Abstract
Objective
Low-frequency vibration accelerates cartilage degeneration in knee osteoarthritis (KOA) rat model. In this article, we investigated whether whole-body vibration (WBV) increases cartilage degeneration by regulating tumor necrosis factor-α (TNF-α) in KOA.
Design
Proteomics analysis was used to filter candidate protein from synovial fluid (SF) in KOA people after WBV. Enzyme-linked immunosorbent assay (ELISA) was used to estimate changes in TNF-α levels in SF. The C57 mice and TNF-α knock-out mice were sacrificed for the KOA model and WBV intervention. The cartilage was tested by ELISA, histology, terminal-deoxynucleotidyl transferase mediated nick end labeling (TUNEL), immunohistochemistry, and reverse transcriptase polymerase chain reaction. Luciferase activity test in vitro study was conducted to confirm the relationship between TNF-α and the candidate protein.
Results
Differentially expressed proteins were enriched in the glycolytic process, glucose catabolic, and regulation of interleukin-8 (IL-8) secretion processes. Phosphoglycerate kinase, triosephosphate isomerase 1, T cell immunoglobulin- and mucin-domain-containing molecules 2, fumarylacetoacetate hydrolase (FAH), and TNF were the hub node. TNF-α expression increased in SF after WBV (P < 0.05). The cartilage was more degenerated in the TNF-α−/− mice group compared to controls. A significant change was observed in collagen II and FAH (P < 0.05). TNF-α expression improved in C57 mice (P < 0.05). Apoptosis of chondrocytes was inhibited in TNF-α−/− mice by the TUNEL test. Luciferase activity significantly increased in TNF-α + FAH-Luc cells (P < 0.05).
Conclusion
A novel mechanism underlying WBV-triggered cartilage degeneration was found in KOA that demonstrated the critical regulatory function of TNF-α and FAH during WBV.
Introduction
Whole-body vibration (WBV) reduces pain and improves function in individuals with knee osteoarthritis (OA).1,2 In our previous randomized controlled study, similar benefits of WBV were observed in people with knee OA. 3 However, the side effects of WBV have not been well defined in all clinical trials. It was previously found that repeated high-frequency and low-amplitude vibration induce degeneration of knee joints in mice. 4 Similar negative effects are observed in rat models of knee OA following WBV intervention. 5 However, the molecular mechanism of WBV intervention is not well known.
Examination of inflammatory pathways revealed that vibration stimulation may affect cartilage metabolism. Intervertebral disc degeneration induced by vibration upregulates the expression of interleukin-1β (IL-1β). 6 Tumor necrosis factor-α (TNF-α) may orchestrate the effects of vibration on cartilage degradation in ovariectomized rats. 7 WBV inhibits the LPS-mediated secretion of inflammatory cytokines TNF-α in human mesenchymal stem cells. Proinflammatory cytokine TNF-α plays an important role in the pathophysiology of OA. 8 It is still unclear whether vibration increases cartilage degeneration through TNF-α in knee OA.
We hypothesized that WBV may induce cartilage degeneration in patients with OA through the TNF-α pathway. We set up a self-control clinical study to observe changes in TNF-α in synovial fluid (SF) of knee OA after WBV intervention and performed proteomics analysis to identify candidate biomolecules or pathways. The effects of WBV and TNF-α on cartilage degeneration were investigated using a knockout mouse model of OA. Isobaric tags for relative and absolute quantitation (iTRAQ) proteomic analysis and protein-protein interactions (PPI) analysis identified one hub gene, fumarylacetoacetase hydrolase (FAH). Cell experiments were conducted to examine the relationship between TNF-α and FAH.
Methods
Clinical Trial
This study was a single-center, randomized controlled trial. The study design followed the OA clinical trial recommendations from Osteoarthritis Research Society International. 9 Approval for the study was provided by the ethics committee of West China Hospital, Sichuan University (Approval Number: 2016-286). All patients provided informed consent for the publication of data collected during the study. This clinical trial was registered on the public trial registry (http://www.clinicaltrials.gov/, Number: ChiCTR-ONC-16010176). Patients who met the eligibility criteria and signed informed consent form were assigned to the WBV + Exercise group and Exercise group. The randomization list was generated by SPSS21.0 software and allocation concealment was conducted using sealed opaque envelopes. Synovial fluid was collected before and after WBV intervention under ultrasound-guided in the WBV + Exercise group.
Those included in the study were outpatients who met the diagnostic standard of American OA of rheumatology 10 ; classified in grade II to IV in Kellgren & Lawrence (K/L) X-ray imaging; aged between 50 and 75 years old; and had no history of knee trauma and surgery. Patients were excluded from the study if they had combined rheumatic diseases; other severe systems disease and hence could not complete the treatment; knee neoplasms, infections, fractures, and metabolic osteopathy; and severe pain in the knee and hence could not complete the WBV treatment. The sampled patients were not included if there was a misdiagnosis or the required treatment failed.
