Abstract
Objective
Osteochondral surgical procedures have been described for the treatment of unfixable osteochondritis dissecans (OCD), but only few of them have been studied for juvenile OCD (JOCD) lesions. A cell-free biomimetic osteochondral scaffold showed positive results in adult patients. The aim of this study was to evaluate the results of this scaffold for the treatment of knee JOCD at mid-term follow-up.
Design
Twenty patients (14 males, 6 females) were included in this study. Mean age was 16.2 ± 1.4 years, average defect size was 3.2 ± 1.8 cm2, and mean symptoms duration was 20.2 ± 17.9 months. After the implantation of the osteochondral collagen–hydroxyapatite scaffold (Maioregen, Fin-Ceramica, Faenza, Italy), patients were evaluated preoperatively and prospectively at 1, 2, and at final mean follow-up of 6 years (range 5-7 years) with International Knee Documentation Committee (IKDC) subjective and objective, Tegner, and EuroQol visual analogue scale (VAS) scores. MRI evaluation was performed with the MOCART 2.0 score.
Results
All scores showed a significant improvement. IKDC subjective score went from 50.3 ± 17.4 preoperative score to 75.3 ± 14.6 at 1 year (P = 0.002), 80.8 ± 14.6 at 2 years and 85.0 ± 9.3 at 6 years. The Tegner score improved from the preoperative evaluation of 2.6 ± 1.4 to 5.5 ± 2.0 at 6 years (P < 0.0005), although without reaching the level registered before the onset of symptoms. A longer symptoms duration influenced negatively IKDC subjective and Tegner scores up to 2 years (P = 0.003 and P = 0.002, respectively) but did not affect the final outcome. Lesion size did not affect the final result. The MOCART 2.0 score showed a significant improvement between 1-year and final follow-up, but with persisting subchondral alterations.
Conclusions
This study demonstrated a clinical improvement stable over time with a high survival rate, although with persisting abnormal MRI findings, especially at subchondral bone level. This procedure can be considered a suitable option for the treatment of young patients affected by knee OCD. Level of evidence. Case series, level IV.
Introduction
Osteochondritis dissecans (OCD) is an acquired pathological condition of the osteochondral unit primarily affecting the subchondral bone, which can eventually result in chondral involvement, fragment detachment, severe loss of function, joint lockage, and premature osteoarthritis.1-4 OCD was first named by Konig in 1888 but, despite being known for centuries, its etiology is still poorly understood.1,5,6 Multiple factors like trauma, ischemia, abnormal ossification patterns, genetic disorders, and endocrine aspects may play an interconnected role and concur in the development of the disease. 7 This more often affects sport-active young male patients, especially those between 12 and 19 years old, with an incidence of 11.2 per 100,000 in this age group and a 3.3-fold increased risk of knee OCD compared with younger patients.8-10 Age is probably the most important prognostic factor, key for the treatment decision. 11 In fact, while adult OCD often requires surgical treatment, juvenile OCD (JOCD) lesions are characterized by an overall better prognosis also with conservative treatment.11,12 However, another crucial prognostic factor is the stability of the lesions. While stable fragments treated conservatively will heal in most of the cases, 11 an unstable lesion needs a surgical treatment to restore the articular surface, possibly through fragment fixation or by reconstructive procedures for unfixable lesions. In this case, the ideal approach should address both subchondral bone and overlying cartilage, to restore the entire osteochondral unit affected by OCD.
Several osteochondral surgical procedures have been described for the treatment of unfixable OCD,13-17 but only few of them have been studied for JOCD lesions. Moreover, the available osteochondral techniques are doomed by important limitations: intrinsic morbidity at the donor site for osteochondral autologous transplantation (OAT), 18 costs and availability for fresh osteochondral allograft (FOCA), 17 and size limitation for matrix-assisted autologous chondrocyte transplantation (MACT). 19 A cell-free biomimetic osteochondral scaffold has been developed to overcome these limitations. Its use has already shown positive results in the clinical practice for different types of lesions, including OCD, although mainly documented in adult patients.20-22 Analogously, this osteochondral approach could prove to be suitable also for the treatment of unfixable knee OCD lesions in young patients.
The aim of this study was to evaluate at mid-term follow-up the results of a cell-free biomimetic osteochondral scaffold for the treatment of knee JOCD.
Methods
Patient Selection
The treatment was approved by the Local Ethical Committee and the Internal Review Board of the Rizzoli Orthopaedic Institute, Bologna, Italy (prot. gen. n. 39667) and all patients gave their informed consent. Between October 2008 and January 2015 patients were consecutively enrolled according to the following criteria: ≤18 years old, affected by a symptomatic knee OCD lesion, graded III or IV on the ICRS (International Cartilage Repair Society) scale, 23 involving femoral condyles and trochlea, and not suitable for fragment refixation. According to previous literature,19,24 the age cutoff between adult and juvenile OCD was set from a regenerative perspective, based on the findings of Schmal et al., 25 who demonstrated that age can still influence the regenerative potential regardless of epiphyses closure. Moreover, trochlear lesions were considered separately from patellar lesions, according to previous literature suggesting to not consider them together when analyzing results of cartilage treatments because of the different characteristics and clinical results of these locations. 26
Exclusion criteria were >18 years old, patella involvement, untreated misalignment, or instability (patients who received realignment procedures concurrently with the implantation were included in the study). Patients with infectious, neoplastic, metabolic, and inflammatory diseases were also excluded.
