Abstract
Objective
Chronic inflammation plays an important role in the osteoarthritis (OA) pathology but how this influence OA disease progression is unclear. Leukotriene B4 (LTB4) is a potent proinflammatory lipid mediator generated from arachidonic acid through the sequential activities of 5-lipoxygenase, 5-lipoxygenase–activating protein, Leukotriene A4 hydrolase (LTA4H) and its downstream product LTB4. The aim of this study is to investigate the involvement and the potential therapeutic target of the LTB4 pathway in OA disease progression.
Design
Both clinical human cartilage samples (n = 7) and mice experimental OA models (n = 6) were used. The levels of LTA4H and leukotriene B4 receptor 1 were first examined using immunostaining in human OA/non-OA cartilage and mice experimental OA models. We also determined whether the LTA4H pathway was associated with cartilage degeneration and synovitis inflammation in OA mice models and human articular chondrocytes.
Results
We found that both LTA4H and LTB4 receptor (BLT1) were highly expressed in human and mice OA cartilage. Inhibition of LTA4H suppressed cartilage degeneration and synovitis in OA mice model. Furthermore, inhibition of LTA4H promoted cartilage regeneration by upregulating chondrogenic genes expression such as aggrecan (ACAN), collagen 2A1 (COL2A1), and SRY-Box transcription factor 9 (SOX9).
Conclusions
Our results indicate that the LTA4H pathway is a crucial regulator of OA pathogenesis and suggest that LTA4H could be a therapeutic target in combat OA.
Introduction
Osteoarthritis (OA) remains the leading cause of disability, morbidity, and mortality worldwide with no cure. 1 OA is characterized by cartilage degradation, osteophytes, subchondral bone sclerosis and synovitis, causing pain and reduced function of the affected joints. In the past few years, growing evidence has shown that non-resolved persistent synovitis is important in OA radiographic progression. 2 The proper shift from inflammatory to non-inflammatory status is essential to avoid tissue damage and the development of chronic inflammation. 3 Resolution of inflammation is an active process involving a molecular pathway switching from inflammatory eicosanoids and pro-resolving mediators. We demonstrated that promoting inflammation resolution by a pro-resolving lipid mediator, Resolvin D1, attenuated OA-like changes in a mouse model of post-traumatic OA. 4 Although the mechanism of persistent chronic inflammation in OA is incompletely understood, improper/inactivated resolution pathways may play an important role.
Inflammatory cascades in OA cartilage and synovial joints are associated with various cytokines and lipid mediators, mainly the eicosanoids and pro-inflammatory mediators. 5 Eicosanoid molecules produced mediators have been highlighted and linked to inflammatory and cellular metabolic responses in many studies, of which leukotrienes (LTs) are recognized as having a pivotal role.6 -10 Eicosanoids, including prostaglandins and LTs, are biologically active lipids implicated in various pathological processes, such as inflammation and cancer. 11 Leukotriene A4 hydrolase (LTA4H) is classically recognized as an epoxide hydrolase to generate leukotriene B4 (LTB4) from leukotriene A4 in response to inflammatory stimulus. 12 This enzyme is expressed mainly in neutrophils and other cell types, including endothelial cells and articular chondrocytes.13 -15 Leukotriene A4 is a pro-inflammatory lipid mediator generated from membrane phospholipid by the 5-lipoxygenase (5-LOX) enzymatic action at arachidonic acid (AA) during the initial phases of inflammation. LTB4 regulates inflammatory cytokine expression and recruitment of neutrophils primarily through its G protein-coupled receptor BLT1, which is only activated by LTB4.16,17 The involvement of LTB4 has been studied in various diseases, such as inflammatory arthritis and metabolic disorders.18,19 Given the central role of LTB4 in regulating inflammatory response mechanisms through recruitment and activation of leukocytes, modulation of the LTB4 pathway has been considered a new therapeutic approach in treating several diseases.
Previous studies have shown that an elevated level of LTB4 is found in the synovium, synovial fluid, articular cartilage, and subchondral bone from patients with OA.20,21 It was also revealed that increased expression of LTA4H occurs in human OA synovial fluid. 5 Although the activity of LTA4H and expression of BLT1 was found in human cultured chondrocytes,22,23 the relationship and mechanism of LTA4H and OA have not been well studied. In the current study, we firstly investigated the expression of LTA4H and BLT1 in OA cartilage. Based on these observations, we further studied the involvement of leukotriene in disease pathogenesis using an established OA model. We tested whether LTA4H inhibitor can affect OA development and progression using both in vivo and vitro models.
Materials and Methods
Study Approval
Animal studies were approved by the Institutional Animal Care and Use Committees and Institutional Biosafety Committees of Central South University, China (CSU; 2013-05). Six-week-old male C57BL/6 mice were purchased from the Animal Centre of Central South University (Changsha, Hunan, China). Collection of human OA tissues from discarded knee replacement surgeries was approved by the Queensland University of Technology (QUT) and the St Vincent Private Hospital Ethics Committees (Human ethics number: #1400001024). Informed consent was obtained from all human subjects.
