Abstract
Importance:
The promising therapeutic potential and regenerative properties of platelet-rich plasma (PRP) have rapidly led to its widespread clinical use in musculoskeletal injury and disease. Although the basic scientific rationale surrounding PRP products is compelling, the clinical application has outpaced the research.
Objective:
The purpose of this article is to examine the current concepts around the basic science of PRP application, different preparation systems, and clinical application of PRP in disorders in the knee.
Evidence Acquisition:
A systematic search of PubMed for studies that evaluated the basic science, preparation and clinical application of platelet concentrates was performed. The search used terms, including platelet-rich plasma or PRP preparation, activation, use in the knee, cartilage, ligament, and meniscus. Studies found in the initial search and related studies were reviewed.
Results:
A comprehensive review of the literature supports the potential use of PRP both nonoperatively and intraoperatively, but highlights the absence of large clinical studies and the lack of standardization between method, product, and clinical efficacy.
Introduction
Musculoskeletal injuries limit the activity level and affect lifestyles of athletes at all levels. In addition, they representing a substantial health care burden. 1 Not only are acute injuries associated with immediate inflammation and pain, but chronic or poorly healing musculoskeletal injuries often are associated with increased inflammation and tissue catabolism that outpace anabolic reconstruction. The use of platelet-rich plasma (PRP) as a therapeutic treatment to control inflammation and enhance repair of musculoskeletal injuries is an approach that has recently grown in popularity.
The systematic search of PubMed performed between June 2011 and January 2013 for studies that evaluated the basic science, preparation, and clinical application of platelet concentrates revealed that although there is an abundance of published literature on the therapeutic use of PRP, clinical reports are predominantly case studies on various conditions that report mixed findings on efficacy. Taken as a whole, this body of evidence highlights a lack of standardization with respect to the devices and methods used for preparation and application of PRP across fields, including sports medicine. The absence of statistically significant randomized controlled trials makes it difficult to derive firm recommendations regarding the clinical utility of PRP therapeutics. Nonetheless, positive effects reported for PRP in multiple medical capacities, from its early use in maxillofacial and plastic surgery to sports medicine, and the underlying hypotheses for its mechanism of action are interesting, if not compelling.2-8 It is therefore worth considering that mixed reports on efficacy relate to a lack of standardization rather than an absence of therapeutic potential. This review examines the development and continuity of thought around the use of PRP based therapeutics through the examination of the proposed mechanisms of action, differences in PRP preparations, and published clinical findings on the use of PRP for injuries and disorders of joint tissues in the knee.
What Is Platelet-Rich Plasma?
Classification of Platelet-Rich Plasma
All PRP is not the same. Currently, PRP is a generic term used to describe a broad range of plasma products derived from a sample of whole blood. Depending on the methods and devices used to produce PRP, preparations differ significantly in cellular composition of red blood cells, platelets, leukocytes, and plasma proteins.
9
For example, the American Association of Blood Banks defines PRP as the resultant plasma fraction following a single light spin of whole blood, in which platelets are enriched in comparison with other cell types.
10
This definition refers to the methodology that produces a plasma fraction containing platelet concentrations close to those found in a whole blood sample and having greatly reduced, or undetectable levels of red blood cells and leukocytes. In stark contrast, the seminal publication by Marx
In attempt to distinguish PRP products, a recent publication proposes a classification system based on the absolute number of platelets, platelet activation method and leukocyte content. 12 While this classification may prove beneficial for meta-analysis of studies and provide some guidance with respect to the choice of a PRP system, to avoid confusion, it remains reasonable for investigators to provide quality control (i.e., cellular composition and platelet concentration) of the final therapeutic PRP preparation and to examine results with regard to efficacy and tissue metabolism.
