Abstract
Objective
Precise graft-host interference fit is a potentially import factor for the successful incorporation of osteochondral allograft (OCA) transplants for the treatment of symptomatic focal cartilage defects. It was hypothesized that selection of OCA plug instrumentation set has a significant effect on graft integration and morphology after OCA transplantation
Methods
Thirty-one patients who underwent OCA transplantation between July 2013 and July 2016 were identified for this comparative magnetic resonance imaging (MRI) analysis. Patients were stratified into two groups based on the treatment with the
Results
At a mean follow-up of 11.39 ± 1.98 months, 95.5% of all grafts showed osseous integration into the recipient bone with 68.2% presenting without cystic changes of the graft or host-graft junction. No differences were seen in any OCAMRISS subscale besides cartilage signal, which demonstrated minimal differences (
Conclusion
OCAs show excellent osseous integration at the graft-host junction at 12 months postoperatively. The comparative MRI analysis of two instrumentation sets yielded no significant differences in terms of graft integration, bone marrow edema, or cystic formation. Hence, this study cannot recommend one instrumentation set over the other. Consequently, the individual preference should guide the surgeon’s selection of instrumentation for OCA transplantation.
Introduction
Fresh osteochondral allograft (OCA) transplantation has evolved as a safe and effective surgical option in treating patients with symptomatic osteochondral lesions. 1 Typically, a cylindrical OCA plug is inserted into a matching osteochondral recipient site to achieve a press-fit that does not require additional fixation. During insertion, force is applied to the articular surface of the OCA plug, which can diminish chondrocyte viability.2,3 Excessive force has been reported to result in poor cartilage function and matrix content, ultimately leading to inferior clinical outcomes.4-8 Prior studies showed that the force needed to securely fit the OCA plug into the osteochondral recipient site is associated with the graft-host interference fit (OCA plug radius – OC recipient radius).3,9 Additionally, graft-host inference fit is directly related to the stability of the implanted OCA plug and may influence osseous integration and subchondral cyst formation, both of which were shown to result in inferior clinical outcomes after cartilage repair.10,11
Generally, 2 hypotheses have been proposed that may explain the formation of subchondral bone cysts. The
With the goal to ensure optimal fit of the graft into the recipient site, the surgeon has different cutting tools and guides to precisely measure, harvest and prepare the OCA plug and recipient site during surgery. After determination of the defect location and the site of allograft harvest, the recipient site is prepared using a cylindrical coring reamer to remove the entire osteochondral lesion, followed by harvesting a size-matched plug from the donor femoral condyle. Instrumentation sets differ in the amount of oversizing of the plug relative to the recipient hole, resulting in different degrees of interference fit and hence, mechanical stability. Hence, it can be hypothesized that osseous integration and particularly subchondral bone cyst formation after OCA transplantation may differ between instrumentation sets.
Accordingly, we sought to assess the subchondral bone and graft integration using the comprehensive osteochondral allograft magnetic resonance imaging scoring system (OCAMRISS).15,16 in patients after OCA transplantation for symptomatic osteochondral defects in the knee and comparatively analyze bone morphology in dependence of the intraoperatively utilized instrumentation for OCA transplantation. It was theorized that selection of OCA instrumentation set has significant effects on graft integration, bone cyst formation, and bone marrow edema after OCA transplantation.
Materials and Method
After our institutional review board approved this study, our prospectively collected database for all patients undergoing cartilage repair was used to identify patients who underwent cartilage repair with OCA for focal osteochondral defects in the knee by a single surgeon. OCA transplantation was indicated in patients with large focal full-thickness chondral and osteochondral defects (>2 cm2) in the tibio- and patellofemoral joint, osteonecrosis, and after primary failed cartilage repair. Contraindications included inflammatory arthritis and/or advanced osteoarthritis.
For the comparative MRI analysis of this study, patients who underwent treatment between July 2013 and July 2016 were included in this study. In October 2015, the senior author started augmenting OCA plugs with freshly harvested autologous bone marrow aspirate (BMA) from the ipsilateral distal femoral metaphysis which was rooted in a change of practice to optimize surgical treatment. Thus, patients receiving BMA augmentation at the time of OCA transplantation, or had premanufactured 10 mm OCA cylinders implanted, and/or had missing MRI of the involved knee at the time of follow-up at 12 months postoperatively were excluded from this study.
For each part, patients were stratified into 2 groups based on the utilized osteochondral allograft plug instrumentation, namely the
Patient’s age at the time of surgery, body mass index (BMI), sex, smoking status, involved side, and whether the patient had concomitant surgeries such as osteotomy or ligamentous repair/reconstruction and meniscus allograft transplantation (MAT), and/or previous surgeries on the index knee were recorded. OCA plug morphology, including the size, number, and location were collected from surgical notes.