Human Cartilage Specimen Preparation
Human cartilage was obtained from patients with OA (Kellgren-Lawrence grade 4) undergoing total knee replacement. Following our previous publication, the cartilage tissues collected from the relatively smooth region were classified into the grade 1 (G1, non-OA) group, and the cartilage collected from the severely damaged region were classified into the grade 4 cartilage (G4, OA) group. 24 Osteochondral plugs (including cartilage and subchondral bone) were taken from the tibial plateau region. All samples were prepared for histology and immunohistochemical analysis by washing in phosphate-buffered saline (PBS) and immediately fixed in 4% paraformaldehyde. The samples were then decalcified in 10% ethylenediaminetetraacetic acid (EDTA, pH = 7.2–7.4). The specimens were embedded in paraffin, and 5-µm-thick sections were obtained as described in our previous study. 25
Cell Culture and Bestatin Treatment In Vitro
Human chondrocyte cell line C28I2 was seeded into 6-well plates at a density of 1 × 106 cells/well and cultured in DMEM containing 10% fetal bovine serum (FBS) and 1% of penicillin and streptomycin (PS). At 90% confluence, cells were serum-starved for 24 hours and then were incubated in the presence or absence of 10 ng/ml interleukin-1β (IL-1β) or 50 μM bestatin with culture medium for 24 hours. Bestatin is a dipeptide immunomodulator that was discovered in the culture supernatant fraction from Streptomyces olivoreticuli, and it is a well-characterized inhibitor of LTA4H to attenuate LTB4 synthesis in many studies and particularly in animal models.26 -28
Cell Viability Test
Human chondrocyte cell line C28I2 was seeded into 96-well plates at a density of 5000 cells/well with culture medium. After 24 hours, cells were serum-starved for 24 hours and then were incubated in 0, 10, 50, 100, and 200 μM of bestatin separately. Cell viability test and cell count were performed according to our previous publication using CyQUANT proliferation assay (Life Technologies, Thermo Fisher). 29 Briefly, 100 μl of lysis buffer containing the CyQUANT GR dye was added to each well and incubated at 37 °C for 30 minutes. Fluorescence was measured using a fluorescence microplate reader with an excitation of 485 nm and emission of 530 nm.
Preparation of Bestatin
Bestatin was purchased from Cayman Chemical (Michigan, USA), and 2-hydroxypropyl-β-cyclodextrin (HPCD) and polyethylene glycol 400 (PEG-400) were obtained from Sigma Aldrich (Missouri, USA). The oral formulation for bestatin was prepared as previously described. 13 Briefly, 100 µl PEG-400 and 100 mg of bestatin were gently mixed before dissolving in a 20% (w/v) aqueous solution of HPCD. Then, the solution was mixed at room temperature to obtain a homogeneous mixture at 2 mg/ml concentration.
Induction of OA in Mice and Bestatin Treatment
Mice were group-housed (5 animals/cage) in ventilated cages in a temperature-controlled room on 12-hour light/dark cycles with ad libitum access to standard mouse food and water and routine veterinary assessment. Six-week-old male C57BL/6 mice were purchased following our previous publications.4,25 After that, all the animals were fed with the standard diet from the Animal Center (control diet) for at least 2 weeks as an observation period. Eight-week-old C57BL/6 mice (similar to other studies,30 -32 while 8-week-old of mice were considered as around 18-year-old in human age) 33 were randomly assigned and underwent destabilization of the medial meniscus (DMM) surgery to induce OA as previously described. 25 Briefly, OA was induced by dissecting the medial meniscus ligament to destabilize the medial meniscus in the knee joint of the hind limb on the right side. Mice in the sham group were surgically treated in which the ligament was visualized but not dissected. One week after experimental OA induction, mice were treated orally with bestatin 34 or vehicle (PEG-400 and HPCD). All experimental mice were then received twice-weekly oral bestatin after DMM surgery at a dose of 5 mg/kg for a total of 8 weeks. Bestatin (5 mg/kg) significantly decreased serum levels of LTB4 and BLT1 production in a mouse model and showed no difference to high dose of bestatin (15 mg/kg). 27 Mice were sacrificed 8 weeks after surgery for histological analysis. Bestatin is well absorbed in the gastrointestinal tract and is widely distributed in the body, including the kidney, liver, lung, spleen, and heart. It is metabolized in the liver, and its active metabolites are further metabolized and excreted in the urine.35,36 Overall, bestatin is rapidly absorbed and metabolized and is safe for in vivo experiments.
