Abstract
Objective
Advances in research have shown that the subchondral bone plays an important role in the propagation of cartilage loss and progression of osteoarthritis (OA), but whether the subchondral bone changes precede or lead to articular cartilage loss remains debatable. In order to elucidate the subchondral bone and cartilage changes that occur in early OA, an experiment using anterior cruciate ligament transection (ACLT) induced posttraumatic OA model of the rat knee was conducted.
Design
Forty-two Sprague Dawley rats were divided into 2 groups: the ACLT group and the nonoperated control group. Surgery was conducted on the ACLT group, and subsequently rats from both groups were sacrificed at 1, 2, and 3 weeks postsurgery. Subchondral bone was evaluated using a high-resolution peripheral quantitative computed tomography scanner, while cartilage was histologically evaluated and scored.
Results
A significant reduction in the subchondral trabecular bone thickness and spacing was found as early as 1 week postsurgery in ACLT rats compared with the nonoperated control. This was subsequently followed by a reduction in bone mineral density and bone fractional volume at week 2, and finally a decrease in the trabecular number at week 3. These changes occurred together with cartilage degeneration as reflected by an increasing Mankin score over all 3 weeks.
Conclusions
Significant changes in subchondral bone occur very early in OA concurrent with surface articular cartilage degenerative change suggest that factors affecting bone remodeling and resorption together with cartilage matrix degradation occur very early in the disease.
Introduction
Osteoarthritis (OA) have been historically considered a “wear and tear” disease of the joint that is hallmarked by articular cartilage degeneration as well as subchondral bone sclerosis, osteophyte formation, synovial inflammation, and deterioration of other surrounding structures. 1 Various risk factors are thought to give rise to OA through an altered mechanical loading across the joint. 2 This altered mechanotransduction initiates a cascade of biological changes that upsets the normal bone-cartilage homeostasis 3 and identification of a common pathway in early disease progression may offer therapeutic potential.
The theory of altered mechanotransduction is closely linked to the role of the subchondral bone as the natural absorber and dampener of mechanical stresses that effect the cartilage. Radin et al.4,5 were the first to suggest the possible role of the subchondral bone in OA initiation and progression. The subchondral bone is an effective shock absorber and maintenance of joint integrity depends on the load adaptation of cartilage and bone. In areas of aberrant mechanical loading, it is hypothesized that microfractures occur in the subchondral bone, which in turn will initiate bone remodeling in an effort to repair the damage.6,7 Bone remodeling leads to the formation of new, less-mineralized bone, which is softer and more liable to deformation when loaded. It is thought that this results in the changes in transmission of mechanical strain to the cartilage, disturbing the structural support of the cartilage and therefore resulting in cartilage degradation. 8
The structural changes in established OA have long been defined through the analysis of specimens obtained during joint replacement surgeries. Cartilage fissuring and eburnation, subchondral bone thickening and sclerosis, subchondral bone cysts and osteophyte formation, as well as synovial hypertrophy are all changes of moderate to advanced OA and cannot be used to extrapolate the changes that occur in early OA. In humans, early stages of the disease are rarely symptomatic and goes undetected until much later, making the study of early OA challenging. Studies that examine sections of subchondral bone with intact overlying cartilage from human advanced OA samples also may not accurately represent early OA bone and cartilage change. 9 One established way of studying the initiation and early progression of the disease is via the utilization of animal models of OA.
A widely used model of animal OA is through surgically induced instability of the knee. 10 This model is characterized by an acute injury to the joint that causes mechanical instability, resulting in OA. Many types of operations on various animals have been done including cruciate or collateral ligament transections and partial or total meniscectomies on dogs, goats, rabbits, and rodents. Kamekura et al. have proposed 4 models of surgically induced instability knee OA in the rodent and have shown that the anterior cruciate ligament transection (ACLT) model is the mildest form with the slowest rate of disease advancement. 11 In his study, he showed that mild changes in the articular cartilage was detectable at 2 to 4 weeks postoperatively and cartilage destruction in the ACLT model rarely reached the middle layer even at 8 weeks. Osteophyte formation was hardly visible until 8 weeks after surgery. Another widely used mild form of surgically destabilized model is the destabilization of medial meniscus model where OA is surgically induced by transection of the medial meniscotibial ligament. In this model, cartilage legions were observed as early as 2 weeks after surgery and cartilaginous osteophytes noted in the medial tibial plateau 2 weeks after surgery, which ossifies at 4 weeks. 12
Many studies focused on articular cartilage degeneration and failed to characterize the accompanying subchondral bone change. There still exists a knowledge gap as to whether tissue changes in the subchondral bone precede, accompany, or follow cartilage damage at the onset and very early stages in OA. Therefore, we hypothesized that subchondral bone changes occur earlier or in parallel with cartilage degeneration in early OA. Traditionally, histopathological analysis has been the gold standard for assessing joint structural changes and quantification of subchondral bone changes were evaluated via bone histomorphometry. Current advances in high-resolution micro-computed tomography systems has allowed for the direct measurement of 3-dimensional (3D) bone architecture without relying on stereologic models 13 and is gaining popularity over histomorphometry in quantification of bone microstructural changes. In an effort to characterize initial tissue-level changes in early OA, the ACLT model, which is the mildest form of established rat knee model of posttraumatic OA, was adopted in this experiment. Quantitative changes in subchondral bone microarchitecture were evaluated longitudinally and cross-sectionally using a high-resolution peripheral quantitative computed tomography (HR-pQCT) system, and cartilage degradation was assessed via histology. These changes were studied across a short time frame to closely evaluate the minute changes that occur in early OA versus an unoperated control.
Methods
Animals
Forty-two 12-week-old Sprague-Dawley (SD) male rats weighing between 300 and 400 g obtained from the Animal Experimental Unit (AEU), Faculty of Medicine (FOM) University of Malaya (UM), were used in this study. The rodents were maintained in open cages in the animal house in AEU, FOM UM with water and food provided ad libitum and on a 12-hour dark-light cycle. All animal experiments were conducted according to the guidelines for animal handling and welfare set by the University of Malaya. Approval to conduct this study was applied thorough the FOM Institutional Animal Care and Use Committee (IACUC) (Ethics Reference no. 2015-181201/ORTHO/R/NANF).