The WBV intervention strategy was carried one time each day for 5 times a week for 4 weeks. It was done in a vibration training instrument (Fitvibe, Belgium) under a 20 Hz vibration frequency, 2 mm amplitude for 15 minutes as described in detail by Wang et al. 3 Patients were requested to face the screen of the vibrator in the vibration platform with the knee flexed from 0° to 90°. The knee range motion during WBV training was adjusted according to the pain and motor control ability maintaining 10 seconds at flexion position, 6 times/group, with the rest of 30 seconds every time, for a total of 3 groups in total with an interval of 1 to 2 minutes between each group.
iTRAQ Proteomic Analysis and Protein-Protein Interactions
Four differentially expressed protein sets identified by iTRAQ and enrichment analysis was used to infer an association to a collection of the pathway, disease, drug-gene interactions, and trans-factors binding related gene-set library from WikiPathways2016, GeneOntology2018, and MSigDB databases. Protein-protein interactions were predicted in the Search Tool of the Retrieval of Interaction Genes/Proteins (STRING) database and expand the PPI network of differential proteins by pushing the “more” button in STRING.
Animal Study
Group and Modeling
All animal experiments were performed according to the guidelines of the animal ethics committee regulations recommended by the Institutional Animal Care and Use Committee at the West China Hospital, Sichuan University and National Institutes of Health Guide for the Care and Use of Laboratory Animals. 11 Three-month old male C57BL/6J (wild type [WT] mice and TNF-α knock-out [TNF-α−/−, C57BL/6J background]) mice were obtained from the endocrine laboratory of Sichuan University. All mice were provided with a standard chow diet and clean water. They were kept in ventilated racks with pathogen-free barrier conditions at a constant temperature (22°C) under a 12-hour light/darkness cycle. A total of 16 healthy male C57 mice (3-month old) and 16 healthy males TNF-α−/− (3-month old) were used in this trial. All the mice build the destabilization medial meniscus (DMM) model. DMM model was made by removing the tibial medial meniscus ligament. 12 The detailed modeling method followed the method the OA Research International Society provided. 13 All mice were randomly allocated after 4 weeks of the surgery. All the 16 C57 mice were randomized into the WBV vibration group (C57 + DMM + WBV) and the control group (C57 + DMM), 8 for each group. The 16 TNF-α−/− mice were randomized into the WBV vibration group (TNF-α + DMM + WBV) and the control group (TNF-α + DMM), 8 for each group. All the mice were sacrificed by cervical dislocation.
Enzyme-Linked Immunosorbent Assay (ELISA)
For the proteomic analysis and ELISA test, at least 15 mL of the SF of each patient was collected, marked, and stored in liquid nitrogen for less than 3 months. Detection was carried out using an ELISA kit according to the manufacturer’s instructions (Boster Biological Technology, Wuhan, China).
Red O-Solid Green and Terminal-Deoxynucleotidyl Transferase Mediated Nick End Labeling (TUNEL) Detection
The cartilage of the knee joint was collected, fixed with 4% triformol, decalcified using 20% formic acid, embedded with wax, and finally stained with a red O-solid green kit (Solarbio, Beijing, China) and TUNEL kit (Enogene, Nanjing, China) for apoptosis detection.
Western Blot
The cartilage of the knee joint was mashed with the lysis buffer. The lysate supernatant was collected and the protein concentration quantified using a BCA kit. The proteins were separated using SDS-PAGE and then transferred to a polyvinylidene fluoride membrane. The membranes were blocked with 5% nonfat milk and incubated with a primary Ab at 4°C overnight. The antibodies used included the followings: TNF-α (anti mice), FAH (Proteintech, 14928-1-AP0), CRP (Affinity, DF6027), α-SMA (Affinity, AF1032), Collagen II (Affinity, AF0135), TIMP (Affinity, AF0264), TNF-α (anti-human, Abcam, ab6671), IL-1β (Affinity, AF5103), NF-κB (Omnimabs, OM286071), p-p38 (Affinity, AF3456), C-Casp3 (Abcam, ab32042), Bax antibody (Affinity, AF0083), Fas (Affinity, AF5342).
Cell Experiment
Detection of the Direct Effect of TNF-α on FAH Transcriptional Activity on 293T Cell
Construction of plasmid pGL3-FAH, pcDNA3.1-TNF-α vector, and pcDNA3.1-TNF-α vector. Preparation of DNA fragments and vectors. Plasmids puc57-p-FAH and pGL3-basic were extracted and XhoI (Takara, 1094A) and HindIII (Takara, 1060A) used for double digestion. The recovered and purified target fragment was then connected with the recovered and purified vector. The cultured solution was identified by colony PCR (polymerase chain reaction). The primer sequence of FAH was: CCCAAGCTTATGTCCTTCATCCCGGTGG (forward) and CCCTCGAGTCATGATGGCAGGAGAGC (reverse); and for TNF-α: CGGGGTACCATGTCCTTCATCCCGGTGGC (forward) and CCGCTCGAGTCATGATGGCAGGAGAGCAG (reverse).