Histologic Evaluation of Knee Joint in Mice
After euthanasia, the knee joints of hind limbs were fixed in 4% paraformaldehyde, decalcified in 10% EDTA, embedded in paraffin, and sectioned for histological and immunohistochemical analysis. Sections were stained with Safranin-O (Saf-O)/Fast green to evaluate cartilage degradation and synovitis. The severity of OA was assessed in the medial compartment of the knee using a modified Mankin scoring system (0–14) as previously described. 25 The degree of synovitis was scored using a 0 to 6 scoring system that measured the thickness of the synovial lining cell layer on a scale of 0 to 3 (0 = 1–2 cells, 1 = 2–4 cells, 2 = 4–9 cells, and 3 = 10 or more cells) and cellular density in the synovial stroma on a scale of 0 to 3 (0 = normal cellularity, 1 = slightly increased cellularity, 2 = moderately increased cellularity, and 3 = greatly increased cellularity) as previously described. 25 Both the Mankin scoring and synovial inflammation scoring were carried out by 2 independent blinded readers (Indira and Antonia). The sum of both parameters was used for analysis. Synovitis scores obtained from all 4 quadrants (lateral femur, lateral tibia, medial femur, and medial tibia).
Immunohistochemistry
Immunohistochemistry was performed according to our previously published methods.37,38 Briefly, after dewaxing, sections were incubated with proteinase K for 20 minutes for antigen retrieval. The endogenous peroxidase activity was eliminated with 3% H2O2 in PBS for 20 minutes. Sections were then eliminated non-specific protein binding by incubation with 1% Bovine serum albumin for 1 hour at room temperature. Sections were incubated overnight at 4 °C with antibodies against LTA4H (Abcam, Melbourne, VIC, Australia; dilution 1:250) and LTB4 receptor (BLT1, LSBio, Sapphire Bioscience, Redfern, NSW, Australia; dilution 1:50) followed by incubation with anti-rabbit or anti-rat secondary antibody (1:5000 dilution; LI-COR Biosciences, Nebraska, USA) and visualization with 3,3-diaminobenzidine chromogen. To exclude antigen-independent staining, negative controls for which the incubation step with the primary antibody was omitted were examined. The percentage of positive cells was determined by counting the number of immunostained cells and dividing by the total number of cells (100 cells per sample) visualized by a hematoxylin counterstain using ImageJ (National Institute of Health, Bethesda, BA, USA).
Quantitative Real-Time PCR
Total RNA was extracted with TRIzol, and cDNA was synthesized using the SensiFAST cDNA Synthesis Kit according to the manufacturer’s protocol. SYBR Green detection chemistry was used on the QuantStudio Real-Time PCR system (Applied Biosystems, Thermo Scientific, Scoresby, VIC, Australia). Quantitative measurements of all primers used in this study were determined using (2−ΔΔ Ct) method, and GAPDH expression was used as the internal control,27,39 as described previously by our group.37,40 -42
Statistical Analysis and Sample Size Justification
All experiments were performed in triplicate using different samples: 7 donors of human knee cartilage for histologic evaluation; 5 donors for immunohistochemistry; and 6 mice per group were used for animal study. Statistical difference of the results was tested using the unpaired Student’s t-test comparison of 2 variables. One-way analysis of variance followed by post hoc test was used for comparison of more than 2 variables. All analyses were performed using GraphPad Prism 7, and P < 0.05 is significant. All data are presented as mean ± SD. Sample size for each experiment was determined using G power program (https://clincalc.com/stats/samplesize.aspx). Eighty percent power and an alpha level of 0.05 were used for the calculation. Given concerns about the ability to complete the experiments, we increased the sample sizes in the experiments above.
Results
Increased LTA4H and BLT1 in OA Cartilage
To study the leukotriene A4 pathway in the articular cartilage, we first divided the cohort of OA patients into 2 groups based on a histomorphometry analysis for the semi-quantitative estimation of OA extent on Safranin-O-stained cartilage sections. As exemplarily shown in Fig. 1A for intact smooth cartilage surface (grade 1, G1) and severely degenerated and fibrillated OA regions (grade 4, G4), significant cartilage damage associated with elevations of Mankin score (12.14 ± 1.574, n = 7) were observed in G4 OA sites; compared with G1 group (Mankin score = 3.286 ± 0.951; n = 7) ( Fig. 1B ).