Baseline bone microstructural analysis of the right knees were conducted for all the animals at 12 weeks of age via HR-pQCT (XtremeCT, Scanco Medical AG, Brüttisellen, Switzerland). The animals were subsequently randomized into 2 groups, the ACLT group (n = 21) and the control group (n = 21). All animals in the ACLT group then underwent surgery to induce OA on their right knees. All animals were allowed normal activity, and nonprotected weight bearing on the ACLT limbs was observed immediately postoperation. Repeated HR-pQCT analysis of the right knees of 7 (n = 7) animals from each group were done at week 1, week 2, and week 3 postsurgery followed directly by euthanization and harvest of their right knees for histopathological analysis.
Surgical Induction of Osteoarthritis by Anterior Cruciate Ligament Transection
All ACLT procedure was conducted under aseptic conditions by a single surgeon. Animals were anaesthetized prior to surgery with an intramuscular injection of ketamine (50 mg/kg) (Vetoquinol, Lure Cedex, France) and xylazine (5 mg/kg) (Vetoquinol, Lure Cedex, France). The right knees were shaved and prepared using chlorhexidine. A 1-cm midline incision was made over the right anterior knee and the skin mobilized to expose the patellar tendon. A medial parapatellar approach was done to enter the knee joint with the patella subluxed laterally to expose the ACL. The ACL was carefully sectioned using micro-scissors with the knee in full flexion under 4× magnification with a loupe. Care was taken not to damage the surrounding cartilage or other structures. Complete transection of the ACL was confirmed by manually testing the joint for anterior laxity and the joint was washed with sterile saline. The peripatellar capsular incision and skin incision were then closed using 5-0 Vicryl undyed braided absorbable sutures (Ethicon, Edinburgh, UK). Immediately postsurgery and the following 2 days postoperatively, all animals were given subcutaneous injections of trimethoprim-sulfamethoxazole (Vetoquinol, Lure Cedex, France) and meloxicam (Vetoquinol, Lure Cedex, France) as antibiotics and analgesia, and allowed no restriction to weight bearing.
Bone Microstructural Analysis with High-Resolution Peripheral Quantitative Computed Tomography
All animals underwent in vivo bone microstructural analysis of the right knee using a HR-pQCT at 12 weeks old prior to surgery and subsequently prior to necropsy. Animals were sedated with an intramuscular injection of ketamine (50 mg/kg) (Vetoquinol, Lure Cedex, France) and xylazine (5 mg/kg) (Vetoquinol, Lure Cedex, France) and placed within the scanner in a modified plastic holder with their right knees aligned with the vertical axis of the scanner to obtain sagittal slices of the right knees.