Twenty patients (14 males and 6 females) were included in this study. The mean age at treatment was 16.2 ± 1.4 years, with a mean body mass index of 21.9 ± 2.1 kg/m2. The lesion was located at the medial femoral condyle (MFC) in 9 patients, at the lateral femoral condyle (LFC) in 8, and at the trochlea in 3. The average size of the defects was 3.2 ± 1.8 cm2. Mean symptoms duration was 20.2 ± 17.9 months. Five patients underwent previous surgeries: 2 loose body removals, 1 lateral meniscectomy, 1 microfracture, and 1 arthroscopic shaving. In 4 patients the procedure was combined with another treatment: 2 realignment osteotomies, 1 osteochondral autograft for a concomitant osteochondral lesion, and 1 lateral meniscal allograft implantation.
Surgical Technique and Postoperative Management
The surgical procedure was performed in a single step as previously described. 27 The defect was exposed using a medial or lateral parapatellar approach depending on the lesion site. The sclerotic subchondral bone was removed and a 6- to 7-mm deep area with a flat bottom and stable sides was created to house the implant. The osteochondral scaffold (Maioregen, Fin-Ceramica, Faenza, Italy) is a biomimetic implant composed of a porous 3D composite trilayered structure with type I collagen and hydroxyapatite in different proportions: 100% versus 0% at the cartilage surface, 60% versus 40% in the middle, and 30% versus 70% at the lower subchondral bone layer. 28 The graft was prepared by measuring the exact size of the lesion and was then implanted with a press-fit technique, applying fibrin glue when additional stability was required. 29 The stability of the implant was tested by cyclic bending of the knee while the graft was visualized, before and after tourniquet removal. The postoperative stage began with an early mobilization of the joint: active and passive knee range of motion exercises were allowed the day after surgery, as well as early isometric and isotonic exercises. No weightbearing was allowed for walking in the first 4 weeks after surgery; complete load was allowed after 6 to 8 weeks. 20 Unrestricted sports participation was allowed at 8 to 12 months of follow-up, tailored on patients’ evaluation.
Clinical and Radiological Evaluation
Patients were clinically evaluated preoperatively and prospectively at 1, 2, and at final mean follow-up of 6 years (range 5-7 years). No patient was lost at follow-up. Clinical parameters were rated using the IKDC (International Knee Documentation Committee) subjective and objective scores. The activity level was recorded using the Tegner score and the patient’s self-rated health was assessed with the EuroQol visual analogue scale (VAS). Treatment failures were defined according to both a surgical and a clinical criteria: the operation was considered failed if the patient needed further surgery due to symptoms of the primary defects; moreover, patients without a clinically significant improvement versus basal evaluation were considered clinical failures (10 IKDCsubj points). 30
At 1, 2, and at a mean of 6 years (range 5-7 years) after transplantation, repair of the articular surface and subchondral bone was evaluated in 18 knees with high-resolution magnetic resonance imaging (MRI). MRI evaluation was performed using a 1.5-T superconducting magnet (General Electric Co., Fairfield, CT) with a dedicated phased array (8 channels) HD knee coil. The following sequences were used for graft evaluation: sagittal fast spin echo proton density weighted with fat saturation (repetition time [TR] 3500 ms, echo time [TE] 22 ms, field of vies [FOV] 16 cm, slice thickness 3 mm, matrix 320 × 256); sagittal 3D Cube proton density weighted (TR 2500 ms, TE 30 ms, FOV 20 cm, slice thickness 0.8 mm, matrix 288 × 288); coronal fast spin echo proton density weighted with fat saturation (TR 2600 ms, TE 40 ms, FOV 16 cm, slice thickness 4 mm, matrix 320 × 224); axial fast spin echo proton density weighted with fat saturation (TR 2600 ms, TE 40 ms, FOV 16 cm, slice thickness 4 mm, matrix 320 × 224). The Magnetic Resonance Observation of Cartilage Repair Tissue 2.0 (MOCART 2.0) score was applied for the evaluation of the grafts. 31 All imaging evaluations were performed by an orthopedic surgeon and a musculoskeletal radiologist experienced in cartilage regeneration procedures. After an initial independent assessment, all images were reviewed in consensus.
Statistical Analysis
All continuous data were expressed in terms of mean ± SD, categorical variables were expressed as proportions or percentages. The Shapiro-Wilk test was performed to test normality of continuous variables. Repeated-measures generalized linear model with post hoc Sidak correction for multiple comparisons was performed to compare the scores at different follow-up times. The repeated-measures generalized linear model with the studied factors, as fixed effects were used to assess their influence on the scores evolution during the follow up times. The analysis of variance test was performed to assess the between group differences of continuous and normally distributed and homoscedastic data; the Kruskal-Wallis test was used otherwise. The Spearman rank test was used to assess correlations between scores and continuous data while the Kendall tau correlation test was used to assess the relationship between ordinal data. Pearson chi-square exact test was performed to investigate relationships between grouping variables. For all tests, P < 0.05 was considered significant. All statistical analysis was performed using SPSS v.19.0 (IBM Corp., Armonk, NY, USA).
