Abstract
Objective
This study aimed to test the hypothesis that administration of increasing doses of Sinovial (hyaluronic acid [HA]), would exhibit a dose-dependent effect on the prevention of cartilage degradation, without local and systemic toxicity.
Methods
Twenty-seven adult rabbits were subjected to anterior cruciate ligament transection (ACLT). Two Sinovial products containing HA concentrations of 1.6% and 2.4% were used as active treatment, and 0.9% saline was used as control and injected intra-articularly 7 days post ACLT. Radiographs were taken prior to surgery, at injection and sacrifice times. After euthanasia, 8 weeks postsurgery, knee joints were observed macroscopically using India ink staining with OARSI (Osteoarthritis Research Society International) scoring and histologically using modified Mankin scoring. The synovial membranes were analyzed using Cake classification.
Results
No intraoperative complications were observed. One week postinjection, 4 animals in the HA 2.4% group developed subcutaneous nodules that disappeared spontaneously. No inflammation of the synovial membrane was ever observed. The control group exhibited the maximum uptake of India ink 2.22 ± 0.14. HA groups exhibited a dose-dependent (
Conclusions
A dose-dependent protective effect on cartilage was observed after injection of both HA solutions.
Introduction
Osteoarthritis (OA) is the predominant form of arthritis and the leading cause of disability in approximately one-third of all adults worldwide who have radiological signs of OA. 1 As there is no disease-modifying treatment for OA, the disease leads to the progressive degeneration of articular cartilage with associated remodeling of subchondral bone and synovitis, which contribute to chronic pain, physical disability and impairment in quality of life.
Hyaluronic acid (HA) is a high-molecular-weight glycosaminoglycan present in the synovial fluid. Molecular weight distribution of HA ranges from 0.03 to 7 megadaltons (MDa) and the normal concentration ranges from 1 to 4.0 mg/mL.2-5 In arthritic conditions, the molecular weight distribution shifts lower and the concentration of HA is reduced, thereby contributing to the abnormally low viscoelasticity of the synovial fluid.4,6,7 To counter this problem, high-molecular-weight HA solutions, also known as viscosupplements, were developed for intra-articular injection.8,9 The administration of these products (viscosupplementation) increases the lubrication, shock absorption, hydration, and nutrition of the joint tissue, relieving disease-associated pain.
7
In addition HA may contribute to restoration of the rheological properties (visco-induction) of the arthritic synovial fluid since HA acts as a regulator of cells, enzymes, and inflammatory factors.10,11 In 1974, the first clinical study performed with HA showed that the elastoviscosity of the synovial fluid regained normal levels and the pain associated with the movement of the joint decreased.
12
Since that time, the beneficial effect of the intra-articular injection of HA formulation for the treatment of OA has been confirmed several times in clinical trials, as described in the review written by Bannuru et al.13,14 Conversely, the field is still controversial given the proficiency of HA-derived formulations allowed on the market in many countries. A large part of competing formulations is being allowed on the market without the need for presenting
Different intra-articular HA preparations are currently commercially available in Europe and/or the United States.16-21 They differ from HA origin, method of preparation, molecular weight, concentration, residence time in the joint, rheological properties, treatment schedule, pharmacodynamics, and costs. To date, the superiority of one product over another for the treatment of OA has not yet been proven.
Sinovial (IBSA Institut Biochimique SA, Switzerland; other available trade names Intragel, Yaral, and GELSYN-3, United States) is a chemically nonmodified physiological solution of 0.8% to 2% highly purified (chromatography) HA with a low to medium molecular weight (0.8-1.2 MDa). Sinovial is made available as a sterile, buffered solution of sodium hyaluronate obtained through fermentation of streptococci of Lancefield groups A and C and bears no chemical modification whatsoever.
In clinical studies, administration of Sinovial at 0.8% or 1.6% has shown a beneficial effect in the treatment of pain, or reduced mobility due to degenerative diseases, post-traumatic diseases or joint and tendons alterations.22-24
The demonstration that different concentrations of Sinovial can mitigate pain and mobility issues, made us question, whether such a modality might also mitigate cartilage degradation in a dose-dependent manner. In the present study, we evaluated the effect of increasing concentrations of Sinovial (1.6% and 2.4% HA) intra-articular injection, on the development of osteoarthritis induced by anterior cruciate ligament transection (ACLT), and on local and systemic toxicity.