Basic Scientific Rationale
PRP products are thought to facilitate the recruitment, proliferation, and maturation of cells that participate in the regeneration of tendon, ligament, muscle, bone, and cartilage. This is based on our understanding of the normal physiological role of platelets as first responders to injury. 13 In addition to hemostatic activity, platelets are known to release biomolecules that control myriad different biological activities. Through the efforts of the platelet proteome project, more than 1,500 different proteins have been identified in platelet releasate, and many of the important growth factors (GFs), cytokines, and chemokines have been defined as critical for processes necessary for effective tissue regeneration ( Table 1 ).3,14-16
Growth Factors, Cytokines, and Bioactive Molecules Associated with Platelets. a
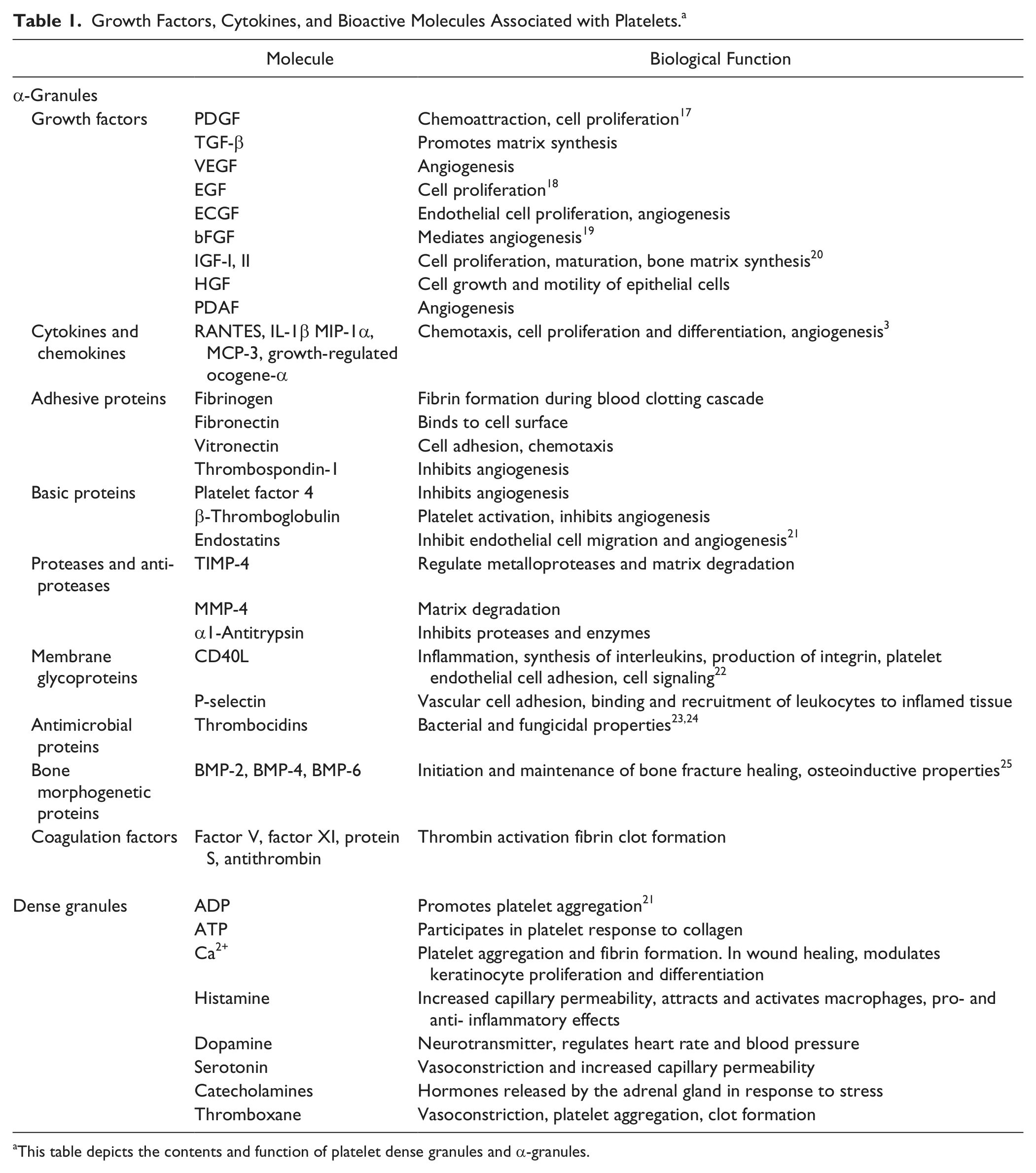
This table depicts the contents and function of platelet dense granules and α-granules.