Surgical Technique
The senior author utilizes fresh OCAs acquired from the Joint Restoration Foundation (JRF). Allografts were harvested within 24 hours of donor’s death and stored at 4°C between 7 and 28 days after undergoing viral and bacteriologic screening tests. All surgeries were performed using a medial or lateral peripatellar arthrotomy. The dowel technique uses coring reamers of variable diameter to create osteochondral allograft plugs. Before February 2016, the senior author used the Arthrex (Naples, FL, USA) Allograft OATS Instrument Set for OCA transplantation (

Arthrex Allograft OATS Instrument Set.
Arthrex Allograft OATS Instrument Set
After identifying the lesion, cannulated sizers of a diameter appropriate to cover the lesion were selected and placed over the defect, staying perpendicular to the articular surface. A guide pin was drilled through the sizer into the bone and a circumferential mark around the cylinder was created. The sizer was removed and the peripheral cartilage was scored to the underlying subchondral bone. The recipient site was then reamed under constant cold irrigation, creating a 5- to 8-mm deep bed. Utilizing an appropriately sized dilator, the recipient’s site was dilated. The created recipient bed was measured in depth in all 4 quadrants. The donor graft was then secured in the Allograft OATS Workstation (
JRF Ortho Osteochondral Allograft Plug Instrumentation
The JRF cutter/guide was placed over the cartilage defect ensuring full coverage of the lesion and a pin was inserted perpendicular to the articular surface holding the guide in place. By hand, the cutter/guide was rotated until the complete thickness of the articular cartilage was cut. The cutter/guide was removed and a drill bit reamer was mounted on a cannulated drill, placing it over the pin in the patient’s defect site. With constant irrigation, the drill reamer was slowly advanced, creating a 5- to 8-mm deep recipient bed. The recipient bed’s depth was measured and noted in all four quadrants. Now, the appropriate size centering ring was placed on the allograft at the corresponding location of the patient’s condylar defect and secured with several short guide pins. A coring reamer was slowly advanced into the allograft under constant cold irrigation. The plug was excised by transecting the allograft with an oscillating saw approximately 20 mm below the cartilage surface. On internal validation by the manufacturer, the clearance between the plug and the recipient hole can be anywhere between 0.001 and 0.003 mm, which in fact can be considered a line-to-line graft-host interference fit. The harvested plug was then trimmed to the recipient site’s corresponding depth. Again, marrow elements were pulse lavaged in order to decrease the immunogenicity and the graft was then manually placed press-fit into the recipient site until it was seated flush circumferentially (

JRF Ortho Osteochondral Allograft Plug Instrumentation.
Postoperative Rehabilitation
Patients were maintained on touchdown weightbearing on crutches for 4 weeks and then advanced to full weightbearing by 6 weeks. Continuous passive motion (CPM) was started on postoperative day 3, advancing from 0° to 90° as tolerated. At 6 months after surgery, patients were allowed to return to recreational activity.
MRI Acquisition
Postoperatively, patients returned to our clinic for routine follow up during which MRI of the involved knee were obtained. As complete osseous integration and settlement of acute bone reaction is expected at approximately 12 months postoperatively, patients with MRIs obtained at 12 months after surgery were included in this comparative MRI analysis.
Imaging analysis was performed using the previously published and validated comprehensive OCAMRISS.15,16 The original scoring system includes 5 primary cartilage features, 4 primary bone features, and 4 ancillary features. Because of the lack of ultrashort echo (UTE) in clinical MRIs, which are needed for assessing the integrity of the calcified cartilage layer,
17
this feature was not included in the analysis of the current study. Based on the study by Sirlin
Statistical Analysis
Descriptive statistics and frequencies were utilized to determine sociodemographic and clinical characteristics of patients. Based on distribution of data, the Mann-Whitney
Results
The senior author treated a total of 140 patients with OCA transplantation for focal symptomatic osteochondral lesions within the knee joint during the study period. Of these patients, 31 patients (22.1%) were enrolled for the present study. Of the 109 excluded, 35 patients (32.1%) received premanufactured 10-mm OCA cylinders (JRF Ortho, Centennial, CO, USA), 5 (4.6%) received BMA augmentation at the time of OCA transplantation, and 69 patients (63.3%) had missing postoperative MRIs at 12 months. Of the included patients, 19 (61.3%) were female, 1 (3.2%) was an active smoker, 16 (51.6%) had previous operations on the index knee, 1 (3.2%) underwent concomitant high tibial osteotomy (HTO), 1 (3.2%) underwent concomitant lateral MAT, and 4 (12.9%) underwent juvenile particulated cartilage allograft (DeNovo NT, Zimmer, Warsaw, IN) transplantation for the treatment of other, smaller defects elsewhere in the knee. The number of OCA plugs ranged from 1 to 3 with 48.4% of patients treated with 1 plug, 32.2% with 2 plugs, and 19.4% with 3 plugs. Twenty-one of the evaluated plugs were implanted in the medial femoral condyle, 9 in the lateral femoral condyle, 11 in the trochlea, and 3 in the patella. The mean age was 34.61 ± 9.52 years with an average BMI of 27.99 ± 4.86 kg/m2. The combined size of all implanted OCA plugs in a patient averaged 4.15 ± 2.15 cm2. Mean time between surgery and evaluated MRI was 11.39 ± 1.98 months (
Patient and Osteochondral Allograft Characteristics.
