Abstract
Objective
We aimed to demonstrate that electroarthrography (EAG) measures streaming potentials originating in the cartilage extracellular matrix during load bearing through electrodes adhered to skin surrounding an articular joint.
Design
Equine metacarpophalangeal joints were subjected to simulated physiological loads while (1) replacing synovial fluid with immersion buffers of different electrolyte concentrations and (2) directly degrading cartilage with trypsin.
Results
An inverse relationship between ionic strength and EAG coefficient was detected. Compared to native synovial fluid, EAG coefficients increased (P < 0.05) for 5 of 6 electrodes immersed in 0.1X phosphate-buffered saline (PBS) (0.014 M NaCl), decreased (P < 0.05) for 4 of 6 electrodes in 1X PBS (0.14 M NaCl), and decreased (P < 0.05) for all 6 electrodes in 10X PBS (1.4 M NaCl). This relationship corresponds to similar studies where streaming potentials were directly measured on cartilage. EAG coefficients, obtained after trypsin degradation, were reduced (P < 0.05) in 6 of 8, and 7 of 8 electrodes, during simulated standing and walking, respectively. Trypsin degradation was confirmed by direct cartilage assessments. Streaming potentials, measured by directly contacting cartilage, indicated lower cartilage stiffness (P < 10−5). Unconfined compression data revealed reduced Em, representing proteoglycan matrix stiffness (P = 0.005), no change in Ef, representing collagen network stiffness (P = 0.15), and no change in permeability (P = 0.24). Trypsin depleted proteoglycan as observed by both dimethylmethylene blue assay (P = 0.0005) and safranin-O stained histological sections.
Conclusion
These data show that non-invasive EAG detects streaming potentials produced by cartilage during joint compression and has potential to become a diagnostic tool capable of detecting early cartilage degeneration.
Keywords
Introduction
Osteoarthritis (OA) is characterized by progressive deterioration of articular cartilage eventually resulting in loss of joint function and pain.1,2 No treatments presently exist to modify or reverse the disease course at later, symptomatic stages, and late-stage OA can only be remedied by joint replacement. Identifying earlier, asymptomatic, stages of the disease, where cartilage deterioration has recently commenced, may present opportunities for treatment aimed at slowing disease progression.1,3 To accomplish this, sensitive diagnostics of cartilage quality are required, as current methods, such as radiography, are not capable of detecting subtle changes in cartilage that may occur during early OA. 4 Imaging methods, including ultrasound and magnetic resonance imaging are promising candidates; however, they are limited by high costs and technical expertise requirements.4,5 Electroarthrography (EAG) is a non-invasive method for measuring cartilage streaming potentials, which result when the hydrated cartilage extracellular matrix (ECM) responds to compressive loading. The cartilage ECM consists of large hydrophilic proteoglycan molecules which are immobilized by a highly organized, predominantly type II collagen network. When cartilage is in equilibrium, the fixed, negative charge density of the glycosaminoglycan chains is electrically balanced by mobile positive ions distributed within interstitial fluid. During cartilage compression, a pressure gradient develops that displaces mobile positive ions relative to proteoglycan, which remain immobilized by the fibrillar collagen network, thereby producing an electrical signal known as the streaming potential.6-8
Cartilage streaming potentials have been measured by directly contacting cartilage with electrodes using a variety of benchtop apparatus,6-10 as well as an indentation probe.11-14 These studies have consistently demonstrated that streaming potentials detect changes in cartilage quality, including, for example, biomechanical changes resulting from enzymatic degradation8,10 and acute impact injury. 13 Cartilage streaming potentials correlated to histological assessments, including Mankin score,15-17 collagen architecture, 15 as well as the International Cartilage Repair Society macroscopic score.12,16 Correlations to biochemical composition,8,10,15 chondrocyte viability, and apoptosis 17 have also been demonstrated by several groups. Streaming potentials, measured non-destructively by lightly compressing articular surfaces using an indentation probe, were capable of discerning subtle cartilage degeneration in human knee joints that was not visible to the eye. 11 Indeed, these authors demonstrated greater effect sizes for streaming potentials, indicating greater sensitivity to cartilage degenerative changes, compared to indentation tests, unconfined compression, biochemical assay, or histological appearance. 11 EAG is a non-invasive method for measuring cartilage streaming potentials that involves placing contact electrodes on skin external to an articulation while the joint undergoes compressive loading.18-22 Préville et al. 18 introduced a clinical EAG protocol whereby adhesive electrodes were attached to skin surrounding one knee and the subject applied compressive load to the instrumented leg by shifting their body weight to achieve a one-legged stance. EAG measurements were thus acquired from 20 control subjects, as well as both knees of twenty patients diagnosed with radiographic bilateral knee OA and who had undergone unilateral total knee arthroplasty (TKA). EAG distinguished among these 3 groups of knees and detected statistically significantly higher streaming potentials, indicating better quality cartilage, in control knees compared with osteoarthritic knees, while EAG measurements from TKA knees were statistically null.
The present study aims to demonstrate that EAG originates in articular cartilage by controlling the conditions under which streaming potentials are produced. We used cadaveric equine fetlock (metacarpophalangeal) joints to study EAG response to altering the salt concentration of the solution bathing cartilage surfaces, as well as to enzymatic degradation of cartilage. The equine fetlock joint, consisting of the distal metacarpus, the proximal first phalanx and sesamoid bones, is suitable because it is not surrounded by musculature, which could produce high-frequency electrical signals in live animals, nor fat, which has been shown to diminish cartilage streaming potentials in a finite element simulation. 19 Theoretically, higher ionic strength reduces streaming potentials because the presence of additional mobile ions increases electrical shielding. Streaming potential amplitude, measured directly from cartilage during compression, has indeed been shown to decrease as ionic strength increases6,7,23 and we hypothesize in our first set of experiments that EAG will demonstrate a similar inverse relationship. A second set of experiments examined EAG response to enzymatic cartilage degradation by intra-articular injection of trypsin, a proteinase that digests proteoglycan and has been used to create in vitro models of early OA.9,24-26 Cartilage exposed to trypsin develops a gradient of proteoglycan depletion over time that progresses from the articular surface toward the bone-cartilage interface.27,28 We hypothesize that non-invasive EAG would diminish in response to trypsin degradation similar to direct measurements of cartilage streaming potentials, biomechanical and biochemical properties, and histological appearance.