Many of the chemical messengers and bioactive proteins released by platelets have been well studied in both
The alteration of these biomarkers indicates that PRP appears to trigger multiple biological pathways and also delivers multiple biological factors directly to the injection site. However, there is limited clinical evidence that reports optimum concentrations of the bioactive factors responsible for cytokine and chemokine expression in PRP products. Although the high level clinical evidence has yet to be developed, it is intuitive that PRP may indeed provide a unique therapy that drives physiological regeneration by activating the necessary biological pathways.
Preparation of Platelet-Rich Plasma Products
Many systems are available for the preparation of PRP: This results in significant differences in the composition of PRP products ( Table 2 ). The preparation of PRP begins with collection of autologous peripheral blood containing citrate (sodium citrate, calcium citrate, or acid-citrate dextrose) to inhibit the coagulation pathway. Two basic preparation systems exist; plasma-based PRP systems and buffy-coat–based PRP systems. These systems differ based on centrifugation spin parameters that directly affect the spatial distribution of blood-cell components. The centrifugation time and speed affect the number and concentration of platelets and other cell types within the PRP and thus availability of growth factors, chemokines as well as pro-inflammatory and anti-inflammatory mediators. Several examples of different PRP systems are presented in Table 2 .
Platelet-Rich Plasma (PRP) Preparation Systems. a
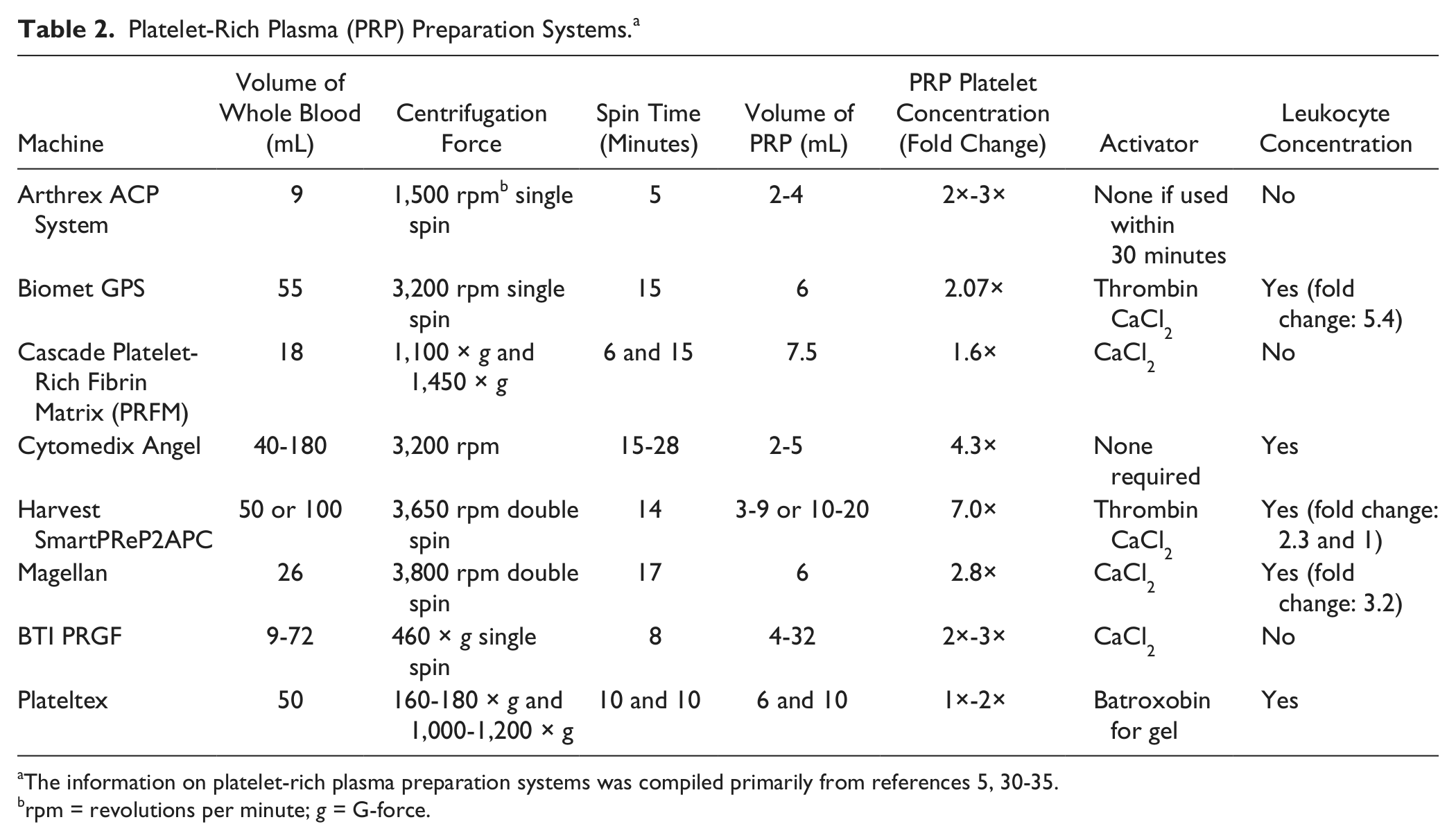
The information on platelet-rich plasma preparation systems was compiled primarily from references 5, 30-35.
rpm = revolutions per minute;
Not only are there differences in PRP preparations introduced by the technological features of individual devices, but there is also wide biological variation between patients. The normal range of platelets count in human whole blood is relatively broad (150,000-350,000 platelets/µL) and can vary considerably day to day.36,37 It is also noteworthy that the range of platelet size and density can vary depending on the size and distribution of megakarocyte precursors from which platelets are derived. 38
Variability of Platelet-Rich Plasma Products