Results
Clinical Evaluation
Four patients presented postoperative complications: 2 cases presented joint stiffness treated by knee mobilization under narcosis, respectively at 3 and 5 months, with complete recovery; 1 patient had persistent swelling solved with 2 arthrocentesis; finally, 1 patient had a trauma that required a medial partial meniscectomy and negatively affected only the 2-year follow-up. No surgical failures were documented; only 1 patient was accounted as failure according to the clinical definition. During the only second-look arthroscopy that was performed (for a meniscal lesion 2 years after scaffold implantation), an optimal filling of the defect with a good integration of the scaffold was documented ( Fig. 1 ).
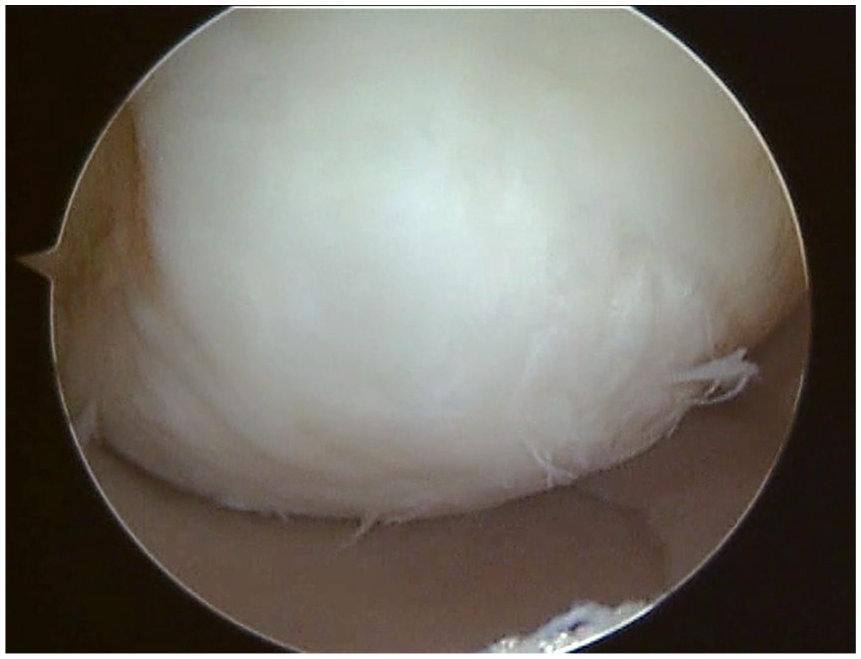
Second-look arthroscopy in a 17-year-old female performed 2 years after scaffold implantation, showing good coverage of the defect with smooth cartilage and good integration.
All the evaluated scores showed a significant improvement from the basal evaluation up to the final follow-up. The IKDC subjective score improved significantly from the preoperative score of 50.3 ± 17.4 to 75.3 ± 14.6 at 1 year (P = 0.002), with a further nonsignificant increase at 2 years and at the final follow-up (80.8 ± 14.6 and 85.0 ± 9.3, respectively, Fig. 2 ).
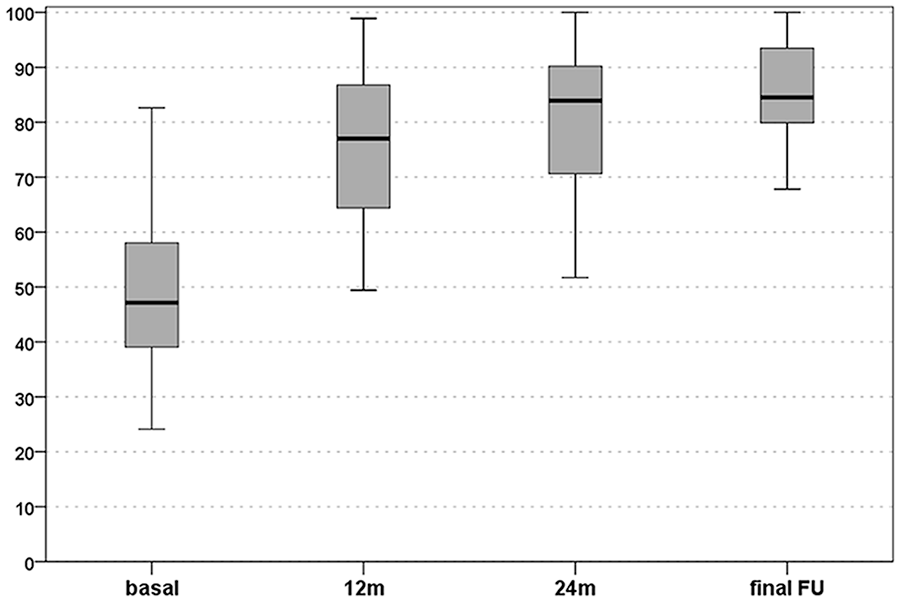
International Knee Documentation Committee (IKDC) subjective score: improvement from preoperative level to 12 months, 24 months, and final evaluation at a mean follow-up of 6 years. The box represents the 25th to 75th percentiles, with the thick line representing the median value and the bars representing the minimum and maximum.
A similar trend was demonstrated with the EuroQol-VAS score, which improved significantly from the basal value of 65.5 ± 14.3 to 84.8 ± 10.7 at 1 year (P < 0.0005), subsequently remaining stable up to the final follow-up (85.5 ± 8.6 at 2 years; 89.3 ± 9.9 at the final follow-up, Fig. 3 ).