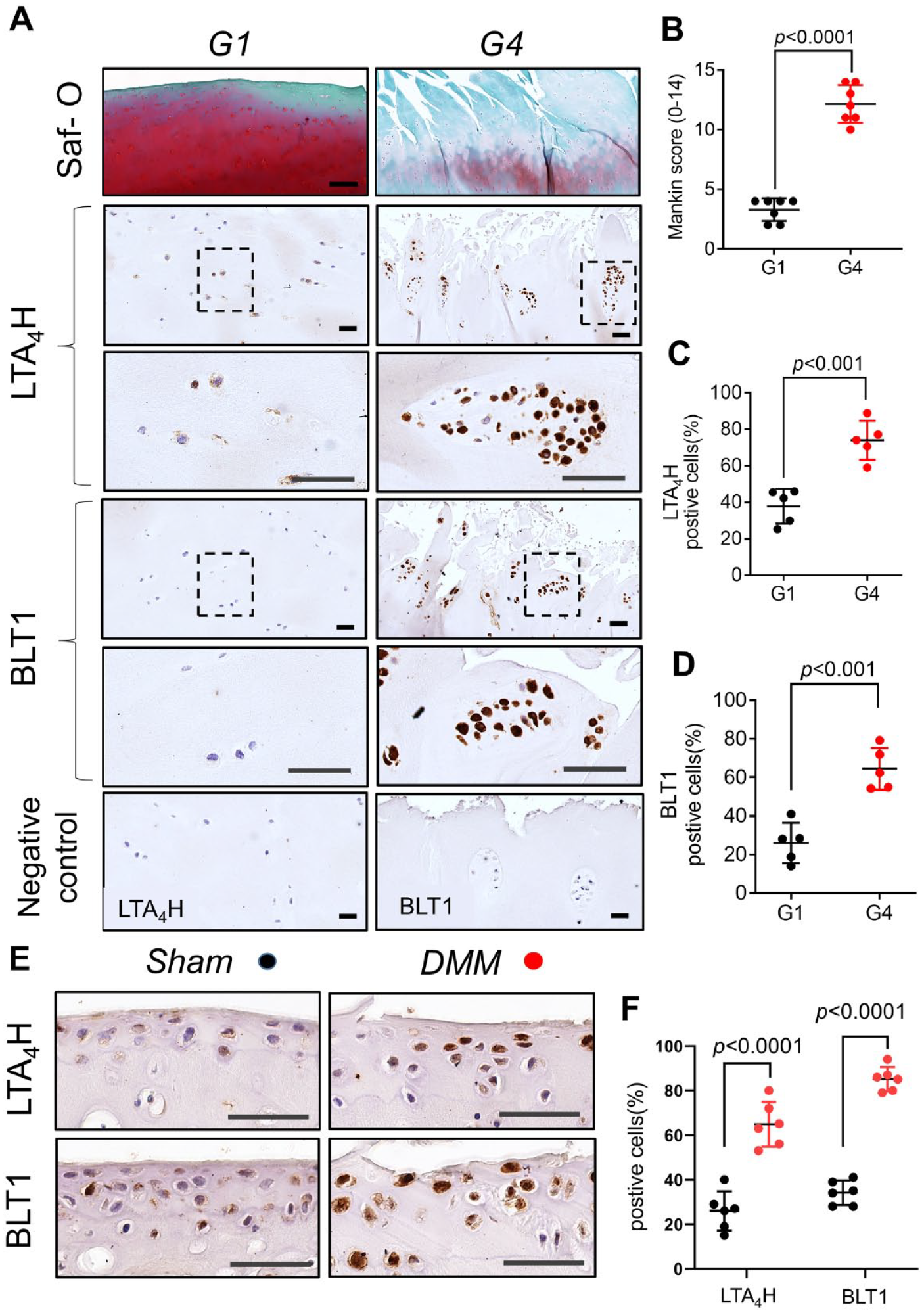
Increased leukotriene A4 hydrolase (LTA4H) and leukotriene B4 receptor 1 (BLT1) in osteoarthritis cartilage.
We then examined LTA4H and BLT1 expression in OA cartilage of humans and mice DMM OA model. The percentage of LTA4H immunostained chondrocytes was markedly elevated in OA-affected G4 cartilage compared with undamaged G1 regions of patient-matched OA cartilages ( Fig. 1A and C ). BLT1 levels were also significantly increased in severe G4 versus mild G1 cartilage ( Fig. 1A and D ). To further ascertain whether leukotriene plays a role in OA pathogenesis, we examined LTA4H and BLT1 expression in OA cartilage of mice. Similar results were found in experimental OA mice, in which LTA4H and BLT1 were significantly increased in knee cartilage from DMM mice compared to Sham surgery mice ( Fig. 1E and F ). These results indicated that the expression of LTA4H and BLT1 is in accordance with the disease severity.
Inhibition of LTA4H Level Suppressed Articular Cartilage Degradation In Vivo
The articular cartilage of mice with DMM-induced OA exhibited pathological OA changes characterized by Safranin-O loss and detectable cartilage roughness ( Fig. 2A ), with a Modified Mankin score of 11.833 ± 1.169 ( Fig. 2B ). In contrast, the knee joint of sham mice exhibited no visible changes on the articular cartilages with a significant lower Mankin score (0.857 ± 0.690) than that in mice with DMM-induced OA ( Fig. 2A and B ). Bestatin is a well-characterized inhibitor of LTA4H and was shown to attenuate LTB4 synthesis in several previous studies.27,43 Inhibition of LTA4H level in mice with DMM-induced OA suppressed OA progression with a lower Mankin score (8.000 ± 1.095) compared with DMM-induced OA mice with vehicle ( Fig. 2A and B ). These results suggested that increased leukotriene may be one of the reasons causing OA as inhibition of which could suppress OA progress in DMM mice.