Multiple studies show that the highest concentration of platelets does not necessarily stimulate and may even suppress cell proliferation and differentiation.36,39,40 Thus, the mechanics of preparation in different devices are likely to contribute to the different outcomes seen in clinical studies. The optimal number and/or concentration of leukocytes in the final preparation that will yield the greatest benefit remains controversial as well. It has been reported that PRP containing pro-inflammatory granulocytes may contribute to the inflammatory responses beneficial for wound healing as well as treating tendinopathy.41,42 On the other hand, the presence of these same pro-inflammatory cells may not be desirable for addressing pathologies related to inflammation, as is the case for inflammatory arthritides,43,44 as neutrophils secrete matrix metalloproteinase (MMP) components that have been shown to be degradative to tenocyte and chondrocyte repair following intra-articular PRP injection. 45
Two recent articles highlight relevant findings in the platelet versus white cell debate.8,46 Authors of the first study hypothesized that the concentration of growth factors and catabolic cytokines are dependent on the cellular composition of PRP. They compared PRP from 11 human volunteers using 2 commercial systems,
8
the PRP-I system (ACP Double Syringe System, Arthrex, Naples, FL), and the PRP-II system (GPS III Miniplatelet Concentration System, Biomet, Warsaw, IN). The PRP-I system had 1.9× platelets and 0.13× leukocytes compared with whole blood whereas PRP-II had 4.69× platelets and 4.26× leukocytes compared with whole blood. Growth factors were increased significantly in PRP-II compared with PRP-I, but catabolic cytokines (MMP-9, MMP-13, and IL-1β) also were increased significantly in PRP-II compared with PRP-I. The second study examined the effects of a single- versus double-spin preparation of PRP on anabolic and catabolic activities of cartilage and meniscal explants
Technical Aspects of Plate-Rich Plasma Application
For PRP to stimulate biological action, degranulation and release of growth factors from platelet α-granules at the site of application must occur. Although no evidence was provided, it has been suggested that PRP can be stored in its anticoagulated state for approximately 8 hours; however, once activated, immediate application is recommended.
47
The activation can occur in the preparation step before application via exogenous activation or
Bovine thrombin is often used as an activator to initiate clot formation before application. One potential complication presented in earlier literature was the potential to develop coaguopathies when using bovine thrombin, but this risk has been largely obviated with the use of highly purified bovine thrombin. However, because of continued perception of a safety risk, methods to activate PRP that do not rely on thrombin have been explored.