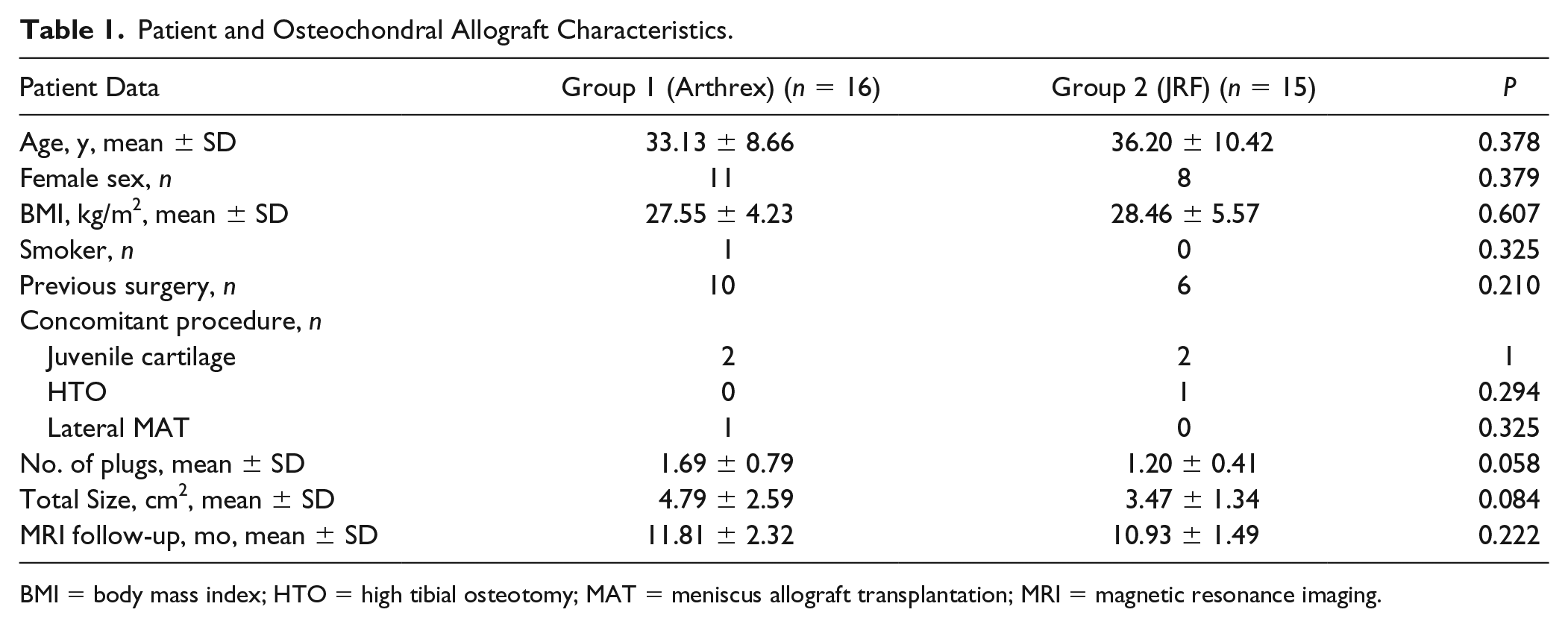
BMI = body mass index; HTO = high tibial osteotomy; MAT = meniscus allograft transplantation; MRI = magnetic resonance imaging.