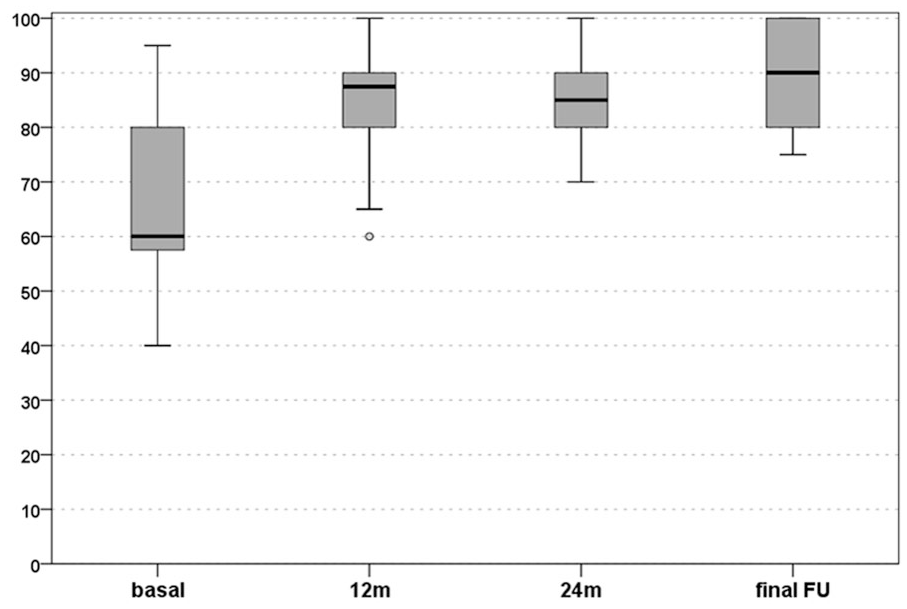
EuroQol visual analogue scale (VAS) score: improvement from preoperative level to 12 months, 24 months, and final evaluation at a mean follow-up of 6 years. The box represents the 25th to 75 percentiles, with the thick line representing the median value and the bars representing the minimum and maximum.
The analysis of the IKDC objective score showed a not significant improvement (P = 0.07). At basal evaluation, 15 knees (75%) were considered “normal” or “nearly normal” (9A, 6B, 4C, 1D) compared to 19 knees (95%, 14A, 5B, 1C) at 2 years, and this result was confirmed at the final follow-up, with 20 “normal” or “nearly normal” knees (17A, 3B).
The Tegner score did not improve significantly from the preoperative evaluation of 2.6 ± 1.4 to 4.0 ± 1.4 at 1 year (P = 0.053), but showed a significant improvement at 2 years (5.2 ± 1.6, P = 0.001) and 6 years (5.5 ± 2.0, P < 0.0005) postsurgery. Moreover, an improvement was documented also from the 1 year to the final follow-up (P = 0.002). However, although the final evaluation was not significantly lower compared with the preinjury level, the activity level before the onset of symptoms (7.1 ± 1.7) was not reached at any follow-up ( Fig. 4 ).