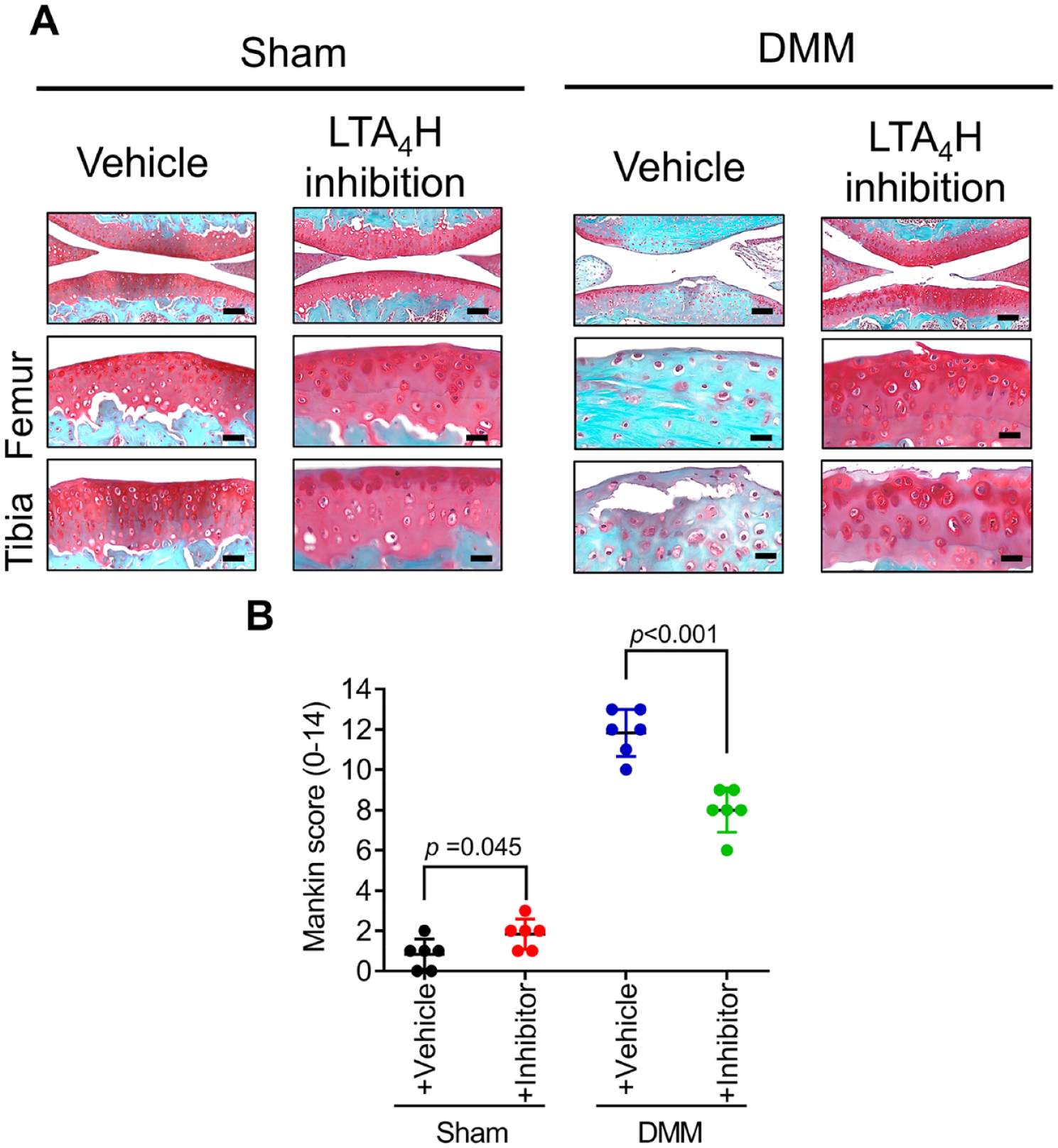
Inhibition of LTA4H level suppressed articular cartilage degradation in a mouse model of experimental osteoarthritis.
Inhibition of LTA4H Level Reduced Synovitis in OA Mice
Mice with DMM-induced OA also revealed an increased influx of synoviocytes and lining layer thickening in synovium compared with the respective control mice ( Fig. 3A ). The synovitis scores reiterated these observations with a significantly higher score in DMM-induced OA mice (Synovitis score: 4.417 ± 0.540 compared with 1.208 ± 0.485) ( Fig. 3B ). The role of leukotriene in synovitis in mice was examined by oral administration of LTA4H inhibitor-bestatin. Bestatin treatment reduced the thickness of the synovial lining cell layer and cellular density in the synovial stroma in mice with DMM (Synovitis score: 2.917 ± 0.585) compared with vehicle-treated mice with DMM (Synovitis score: 4.417 ± 0.540) ( Fig. 3A and B ). However, oral administration of bestatin had no significant effect on synovitis in mice without DMM ( Fig. 3A and B ). These results indicated that bestatin treatment also reduced synovitis.