Methods
OA Model
This study was approved by the Institutional Animal Care and Use Committee (IACUC) and the Committee for Animal Protection of the Ministry of Health of the Czech Republic (project number 27, 2014).
Twenty-seven specific pathogen free (SPF) adult female HYLA rabbits, older than 21 weeks (Genetické Centrum Hyla, Czech Republic), after 7 days of acclimation were subjected to the following surgical procedure. The animals were first sedated by the intramuscular (IM) administration of 100 µg/kg medetomidine (Domitor, 1 mg/mL, Orion Pharma) and 50 mg/kg ketamine (Narkamon, 50 mg/mL, Bioveta) and subsequently induced with IM medetomidine and ketamine in identical doses. Anesthesia was maintained with 2% propofol (MCT/ACT Fresenius Kabi) administered at a dose between 4 and 12 mg/kg that was adjusted intravenously according to the required level of anesthesia. Under aseptic conditions, a 4 cm skin incision was made precisely medially to the midline from the tibial crest to the base of the patella of the rabbit’s right knee. This was followed by a stab incision on the medial aspect beneath the patella tendon into the femorotibial joint. The patella was luxated laterally and the joint flexed. With the joint flexed, the fat pad was retracted distally, the anterior cruciate ligament was clearly exposed and isolated with the help of a surgical hook. The ACL was transected with a No. 15 blade and sterile microsurgical scissors. To ensure that the entire ACL was transected, the patella was replaced at its initial position and the articular joint was checked via the Lachman test to ensure a positive anterior movement. Connective tissues were closed using No 3.0 monocryl and simple interrupted sutures. Thereafter, the skin wound was closed with sutures using No. 4.0 vicryl and a sterile aerosol solution bandage (OpsiteTM) was sprayed over the wound.
Postoperatively, the animals were permitted cage activity without immobilization; analgesia was promoted by the subcutaneous administration of 0.1 mg/kg Metacam (Meloxidyl 20 mg/mL, Ceva Santé Animale) per day for 3 days. Infection was prevented by the subcutaneous administration of 10 mg/kg enrofloxacin (hypralona, Hypra S.A.) and the treatment of the closed wound with Alamycin aerosol spray (oxytetracyclin hydrochloride, Norbrooc Laboratories) both administered daily for 14 days after the surgery.
Treatment Regimens and Study Design
This study utilized 3 treatments: 2 Sinovial products containing HA concentrations of 1.6% and 2.4%, respectively, were used as the active treatments and 0.9% saline (Sigma-Aldrich) was used as the control. Nine animals were randomly assigned to each group (saline, HA 1.6% and HA 2.4%). Treatment was one single intra-articular injection 1-week postsurgery and explantation was performed 7 weeks posttreatment.
A week postsurgery, the animals were sedated following the procedure previously described, the operated (right) knee of the rabbit was cleaned and sterilized with betadine solution before the intra-articular injection. A syringe filled with 1 of the 3 treatments was selected, the syringe tip cap was removed and replaced with a sterile 21 G needle. The actual administration procedure was performed as described elsewhere, 24 volume of injection was based on our expertise and literature.25,26 Briefly, an intra-articular injection of 600 µL of the applicable treatment was directly administered into the right knee, pushed gradually and with constant force. No injection was administered on the contralateral joint.