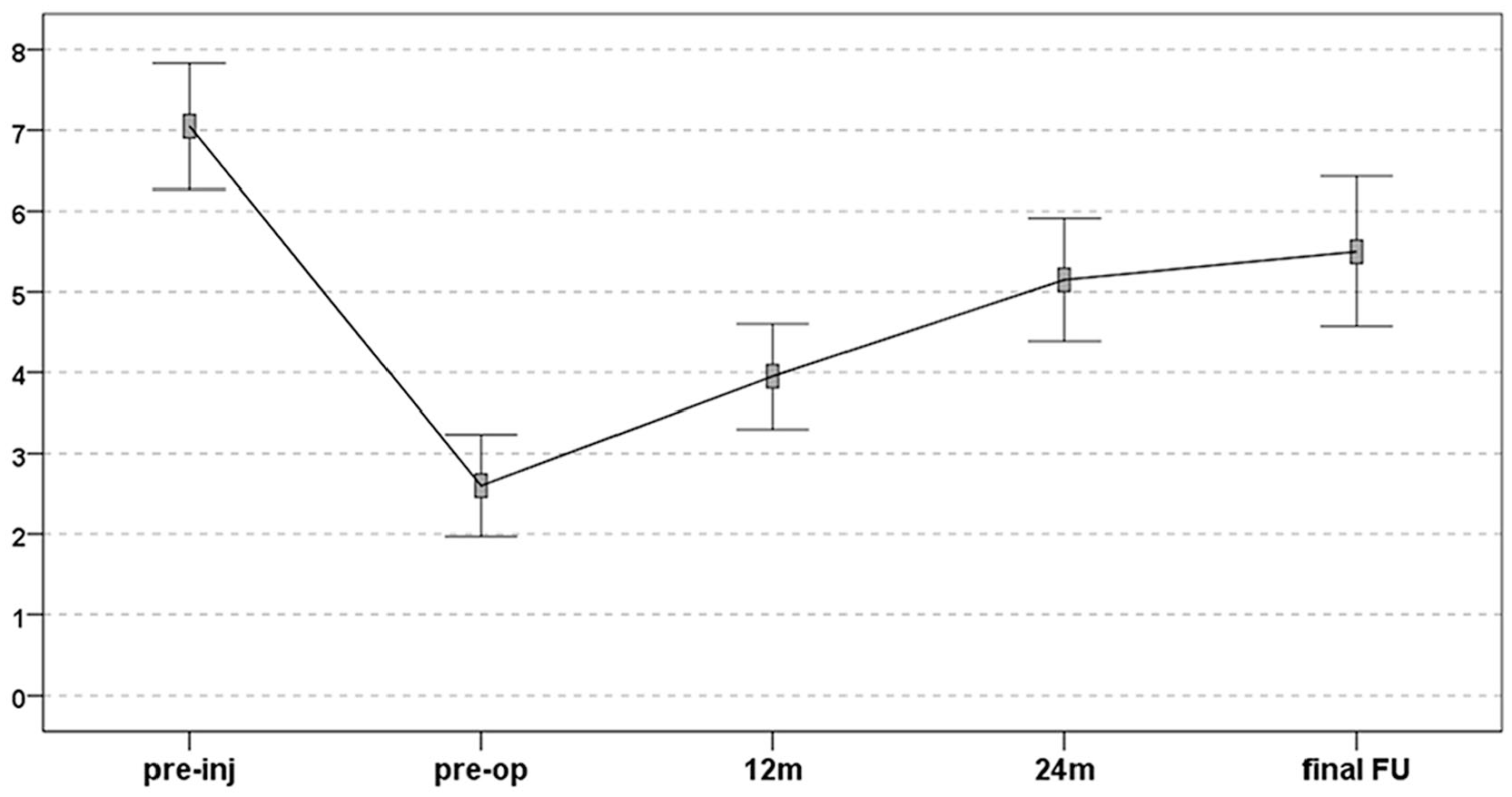
Tegner score: improvement from pretreatment level to 12 months, 24 months, and final follow-up levels. The final follow-up did not reach the preinjury level, despite being not significantly lower compared to it. The points represent the mean values and the bars represent the 95% confidence interval of the mean.
Further analysis was performed to evaluate the parameters which might influence clinical results. No significant correlation was found between clinical outcomes and patients’ sex, age, combined and previous surgery. A longer symptoms duration influenced negatively the IKDC subjective scores and the Tegner scores at 1 year (ρ = −0.415, P = 0.069 and ρ = −0.496, P = 0.026, respectively), and at 2 years (ρ = −0.630, P = 0.003 and ρ = −0.647, P = 0.002, respectively), but did not affect the outcome at the final follow-up. The lesion size did not affect the final result: Even patients with larger lesions and lower basal scores were able to reach the same final score compared with those with smaller lesions, with larger IKDC improvement for larger lesions (ρ = 0.468, P = 0.037).
Imaging Evaluation