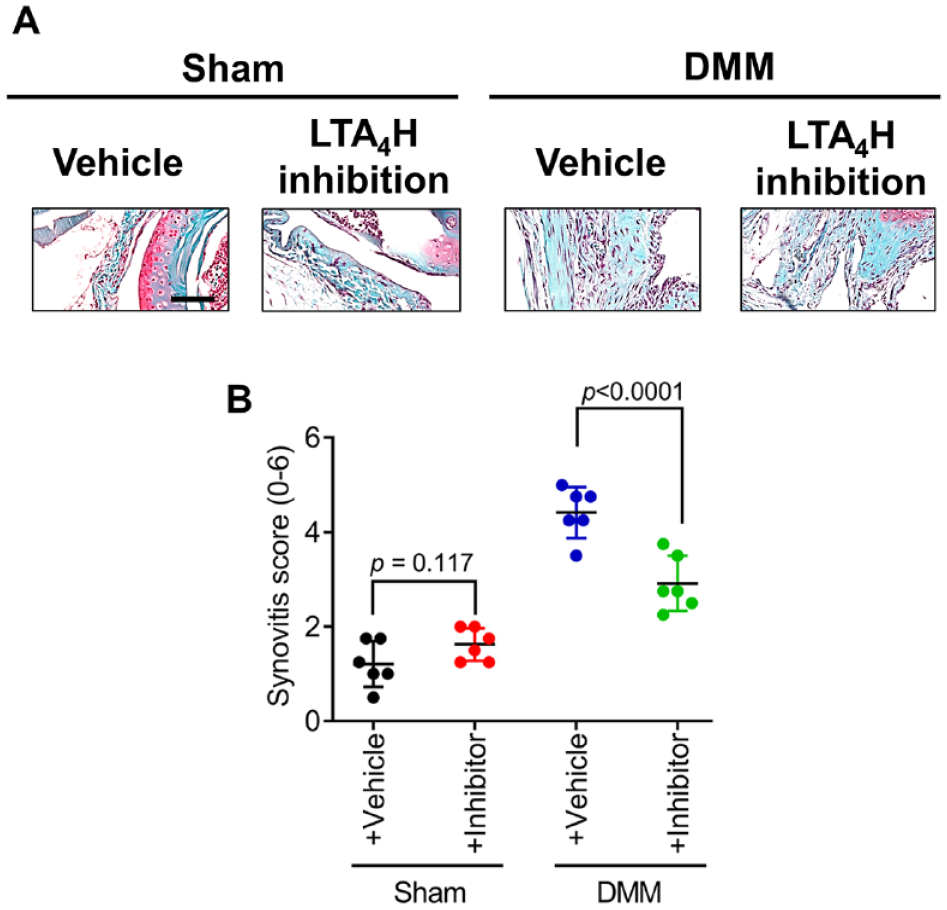
Inhibition of LTA4H level reduced synovitis in osteoarthritis mice.
In Vitro Inhibition of LTA4H Significantly Affect Gene Expression Profile in Chondrocytes
We further tested the effect of LTA4H inhibitor on C28I2 chondrocytes cell line in vitro. To test whether bestatin plays a direct role in chondrocytes, we validated the potential effect of bestatin by gene expression profile analysis. We first tested cell viability under different concentrations of bestatin (
Fig. 4A
and
B
). Our results indicated that 0 to 200 μM of bestatin treatment showed no toxic effect on chondrocytes for 24 hours. Next, we tested the effects of 50 μM bestatin treatment on chondrocytes phenotype. Bestatin treatment significantly upregulated COL2A1 and inhibited SOX9 expression in chondrocytes in the absence of IL-1β (
Fig. 4D
and
F
). No significant effects of bestatin on the expression of aggrecan (ACAN), Runt-related transcription factor 2 (RUNX2), a disintegrin and metalloproteinase with thrombospondin motifs 5 (ADAMTS5) and MMP13 were observed
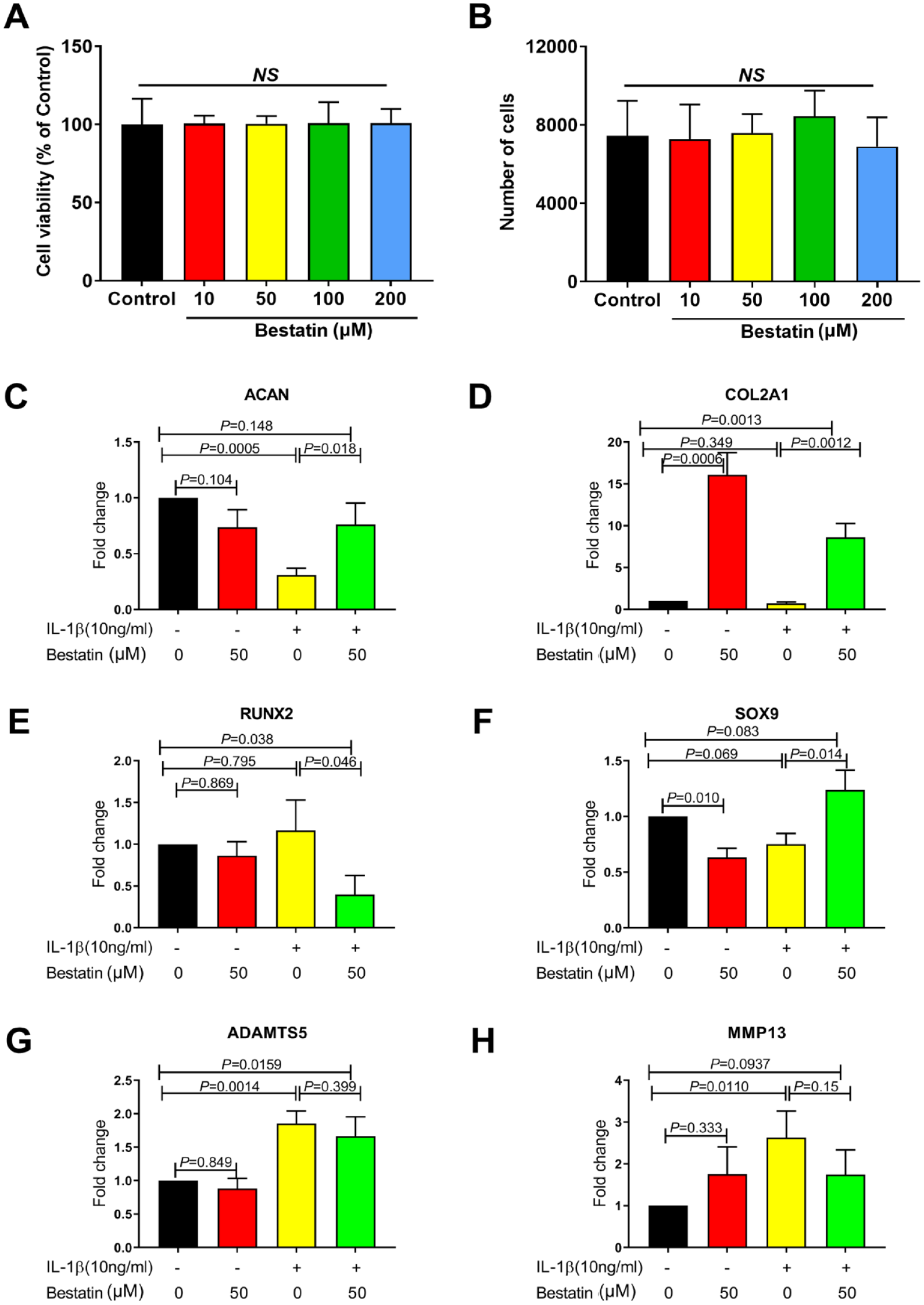
In vitro inhibition of LTA4H significantly affect gene expression profile in chondrocytes. Cell viability
Discussion
Growing evidence shows that the resolution of inflammation represents a critical therapeutic target in OA treatment. 44 The involvement of lipid mediators in inflammatory events has been brought into focus with several studies pointing to its role in a multitude of cellular responses, including cell growth and death and inflammation via receptor-mediated pathways. 45 A constitutive low-grade inflammatory response accompanies the OA, and the leukotriene signalling is now considered a potent chemoattractant agent related to acute inflammatory responses and maintenance of chronic inflammation.46,47 In this study, we examined the role of the leukotriene pathway in the development of OA. We found that the level of LTA4H and LTB4 receptor BLT1 was increased in articular cartilage from OA patients and mice with post-traumatic OA. Moreover, inhibition of LTA4H decreased cartilage tissue damage and synovial inflammation. These results indicate that upregulation of LTB4 could be an important event in OA pathogenesis, and its upregulation may be involved in the mechanisms that trigger both cartilage degeneration and synovial inflammation.