X-Rays
Under anesthesia, X-rays (Poscom 100 HF) were performed prior to surgery, on the day of injection and on the day of sacrifice in 2 different planes (dorso-plantar and latero-medial views). In order to have reproducible orientation and knee position among the different rabbits in the study, a box system was employed to enable a similar position for each animal. 27 The 2-plane radiographs were taken with straight knees. The radiographs were scored in a blind fashion by a qualified veterinarian, reminiscent of the Kellgren and Lawrence scaling28,29 used for scoring radiographs of human knees:
Grade 0: No osteophytes
Grade 1: Doubtful osteophytes
Grade 2: Minimal osteophytes, possibly with narrowing, cysts and sclerosis
Grade 3: Moderate or definite osteophytes with moderate joint space narrowing
Grade 4: Severe with large osteophytes and definite joint space narrowing
Macroscopic Analysis
Macroscopic joint assessment was performed on the right knee joint according to the Osteoarthritis Research Society International’s (OARSI) recommendations. 30 The knee specimens were washed in phosphate buffered saline (PBS), painted with an India ink solution and subsequently washed with PBS for the removal of excess ink. The 4 compartments, medial femoral condyle (MFC), lateral femoral condyle (LFC), medial tibial plateau (MTP), and lateral tibial plateau (LTP) were graded following the application of India ink. A photograph of all joints was taken, and samples were scored blindly by a qualified pathologist among the authors, reminiscent of the Outerbridge classification 31 normally used for scoring human knees:
Grade 1: No uptake of India ink that indicates an intact surface
Grade 2: Minimal focal uptake of India ink that indicates a minimal fibrillation
Grade 3: Evident large focal dark patches of India ink uptake that indicates over fibrillation
Grade 4: Large, general uptake of India ink that indicates cartilage erosion
In addition, presence of osteophytes was quantified according to the following scale:
Grade 0: No osteophyte
Grade 1: Minimal, single, and small
Grade 2: Moderate, small, and multiple or single and large
Grade 3: Severe, large, and multiple
Histological Processing and Histopathological Observations
Joints were fixed in 10% neutral buffered formalin at room temperature. Femur and tibia were decalcified in ethylenediaminetetraacetic acid (EDTA), and subsequently cut into different blocks. The anterior condyle, the posterior condyle, and the condyle trochlea were separated. The tibia was cut into 3 parts (anterior, medial, and posterior). The 6 resulting tissue samples were then embedded into separate paraffin blocks. A total of 5 sets of sections, 5 µm thick each, were cut from each paraffin block. Hematoxylin and eosin, toluidine blue, and safranin O were employed to stain 3 sections. The intensity of safranin O and toluidine blue staining is directly proportional to the proteoglycan content in a normal cartilage. The histopathologist among the authors observed the specimens under a light microscope (Olympus, BX41 at ×20 and ×40 magnification). The surface of the joints was morphologically described, while observing the cartilage and the bone compartment. The histological sections from each compartment, and not only the most severe lesions as sometimes recommended 30 were evaluated using a modified Mankin score ( Table 1 ) 32 in order to assess the effect of the product in the entire joint. All the sections were scored in a blind fashion and the histopathologist documented his observations in digital pictures.
Modified Mankin Histological Score.

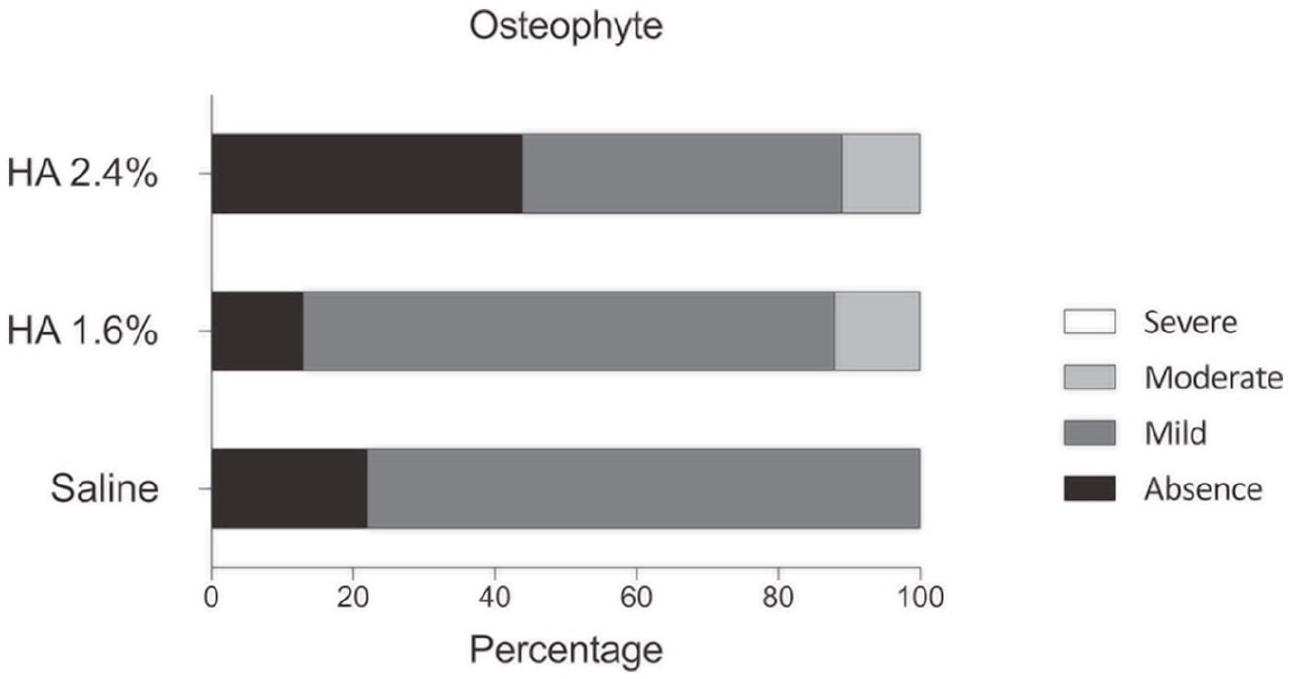
In addition, the synovial membranes were graded based on the following criteria: intimal hyperplasia, lymphocytic/plasmocytic infiltration, subintimal fibrosis and vascularity, according to the Cake classification ( Table 2 ). 33
Synovia Scoring System.