Methods
Procedures were performed with Institutional Animal Care Committee approval in accordance with the Canadian Council on Animal Care (University of Guelph, AUP#1412). Three distal forelimbs, comprising the third metacarpus to the hoof, were obtained from skeletally mature horses ranging from 2 to 7 years of age and weighing 561 ± 49 kg. Forelimbs were frozen until testing. EAG measurements were acquired on each fetlock while it underwent a set of loading sequences in the presence of native synovial fluid followed by three immersion buffers with different electrolyte concentrations.
EAG Measurements
Each forelimb was mounted in a servohydraulic mechanical tester (Instron 8800, Norwood, MA, USA) with joint axes aligned to those of a 6-degree-of-freedom load cell (SI-1500-240, ATI Industrial Automation, Apex, NC, USA). EAG was performed on each fetlock during simulated physiological loading using gold-plated disk electrodes attached to 6 sites around the articulation. Electrodes were placed at the anterior (dorsal) phalanx/metacarpus interface, on the medial (EAG1) and lateral (EAG2) sides of the common digital extensor tendon, as well as at the medial (EAG3) and lateral (EAG4) phalanx/metacarpus interface and the medial (EAG5) and lateral (EAG6) metacarpus/sesamoid interface (

Placement of electroarthrography (EAG) electrodes on a right fetlock joint. Views are (
Adjustment of Electrolyte Concentrations with Different Immersion Buffers
EAG measurements were repeated four times on each joint. First, with cartilage surfaces bathed in native synovial fluid, followed by 1 of 3 immersion buffers, namely 0.1X phosphate-buffered saline (PBS) (0.014 M NaCl), 1X PBS (0.14 M NaCl), or 10X PBS (1.4 M NaCl). PBS solutions were made from a 10X stock solution consisting of 1369.4 mM NaCl, 97.4 mM Na2HPO4, 21.8 mM KH2PO4, and 26.5 mM KCl (BP665, Thermo Fisher Scientific, Waltham, MA, USA). Immersion buffers were adjusted to pH 7.4.
Native synovial fluid was removed by fully flexing the fetlock and inserting an 18-gauge needle into the joint space between the sesamoid and metacarpus bones first on the medial, and then lateral, aspects. To maximize synovial fluid removal, the needle was oriented downward to allow gravity to assist and the joint was repeatedly flexed and extended. Quantities of synovial fluid retrieved were 2.5 mL, 4 mL, and 5.5 mL for each of the 3 fetlocks, respectively.
To transition from synovial fluid to 0.1X PBS, the fetlock was subjected to a minimum of 2 lavages with 0.1X PBS followed by addition of a fresh quantity of 0.1X PBS prior to repeating EAG measurements. Lavage involved accessing the joint space as described above and delivering 5 mL 0.1X PBS medially and 5 mL 0.1X PBS laterally. Distribution throughout the joint was encouraged by manually flexing and extending 30 times followed by sinusoidal loading at 0.1 Hz with amplitude of 3 mm for 20 cycles. The lavage solution was then retrieved and conductivity measured (described below). Lavages were repeated until conductivity stabilized. Then 10 mL of fresh 0.1X PBS was delivered to the joint and it was incubated at 4°C for 1 hour. Lavage solutions were retrieved in quantities ranging from 2.5 mL to 10.5 mL (median 8.5 mL), indicating some solution remained in the joint and surrounding tissues with each lavage. EAG measurements were completed with joint surfaces bathed in 0.1X PBS, then the lavage procedure repeated to transition from 0.1X PBS to 1X PBS and finally from 1X PBS to 10X PBS.
Conductivity Measurements
Conductivity was measured using an Accumet AP85 (Thermo Fisher Scientific, Waltham, MA, USA) calibrated with a 12.88 mS/cm standard. 10X PBS solutions were diluted 10:1 with deionized water prior to conductivity measurement. The conductivity probe was cleaned with a mild soap solution and rinsed with deionized water between measurements. The certificate of analysis for the lot of PBS used reported conductivity of a 1X solution as 16.6 mS/cm at 25°C.
Trypsin Degradation of Cartilage
Fresh distal forelimbs from 1 horse (18 years old, 460 kg) were collected to investigate EAG sensitivity to trypsin degradation of cartilage. EAG measurements (as above) were performed on the right fetlock. Cartilage degradation was subsequently induced by intra-articular injection (as above) of 0.5% w/v trypsin (T9201, Sigma Aldrich, St. Louis, MO, USA) in Tris buffer (BP154-1 Fisher, Waltham, MA, USA) and incubation at 37°C for 3 hours. 27 EAG measurements were repeated post-trypsin. Confirmation of trypsin degradation was sought by directly measuring cartilage streaming potentials, biomechanical properties, histological appearance, and biochemical composition. The untreated contralateral fetlock served as a control for these destructive analyses. Direct measurements of cartilage streaming potentials were made at 75 sites on the articular surfaces of the metacarpus and phalanx with the Arthro-BST (Biomomentum Inc.); an arthroscopic device that records streaming potentials during manual cartilage indentation through an array of 37 microelectrodes (5 microelectrodes/mm2). The device calculates a quantitative parameter (QP, arbitrary units) corresponding to the number of microelectrodes in contact with cartilage when the sum of potentials reaches 100 mV. Cartilage disks, 3 mm in diameter, were subsequently isolated from load bearing regions29,30 for testing in unconfined compression geometry. Each disk was subjected to five stress relaxation ramps of 2% strain. The fibril-reinforced poroelastic model 31 was used to obtain fibril modulus (Ef), matrix modulus (Em), and hydraulic permeability (k). Cartilage disks were then fixed in 10% neutral buffered formalin and embedded in paraffin. Five-micrometer sections were stained with safranin-O/fast green/iron hematoxylin. Biochemical composition was quantified in adjacent cartilage disks submitted to the dimethylmethylene blue (DMMB) and hydroxyproline (HPr) assays. 32
Statistical Analysis
A full factorial analysis of variance with replicates was used to explain variation in EAG coefficients due to immersion buffer (synovial fluid, 0.1X PBS, 1X PBS, 10X PBS) and displacement (8 mm, 15 mm, 25 mm). For each electrode, immersion buffer and displacement were treated as fixed effects with repeated measurement across 5 loading cycles. The experiment was independently replicated 3 times with fetlock treated as a random effect. Tukey’s post hoc honestly significant difference was applied to identify significant differences in average EAG coefficients, accounting for multiple comparisons. Paired t tests were used to compare EAG coefficients, QP, biomechanical parameters, and biochemical parameters. Statistical analyses were performed in Statistica v.8 and reported as least square mean (lower 95% confidence limit, upper 95% confidence limit). 95% confidence intervals = ±1.96 × standard error of the mean. Descriptive data are reported as mean ± standard deviation.