At final follow-up, 95.5% of all grafts showed osseous integration into the recipient bone with 81.8% sitting flush to the adjacent cartilage. A total of 88.6% of grafts had excellent cartilage fill and 68.2% had no presence of cystic changes of the graft or host-graft junction. No difference was seen in any OCAMRISS subscale except cartilage signal, which barely reached significance (
Osteochondral Allograft MRI Scoring System Scores in Patients Who Underwent Osteochondral Allograft Transplantation Using the JRF or Arthrex Instrumentation Set.

Boldfaced value indicates significance at the level of
Of the 31 patients included, 4 (12.9%) presented with an effusion in the index knee at 12 months postoperatively. No association was seen between effusion and instrumentation set (Arthrex:
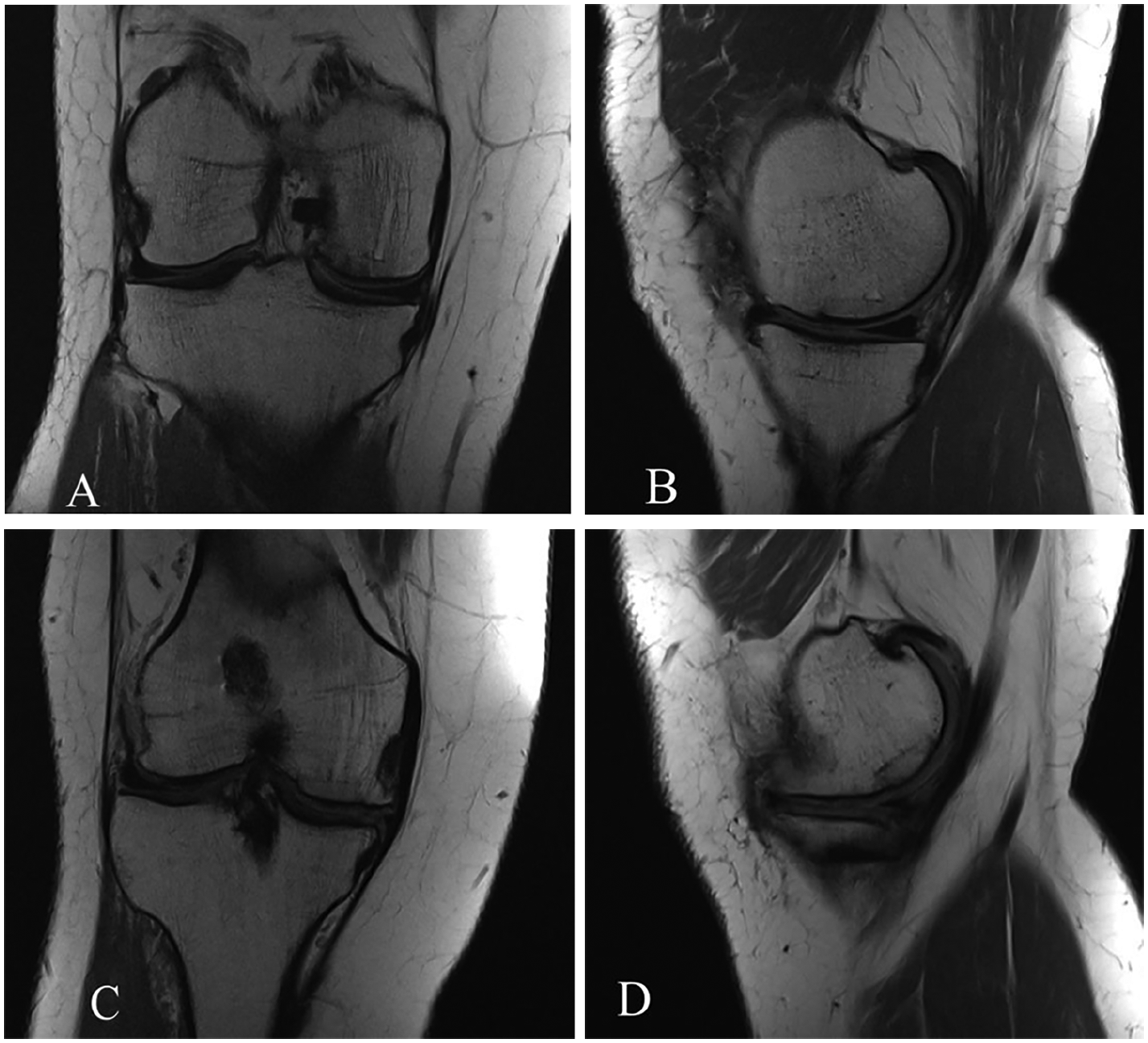
Magnetic resonance imaging at 12 months after osteochondral allograft transplantation showing a discernible cleft at the graft-host interface in the Arthrex group (
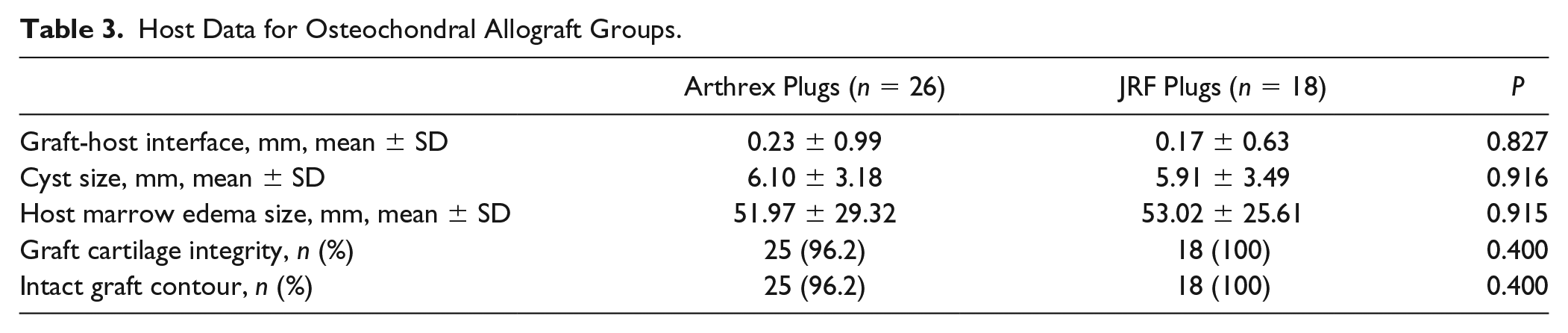
Host Data for Osteochondral Allograft Groups.
Discussion
Achieving a stable press-fit of the OCA plug within the recipient site is deemed desirable to minimize micromotion, thus improving osseous integration. The instrumentation is designed to balance graft stability and the force required for graft insertion. In this study, the instrumentation in group 1 (Arthrex) is designed to provide a press-fit graft-host interference of 0.5 mm, while group 2 (JRF) achieves a line-to-line fit measured at 0.001 to 0.003 mm. Interestingly, this study demonstrated no significant differences in terms of graft integration, bone marrow edema, or cystic formation between groups. At 12 months, only 1 patient from the Arthrex group and 1 from the JRF group demonstrated a discernable graft-host interface gap on MRI (
It should be noted that the senior author uses manual compression by either finger compression or the congruence of the femorotibial articulation to reduce grafts and the grafts are not affected. The existing literature describing chondrocyte damage investigates the influence of direct axial force that occurs during impaction of the grafts. Su
In a study determining the effect of OCA storage on subchondral bone structure after OCA transplantation in a goat model, Pallante-Kichura