Statistical Analysis
Generally, values are reported as mean ± standard error of the mean. The statistical analyses of the macroscopic and histological data along with radiology data were performed using 1-way analysis of variance (nonparametric test), followed by Kruskal-Wallis posttest with 95% confidence intervals.
Results
Clinical Observations
All operations were routine without any intraoperative complications, except for 1 animal that died during the sedation and was replaced.
Some postoperative complications were observed in the treated groups. Two animals from the HA 1.6% group (22.22%) were involved, in either case, without body weight loss or any manifestation of lameness. One animal developed a large periarticular joint nodule and had to be euthanized 15 days postsurgery since care was taken not to let any major inflammation event mask actual physiological processes and flaw statistical analyses. One week after injection, 1 animal developed caseous exudation, which disappeared with antibiotherapy within a week.
Four animals from the HA 2.4% group (44.44%), 1 week after injection, developed small subcutaneous nodules of 1 to 2 cm in diameter, these nodules disappeared within less than a week for 3 animals and within 3 weeks for the fourth one.
The body weight of all animals from both control and treated groups slightly decreased from week 1 to week 3 postoperatively, although normal body weight was regained from week 4 onward until the end of the follow-up period (8 weeks after surgery; data not shown).
Gross Pathology of Knees and Synovial Membranes
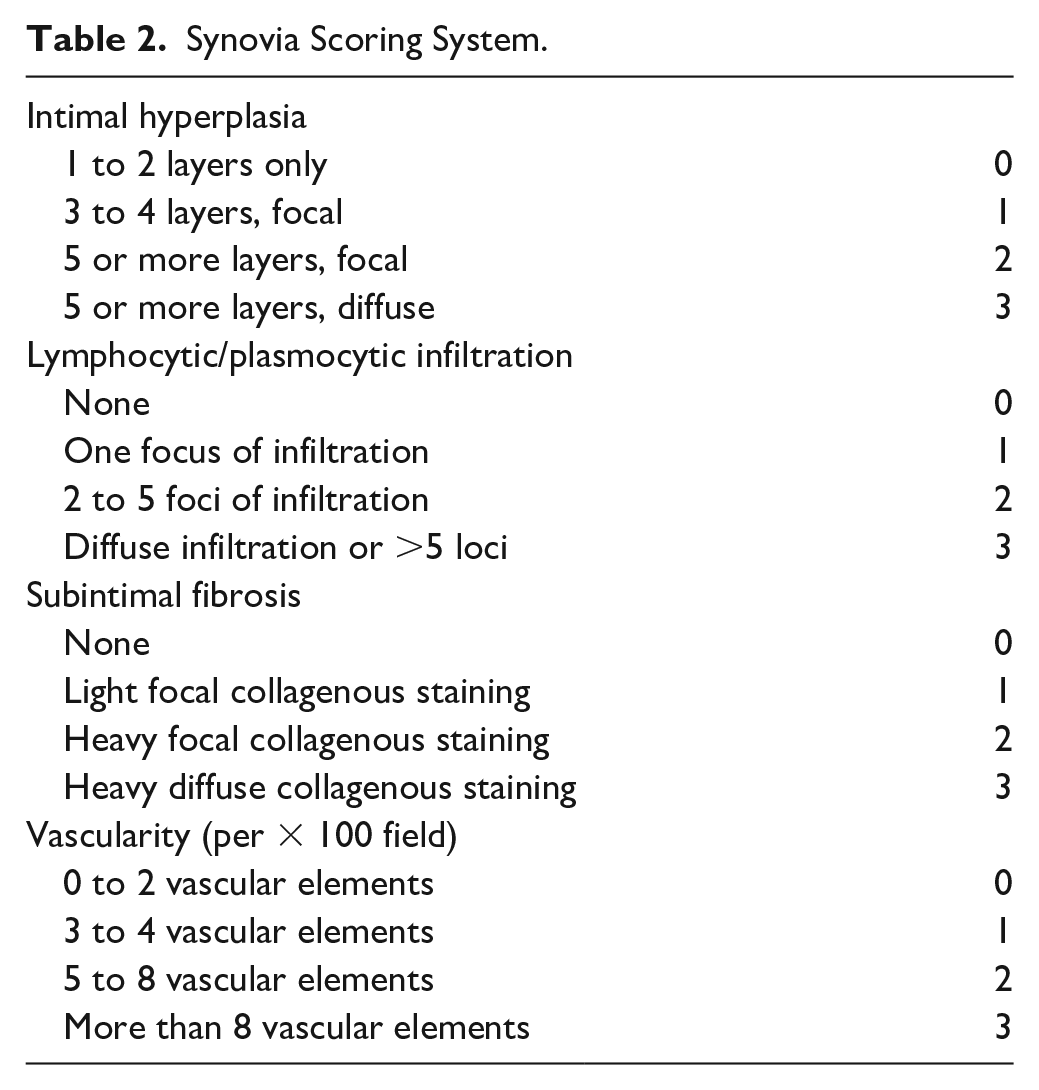