Results
Conductivity measurements were made on synovial fluid and immersion buffers retrieved after EAG measurements ( Table 1 ) and within a temperature range of 20.7°C ± 0.7°C (n = 12). Results from a t test indicated that compared to synovial fluid, 0.1X PBS had significantly lower (P = 0.021) and 10X PBS significantly higher (P < 1 × 10−4) conductivities. There was no statistical difference detected between conductivity measured in synovial fluid and 1X PBS.
Conductivity of Synovial Fluid and Immersion Buffers Retrieved After Electroarthrography Measurements Were Performed [Least Square Mean (Lower 95% Confidence Interval, Upper 95% Confidence Interval), n = 3].
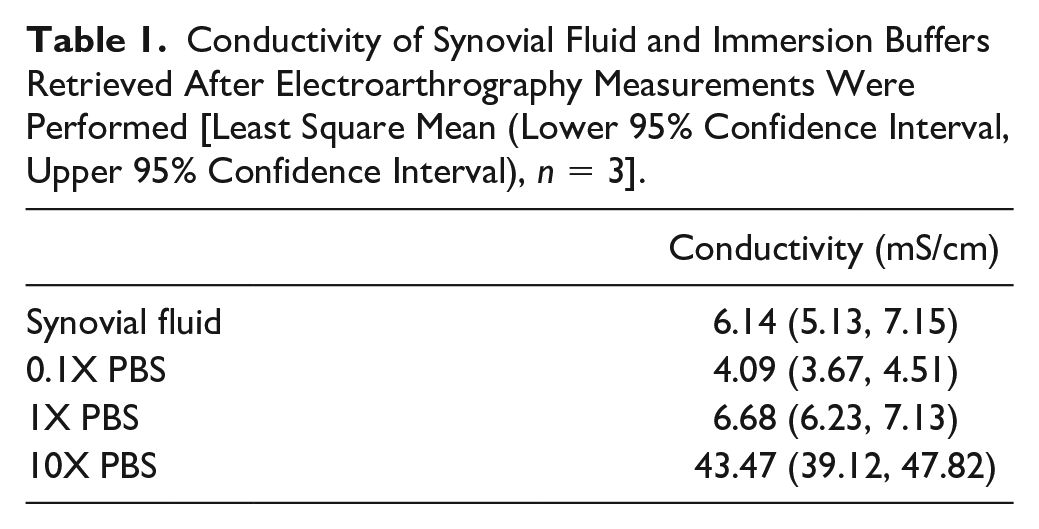
Taken together, across all displacements and fetlocks, average EAG coefficients increased (P < 0.05) at 5 of 6 electrodes in the presence of 0.1X PBS and decreased (P < 0.05) at all electrode sites in the presence of 10X PBS compared with synovial fluid (
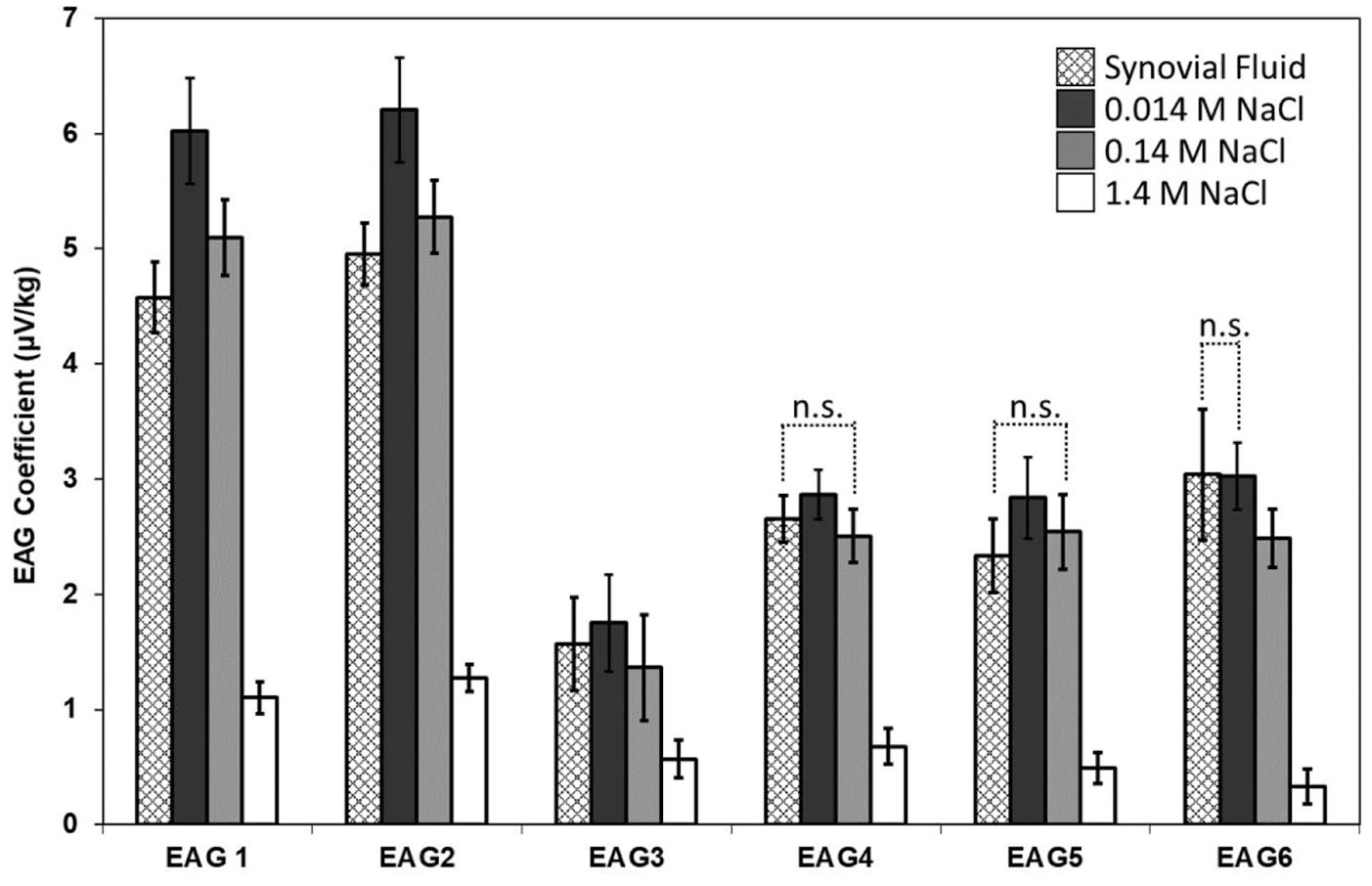
Effect of electrolyte concentration on electroarthrography (EAG) coefficients. EAG measurements were made at 6 sites while cartilage surfaces were immersed in native synovial fluid, 0.1X phosphate-buffered saline (PBS) (0.014 M NaCl), 1X PBS (0.14 M NaCl), or 10X PBS (1.4 M NaCl). Significant differences (P < 0.05) accounting for multiple comparisons were detected between immersion buffers unless otherwise identified as not significant (n.s.). Anterior electrodes, EAG1 and EAG2, as well as EAG3 at the mediolateral interface between the metacarpus and phalanx, demonstrated significantly different EAG coefficients for each solution. EAG coefficients measured at EAG4 and EAG5 were significantly different among synovial fluid, 0.1X PBS (0.014 M NaCl) and 10XPBS (1.4 M NaCl), but not between synovial fluid and 1X PBS (0.14 M NaCl). EAG coefficients measured at EAG6 were significantly different among synovial fluid, 1X PBS (0.14 M NaCl) and 10X PBS (1.4 M NaCl) but not between synovial fluid and 0.1X PBS (0.014 M NaCl). Bars are least square means ± 95% confidence intervals (n = 3 displacements × 3 fetlocks × 5 repetitions = 45).