Dugrillon
Type I collagen can also be used for PRP activation and may be beneficial as endogenous collagen is already present in the environment of PRP application. In some
Another factor that must be considered is the duration of bioactive molecules at the injection site or interface once released from the platelet α-granules. While this can depend on the specific interface and the activation method, a recent study evaluated the kinetics of growth factor release in dental implants following PRP application. 55 Comparing the spatiotemporal relationship between implants treated with activated and nonactivated platelet-rich plasma and platelet-poor plasma, it was reported that activation and subsequent clot formation resulted in greater duration of growth factors at the interface. Furthermore, it was noted that the concentration of PDGFs at the interface was significantly higher in samples where activated-PRP was applied, but this concentration became negligible after 2 to 4 days. The authors concluded that prior to PRP application, products should be activated and allowed to clot to permit the adequate duration of bioactive factors at the injection site. Although this kinetic relationship examined in dental implants remains relevant, further investigation into the activation of PRP products and time-dependent release of growth factors must be considered in the application of PRP, particularly with intraoperative application when the injury site is copiously irrigated.
Rationale for Clinical Application of Platelet-Rich Plasma
The benefits seen early in maxillofacial and plastic surgery led to the use of PRP in myocardial surgery, treatment of ulcers, reproductive pathology, and orthopedics. As happens with novel medical advancements, when beneficial results were demonstrated, the clinical use of PRP expanded and outpaced the research behind the product. However, the application of PRP is experiencing widespread growth in this field and an increasing number of clinical trials as well as well-designed controlled studies are being performed to determine optimal application of PRP in tendon, muscle, ligament, bone, and cartilage injuries.
Basic Scientific Rationale of Platelet-Rich Plasma Use in Articular Cartilage Degradation and Repair
On a molecular level, results have indicated a beneficial effect of PRP on chondrocytes and mesenchymal stem cells. Increased cell proliferation and synthesis of proteoglycans and collagen type II has been demonstrated when cell cultures of chondrocytes56,57 and mesenchymal stem cells
58
are treated with PRP compared with controls treated with platelet-poor plasma or fetal bovine serum. PRP also promotes differentiation of subchondral bone progenitor cells. Kruger
Clinical Application of Platelet-Rich Plasma Use in Articular Cartilage Degradation and Repair in the Knee
Osteoarthritis
In an uncontrolled prospective study, 14 patients with knee OA were treated with PRP produced using anticoagulated blood processed in the GPS III system.
61
These authors reported reduced pain and improved functional outcome after a series of injections with PRP. They also reported improvement in the ultrasonographic measurement of femoral articular cartilage thickness in 6 out of 13 patients at 6 months and in the satisfaction survey at the 1-year follow-up examination, 8 of 13 patients reached their goals at 1 year (
Table 3
). Another study showed that 261 patients presenting with OA and treated with a series of 3 PRGF injections demonstrated statistically significant improvement in knee outcome assessment scales, including visual analog scale (VAS), Health Survey Scoring Demonstration (SF-6), Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), and Lequesne Index, 6 months following the final intra-articular injection (
Table 3
).
62
Napolitano
Published Human Studies of Platelet-Rich Plasma (PRP) Clinical Application in Osteoarthritis.

EQ-VAS = EuroQoL visual analog scale; IKDC = International Knee Documentation Committee; KOOS = Knee Injury and Osteoarthritis Outcome Scores; NRS = Numerical Rating Scale; SF-36 = Health Survey Scoring Demonstration; VAS = visual analog scale; WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index.
Kon
In a study comparing 2 different PRP preparation approaches, Filardo
A retrospective study comparing the effectiveness of 30 patients who received PRP injections versus 30 patients who received HA injections for the treatment of knee OA, yielded encouraging results.
69
Three injections were administered over a 3-week period and by week 5, the success rate for the WOMAC pain subscale reached 33.4% for the PRP group versus 10% for the HA group (
Intraoperative Use of Platelet- Rich Plasma in Cartilage Repair
PRP has also been investigated as a treatment option for cartilage repair and its application to chondral defects using both animal and human models has been evaluated in a clinical setting. In an ovine model, treatment of 15 chronic full-thickness chondral lesions of the knee using microfracture supplemented with PRP and fibrin glue resulted in improved outcomes compared with the microfracture-alone controls.