In this study, both instrumentation sets achieved a clinically acceptable graft-healing rate with osseous integration at the graft-host interface in 25 out of 26 (96%) and 17 out of 18 (94%) in the Arthrex and JRF groups, respectively. Given these findings, the graft-host fit, which is achieved with either instrumentation set, appears to sufficiently stabilize the grafts within the recipient bed and effectively minimizes cyst formation by either synovial intrusion or the bony contusion theory.
This study has certain limitations. It is a retrospective study and subject to the biases of this design. Additionally, due to the small number of patients we were unable to match groups for confounding variables, such as age, BMI, medical comorbidities, concomitant procedures, and the size and location of the grafts. However, considering the lack of larger studies in the literature, we believe that the current results offer valuable insight to cartilage repair surgeons as this, to our knowledge, is the first clinical study to evaluate the influence of instrumentation sets with different amounts of press-fit interference, using the OCAMRISS to evaluate the graft-host interface for osseous integration.
In summary, we found excellent osseous integration at the graft-host interface at 12 months postoperatively. Hence, this study cannot recommend one instrumentation set over the other. Consequently, individual preference and availability should guide the surgeon’s selection of instrumentation for OCA transplantation.
Footnotes
Authors’ Note
The study was performed at the Cartilage Repair Center and Center for Regenerative Medicine, Brigham and Women’s Hospital, Harvard Medical School, Harvard University, Boston, Massachusetts, USA.
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Jakob Ackermann and Alexandre Barbieri both received research support from JRF; outside of this work.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: R.A.D. has received educational support from, Arthrex. A.H.G. receives research support from JRF and is a consultant for JRF.
Ethical Approval
Approval for this study was obtained from Partners Healthcare (2018P000176/PHS).
Informed Consent
Not applicable.
Trial Registration
Not applicable.