During EAG measurements, fetlocks were subjected to displacements of 8 mm, 15 mm, and 25 mm, to approximate, respectively, half the load experienced during standing, standing, and walking. Average axial loads (n = 12) were 946 ± 161 N, 1935 ± 273 N, and 3240 ± 530 N, for 8 mm, 15 mm and 25 mm of displacement, respectively, which are within range of reported peak vertical forces.29,33 Change in flexion angle (n = 12), measured manually with a goniometer, were 6.1° ± 0.4°, 11.4° ± 1.1°, and 18.1° ± 1.9° for 8 mm, 15 mm, and 25 mm of displacement, respectively. Significant differences among EAG coefficients due to displacement were detected for EAG2 and EAG3 but not for EAG4 or EAG5 (
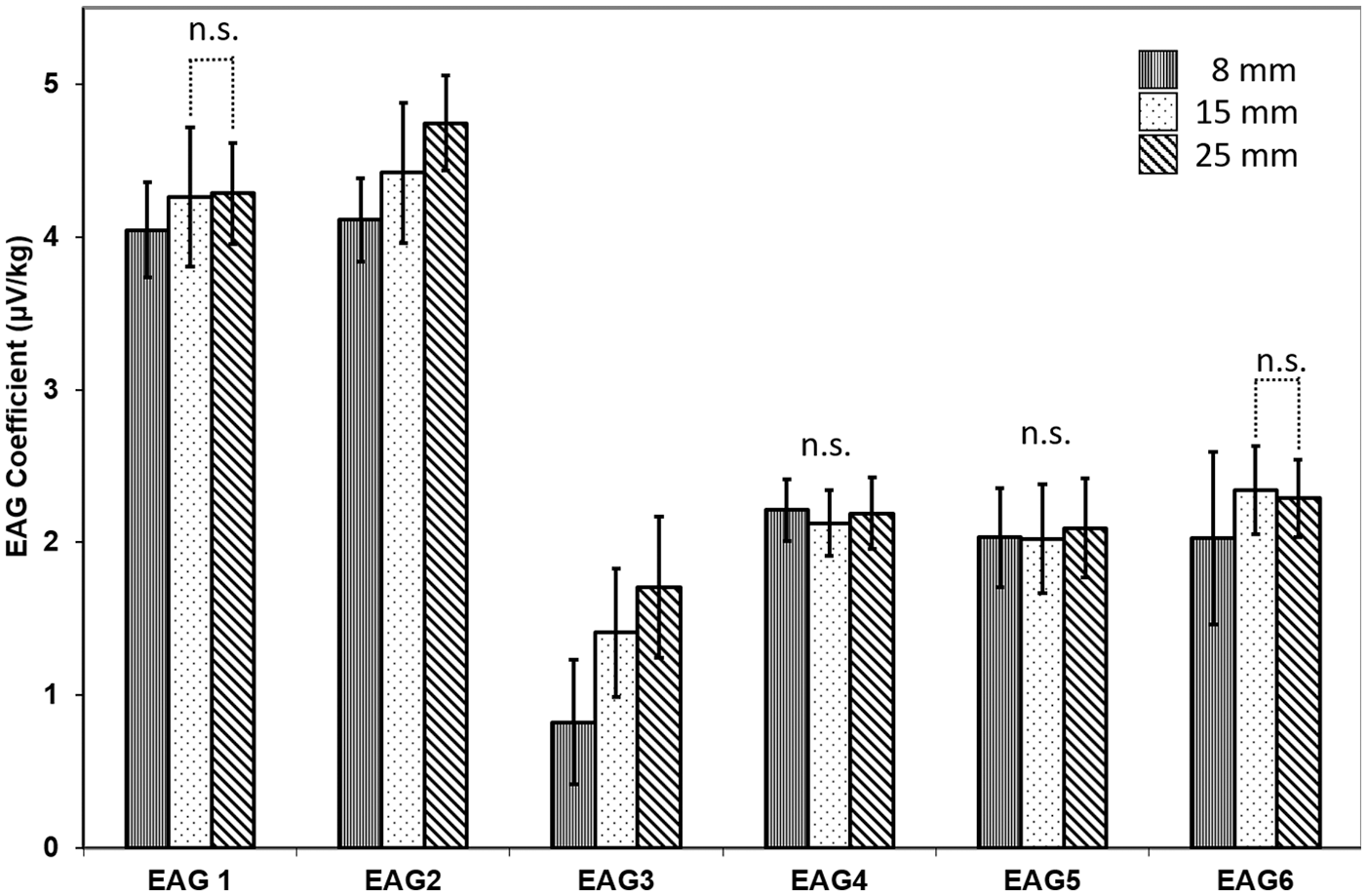
Effect of displacement on electroarthrography (EAG) coefficients. EAG measurements were made at 6 sites while joints were subjected to displacements of 8 mm, 15 mm, or 25 mm, corresponding to average loads of 946 ± 161 N, 1935 ± 273 N, and 3240 ± 530 N, and change in flexion angles of 6.1° ± 0.4°, 11.4° ± 1.1°, and 18.1° ± 1.9°, respectively. Significant differences (P < 0.05) accounting for multiple comparisons were detected between displacements unless otherwise indicated as not significant (n.s.). Significant differences (P < 0.05) were detected for EAG2 and EAG3 but not for EAG4 or EAG5. EAG1 and EAG6 measured significantly different EAG coefficients at 8 mm compared with 15 mm of displacement but not between 15 mm and 25 mm of displacement. Bars are least square means ± 95% confidence intervals (n = 4 immersion buffers × 3 fetlocks × 5 repetitions = 60).