74
The PRP product consisted of 60 mL of autologous blood centrifuged in a 2-step process (2400 rpm 3 minutes, 3000 rpm 12 minutes), resulting in 6 to 8 mL of PRP. Similarly, Sun
Basic Scientific Rationale of Intra-Articular Use of Platelet-Rich Plasma in Meniscal Injuries
Damage to meniscal tissue presents unique challenges because of the absence of healing at the avascular zone, the accelerated degeneration of articular cartilage and increased rate of knee OA that can occur following a meniscal injury.
76
Application of PRP represents a potential therapeutic technique to stimulate proliferation and enhance the healing process of menisci. Although there are limited published studies evaluating the efficacy of PRP application in meniscal injuries, Ishida
Basic Scientific Rationale of Intra-Articular Use of Platelet-Rich Plasma in Ligaments
Several
Perhaps of more interest is the effect of PRP on anterior cruciate ligament (ACL) cells. ACL rupture is a common injury that often requires surgical reconstruction, but the absence of normal knee kinematics and premature osteoarthritic changes that can result following operative treatment
80
and may in part be a result of incomplete healing, call for the investigation into therapeutics that augment the repair process, such as PRP. Cheng
In another study, Yoshida and Murray 83 examined the effect peripheral blood mononuclear cells (PBMCs), a byproduct that includes monocytes and lymphocytes but excludes polymorphonucleocytes and often is removed from PRP products along with other WBC components on ACL fibroblasts cultured on collagen scaffolds with and without platelets and plasma. When exposed to PBMCs in the presence of platelet products, ACL fibroblasts demonstrated increased collagen gene expression, collagen protein expression, and cell proliferation. Not only do these results indicate the positive effect of some leukocyte components in PRP products, but this interaction between PBMCs and platelet products may also be beneficial in ACL reconstruction where collagen production by fibroblasts is desired.
Clinical Application of Intra-Articular Use of Platelet-Rich Plasma in Ligaments
Intraoperative Use of Platelet-Rich Plasma in Anterior Cruciate Ligaments
There are several studies that support the use of PRP products to augment ACL reconstruction in animal models, particularly when PRP is used in combination with a collagen-based carrier.84,85 Still other studies show these combined PRP products to be ineffective. The difference in results may be explained, at least in part, by the different methods of preparation of PRP derivatives used in these studies. Still, the resultant controversy leads to a lack of consensus.86-88
Mixed results and various manufacturing systems are also factors in evaluating human ACL reconstruction studies. In ACL reconstructions the tibial and femoral graft-to-bone healing and ligamentization of the tendon graft represent the primary biological processes that occur postoperatively.89-92 Measurements of these processes are used as indicators of healing following ACL reconstruction.
Many reports of the use of PRP alone in humans are associated with positive outcomes in ACL reconstruction.93,94 Sánchez
In a single-blinded controlled study, Radice
Published Human Studies of Platelet-Rich Plasma (PRP) Clinical Application in Ligament Injuries.