HR-pQCT imaging of the knee joint was performed with an X-ray tube potential of 60 kVp and intensity of 900 µA with an isotropic voxel resolution of 41 µm and 200 ms integration time ( Fig. 1 ). The measuring time for each specimen took approximately 15 minutes. Acquired images were filtered using a Gaussian filter (σ = 0.8, support = 1) and the threshold for segmentation was determined and set by comparing 2-dimensional images from the original and segmented images for some of the acquired scans to ensure that the extracted bone is a good representation of the actual structure. Manual contouring of the tibial subchondral trabecular bone (trabecular bone in the tibia epiphysis) was done on each slice ( Fig. 2 ) and subsequently 3D scans were generated. The volumetric bone mineral density (vBMD, mg HA/ccm), bone volume fraction (BV/TV, %), trabecular number (Tb.N, 1/mm), trabecular thickness (Tb.Th, mm), and trabecular separation (Tb.Sp, mm) were derived from the 3D images according to protocol. 14
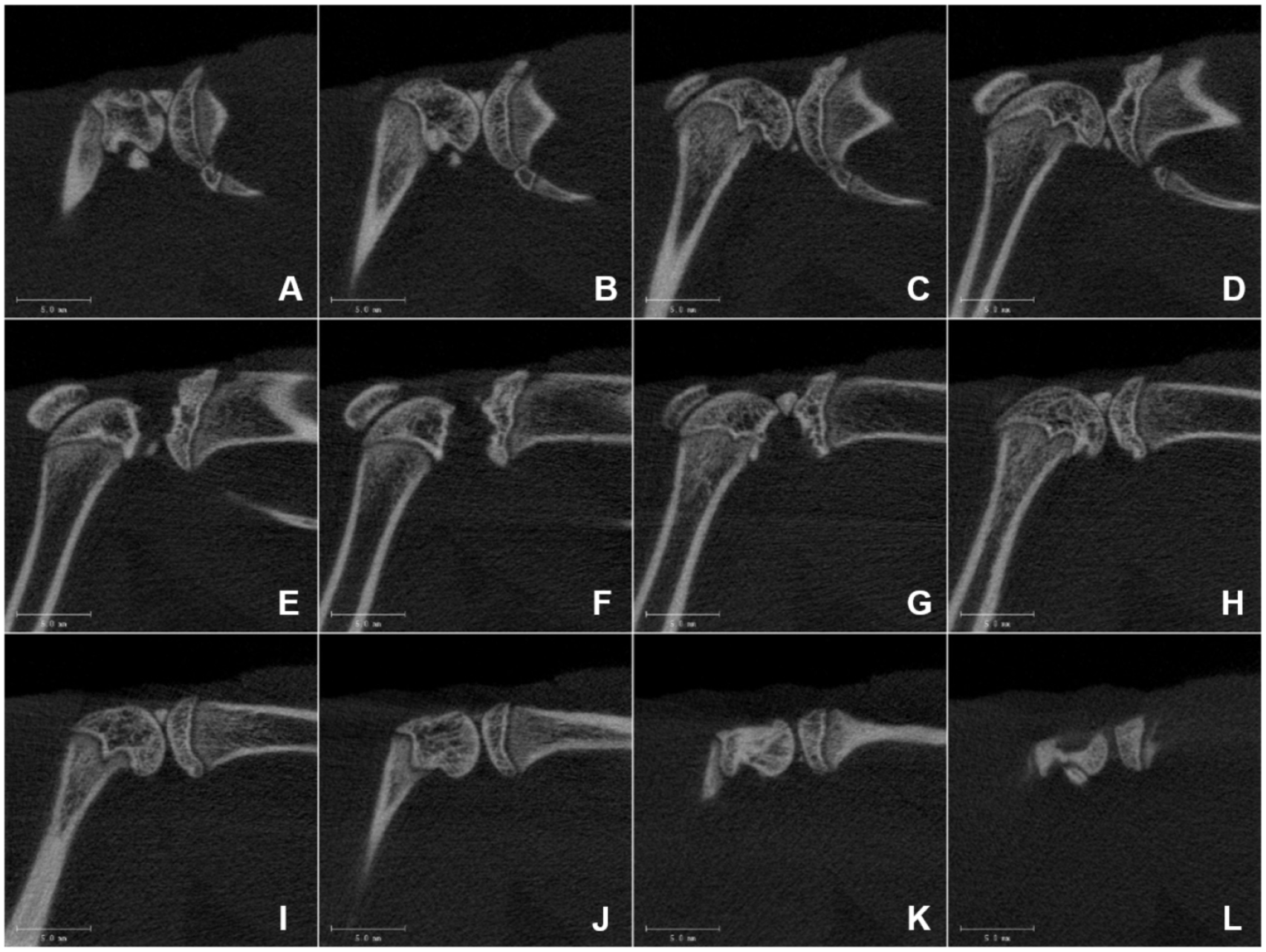
Overview of high-resolution peripheral quantitative computed tomography (HR-pQCT) imaging acquired from a right rat knee. Knees are aligned with the vertical axis of the scanner to obtain sagittal slices cut from lateral to medial (from
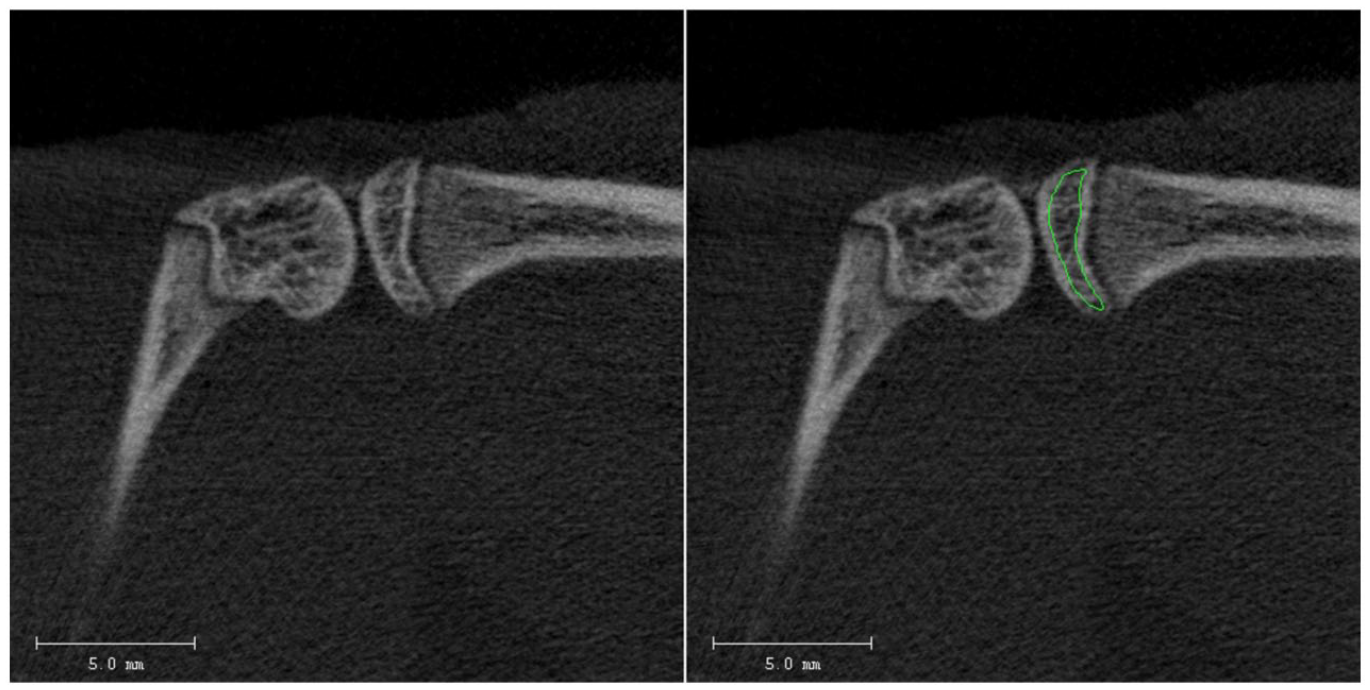
Manual contouring (green line) of the tibial subchondral trabecular bone is performed on each cut and 3D scans are generated. Scale bar = 5 mm.
Tissue Preparation and Histopathological Analysis
All animals at euthanization underwent dissection to obtain the right knee joints. The joints were dissected free of muscles and fixed in 10% formaldehyde (Leica Biosystems, Nussloch, Germany) for 3 days and then decalcified in 10% formic acid (Leica Biosystems, Nussloch, Germany) for 5 days. Subsequently, the tibia was isolated and cut into 2 equal halves in the coronal plane along the medial collateral ligament. Both anterior and posterior halves of the tibia were dehydrated in ethanol series immersion and cleared in toluene and embedded in a single paraffin block with the cut planes facing down. Three 5-µm sections were cut from each block at 100 µm steps to obtain 3 sections from each half (anterior and posterior) of the tibia. Serial sections were mounted on glass slides and stained with Safranin-O and fast green counter stain.
Sections were examined under a light microscope (Olympus, Tokyo, Japan) and histopathological scoring of the tibial plateau performed according to the modified Mankin scoring system15,16 by a single histopathologist blinded to the group identities. Histopathologic scoring was performed on the 3 most severely affected sections and the values averaged across the 3 scored sections.