EAG coefficients were significantly reduced (P < 0.05) following trypsin treatment in 6 of 8 electrodes during standing and 7 of 8 during walking (
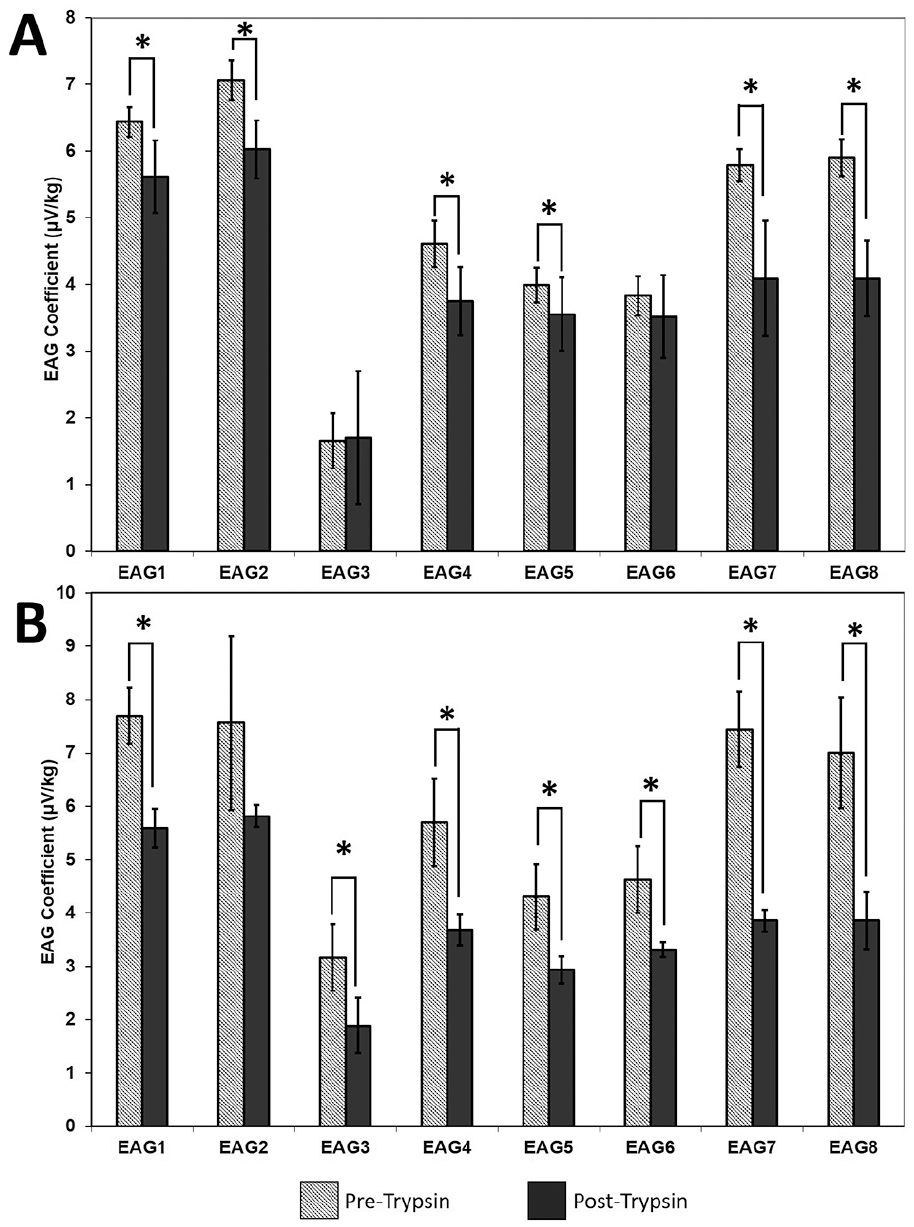
Effect of trypsin degradation on electroarthrography (EAG) coefficients. EAG measurements were made at 6 sites pre- and post-trypsin on a single fetlock during (
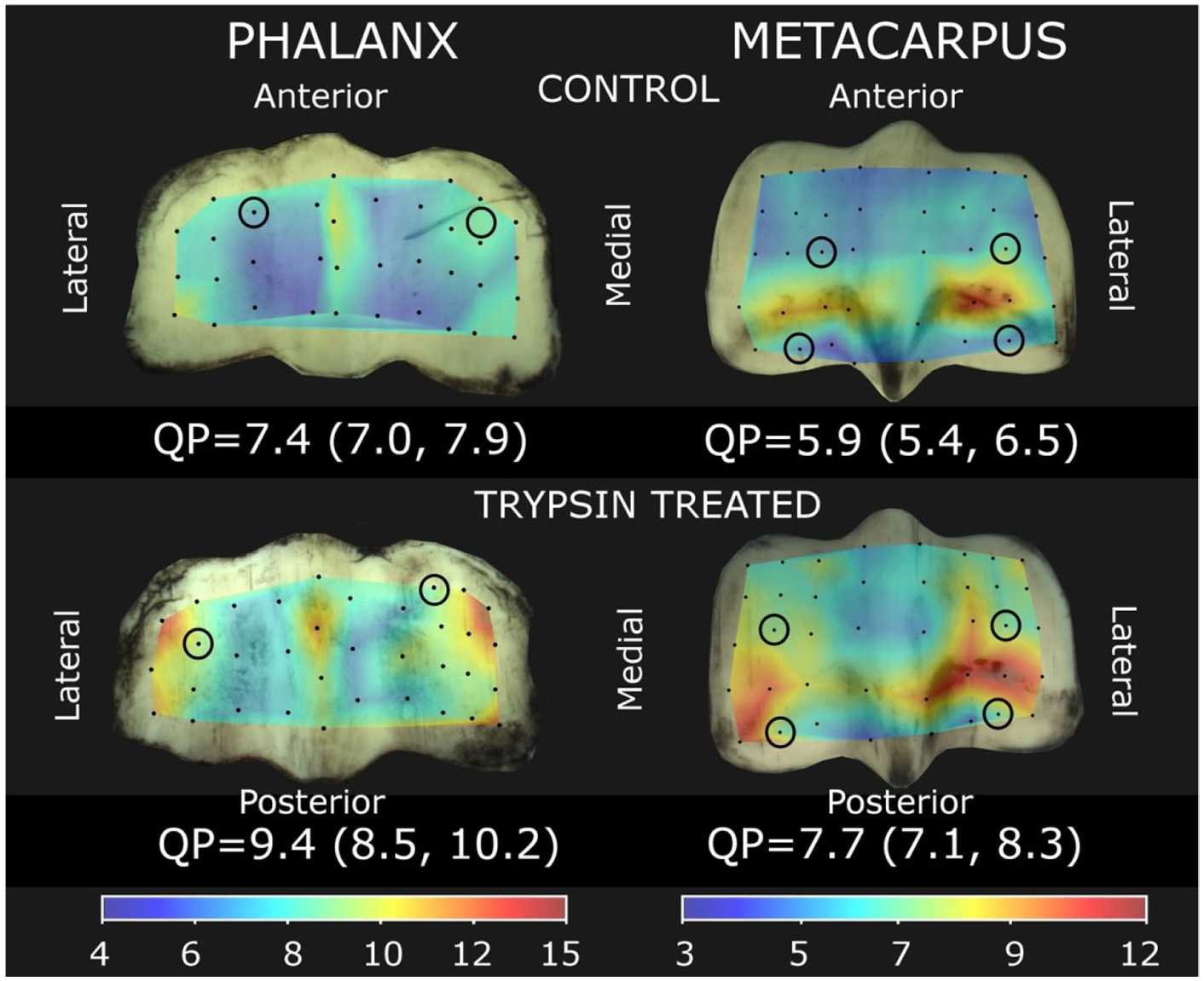
Direct measurements of cartilage streaming potentials. Quantitative parameter (QP) maps were calculated from streaming potentials obtained with the Arthro-BST device at 75 locations on the phalanx and metacarpus in control and trypsin-treated fetlocks. QP is inversely proportional to cartilage stiffness so that higher values are interpreted as degradation. Joint surfaces from the trypsin-treated fetlock have higher QP, inferring degraded