All animals knees were stained with India ink and graded 8 weeks post-ACLT, using a previously described grading system.30,31 The saline group exhibited the maximum overall uptake of India ink (2.22 ± 0.14), indicating more significant cartilage damage and thus a major cartilage porosity, in keeping with the principle of the India ink test. The HA groups (1.6% and 2.4%) exhibited reduction in India ink uptake (1.75 ± 0.17 and 1.58 ± 0.14, respectively), with the 2.4% group achieving a statistically significant reduction as compared to the control group (
Fig. 1
,
(
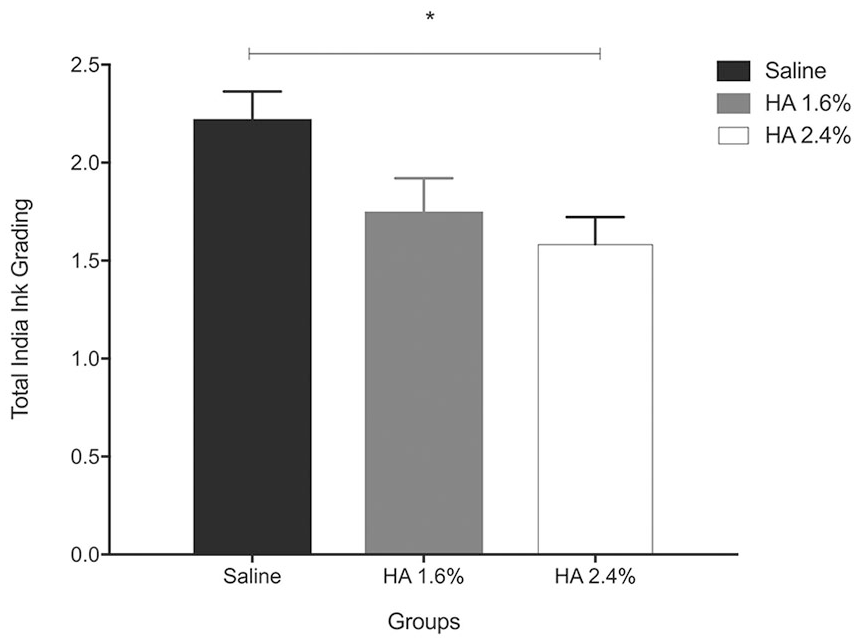
These results were elaborated by the examination of the different anatomical locations within the knee. Indeed, significant cartilage erosion was observed in the lateral condyle, moderate erosion in the medial condyle, and fibrillation in the medial tibial plateau, as evidenced by India ink uptake; however, there was no obvious effect on the lateral tibial plateau ( Fig. 2 ). In the affected locations, the HA-treated groups exhibited dose-dependent reductions in cartilage damage ( Fig. 2 ).
Graphic representation of the macroscopic assessment of the different compartments (LFC, MFC, LTP, and MTP) in each group 8 weeks after surgery. Cartilage degeneration in HA 2.4% group was generally less severe than that in either saline or HA 1.6% group (n = 8-9 per group). The degenerative changes were more pronounced in MFC and LFC. HA = hyaluronic acid; LFC = lateral femoral condyle; MFC = medial femoral condyle; LTP = lateral tibial plateau; MTP = medial tibial plateau.