Experimental grouping: pcDNA3.1 + pGL3 group, pcDNA3.1-TNF-α + pGL3 group, pcDNA3.1 + pGL3-FAH group, and pcDNA3.1-TNF-α + pgl3-FAH group.
Real-Time PCR Analysis
RNA was extracted using the classic Trizol (Invitrogen) method. A reverse transcription kit was used to generate complement DNA (cDNA) that was used as a template for Q-PCR analysis. The expression of protein from mRNA was detected through a real-time fluorescent quantitative PCR (ABI, V7) analysis using SYBR Green qPCR kit following the manufacturer’s recommendations.
Luciferase Activity Detection
The plasmid pGl3, pRL, and pcDNA 3.1 were diluted at the ratio of 0.5:0.1:1.9 in serum-free opti-MEM. The plasmid mixture was transfected into 293T cells using Lipofectamine 2000 reaction reagent following the recommended procedure. The 293T cell line (human embryonic renal cell lines) were purchased from Shanghai Cell Bank. After 4 to 6 hours of incubation, the media was changed with normal media and incubated for 48 hours. Luciferase activity was detected using a dual-luciferase kit (Beyotime, RG008)
Detection of the Inflammation Biomarker Change after TNF-α Influence the FAH Transcriptional Activity on SW1353 Cell
siRNA knockdown FAH and grouping
SW1353 (human osteosarcoma cell line) was purchased from Shanghai Cell Bank and the siRNA synthesized in Shanghai Bioengineering Co. Ltd. The SW1353 cells were transfected with the pcDNA3.1 and pcDNA3.1-FAH vectors for pcDNA3.1 and pcDNA3.1-FAH group, respectively. The SW1353 cells in the logarithmic growth stage and good growth state were inoculated into a 12-well plate for cell culture with 2 × 105 cells per well. They were then cultured overnight in an incubator at 37°C and 5% CO2 concentration and changed to serum-free DMEM medium 2 hours before transfection.
For the control group, SW1353 cells were cultured for 48 hours. For experimental group 1, pcDNA3.1-TNF-α vector and pcDNA3.1-FAH vector transferred into SW1353 cells, and then cultured for 48 hours. The pcDNA3.1-TNF-α vector and FAH-siRNA plasmid transferred into SW1353 cells and further cultured for 48 hours for experimental group 2. Western blot experiments were used to detect TNF-α, FAH, IL-1, NF-κB, C-Casp3, Bax, p-p38, and Fas protein expression.
Statistical Analysis
Statistical Package for Social Science (SPSS) version 21.0 was used in data analysis. All the data were expressed as a mean ± standard deviation. Differences between groups were assessed through analysis of variance (ANOVA) followed by Bonferroni or Fisher LSD post hoc tests with the significance level of P < 0.05.
Results
WBV Can Significantly Increase the TNF-α Level in the SF of OA Patients
Among the 277 patients assessed for eligibility, 56 met the inclusion criteria for the study and 37 of them completed the WBV intervention. The SF (10 mL at least) that met the proteomic analysis standards was successfully collected from 6 patients. Four patients had their SF (5 mL at least) meeting the ELISA requirements. From these analyses, differentially expressed proteins ( Table 1 ) were found to be significantly (P < 0.05) enriched in the glycolytic process through glycolysis, glucose catabolic, and regulation of interleukin-8 (IL-8) secretion processes ( Fig. 1 ). PGK, fumarylacetoacetate hydrolase (FAH), phosphoglycerate kinase (PGK), triosephosphate isomerase1 (TPI1), T cell immunoglobulin-and mucin-domain-containing molecules 2 (TIM2), and TNF-α were hub genes, which might connect with important proteins in expanding the PPI network. PGK and FAH were the important metabolizing enzyme of glycolysis and glucose catabolic in cartilage degeneration. 14 IL-8 regulates TPI1, TIM2, and TNF pathways. 15 These proteins had tight interactions suggesting that they function in a network manner.
Results of Difference Expression Protein after WBV Intervention (>1.5-Fold).

