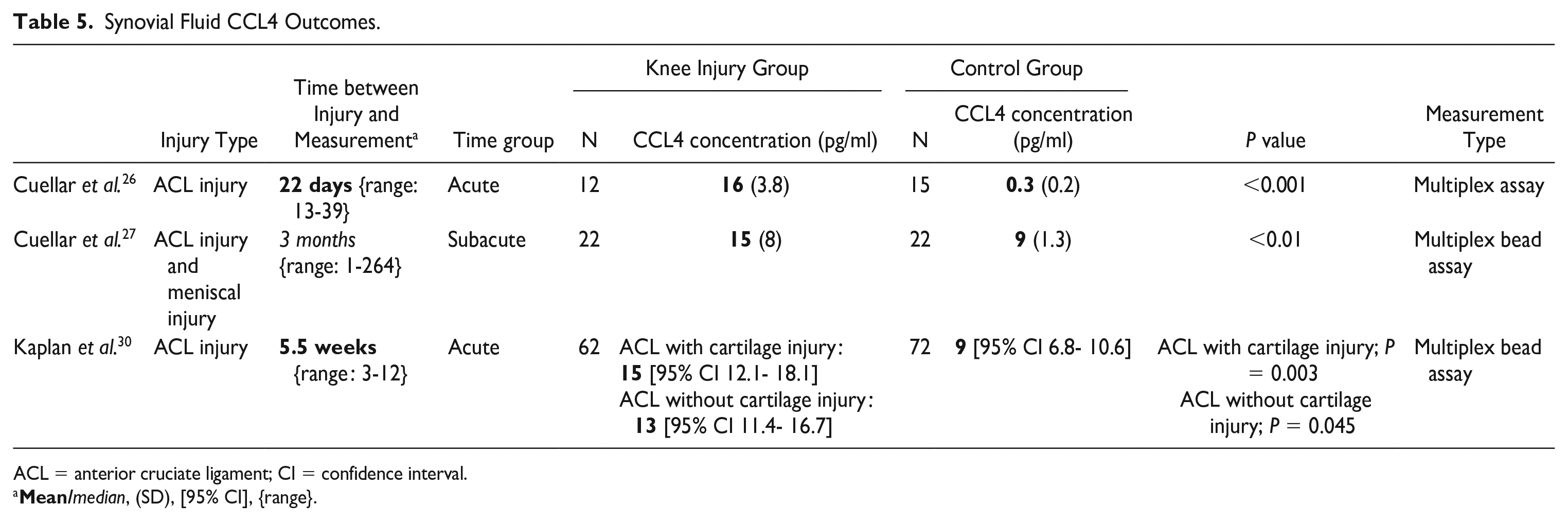
ACL = anterior cruciate ligament; CI = confidence interval.
No increase in concentration after knee injury
Limited evidence by 1 study with a low risk of bias was found for similar IL-2, G-CSF, and GM-CSF concentrations in patients with or without an acute knee-injury (Table 6). 26 No evidence was available regarding the subacute measurements for IL-2, G-CSF, and GM-CSF.
Synovial Fluid IL-2, G-CSF and GM-CSF Outcomes.
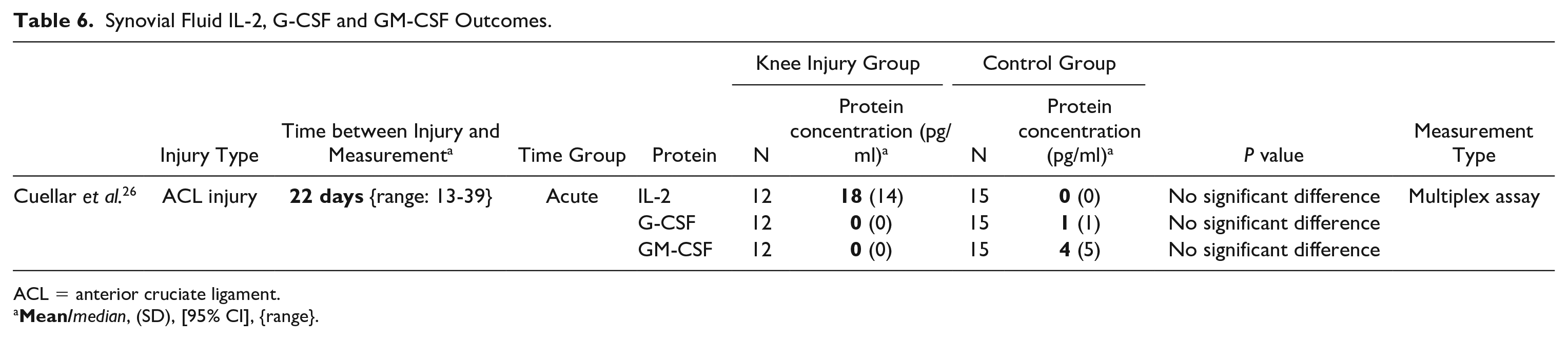
ACL = anterior cruciate ligament.