Statistical Analysis
All statistical analyses were performed using SPSS (v23, IBM, Armonk, NY). Normality testing were assessed by box plots, skewness and kurtosis, and the Shapiro-Wilk test. Equal variance assumptions were evaluated with Levene’s test. For statistical analysis of HR-pQCT data, baseline bone scans were taken as covariates. After adjustment for baseline covariates, the differences in all variables as a function of treatment group and time point were separately assessed using 2-way analysis of covariance (ANCOVA). Two-way ANCOVA was the chosen method of analysis as it enables the elimination of the effect of the confounding variable (baseline bone scan results) that may be influencing the results. Two-way analysis of variance (ANOVA) was used to assess the modified Mankin scores. P values lower than 0.05 were considered as significant.
Results
Quantitative Changes to Tibia Subchondral Trabecular Bone and Articular Cartilage
Normality testing and equal variance assumptions were met for all data sets. Mean comparison was done using Duncan test for each dependent variable separately at 0.05 level. Summaries of the results for all quantitative variables are provided in Table 1 and Figure 3 . Initial changes in the subchondral bone was observed as early as 1 week post-ACLT with a significant reduction in the Tb.Th (P < 0.005) and increase in the Tb.Sp (P < 0.05 at week 1, P < 0.005 at week 2 and week 3) across all 3 time points compared to control. Longitudinally, increase in the Tb.Sp is significant in the week 3 ACLT group compared to week 1 and week 2, with no significant longitudinal change observed in the control group. The ACLT and control groups demonstrated no significant longitudinal change in Tb.Th.
Subchondral Trabecular Bone Microstructure Parameters in ACLT versus Control Groups across the Time Points (Mean Values ± Standard Error) After Adjustment for Baseline Covariates.
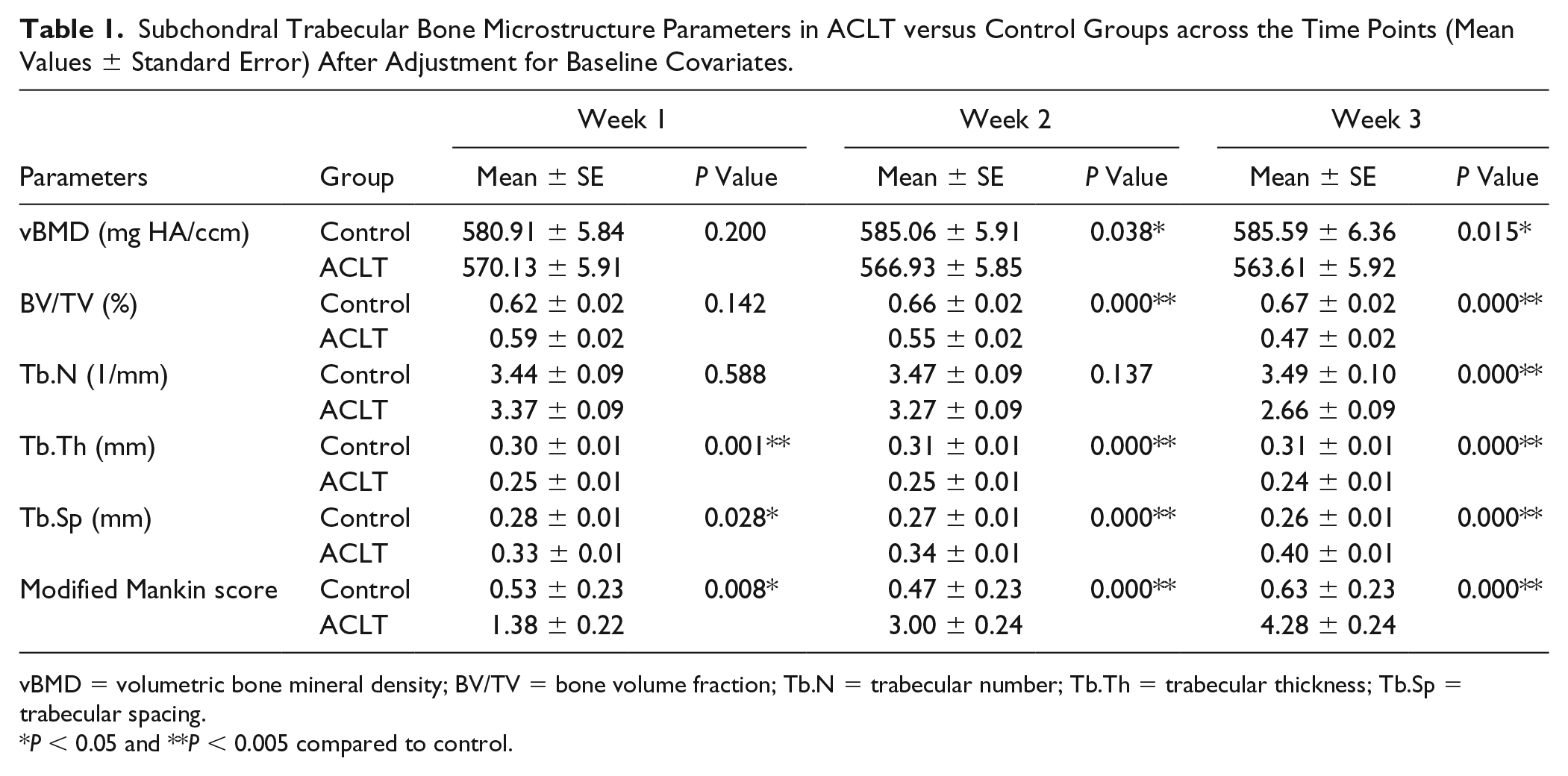
vBMD = volumetric bone mineral density; BV/TV = bone volume fraction; Tb.N = trabecular number; Tb.Th = trabecular thickness; Tb.Sp = trabecular spacing.
P < 0.05 and **P < 0.005 compared to control.