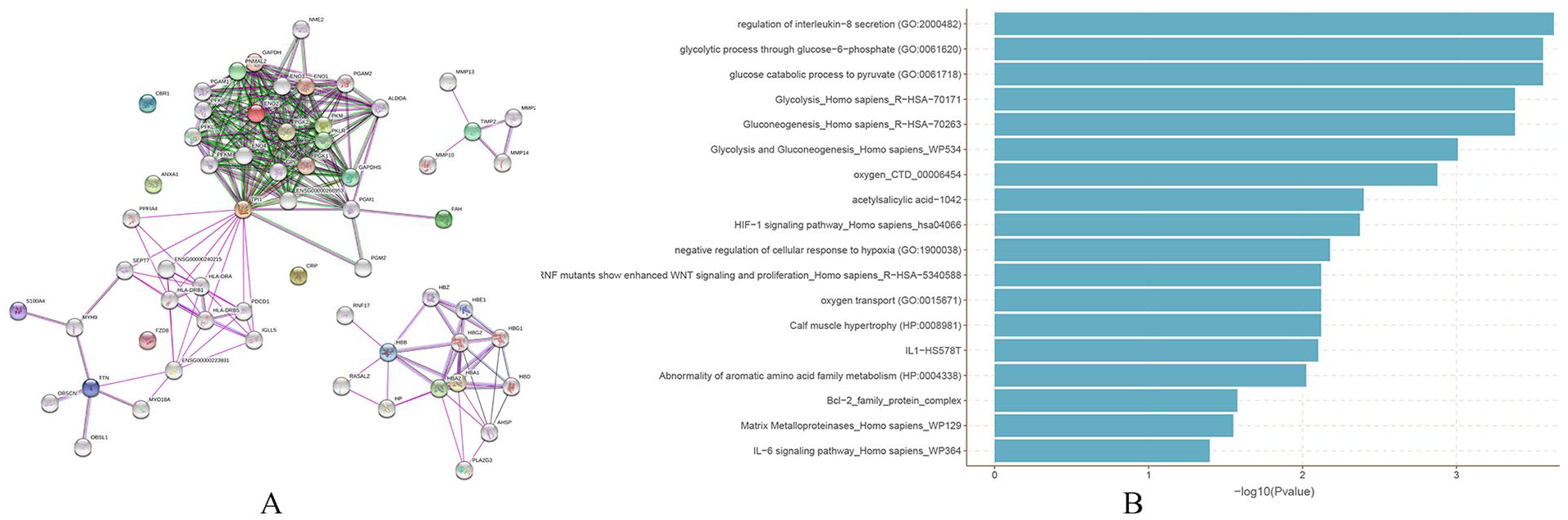
Isobaric tags for relative and absolute quantitation (iTRAQ) proteomic analysis and protein-protein interactions (PPI).
The TNF-α expression level in SF significantly increased after WBV treatment (P < 0.01). There was a significant difference before (0.13 ± 0.02) and after (0.24 ± 0.03) WBV intervention, with the mean difference of 0.11, 95% confidence interval = (0.07, 0.15).
TNF-α Plays an Important Role in the WBV Treatment of OA in Mice Models
Cartilage matrix of 4 groups stained Red O-solid green, and cartilage damage was evaluated by Osteoarthritis Research Society International (OARSI) score. Figure 2A showed the cartilage was severely destroyed in the C57 experimental group compared to the C57 control group. The staining of cartilage was smooth in both the TNF-α knock-out mice model and the TNF-α knock-out mice control group. A Kruskal-Wallis H analysis revealed a significant difference in OARSI score distribution among the 4 groups (F = 22.624, P < 0.001). OARSI score in the C57 + DMM + WBV was significantly higher than the C57 + DMM group (F = 16.668, P = 0.001), and that in TNF-α + DMM + WBV significantly higher than TNF-α + DMM group (F = 11.938, P = 0.043). The cartilage matrix of 4 groups stained by TUNEL is shown in Figure 2B . A higher level of cell apoptosis was induced in cartilage in the C57 + DMM + WBV group than in the C57 + DMM group. This indicated that vibration therapy might enhance the cartilage cell apoptosis of KOA. The cell apoptosis in the TNF-α + DMM + WBV group was significantly lower (P < 0.05) than in the C57 + DMM + WBV group. The TNF-α + DMM group and the TNF-α + DMM + WBV group had a similar level of apoptosis.