The MRI analysis included the images of 18 patients: 17 patients were evaluated at 1 year, 14 at 2 years, and 13 at the final follow-up. The MRI evaluation at the final follow-up showed a complete filling of the defects in most of patients with a high rate of integration. The articular surface of the repair tissue was found to be intact in two-thirds of the MRIs, and the signal of the cartilage layer was normal in more than half of the examinations. On the other hand, the presence of subchondral alterations, such as bony defects or bony overgrowth and edema-like marrow signal, was still detectable in a large part of patients ( Fig. 5 ). Details of the MOCART 2.0 evaluation are reported in Table 1 .
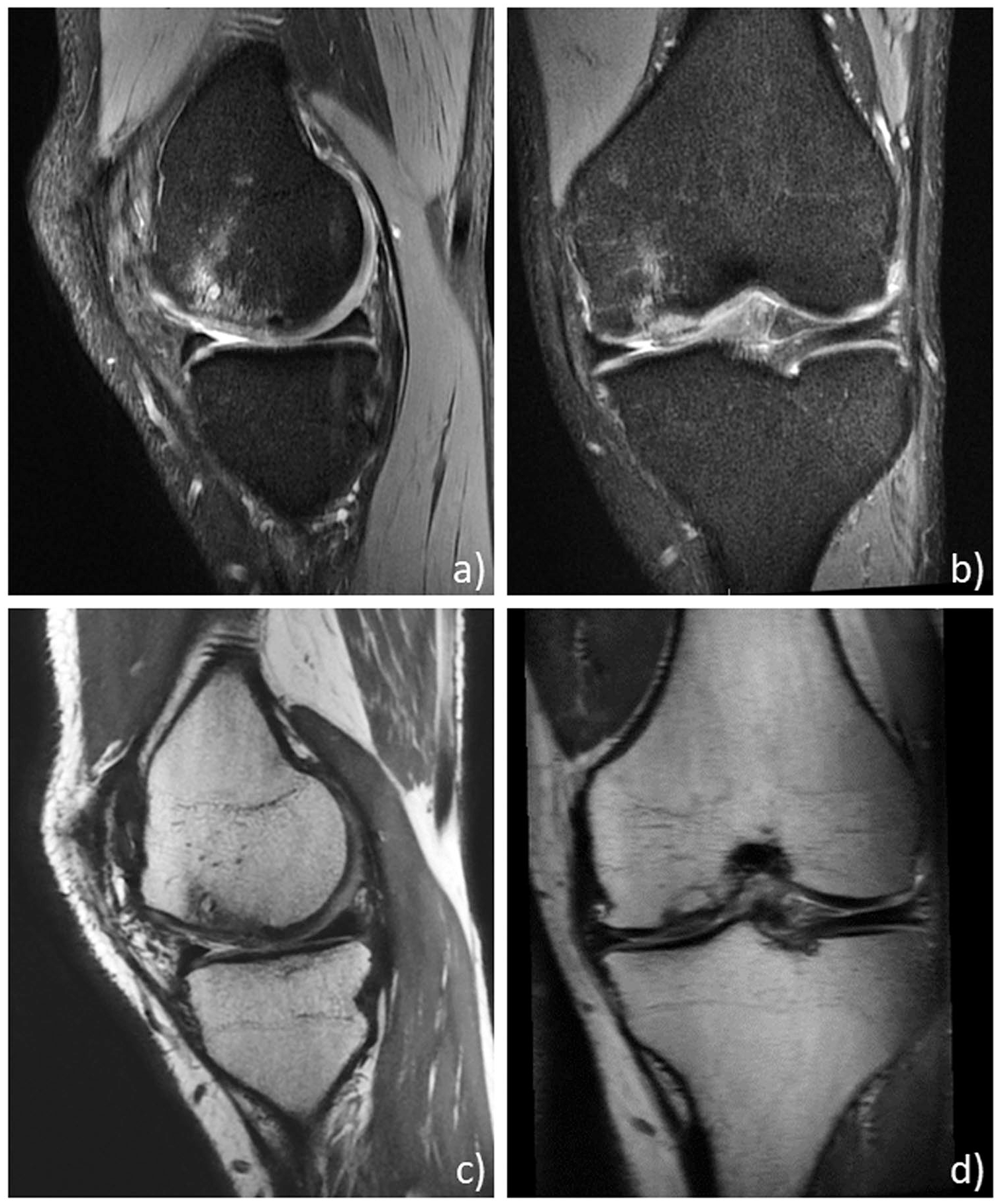
Magnetic resonance imaging (MRI) evaluation at 6 years of follow-up of a medial femoral condyle (MFC) knee lesion treated with an osteochondral cell-free scaffold in a 23-year-old man (
Evaluation of MOCART 2.0 Score at 12 Months, 24 Months, and Final Evaluation at a Mean Follow-up of 6 Years.
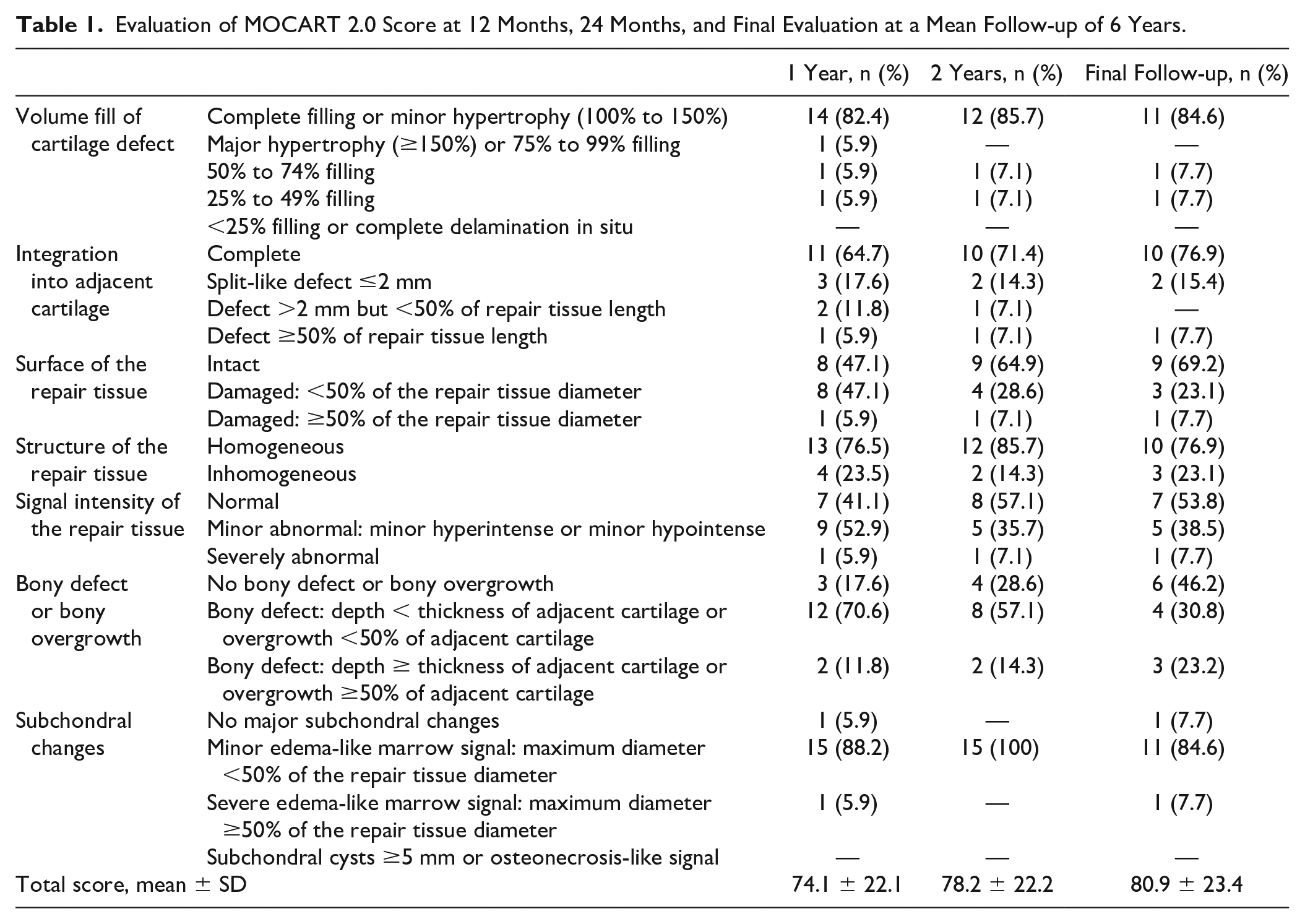
Eleven patients underwent all MRI follow-ups: the total MOCART 2.0 score showed a significant improvement between 1-year and final follow-ups (75.5 ± 21.6, 77.3 ± 22.1, and 80.0 ± 23.0, respectively, P = 0.025).