cartilage, compared with the contralateral control. Values are least square mean (lower 95% confidence interval, upper 95% confidence interval) for each joint surface. Circles indicate locations where cartilage disks were subsequently isolated for biomechanical testing and histology.
Cartilage Thickness, Biomechanical Parameters, and Biochemical Composition Measured on Cartilage Disks Isolated from Weightbearing Areas of Trypsin-Treated and Contralateral Control Fetlocks. a .
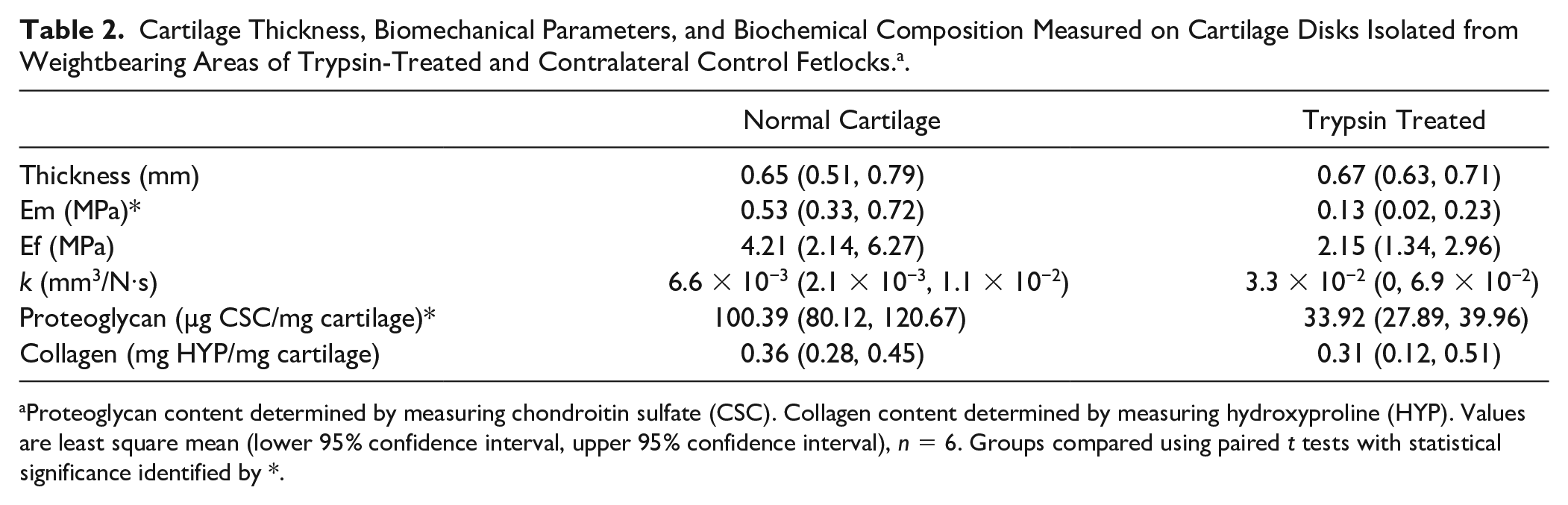
Proteoglycan content determined by measuring chondroitin sulfate (CSC). Collagen content determined by measuring hydroxyproline (HYP). Values are least square mean (lower 95% confidence interval, upper 95% confidence interval), n = 6. Groups compared using paired t tests with statistical significance identified by *.