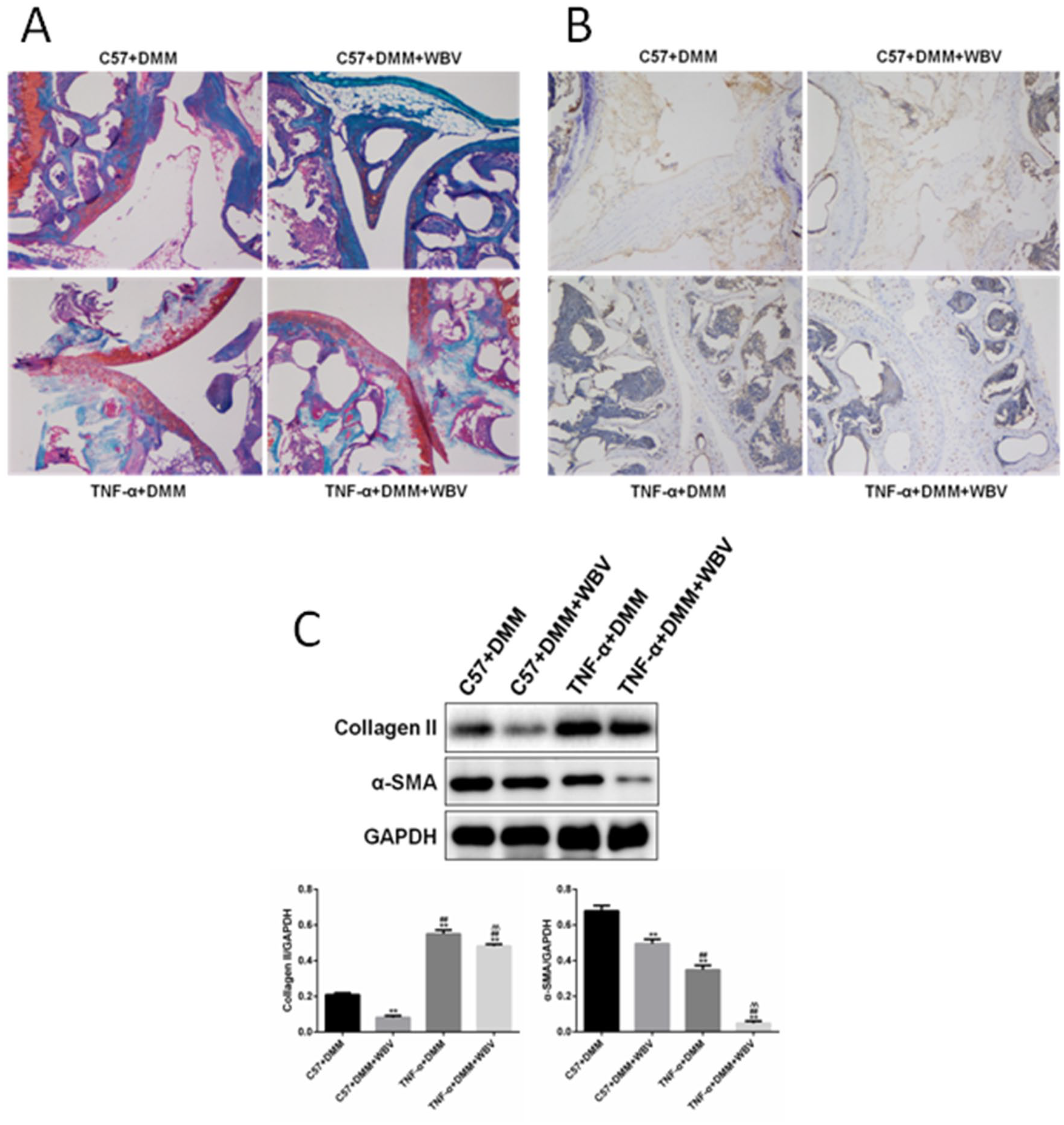
Representative results of WBV treatment in vivo. (
The western blot result is shown in Figure 2C . The expressions of collagen II and α-SMA in cartilage reduced severely in both the TNF-α + DMM + WBV group and C57 + DMM + WBV group but were lower in the TNF-α + DMM + WBV group.
Roles of FAH in the TNF-α Induced WBV
The expression of FAH, TIM2, and CRP in the cartilage was detected in different groups ( Fig. 3 ). The levels of FAH, TIM2, and CRP expression in experimental groups significantly decreased compared to the control group. The level of CRP and TIM2 was lower and higher, respectively, in the TNF-α + DMM + WBV group than the TNF-α + DMM group. There was no significant difference in the expression level of FAH.