First, the production of LTA4H and LTB4 in OA has been previously shown. Paredes et al. have shown that human OA subchondral osteoblasts constitutively produce LTB4, whereas He et al. and Lascelles et al. have demonstrated a high amount of LTB4 in the synovial tissue from human and animal models of OA, respectively.20,48,49 Overexpression of LTA4H in human OA synovial fluid was also observed, and treatments with chemical inhibitors of 5-lipoxygenase- activating protein/5-lipoxygenase (FLAP/5-LOX) were shown to inhibit the severity of OA.5,50,51 Our experiment found that human OA cartilage/chondrocytes expressed increased LTA4H and LTB4 receptor BLT1. These findings are in corroboration with a study performed by Martel-Pelletier et al. that revealed the presence of FLAP and 5-LOX (as determinants of LTB4 synthetic capacity) in chondrocytes. 51 Besides, Hansen et al. found a decreased level of BLT1 in OA chondrocytes. This might be because the samples they compared were cultured cells, which showed a significant variation between cartilage and cultured chondrocytes in their study. 22 In the mouse model of post-traumatic OA, we observed significant changes in cartilage damage and synovitis and correlated with an increase in the expression of LTA4H and BLT1. Similarly, Attur et al. showed that the level of lipoxygenase-derived products such as LTB4 was found to be significantly increased in OA cartilage. 52 Although LTB4 expresses mainly in inflammatory and immune cells, including macrophages, granulocytes, mast cells, and T and B lymphocytes, our study and other studies all demonstrate that the inflammatory leukotrienes are also produced by human OA chondrocytes. 52 LTB4 is stimulated by diverse signalling molecules, including pro-inflammatory cytokines, such as tumour necrosis factor-α (TNF-α) and IL-1β, and itself. 12 The LTB4 synthesis in OA chondrocytes is increased by growth factors such as transforming growth factor β1 (TGFβ1) and 1,25(OH)2D3 in the inflammatory response. 51 Thus, LTA4H and LTB4 in OA tissues could be considered an important event in OA pathogenesis and pathophysiology.