Limited evidence was found for no increase of concentration after knee injury for CCL3, CCL5, and CCL11 concentrations in 2 separate studies with high risk of bias at, respectively, 5.5 weeks (range 3-12 weeks) and 3 months after trauma (range 1-264 months).27,30 When subdividing in acute and subacute measurements, no conclusions could be made, because of the low quality of included studies and only 1 study available per group.
For IL-10 limited evidence showed no difference between the control group and knee injury group (
Conflicting findings
Conflicting findings were found for IL-1β, CXCL8, CCL2, TNF-α, and IFN-γ (see
No evidence
For IL-4, IL-5, IL-7, IL-12, IL-13, and C-reactive protein (CRP), not enough evidence was available.26,35 Concerning IL-1α, IL-1Ra, IL-1, IL-3, IL-9, IL-15, CCL7, CCL8, CCL12, CCL13, CCL22, CX3CL1, CXCL1, CXCL2, CXCL3, CXCL10, and M-CSF no evidence was available.
Serum Measurements
Limited evidence was found for no upregulation of IL-6 and CRP in serum after knee injury.32,33,35 Also IFN-γ, CXCL8, and IL-10 concentrations were not different in serum between the knee injury group and healthy control group.29,33 Conflicting evidence was found for TNF-α.29,33,34 For the other serum biomarkers included in this review, no evidence was available.35,36
Discussion
Summary of Synovial Fluid Biomarkers after Knee Trauma
In this review, we found that IL-6 and CCL4 are upregulated in SF after acute knee injury. All studies which included IL-6 and CCL4 found a significant difference in concentration between the knee injury group and control group (See
In contrast, we found unchanged concentrations in SF after trauma for IL-2, IL-10, CCL3, CCL5, CCL11, G-CSF, and GM-CSF in SF. For IL-10, the evidence was conflicting, but >75% of available studies using different datasets, showed no significant difference between the control group and knee injury group.26,27,30,31,33 For IL-2, G-CSF, and GM-CSF 1 high-quality study was available. 26 No significant difference between healthy controls and injured knees was shown in this study.
For IL-1β, CXCL8, CCL2, TNF-α, and IFN-γ the findings were conflicting. Regarding IL-1β, CXCL8, and TNF-α, 2 studies had a low or moderate risk of bias (see
Summary of Serum Biomarkers after Knee Trauma
There was limited evidence for no change in serum levels for IL-6, IL-10, CXCL8, IFN-γ, and CRP between controls and the knee injury group reported for each protein by at least 2 independent studies.29,33,35,37 Other serum biomarkers such as IL-1β, IL-2, IL-13, CCL2, CCL3, CCL4, and CCL5, were also not higher in the knee-injury group than in the control group. This was only reported by 1 study for each separate protein, therefore no conclusions could be made about these proteins.33,35,37 For TNF-α, the data were conflicting.29,33 Hagemans et al. 29 found a significant increase of concentration in serum after ACL rupture (3-25 weeks after trauma). This remained significantly higher than the healthy control group until the 2-year follow-up. This was not supported by data from Struglics et al. The low number of available studies and high risk of bias requires further research, but the absence of any increased concentration of inflammatory cytokines in serum debates the use of these serum markers in future research considering knee injury and onset of PTOA.
Time between Injury and Measurement
With the limited available evidence, subgroup comparison in terms of acute (0-6 weeks after trauma) and subacute measurements (6 weeks to 1 year after trauma) further decreases the amount of evidence.