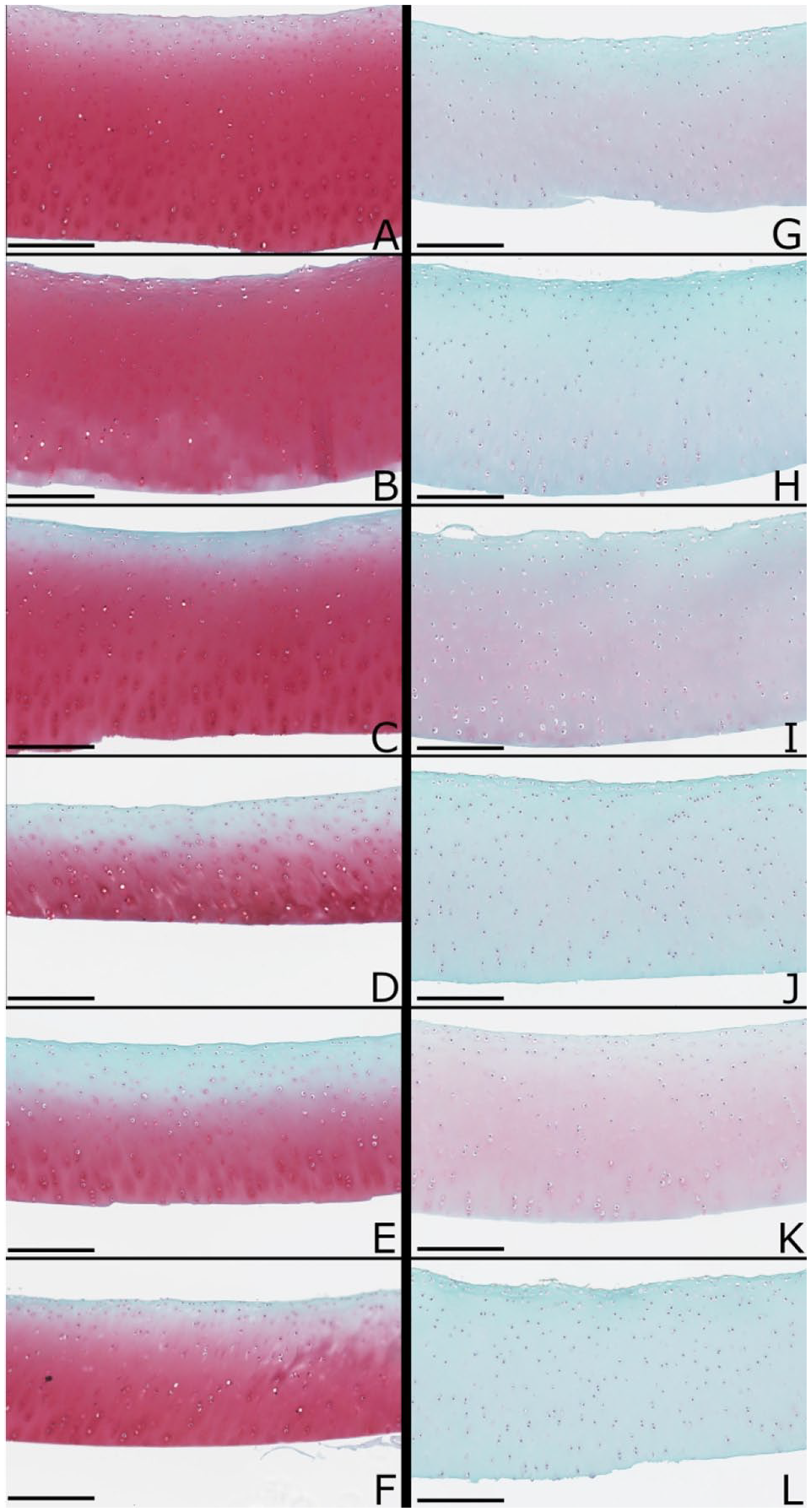
Safranin-O/fast green/iron hematoxylin stained histological sections. Histology was performed on cartilage disks isolated from the contralateral control fetlock (
Discussion
The conditions under which cartilage streaming potentials are produced were controlled first, by changing the ionic strength of the immersion buffer, and second, by directly altering cartilage quality with trypsin. EAG responded in the same manner as direct measurements under similar conditions where increasing salt concentration or trypsin degradation reduces cartilage streaming potentials. These results demonstrate that EAG, measured through skin electrodes, is indeed measuring streaming potentials produced by cartilage during compression. These experiments were conducted on a total of 5 metacarpophalangeal joints, which is a small sample size and a limitation of the study.
EAG Responds Inversely to Ionic Strength of Immersion Buffer
EAG coefficients decreased as conductivity, reflecting ionic strength, of the immersion buffer increased (
Frank and Grodzinsky
6
used silver–silver chloride electrodes to measure streaming potentials produced by mature bovine cartilage disks during sinusoidal compression in confined compression geometry. These measurements were performed in real time while the ionic strength of the bath was increased from 10−4 to 10 M NaCl. These authors found that streaming potentials decreased monotonically as bath ionic strength increased but not at a constant rate. The greatest change in streaming potentials occurred when ionic strengths were in the range of the fixed charge density of the bovine cartilage used, approximately 0.12 to 0.15 M. The present study used PBS solutions with total salt concentrations of approximately 0.015 M, 0.15 M, and 1.5 M for 0.1X PBS, 1X PBS, and 10X PBS, respectively. Similar to Frank and Grodzinsky,
6
EAG coefficients (
Similar relationships between streaming potentials and ionic strength were also obtained from experiments where spatially resolved streaming potentials were measured. Quenneville et al.