The inflammatory process in diseases is due to an imbalance between pro-inflammatory and anti-inflammatory molecules and a deficiency of bioactive lipid mediators in the resolution phase of inflammation. 17 It has been reported that LTB4 contributes to persistent inflammation in arthritis by delivering IL-1 into the joint through neutrophils. 53 More recent studies have shown that BLT1−/− mice were protected entirely from collagen-induced arthritis development and systemic insulin resistance in diet-induced obese mice.18,19 Our previous study has shown that intra-articular treatment of pro-resolving lipid mediator resolvin D1 diminishes the progression of OA in mice. 4 Leukotriene has also been shown to increase the production of pro-inflammatory cytokines, IL-1β, and TNF-α in dose-dependent human OA synovial membrane.48,54 Modulating the secretion of lipid mediators may therefore attenuate the severity of the disease. In this study, we considered the LTA4H as the candidate to investigate the effect of leukotriene activity on OA progression since it is the essential enzyme that catalyzes the distal step in LTB4 generation. Blocking LTA4H by bestatin in vivo has shown protective and preventive effects in various diseases such as pulmonary hypertension and oesophagal adenocarcinomas.13,26 Another advantage of bestatin is the low toxicity. 26 Therefore, we used the LTA4H inhibitor bestatin and tested if inhibiting LTB4 production has beneficial effects in mice with post-traumatic OA. In our study, bestatin significantly inhibited the presence of LTA4H and BLT1 production in articular cartilage of mice with DMM-induced OA. Our results indicate that LTA4H inhibition decreases cartilage degradation and synovitis in a mouse model of post-traumatic OA. This decrease in synovial inflammation could be because LTB4 is a critical mediator in the cascade of complement, lipids, cytokines, and chemokines. In BLT1 knock-out mice, LTB4 has shown crucial roles in T helper 2 (Th2)-type cytokine interleukin-13 (IL-13) production from lung T cells and recruitment of antigen-specific effector CD8+ T cells. 55 Therefore, blockade of the LTB4-BLT1 pathway might be a promising therapeutic strategy in OA patients. However, clinical trials to date have shown that compared with traditional disease-modifying antirheumatic drugs, oral LTB4 receptor and 5-LOX inhibitors have little clinical benefit 56 ; however, whether or not this is the case with OA patients needs further investigation. The limitations of this study were that our results couldn’t reveal that the mechanism-of-action of bestatin is via attenuated synovitis or reduced synovial-driven cartilage degeneration. Further experimental design was needed to investigate the role of bestatin, synovitis, and cartilage degeneration. Another limitation was that DAPI staining for nuclei detection was no used in our study. The quantification of the immunohistochemistry (IHC) staining in this study was calculated by counting the number of positively stained cells per 100 cells per slide visualized by hematoxylin counterstain to avoid difference of cellularity between samples. This is also one of the well-established methods for IHC quantification in several publications.57 -60 We have been trying to quantify by using staining intensity or IDO value. However, because chondrocytes only accounts for around 5% of the cartilage area, it would bring us much background signal for quantification.
In conclusion, our study applied oral LTB4 receptor inhibitor bestatin for feeding mice after OA surgery. Reduced cartilage damage and synovium inflammation were observed in mice joints after 8 weeks, indicating that oral bestatin uptake in early OA might be a potential drug for OA therapy targeting Leukotriene A4 hydrolase.
Footnotes
Author Contributions
Conceptualization: I.P., X.M., R.C., Y.X., and Y.W.; methodology and formal analysis: X.W., I.P., and A.S.; drafting of the article: A.S., X.W., and I.P.; review and editing: all authors; supervision: I.P.; acquisition of funding: I.P., X.M. and Y.W. All authors have read and agreed to the published version of the manuscript.
Acknowledgments and Funding
The authors thank the team of the central animal facility of the Central South University for expert animal care. The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was funded by Hunan Provincial Science and Technology Department, China (2018JJ2579, 2015JC3034) and National Natural Science Foundation of China (81371997), and The Prince Charles Hospital Research Foundation fellowship (RF-01) and NHMRC Investigator Fellowship (APP1176298) for Indira Prasadam.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Animal studies were approved by the Institutional Animal Care and Use Committees and Institutional Biosafety Committees of Central South University, China (CSU; 2013-05). Collection of OA tissues from discarded knee replacement surgeries were approved by the Queensland University of Technology and the St Vincent Private Hospital Ethics Committees (Human ethics number: #1400001024).