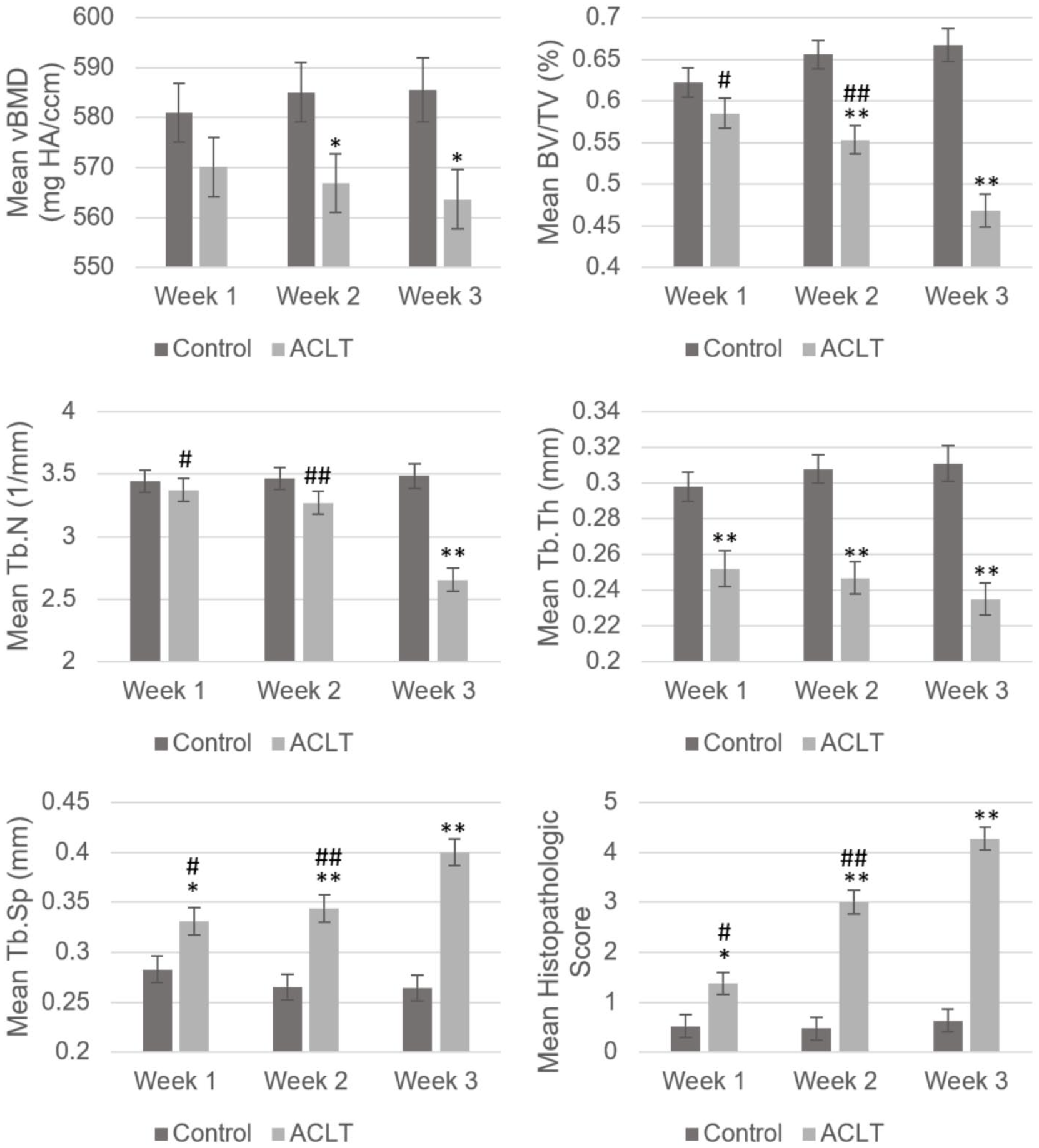
Comparison of subchondral trabecular bone microstructure parameters in ACLT and control groups across the time points. Error bars represent ± 1 standard error of the mean. ACLT = anterior cruciate ligament transection; vBMD = volumetric bone mineral density; BV/TV = bone volume fraction; Tb.N = trabecular number; Tb.Th = trabecular thickness; Tb.Sp = trabecular spacing; *P < 0.05 and **P < 0.005 compared to control; #P < 0.05 week 1 versus week 3 ACLT; ##P < 0.05 week 2 versus week 3 ACLT.
Subsequently, a significant reduction in the vBMD (P < 0.05) and BV/TV (P < 0.005) was seen at 2 and 3 weeks post-ACLT against control. Longitudinally, there were no significant change in the vBMD across time for both groups. There, however, was a significant reduction in BV/TV of week 3 ACLT rats compared to week 1 and week 2, but no significant change in BV/TV across time in the control group.
Last, a significant reduction of Tb.N was observed at week 3 post-ACLT versus control (P < 0.005), which was also significant longitudinally at week 3 compared to week 1 and week 2 in the ACLT group.
Analysis of modified Mankin scores exhibit a significant increase in ACLT group scores compared to control across all time points (P < 0.05 at week 1, P < 0.005 at week 2 and week 3), with the increase being significant longitudinally as well for the ACLT group.
Qualitative Changes to Tibia Subchondral Trabecular Bone and Articular Cartilage
Visually, it was hard to discern any objective changes among the ACLT group scans over the short time frame. Of note, there is an appearance of increased radiolucency in the medial tibial epiphysis most obvious in ACLT week 3 scans (5 out of 7 subjects) ( Fig. 4 ). Otherwise, no hallmarks of established OA such as subchondral sclerosis, osteophyte formation, or subchondral cysts were seen.