23
measured streaming potentials with a microelectrode array directly compressing bovine cartilage disks immersed in salt solutions at various ionic strengths. Streaming potentials were approximately 4 times higher at 0.01 M NaCl and 5 times lower at 0.5 M NaCl compared to measurements obtained at physiological ionic strength, 0.15 M NaCl. Additionally, the relationship between spatially resolved streaming potentials, explored by using a linear array of microelectrodes in contact with isolated cartilage disks subjected to compression, demonstrated that streaming potentials produced by cartilage immersed in 0.15 M NaCl were 4 to 6 times lower than those produced by the same cartilage disks equilibrated in 0.01 M NaCl.
7
Comparable relationships were observed in the present study, notably in EAG coefficients measured in anterior electrodes (
The streaming potential is a dynamic phenomenon that develops during cartilage compression. Interstitial fluid flows through the pores of the charged ECM in response to compressive load thereby displacing mobile positive ions relative to the fixed, negatively charged proteoglycan. The resulting electrical signal is influenced by the ionic strength of the bathing solution because of changes to the Debye length, defined as the thickness of the electrical double layer. At physiological ionic strength (~0.15 M NaCl), high electrostatic repulsion forces are generated because the Debye length is 0.8 nm, which coincides with the distance between fixed negative charge groups on chondroitin sulphate and half the distance between fixed negative charge groups on keratin sulfate. Lower ionic strengths mean that net charge occurs in the fluid phase at a greater distance from fixed glycosaminoglycan due to an increase in the Debye length resulting in higher streaming potentials, while higher ionic strengths confine charge density closer to glycosaminoglycan, resulting in shorter Debye lengths and decreased streaming potentials.6,7,34,35
EAG Responds to Trypsin-Induced Cartilage Degradation
EAG coefficients, measured in the same fetlock before and after intra-articular injection of trypsin, were reduced when compressed to axial loads that approximated both standing and walking (
Trypsin degradation has been used in a variety of models to simulate proteoglycan loss observed in early OA,8,24,26,28,36 where proteoglycan depletion begins at the articular surface and proceeds toward the subchondral bone. 27 Trypsin is a serine protease and cleaves proteoglycan at multiple sites along its core protein to produce 2 fragments. 37 The first fragment contains the G1 domain, which is a protein-hyaluronic acid complex ranging from 70 kDa to over 100 kDa,38,39 and is critical to the aggregation of proteoglycan into large macromolecules. The second protein fragment includes a lengthy region between the G2 and G3 domains containing both chondroitin sulfate and keratin sulphate, which bear negative fixed charge groups.37,38,40 Trypsin targets collagen to a lesser extent,41,42 with Harris et al. 41 measuring depletions of approximately 5% for hydroxyproline, compared with 70% for proteoglycan, in cartilage incubated in trypsin for 24 hours.
The present study is the first to demonstrate that EAG, acquired through electrodes placed on skin surrounding an articulation, is sensitive to cartilage degradation. Joint compression resulting in loads approximating standing and walking (
EAG Demonstrates Potential to Provide a Non-invasive, Spatially Resolved Indicator of Cartilage Quality
Electrodes were adhered to skin symmetrically around the fetlock at the joint line and we observed that the amplitudes of EAG coefficients varied in a location-specific manner. EAG coefficients at anterior (dorsal) electrodes (EAG1 and EAG2) consistently had higher amplitudes than other sites. These electrodes were in closer proximity to cartilage surfaces undergoing compression compared to electrodes placed mediolaterally (EAG3 and EAG4) or at the metacarpus/sesamoid interface (EAG5 and EAG6). This pattern corresponds to reported load distribution in the fetlock, 29 where cartilage in the dorsal margin of the first phalanx, located immediately behind electrodes EAG1 and EAG2, experiences higher pressures during joint compression compared with the palmar margin. These data are also supported by a validated finite element model developed to better understand the forward problem of EAG. 19 The model, which imposed current density sources of varying configurations in the medial and lateral knee joint compartments, demonstrated that EAG signals obtained at the skin surface were altered when the locations of source current density were changed. Sources placed close to the joint periphery produced well-defined maxima while sources located more internally resulted in broader maxima. EAG appears to reflect joint specific loading and is capable to a certain degree of providing spatially resolved information about cartilage quality.
As cartilage contact area is understood to increase progressively from the dorsal toward the palmar margins as compressive joint loads increase,29,30 EAG coefficients were expected to be relatively stable at different displacements because they are normalized to axial load resulting from whole joint compression. However, we detected statistical differences due to displacement (
Conclusion
This study demonstrates that externally measured EAG is indeed detecting streaming potentials originating in cartilage during joint loading. The non-invasive EAG method has the potential to be developed into a sensitive diagnostic of cartilage health that may contribute to the early detection and treatment of OA and other degenerative joint diseases.
Footnotes
Authors’ Note
This study was performed at the Comparative Orthopaedic Research Laboratory, Department of Clinical Studies, University of Guelph.
Author Contributions
AC, MG, EQ, KG, PS, MDB, and MBH participated in study conception and design. AC and MG acquired and analyzed data. AC and SBB carried out statistical analyses. All authors participated in data interpretation. AC drafted the manuscript and it was critically revised by all authors. All authors approved the final version of the manuscript. AC takes responsibility for the integrity of the work as a whole.
Acknowledgments and Funding
We acknowledge technical assistance provided by Michele Beaudoin-Kimble, Felipe Garcia, Mohamed Hoba, and Sotcheadt Sim. Salary support for AC provided by the Natural Sciences and Engineering Research Council of Canada.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MG and EQ are the owners of Biomomentum Inc.
Ethical Approval
Procedures were performed with Institutional Animal Care Committee approval in accordance with the Canadian Council on Animal Care (University of Guelph, AUP#1412).
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