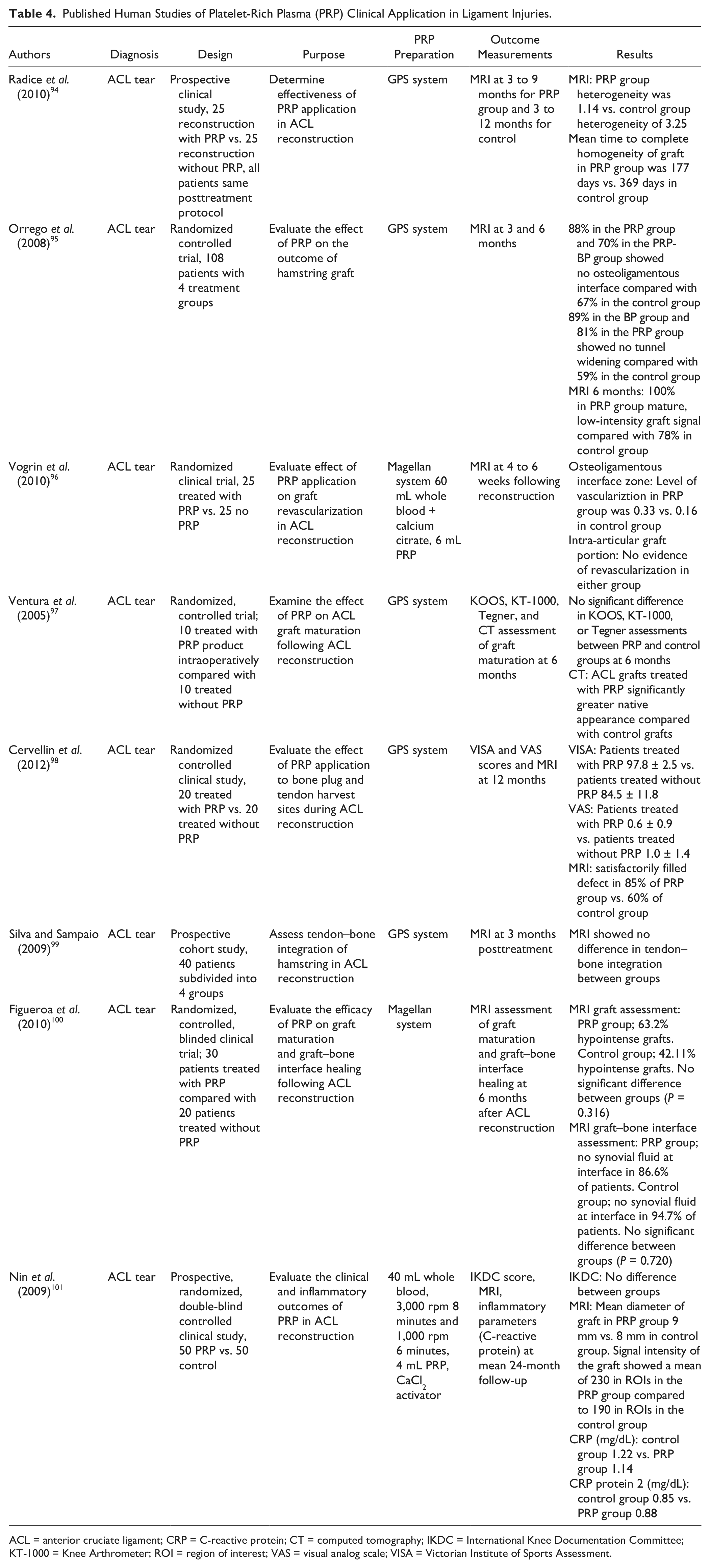
ACL = anterior cruciate ligament; CT = computed tomography; IKDC = International Knee Documentation Committee; KT-1000 = Knee Arthrometer; ROI = region of interest; VAS = visual analog scale; VISA = Victorian Institute of Sports Assessment.
In another clinical study, at the 12-month follow-up examination, Cervellin
On the other hand, some clinical studies have shown no beneficial effect of PRP use during ACL reconstruction. Silva and Sampaio
99
failed to find a significant difference in MRI assessment of the fibrous interface of the reconstructed ACL hamstring graft and the femoral tunnel treated with GPS system–produced PRP at 3 months (
Table 4
). Also, in a study on 50 patients following ACL reconstruction, Figueroa
Three different products with differing cellular components and various outcome measurements were used between the ACL studies, making it difficult to compare the overall efficacy of treatment ( Table 4 ). Moreover, the limited sample size resulted in a beta error that may account for equivocal results reported in some studies. Although some results suggest that PRP can enhance graft remodeling and contribute to improved interface healing, collectively, the outcomes of the effect of PRP on ACL reconstruction both short term and long term is still in need of further exploration.
Further Considerations
The promising potential of PRP has led to its rapidly expanding use in sports medicine. The inconsistent results between studies, however, demonstrate that, although the groundwork is laid, the true efficacy of PRP is yet to be determined. The use of PRP in sports medicine is supported in some clinical studies, showing a clear trend toward patient or lesion improvement; but, there are also well-designed controlled studies that fail to demonstrate any significant effect of PRP.