Higher concentrations of IL-6 and CCL4 are only found in the acute setting, whereas we could not prove this for the subacute setting
The study by Swärd et al. did the harvest of SF mostly on the first day after trauma. They found increased concentrations in SF for IL-1β, IL-6, CXCL8, and TNF-α in the knee injury group, where other studies failed to show a significant difference with longer time intervals between injury and sampling
We therefore question the use of SF sampling for inflammatory proteins in a subacute phase after knee injury. Our findings imply only increased concentrations of IL-6 and CCL4 in the acute phase and not in the subacute phase
Inflammatory Markers and PTOA
A first step to prevent PTOA is to understand the role of inflammation in the pathogenesis. This has been thoroughly investigated with animal model studies. A variety of animal studies can be used to provoke joint injury either surgically, traumatic or chemically. 43 An ovine model was used to compare ACL detachment and direct reconstruction to prevent instability, with sham surgery and healthy controls.44,45 Cartilage changes and osteophyte formation were found 2 weeks after ACL repair when compared to healthy controls.21,44,45 At 20 weeks from ACL repair changes were consistent with early OA, although progression from 2-week follow-up was minimal. Changes were significantly different with early osteophyte formation and cartilage damage in the ACL reconstruction group versus sham surgery and healthy controls. Synovium samples were taken at 2 and 20 weeks. Messenger RNA expressions of IL-1β and IL-6 were both significantly upregulated in synovium for ACL repaired knees versus the healthy contralateral control. The inflammatory response normalized at 20 weeks. This article shows that in absence of instability the immediate postinjury inflammatory response in an ovine model contributes to early cartilage degeneration. 44 In another rabbit model, 2 holes were drilled into subchondral bone in the intercondylar notch. 46 Creating an injury which neither changes joint loading or mechanics of the joint. Synovium was examined for histology and changes in mRNA expression for IL-1β, IL-1Ra, IL-6, IL-8 and TNF-α at 72 hours, 3, 6, 8, and 52 weeks compared to sham surgery and unoperated healthy controls. All surgical damaged joints showed gross and histological cartilage damage after surgery with significant worsening until 52 weeks. The sort-term synovial inflammatory expression of IL-1β, IL-1Ra, IL-6, IL-8, and TNF was increased 3 to 4 times at 72 hours. This resolved to baseline levels by 3 weeks. The authors conclude that intra-articular bone injury creates an early joint inflammation with progressive cartilage damage consistent of OA in a rabbit model. 46 Both studies show that without altering joint loading, stability, or mechanics of the joint an early inflammation phase exist after knee injury. Resolving in cartilage changes consistent with early OA in animal models.
Second, we need to identify important biomarkers which could be a potential target for treatment in humans. Which is in the scope of this review. Third, correlation between these inflammatory markers and PTOA should be assessed. The lack of available data correlating biomarker data and long-term follow-up radiographic data makes this difficult. One study is available correlating biomarkers taken after knee trauma followed by ACL reconstruction with radiological follow-up.
47
Biomarkers were measured 64 days (standard deviation [SD] = 27.1) after trauma and correlated with magnetic resonance imaging (MRI) findings of different time points after surgery (6 months, 1, 2, and 3 years). The authors performed a cluster analysis with 2 groups. One high inflammatory group (with increased concentrations of IL-rα, IL-1α, IL-6, IL8, IL-10, TNF-α, and IFN-γ
A possible explanation may be the time course of the inflammatory markers. Biomarkers were measured at 64 days (SD = 27.1), which is relatively late after trauma, the level of these markers may have normalized by then. For IL-10, CXCL8, and IFN-γ the available data from especially the KANON trial showed these markers were elevated up to 6 weeks after knee injury.31,33 After this timeframe of 6 weeks, no significant difference were found when comparing the knee injury group with healthy controls (see
No other studies available determined the association between inflammatory markers and the radiographical development of PTOA. Further research with radiological follow-up is needed to clarify the relationship between the inflammatory response after knee injury and PTOA.