A larger lesion area presented a negative correlation with the MOCART 2.0 score at 1 year (P = 0.002), but not at the subsequent follow-ups. Three cartilage parameters (volume fill, integration, and surface of repair tissue) at the last MRI significantly correlated with the objective clinical outcome at the final follow-up (P = 0.001, P = 0.005, P = 0.015, respectively).
Discussion
The main finding of this study is that the implantation of this cell-free osteochondral scaffold is safe and provides improvements in terms of symptoms and function in young patients affected by knee OCD, with a high survival rate and satisfactory clinical and radiological results at short term, persisting up to a mid-term follow-up.
Several treatments have been historically proposed for JOCD.12,32,33 The main determinant factor for treatment choice is the stability of the lesions. For stable lesions, nonoperative management is recommended as first-line treatment 11 and arthroscopic drilling is indicated in stable lesions that fail nonoperative treatment. 34 If there are doubts about stability, internal fixation is used to stabilize the fragment into the original position. 35 In case of unfixable fragments or when the articular cartilage or the subchondral bone are excessively deteriorated, primary fixation may not be a viable option. 36 In these cases, the excision of loose fragments has been recently abandoned due the poor results documented: Sanders et al. 37 analyzed the results of 134 patients who underwent fragment excision, reporting a 70% cumulative incidence of osteoarthritis and 32% of arthroplasties after 30 years. Accordingly, restoring the articular surface is now considered important to provide satisfactory and long-lasting results.
Several techniques have been described, like OAT and FOCA, or chondrocyte-based procedures associated with bone grafting. Although these treatments have been largely investigated in the adult population with good results,38-42 the literature is extremely scarce on their outcome in JOCD. Among the above mentioned treatments, OAT showed good results for the treatment of OCD in the juvenile population. 43 In particular, Gudas et al. 24 compared the outcomes of OAT and microfracture for the treatment of JOCD in 50 children with a mean age of 14.3 years, and demonstrated superiority of the mosaic-type OAT at a 4-year mean follow-up. In larger defects, where OAT is not indicated 44 due to the risk of donor-site morbidity, 45 a valid alternative option is FOCA, but very little is known about its use in JOCD. Only a retrospective series of 11 children, with average lesions size of 5 cm2, reported full graft incorporation and satisfactory clinical results at short term. 46
Regenerative procedures have been developed to overcome the limits of more traditional approaches. Among these, the oldest strategy is the first-generation autologous chondrocyte implantation (ACI),41,47 and in particular its combination with an autologous bone graft in the so-called “sandwich” technique. 48 Few studies are available including JOCD patients, generally reporting mixed surveys. Mithofer et al. 49 treated 14 cases of OCD within a survey on 23 knees in adolescent athletes. They reported an overall high rate of functional success at a mean follow-up of 47 months, but with 15% of graft hypertrophy requiring reintervention. Similar results were reported by Micheli et al. 50 in a cohort of 37 patients (mean age 16 years), only 14 of which were OCDs, at a minimum of 2 years after implantation. Recently, Beck et al. 51 performed a long-term evaluation on 10 patients aged between 15 and 22 years affected by OCD and treated with ACI, and reported that, despite initial satisfactory outcomes, 40% of patients required further surgery and 20% were considered failures. To address ACI-related limitations, MACT techniques have been developed, relying on the use of 3-dimensional scaffolds to provide a support for cell proliferation and favor the formation of cartilage-like tissue.52,53 In a study focused on JOCD, Roffi et al. 19 observed a significant improvement in all clinical scores in 16 of 19 patients evaluated at 10 years of follow-up after MACT combined with autologous bone grafting. However, lower results were documented for larger lesions, underlying the limitations of the aforementioned cartilage regeneration procedures adapted to the osteochondral challenge of OCD lesions.
In this light, a new osteochondral approach was developed: Biphasic cell-free scaffolds have been introduced in the clinical practice for the treatment of osteochondral lesions with a single step procedure, avoiding the risks and the costs related to cell manipulation. In particular, several clinical studies demonstrated the safety and efficacy of this collagen-hydroxyapatite osteochondral scaffold, with good results maintained over time.20,54,55 Promising results were shown even in more complex cases like tibial plateau fractures, 56 osteonecrosis, 57 and early osteoarthritis.58,59 This scaffold was also successfully applied for the treatment of knee OCD in the adult population. At short-term follow-up, Verdonk et al. 22 described a significant improvement for up to 2 years in a mixed survey of 38 patients, among which 19 were affected by OCD, with 2 surgical failures (5.3%). Similar positive short-term results were reported by Delcogliano et al. 21 in a series of 23 patients treated with the same scaffold for knee OCD, and by Filardo et al. 60 who observed a significant improvement at 24 months in 27 patients, regardless of the OCD defect size. In a later evaluation of the same survey, Perdisa et al. 15 reported good results at 5 years, with an almost complete recovery of the preinjury sport level without failures.