A recent meta-analysis that evaluates the use of PRP in orthopedic indications highlights the inconsistencies across studies that must be remedied to determine the efficacy of PRP. 102 This systematic review quantitatively assessed methodological quality and functional outcome measurements of 33 randomized controlled trials or prospective cohort studies. Of these, only 22 studies (61%) reported the manufacturer of the platelet separation system used, only 20 were considered of high methodological quality, and 27 different functional outcomes were used. The authors concluded that considerable uncertainty about the benefit of PRP remains and that future studies must address the deficiencies found in the current body of literature and the cases presented in this review. Thus, it remains difficult to draw conclusions in comparing current studies; for not only is the very definition of what constitutes PRP unclear, but the machine, processing, volume, concentration, contamination with other blood cells, treatment interval and frequency, posttreatment therapy, and many other factors, vary between reports.
Since the preparation methods and PRP products are not the same, it seems reasonable to first standardize the preparation of PRP to study its effect. Individual patients blood counts likely contribute to the effectiveness of PRP and these variable counts in both humans and animals, may, in turn, ultimately lead to optimization of individual treatment. Although an optimum concentration of PRP is yet to be determined, certain studies have demonstrated that positive outcomes may be achieved when platelet concentrations fall within a certain range.36,39,103,104 In the study performed by Torricelli
In addition to variation of blood cell components contained within each product, an individual’s health, age and comorbidities may also reflect the effectiveness of PRP. These factors cannot be overlooked, especially in the bone-healing process. For example, circulating estradiol or testosterone levels may account for some of the varying results between age groups, and should be corrected in order to optimize the effects of PRP growth factors on bone.
Furthermore, application of PRP to different sites of injury requires different techniques in processing and delivery. In a study to determine whether shear force on platelets during the injection of PRP has a significant effect, 105 a comparison of injection of equine PRP with high pressure, using a small-bore needle (25 guage), and injection with a 21-gauge needle was conducted. The authors concluded that shear force did not have a significant effect on growth factor concentration. Products also are applied to the region of interest using multiple methods; PRP is injected as bolus, applied with a scaffold, or administered in microinjections. Superiority of a certain administration process over another or the optimal number of injections has not been determined and may depend on the microenvironments of the target lesion. 106
Following the application of PRP, the posttreatment protocol must be optimized as well. The mobility, stretching, and activity levels after treatment play an important role. 107 The protocol that is prescribed, and followed by the patient, varies between studies and patients respectively.
The variety of injuries seen in sports medicine encompasses multiple tissue types and different microenvironments. The environment of the lesion influences the outcome of treatment and tissue-specific requirements that promote optimal conditions for the healing process. Available oxygen, hydrostatic pressure, and the pH in the region of interest should be considered in determining treatment.
107
Kalen
Furthermore, acute and chronic injuries respond differently to the present metabolic state of the lesion. For instance, generally, tendinitis is characterized by an acute inflammatory response, whereas tendinosis does not necessarily refer to the inflammation of tendon, but rather, microtears in tendon pathology. Although they may be concomitant injuries, they are categorized under the umbrella of tendinopathies, and will differ in their healing processes and response to platelet components. Therefore, not only are the components that are administered important in PRP but also the milieu of bioactive factors already present within the site of injury. As mentioned before, the leukocyte concentration in PRP has recently attracted much attention, and while leukocytes may be beneficial in some injuries because of their antimicrobial components, WBCs, in particular neutrophils, have also demonstrated deleterious potential in many tissue types.8,109 In fact, it is recommended that for soft tissue engineering scaffolds, such as PRP, a ratio of 2,000:1 platelet to WBC is prepared. 109 This ratio may be critical in instances where PRP and other mixed regenerative cell concentrate products provide growth factors and act as a scaffold. 110 Not only is the variation in blood cell components important but also the variation in growth factor concentration. Currently, clinicians are conducting studies to determine the optimum ratio of the cellular components contained in PRP with respect to the site of injury and degree of tissue degeneration.
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research and/or authorship of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
Ethical Approval
This study was approved by our institutional review board.