Future Treatment Options for PTOA
Instead of late-stage treatment of PTOA, there has been a shift in focus toward preventing or delaying early disease progression. 56 By understanding the inflammatory response after knee injury, we could investigate potential treatment strategies in the prevention of PTOA. Different treatment strategies and approaches for treatment are currently studied. For example, disease-modifying OA drugs (DMOADs), such as dexamethasone and triamcinolone acetonide or intra articular anti-inflammatory treatments such as hylarunoic acid, inhibitors of TNF-α, IL-1ra, IL-1β, IL-6, the complement system or anti-inflammatory cytokines IL-4, IL-10, and IL-13. 57
In this review, we have summarized the direct inflammatory response after knee injury from clinical studies. Two out of 37 included inflammatory proteins were found to be elevated after knee injury and can act as a biomarker. For 13 other inflammatory proteins, results were conflicting or not enough data were available, as while for 15 inflammatory proteins no evidence was available.
In absence of a direct clinical relation between PTOA and most of the included inflammatory proteins combined with the current lack of clinical data, further research is needed to identify the role of these proteins in the development of PTOA.
Limitations
Unfortunately, there was high heterogeneity between studies. The time between injury and measurement of biomarkers varied a lot in different studies. Furthermore, biomarkers were measured with different techniques based either on a protein level (enzyme-linked immunosorbent assay (ELISA), multiplex bead assay, multiplex assay) and with different calibrators resulting in different concentrations. Because of these differences, directly comparing results between studies was not possible. Therefore, we focused our research on studies using a control group, so measurement type and calibration is the same for injury and control groups. The control group could be a healthy control or the contralateral knee. Using the contralateral knee as a healthy control is controversial because of the possible systemic inflammatory reaction. By using the reported statistically significance per study, underpowering and study sizes may influence our outcomes. Also, the overall risk of bias of the included studies was high.
Conclusion
There is strong evidence for IL-6 and limited evidence for CCL4 supporting elevation of these proteins in SF after acute knee injury up to 6 weeks. Further studies are needed to identify the role of these proteins in the development of PTOA. For IL-2, CCL3, CCL5, CCL11, G-CSF, and GM-CSF in SF, there is limited evidence that these biomarkers are not elevated after an acute knee injury. Interleukin 10 and IFN-γ show conflicting results in the acute phase and no increased concentrations after injury in the subacute phase. For other included inflammatory proteins there is not enough available data. Further research must resolve this question considering these inflammatory markers and should focus on SF measurements taken in the acute phase (less than 6 weeks) after knee injury.
There is limited evidence available concluding no increased concentrations of inflammatory proteins IL-6, IL-10, CXCL8, IFN-γ, and CRP in serum after acute knee injury.
In absence of a direct clinical role for most of the included inflammatory proteins and current lack of clinical data, we must not rule out the potential role of inflammatory proteins as a predictor or treatment modality for PTOA.
Supplemental Material
sj-docx-1-car-10.1177_19476035221141417 – Supplemental material for Improved Understanding of the Inflammatory Response in Synovial Fluid and Serum after Traumatic Knee Injury, Excluding Fractures of the Knee: A Systematic Review
Supplemental material, sj-docx-1-car-10.1177_19476035221141417 for Improved Understanding of the Inflammatory Response in Synovial Fluid and Serum after Traumatic Knee Injury, Excluding Fractures of the Knee: A Systematic Review by Michael F. Nieboer, Max Reijman, Marinus A. Wesdorp, Yvonne M. Bastiaansen-Jenniskens and Duncan E. Meuffels in CARTILAGE
Supplemental Material
sj-docx-2-car-10.1177_19476035221141417 – Supplemental material for Improved Understanding of the Inflammatory Response in Synovial Fluid and Serum after Traumatic Knee Injury, Excluding Fractures of the Knee: A Systematic Review