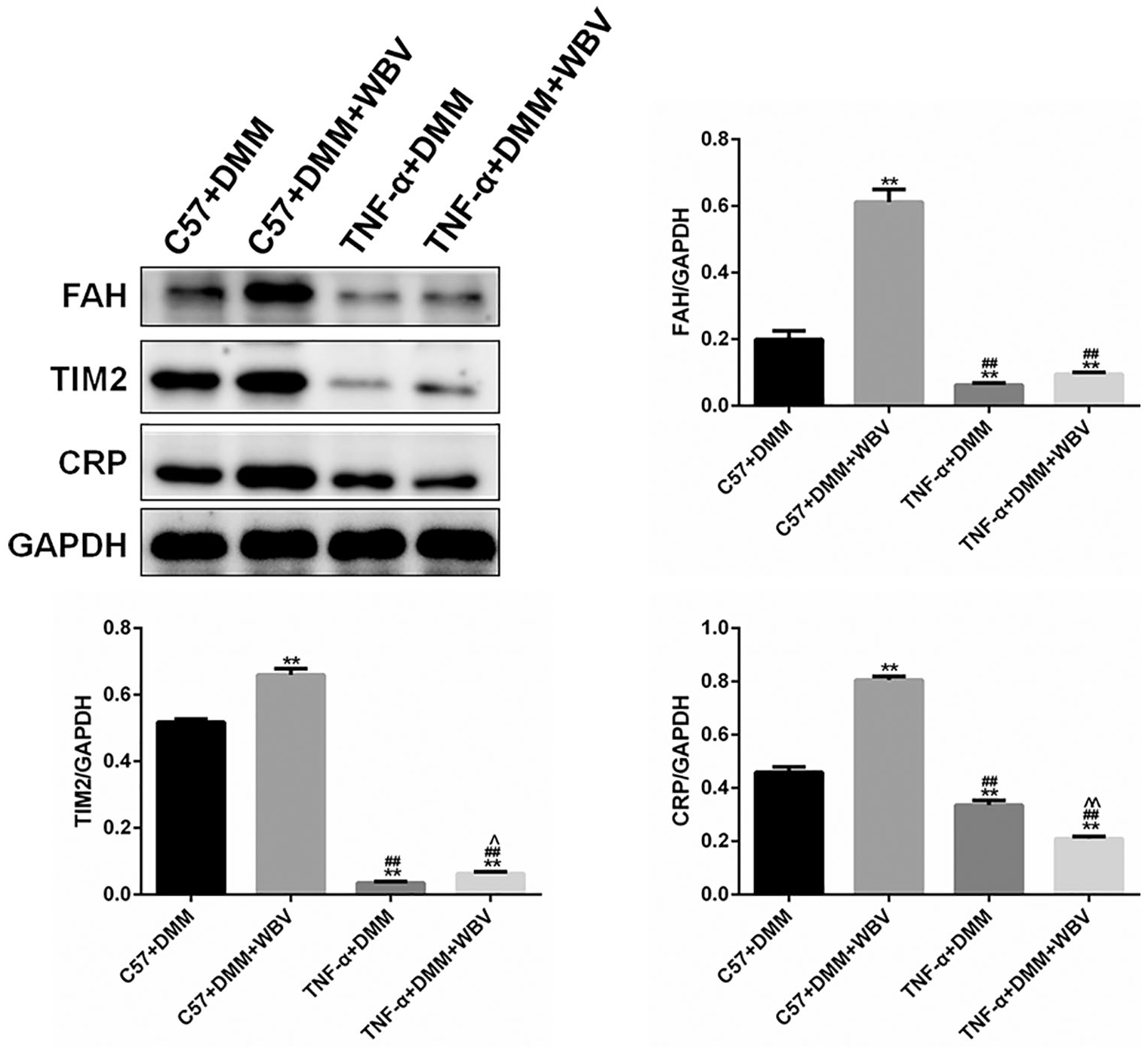
Expression of FAH, TIM2, and CRP in the cartilage in 4 groups. Expression of FAH, TIM2, and CRP in the cartilage was detected in different groups. FAH = fumarylacetoacetase hydrolase; TIM2 = tissue inhibitor of metalloproteinases 2; CRP = C-reactive protein; WBV = whole-body vibration; TNF-α = tumor necrosis factor-α; DMM = destabilization medial meniscus.
The TNF-α Influences the Expression of Inflammation or Apoptosis to Affect the Transcripts of FAH
No activity of luciferase was found in the control group and TNF-α group ( Fig. 4A ). The activity of luciferase significantly increased for the TNF-α + FAH-Luc group compared to the FAH-luc group implying that TNF-α targeted the FAH gene at the transcriptional level.