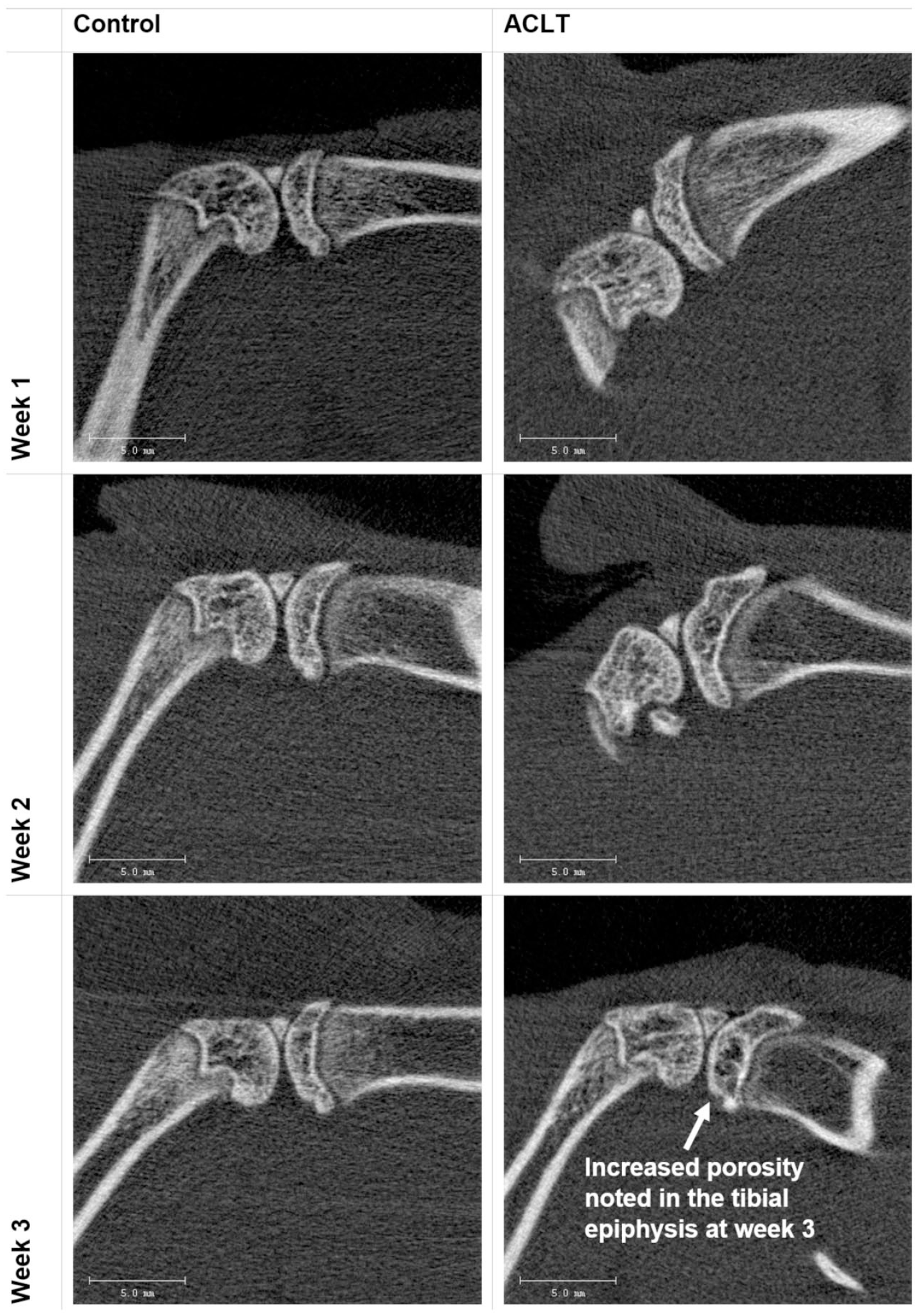
Representative comparison of HR-pQCT (high-resolution peripheral quantitative computed tomography) images acquired from ACLT (anterior cruciate ligament transection) and control groups across the time points. Changes between groups were hard to discern visually over the short time frame. There appears to be an area of increased radiolucency in the medial tibial epiphysis obvious in ACLT week 3 scans. Otherwise, no subchondral sclerosis, subchondral cysts, or osteophytes were seen. Scale bar = 5 mm.
Histopathological examination of the articular cartilage shows that the ACLT group exhibits detectable surface cartilage proteoglycan loss as early as 1 week postsurgery, which increased in surface area at 2 and 3 weeks. There were associated surface irregularities and cell clustering in the week 2 and week 3 samples, with 3 out of 7 samples displaying surface fissures by week 3 postsurgery ( Fig. 5 ). These findings are consistent with early cartilage damage in OA. No area of cartilage eburnation, duplication of tidemark, or osteophytes were observed in any of the samples.