Supplemental material, sj-docx-2-car-10.1177_19476035221141417 for Improved Understanding of the Inflammatory Response in Synovial Fluid and Serum after Traumatic Knee Injury, Excluding Fractures of the Knee: A Systematic Review by Michael F. Nieboer, Max Reijman, Marinus A. Wesdorp, Yvonne M. Bastiaansen-Jenniskens and Duncan E. Meuffels in CARTILAGE
Supplemental Material
sj-docx-3-car-10.1177_19476035221141417 – Supplemental material for Improved Understanding of the Inflammatory Response in Synovial Fluid and Serum after Traumatic Knee Injury, Excluding Fractures of the Knee: A Systematic Review
Supplemental material, sj-docx-3-car-10.1177_19476035221141417 for Improved Understanding of the Inflammatory Response in Synovial Fluid and Serum after Traumatic Knee Injury, Excluding Fractures of the Knee: A Systematic Review by Michael F. Nieboer, Max Reijman, Marinus A. Wesdorp, Yvonne M. Bastiaansen-Jenniskens and Duncan E. Meuffels in CARTILAGE
Supplemental Material
sj-docx-4-car-10.1177_19476035221141417 – Supplemental material for Improved Understanding of the Inflammatory Response in Synovial Fluid and Serum after Traumatic Knee Injury, Excluding Fractures of the Knee: A Systematic Review
Supplemental material, sj-docx-4-car-10.1177_19476035221141417 for Improved Understanding of the Inflammatory Response in Synovial Fluid and Serum after Traumatic Knee Injury, Excluding Fractures of the Knee: A Systematic Review by Michael F. Nieboer, Max Reijman, Marinus A. Wesdorp, Yvonne M. Bastiaansen-Jenniskens and Duncan E. Meuffels in CARTILAGE
Supplemental Material
sj-docx-5-car-10.1177_19476035221141417 – Supplemental material for Improved Understanding of the Inflammatory Response in Synovial Fluid and Serum after Traumatic Knee Injury, Excluding Fractures of the Knee: A Systematic Review
Supplemental material, sj-docx-5-car-10.1177_19476035221141417 for Improved Understanding of the Inflammatory Response in Synovial Fluid and Serum after Traumatic Knee Injury, Excluding Fractures of the Knee: A Systematic Review by Michael F. Nieboer, Max Reijman, Marinus A. Wesdorp, Yvonne M. Bastiaansen-Jenniskens and Duncan E. Meuffels in CARTILAGE
Supplemental Material
sj-docx-6-car-10.1177_19476035221141417 – Supplemental material for Improved Understanding of the Inflammatory Response in Synovial Fluid and Serum after Traumatic Knee Injury, Excluding Fractures of the Knee: A Systematic Review
Supplemental material, sj-docx-6-car-10.1177_19476035221141417 for Improved Understanding of the Inflammatory Response in Synovial Fluid and Serum after Traumatic Knee Injury, Excluding Fractures of the Knee: A Systematic Review by Michael F. Nieboer, Max Reijman, Marinus A. Wesdorp, Yvonne M. Bastiaansen-Jenniskens and Duncan E. Meuffels in CARTILAGE
Supplemental Material
sj-docx-7-car-10.1177_19476035221141417 – Supplemental material for Improved Understanding of the Inflammatory Response in Synovial Fluid and Serum after Traumatic Knee Injury, Excluding Fractures of the Knee: A Systematic Review
Supplemental material, sj-docx-7-car-10.1177_19476035221141417 for Improved Understanding of the Inflammatory Response in Synovial Fluid and Serum after Traumatic Knee Injury, Excluding Fractures of the Knee: A Systematic Review by Michael F. Nieboer, Max Reijman, Marinus A. Wesdorp, Yvonne M. Bastiaansen-Jenniskens and Duncan E. Meuffels in CARTILAGE
Supplemental Material
sj-docx-8-car-10.1177_19476035221141417 – Supplemental material for Improved Understanding of the Inflammatory Response in Synovial Fluid and Serum after Traumatic Knee Injury, Excluding Fractures of the Knee: A Systematic Review
Supplemental material, sj-docx-8-car-10.1177_19476035221141417 for Improved Understanding of the Inflammatory Response in Synovial Fluid and Serum after Traumatic Knee Injury, Excluding Fractures of the Knee: A Systematic Review by Michael F. Nieboer, Max Reijman, Marinus A. Wesdorp, Yvonne M. Bastiaansen-Jenniskens and Duncan E. Meuffels in CARTILAGE
Footnotes
Author Contributions
All authors were involved in drafting the article, and all authors approved the final version to be published.
Acknowledgments and Funding
We thank Wichor Bramer, biomedical information specialist at Erasmus MC, for his assistance with our search. The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethics approval was not required for this systematic review.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