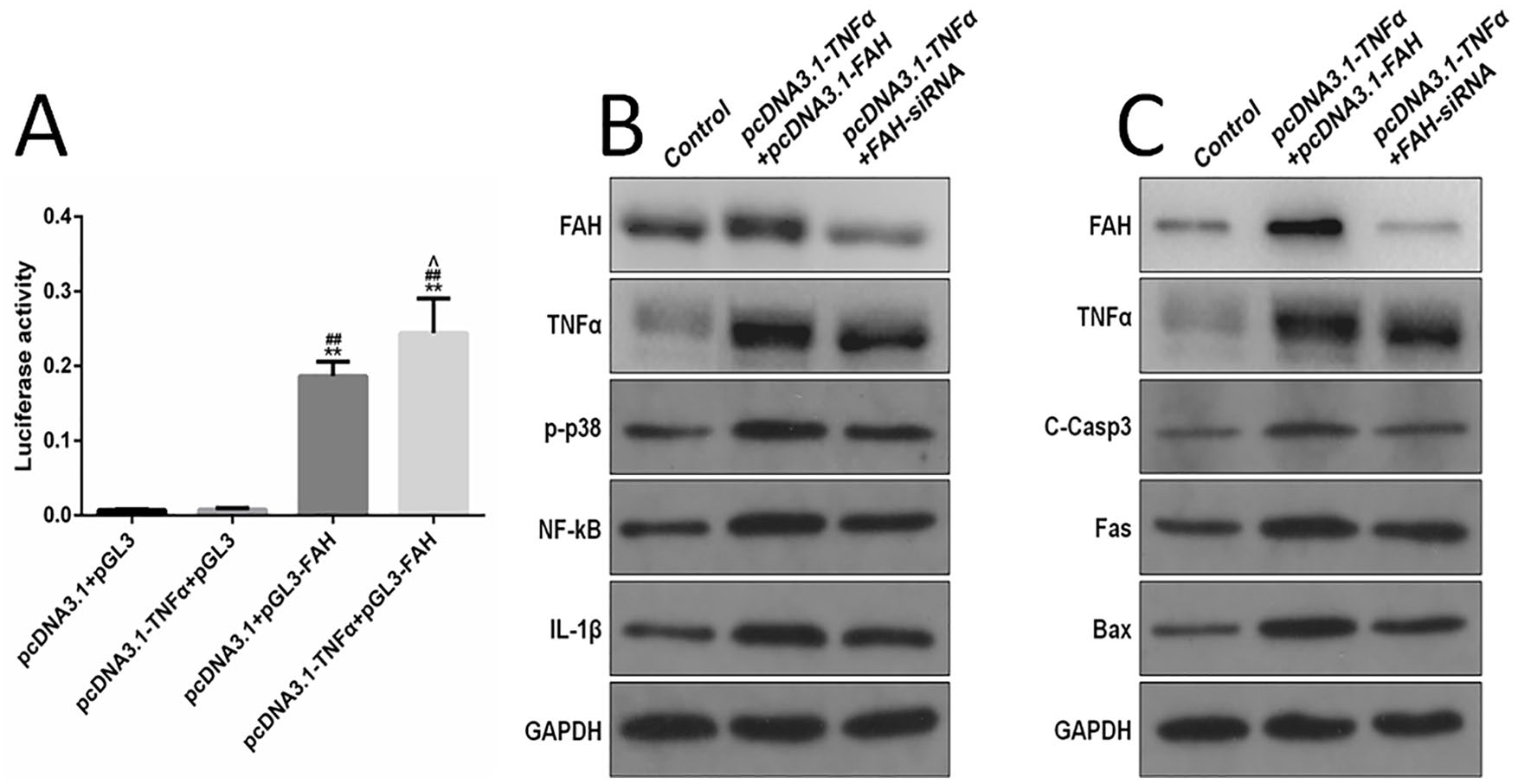
TNF-α upregulates inflammation or apoptosis through FAH. (
Protein expression of TNFα, FAH, C-Casp3, Bax, and Fas was detected by western blot. The level of TNFα, IL-1β, NF-кB, and p-p38 significantly improved in the pcDNA3.1-TNFα + pcDNA3.1-FAH and pcDNA3.1-TNFα + FAH-siRNA group compared to the control ( Fig. 4B ). Cellular expression of FAH increased in pcDNA3.1-TNFα + FAH-siRNA group. Besides, the expressions of TNFα, FAH, IL-1β, NF-κB, and p-p38 were lower in pcDNA3.1-TNFα + FAH-siRNA group than in pcDNA3.1-TNFα + pcDNA3.1-FAH group.
Notably, cell apoptosis and inflammation were enhanced following TNF-α overexpression ( Fig. 4C ). The upregulation of FAH resulted in the overexpression of TNF-α. The FAH knocked out led to a decrease of TNF-α induced inflammation and apoptosis. In summary, TNF-α could influence the expression of inflammation or apoptosis factor in cartilage cells by regulating the transcripts of FAH.
Discussion
This study found TNF-α production to increase cartilage degeneration in KOA through FAH signaling pathways. The detailed mechanism may involve TNF-α, which might promote FAH gene expression to increase chondrocyte apoptosis.