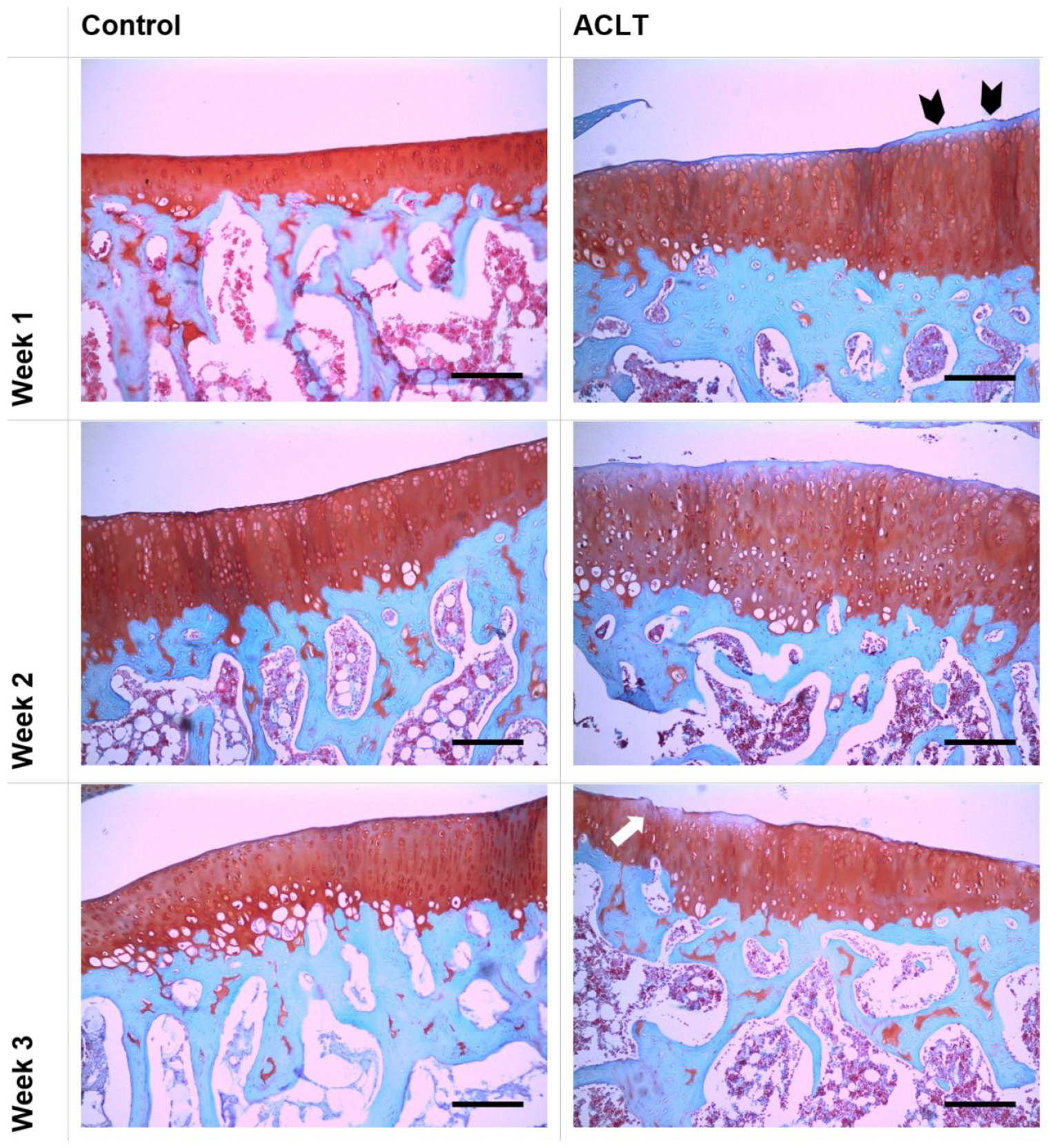
Histological assessment demonstrating articular cartilage changes in ACLT (anterior cruciate ligament transection) and control groups across the time points. Coronal sections from the proximal tibia are fixed and stained with Safranin-O and fast green. Small areas of surface proteoglycan loss (black arrowheads) can be seen at 1 week, which increased in surface area at 2 and 3 weeks. There is associated surface irregularities detected at week 2, which progress into superficial fissures (white arrow) by week 3. Scale bar = 200 µm.
Discussion
In the present study, very early tissue changes in the subchondral bone and cartilage in a mild posttraumatic murine model of OA were captured across a narrow time frame. We found significant changes in the subchondral trabecular bone, which can be observed as early as 1 week post destabilization, characterized by reduced trabecular thickness and increase in trabecular separation. These changes could signify an increase in local osteoclastic activity. Reduction in the BMD and bone volume fraction became significant at week 2 postsurgery, which may suggest a halt in osteoblastic activity. There was also significant reduction in trabecular number seen at week 3 postoperatively. No subchondral sclerosis, subchondral bone cyst, or osteophytes, which signify later disease process, were observed. Collectively, these changes theorize an increase in trabecular porosity and decreased mineralization due to high bone remodeling and turnover that occurs very early in the disease. Subchondral bone remodeling occurred hand-in-hand with mild cartilage degeneration, with no areas of deep fissuring or complete cartilage loss identified nor any osteophyte formation seen. These observations prove our hypothesis, which states that subchondral bone modification is strongly linked to very early articular cartilage degradation at the lowest histopathological scores.
Over the past few decades, much focus has been put on the subchondral bone in the early pathogenesis of OA but the question of whether it is a triggering factor for cartilage wear or whether the changes to the subchondral bone are a secondary consequence of cartilage degeneration still remains open to debate. Anatomically, the subchondral bone consists of 2 parts: the subchondral bone plate and the underlying trabecular bone. Thickening of the subchondral bone plate associated with sclerosis is a hallmark of late stage OA, and has been found to be largely restricted to the areas of severely eroded articular cartilage. 17 There is also an associated increase in the trabecular thickness, decrease in trabecular separation, and transformation of trabeculae from rod-like into plate-like with concurrent increase in the bone volume fraction.18-20 However, despite increase in its bone volume fraction, bone mineralization is significantly lower in severe OA.1,21-23 The quality of this bone was also found to be less stiff when tested mechanically. 24 These data support the hypothesis of an accelerated bone remodeling and turnover in OA, leading to less-mineralized bone tissue. 25 This in turn causes the bone to adapt via increase in bone volume fraction to counteract the increase in bone tissue strain resulting from decreased mineralization and bone material stiffness.26-28
These late changes in humans have also been reflected in animals. In guinea pigs that develop spontaneous knee OA, there is an increase in bone volume fraction with thickening of the trabeculae concurrent with cartilage wear at advanced stages. In guinea pig early OA, however, subchondral cancellous bone had the lowest bone volume fraction together with the lowest mineral concentration, indicating a high bone remodeling with trabecular resorption during the initial stage of OA (at 6 months). 29 Other animal models of OA induced by joint instability also portrayed similar results. Subchondral trabecular bone resorption and decrease in BMD as well as bone volume fraction were found in dogs 30 and rabbits 31 at 1 month after surgical destabilization, and as early as 2 weeks in rats.32,33
In Appleton’s study, subchondral bone changes in an ACLT combined with partial medial meniscectomy model of rat OA were analyzed with relation to the effects of forced mobilization of the joint. 33 Articular cartilage degeneration began as early as 2 weeks after surgery and is worse with forced mobilization. Similarly, subchondral bone loss as expressed by reduction of vBMD and trabecular architecture were seen at 2 weeks in both the nonmobilized and forced mobilized joints compared to the contralateral sides. These findings are reflected in our own study. However, their next measured time points were at 12 weeks and 20 weeks postsurgery, where subchondral bone changes were more likened to late stages of OA.