This study is the first to specifically evaluate the efficacy of this cell-free osteochondral scaffold for the treatment of JOCD. The analysis showed that this 1-step treatment offered good and stable results for up to 6 years of follow-up in both objective and subjective clinical scores, in line with previous studies on two-step cell-based regenerative approaches for OCD lesions.19,42,47 As reported for adult OCD, the Tegner score continued to improve between 1 year and the final follow-up, reaching a final score close (albeit not equal) to preinjury level. 15 Good clinical results were obtained in most patients; the evaluation of parameters influencing the outcome showed that sex, age, combined and previous surgery did not correlate with the final clinical result. On the other hand, symptoms duration was found to negatively influence the activity level for up to 2 years, but this correlation was not confirmed at the final follow-up. Interestingly, the final outcome was not affected by the lesion size. As a matter of fact, patients with larger lesions were found to obtain the largest subjective improvement, but this is probably due to the less severe initial symptoms of patients with smaller lesions. The ability to provide good results even in larger lesions represents an advantage of this procedure compared with other techniques, like MACT and OAT,19,42,44 where size-related limitations have been documented. This also confirms the potential of osteochondral scaffolds in particular for this pathology, which involves both bone and cartilage, and often affects large areas of the articular surface.
No surgical failures were reported in this series, and only one patient was considered a clinical failure. Although the failure rate should be confirmed at longer follow-up, this finding appears positive when compared to studies on ACI and MACT reporting the occurrence of surgical failures at short term.19,61 Roffi et al. 19 interpreted the occurrence of early failures in bone-augmented MACT as caused by a more delicate phase of the regenerated tissue in the first months after implantation. In line with other reports on osteochondral scaffolds, 15 the lack of early failures in the current study suggests that this procedure may provide a more stable regeneration substrate, already in the early phases after surgery.
The MRI evaluation did not demonstrate early outcomes as satisfactory as the clinical ones, as already shown for this surgical procedure in other types of lesions.15,62 A very limited bone formation at 2 years was described by Christensen et al. 62 in 3 patients with osteochondral defect of the femoral condyles. Perdisa et al. 15 also reported the presence of subchondral MRI alterations in the majority of patients, with an ongoing maturation up to 5 years. In the current study, 18 patients underwent at least 1 MRI after surgery, and 11 patients underwent MRI examination at all follow-ups. The total MOCART 2.0 score increased significantly over time, as already reported for OCD lesions in adult patients. 15 However, although the presence of bone alterations improved over time, the majority of patients still presented subchondral changes. Further analysis was therefore performed aiming at investigating if this suboptimal regeneration correlated with an inferior clinical outcome. Lower MRI cartilage parameters significantly correlated with the objective clinical outcome, and bigger lesions were negatively correlated with the MOCART score at 1 year. All these findings underline the challenges in regenerating the articular surface and the complexity of restoring the entire osteochondral unit. In this light, bigger lesions may require longer to integrate and regenerate than what expected for a chondral scaffold or for smaller lesions. 20 The improvement of MRI parameters up to the last follow-up suggests that, even if slowly, the maturation of the scaffold continues to improve over time. Nevertheless, an improvement of the scaffold properties, through the functionalization of the biomaterial or the augmentation with other factors such as cells or bioactive/anti-inflammatory molecules, is warranted to further improve its regenerative potential.
The main limitation of the present study is inherent in its design, a prospective case series that lacks a control group, which did not allow to compare these results with other treatments available for these young patients. Another weak point is represented by the size of the survey, as the number of patients was sufficient to detect an improvement over time, but the sample could be underpowered to detect prognostic factors driving the outcome. Finally, regarding the MRI evaluation, the majority of patients underwent at least one MRI follow-up, but only half of them was available for all 3 follow-ups, which limits a more in depth understanding of the tissue maturation over time.
Despite these limitations, this is the first report about the use of this osteochondral scaffold to address JOCD, and the results at mid-term follow-up can represent the reference for comparison in future studies. Larger surveys at longer follow-up will better explore results and indications of this technique. Still, the clinical and radiological evaluation of this series offered important insights about the potential and limitations of this osteochondral scaffold for the treatment of JOCD.
Conclusions
This study demonstrated a clinical improvement stable over time with a high survival rate, which is paramount especially in these young patients with high functional demands and a long expectancy of active life, although with persisting abnormal MRI findings, especially at subchondral bone level. While further research is needed to address the limitations of this cell-free osteochondral scaffold and improve its subchondral regenerative potential, this procedure can be considered a suitable option for the treatment of young patients affected by knee OCD.
Footnotes
Acknowledgments and Funding
We thank Elettra Pignotti for the statistical analysis, and Lucia Mancini for the English revision service. The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The treatment was approved by the Local Ethical Committee and the Internal Review Board of the Rizzoli Orthopaedic Institute, Bologna, Italy (prot. gen. n. 39667).
Informed Consent
All patients gave their informed consent.
Trial Registration
Not applicable.