A study by Qin demonstrated a cartilage degeneration in the KOA rat model after WBV (35 Hz, 0.3 g) intervention. 5 We observed similar results using lower vibration frequency (20 Hz) mice experiments. Unlike this study that observed degeneration in the sixth week after vibration in rat KOA model, 5 we found degeneration in as early as 4 weeks of vibration in the C57/B mice OA model. After 4 weeks of vibration, Safranin O-fast green staining of cartilage matrix damage increased in the C57/B model. Similar results were reported by Parajuli et al, who found that extent of cartilage damage was higher between 4 and 8 weeks of exposure to WBV. 16 Therefore, cartilage degeneration effects of WBV on mice OA model suggest that the lower frequency vibration still has a negative damage effect on cartilage. Contrary to our findings, Spagnoli reported that low-intensity vibration protected the retention of articular cartilage in a mouse model of diet-induced obesity. 17 This difference can be explained by the different knee OA mice model used. Spagnoli used 5-week-old male C57BL/6 mice while our study used 3-month old male C57BL/6J skeletal mature mice with a destabilization of the medial meniscus surgery.
We used the iTRAQ-based proteomics method and bioinformatics analysis to explore the impact of WBV on people with KOA. The posture of vibration treatment could not be consistently used among subjects because the exact angle was adjusted by patients who could control the balance while feeling no pain. Fortunately, 32 proteins with significant changes were identified after WBV treatment in OA. Differentially expressed proteins were enriched in the glycolytic process through glucose-6-phosphate, glycolysis, glucose catabolic, and regulation of IL-8 secretion processes. FAH, TPI1, TIM2, and TNF were identified as hub genes. TNF-α is a critical inflammatory cytokine associated with cartilage degeneration of OA. 18 It has been reported that TNF-α could increase collagen □ cleavage through matrix metalloproteinases in cartilage. 19 Besides, TNF-α also contributes to chondrocyte apoptosis.20,21 Serum levels of TNF-α are applied in the prediction of knee cartilage loss 22 and have been effectively used to monitor the efficacy of various OA treatment methods. 23 Our animal study found that the degeneration effect after vibration could be induced by TNF-α. The collagen II and cartilage matrix was decreased more severely in the C57 + DMM + WBV group than the TNF-α + DMM group ( Fig. 2 ). For knee OA people, whether vibration could increase TNF-α expression was still unclear. Li 24 observed that dynamic compression (10% and 20% strain amplitude) could protect cartilage by inhibiting TNF-α and IL-6. ELISA results of SF in our study indicated that TNF-α significantly increased after WBV treatment.
The double-luciferase test used in this study required the transfection of multiple plasmids. Therefore, we used 293T, a typical tool cell for luciferase detection, because it has a higher transfection efficiency than chondrocyte. The 293T was used in the first step of our in vitro study to detect whether TNF-α could affect FAH transcriptional activity. After a positive discovery, SW1353 chondrosarcoma cells were further used to identify the effects of TNF-α on FAH transcriptional activity as they have similar properties to chondrocytes and a higher proliferation rate. TIM2 has proved to be an essential biomarker in OA. FAH was a novel finding that associate with TNF-α. FAH is the last enzyme in the tyrosine degradation pathway whose deficiency could cause high tyrosinemia. It contains 14 domains and regulates genes located at 25.1 chromosome area. 25 It also can hydrolyze fumarylacetoacetic acid into acetoacetate and fumarate.25,26 Acetoacetate can induce hepatocyte apoptosis through ROS-mediated MAPKs pathway. 27 Fumarate may decrease cell viability through an apoptotic pathway. 28 Our results ( Fig. 4B and C ) showed that TNF-α upregulated FAH expression in SW1353 cells.
This study has some limitations. The pathogenesis of OA in this DMM model was largely due to joint stability, but WBV could increase muscle contraction to facilitate joint stability. Therefore, the damage effects of vibration on cartilage may not be measured. Besides, the sample size in this clinical research is relatively small since only 6 menopausal female knees OA people were enrolled. Last, our results in clinical and mice research were based on specific WBV intervention parameters and duration, and hence the need to be cautious. Our findings pointed out the side effect of WBV and prudent clinical use of WBV on early OA people. In the progress of WBV clinical use, future studies to explore on inhibition of the degeneration effect of vibration on cartilage needed to be conducted.
Conclusion
Low-frequency vibration promotes TNF-α production, which increases the cartilage degeneration in knee osteoarthritis.
Footnotes
Acknowledgments and Funding
We are grateful to professor De-yin Kang for providing training assistance in statistical analysis. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Natural Science Foundation of China (Grant Numbers 81372110, 81802246) and The Research Fund for the Doctoral Program of Affiliated Hospital of Southwest Medical University (Grant Number 2016-QB-4).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Approval for the study was provided by the ethics committee of West China Hospital, Sichuan University (Approval Number: 2016-286).
Informed Consent
All patients provided informed consent for the publication of data collected during the study.
Trial Registration
Animal Welfare
All animal experiments were performed according to the guidelines of the animal ethics committee regulations recommended by the Institutional Animal Care and Use Committee at the West China Hospital, Sichuan University, and National Institutes of Health Guide for the Care and Use of Laboratory Animals.