In another study more similar to ours, Yu et al. examined the longitudinal progression of OA in a medial meniscal tear SD rat model and characterized subchondral bone structural changes via micro-computed tomography. 32 They found a reduction in BV/TV, Tb.N, and Tb.Th and increase in Tb.Sp significant at weeks 2 and 4 postsurgery. However, their histological analysis reveals matrix and chondrocyte loss in the superficial and upper middle zone at 2 weeks, extending into the deep zone at 4 weeks. When taken together, the rate of OA progression in their model is more rapid, thus limiting the captured data on initial and very early tissue changes in OA. Our model complements their findings but has characterized the early subchondral trabecular bone structural change in more detail, which may give a better indication to the underlying pathophysiological process. Of note, significant subchondral bone thickening only occurred in the late stages of their experiment, concurrent with late-stage OA. The subchondral bone plate thickness was not characterized in our study due to limitations of our imaging modality to measure subchondral plate thickness, but this perhaps is not necessary as the subchondral bone plate thicknesses do not undergo significant changes in early disease as shown in their results.
Many other studies on murine models of OA have used micro-computed tomography imaging to characterize subchondral bone changes.34-37 However, to the best of our knowledge, no study has looked into detail the initiation and early changes of subchondral bone associated with OA. Our study provides a clear support to the findings of previous studies and affirms the recent evidence on the role of the subchondral bone in the early propagation of disease.38,39 Indeed, to date, the underlying mechanism for the increased bone turnover and structural deterioration in the early phases of OA is still not fully understood and several theories have been put forth. Abnormal mechanical forces are implicated to cause an altered mechanotransduction across the joint, 40 initiating microfractures in the subchondral bone plate, which activates a bone remodeling response. 41 This leads to later tidemark advancement and subsequent thinning of cartilage.42,43 Other factors that may play a role in the early increase in subchondral bone turnover include a local increased blood flow stimulated by angiogenic factors, 44 and enhanced bone-cartilage crosstalk via increased porosity in the subchondral bone plate.45,46
The net result of all these factors is an increase in osteoclastic activity and trabecular bone resorption in early OA. In an effort to find a common pathway in developing targeted therapy for OA, the effect of bisphosphonates has also been studied. Bisphosphonates inhibits osteoclast-mediated bone resorption by binding to the surface of mineralized bone. Osteoclasts metabolize the bisphosphonates during bone remodeling, leading to impaired activity and apoptosis.47,48 Several preclinical studies have shown that treatment with bisphosphonates has decreased or halted progression of OA in animal models.49-51 Other subchondral bone targeted therapy such as strontium ranelate and osteoprotegerin (OPG) have also been shown to reduce joint structural degradation and/or pain in animals.52-53
Collectively, these data are supportive of the role of subchondral bone remodeling in the progression of OA and its inhibition, which may be a key therapeutic target for delaying or halting the progression of disease. Future focus on osteoclast activation or the receptor activator of nuclear factor kappa-B (RANK)/RANK ligand (RANKL)/OPG pathway in the initiation and progression of early OA may provide further insight into developing an effective disease modulating drug for early OA.
One limitation to this study is that the changes observed in a murine model of posttraumatic OA may not accurately reflect the changes in human OA. This may be due to several factors. First, the physeal plate in rodents will never fuse throughout their entire lifespan. 54 This has been reflected in Horton et al.’s study on skeletal maturation in the rat. 55 They found that rate of exponential growth of the tibia in rats decayed after 9 weeks of age and became statistically insignificant after 20 weeks of age even though the physeal plate remains histologically present. Similarly, the exponential increase in bone mineral density of the femur decayed after 11 weeks of age. Other studies comparing the age-related differences in a surgically destabilized mouse model of OA showed worsening articular cartilage degeneration in older mice compared to their younger counterparts.56,57 This is consistent with the development of OA after joint injury in humans, where OA changes appear sooner in older patients with meniscal injury. 58 These findings may occur because joint cartilage matrix in older individuals are less able to withstand aberrant mechanical loading, and the chondrocytes of older cartilage may be less able to repair an injured matrix. However, having mentioned this, the model chosen for this study is apt for the purpose of explaining our hypothesis.
Second, this model of OA occurs as a result of mechanical instability produced by a single episode of injury to the joint. This may not reflect the pathophysiology of primary OA, which may not be the result of a single episode of injury. However, this model represents the clinical condition whereby knee instability results in OA. The initial OA changes in surgically destabilized rodent knees have also consistently developed over the medial joint space first, similar to the natural progression of human OA. 59 The advanced annotation of their genomes also aids in subsequent cellular and molecular studies. 60
Many posttraumatic murine models of OA have been proposed with differing rate of OA development depending on the severity of destabilization. 10 As our aim was to capture the changes occurring at the initiation and early stages of OA, it was prudent to use a model with slow OA progression such as the ACLT model, allowing ample time to capture these alterations. Other animal models of spontaneous OA such as in Hartley albino guinea pigs, Syrian hamsters, certain strains of mice, and primates develop OA much more gradually over a longer period and may be more ideal but pose further challenges due to the time commitment and housing costs that are required. 61
In conclusion, significant changes in subchondral bone occur very early in OA, concurrent with mild surface articular cartilage degenerative changes. These findings suggest that factors affecting bone remodeling and resorption together with cartilage matrix degradation occur very early in the disease. Further in-depth research into the cellular and molecular factors affecting bone homeostasis in OA is warranted.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by University Malaya Research Grant (UMRG; Project Number: RP005D-13HTM) and Fundamental Research Grant Scheme (FRGS; Project Number: FRGS/1/2018/STG05/UM/02/10; FP068-2018A).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was obtained from Institutional Animal Care and Use Committee (IACUC), Animal Experimental Unit (AEU), Faculty of Medicine, University of Malaya (Ethics Reference no. 2015-181201/ORTHO/R/NANF).
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