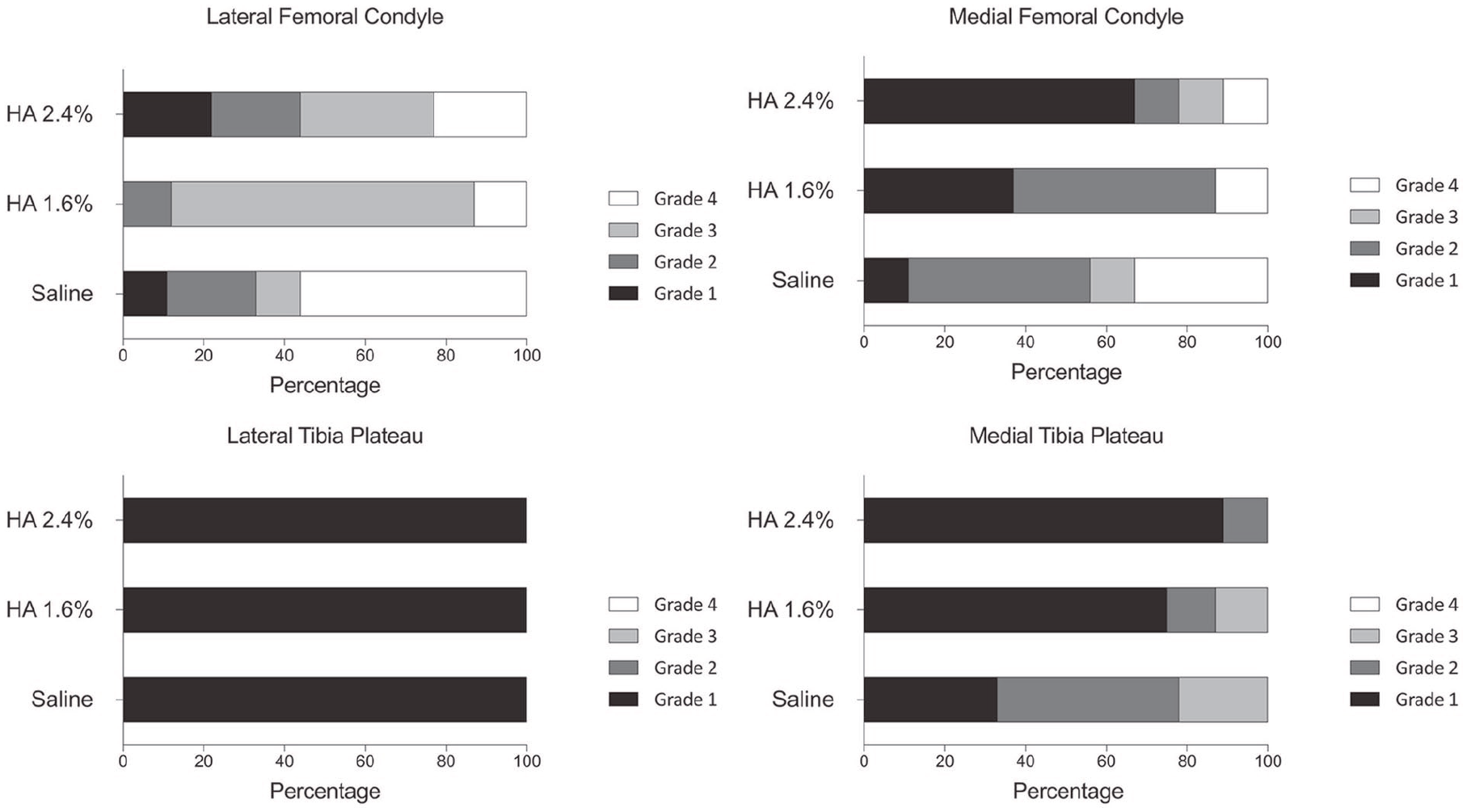
All groups exhibited mostly mild osteophytes (7/9 in group saline and HA 1.6%, 4/9 in group HA 2.4%). One animal from HA-treated groups displayed moderate osteophytes ( Fig. 3 ).
Macroscopical assessment of osteophytes.
Radiological Analysis
The Kellgren and Lawrence score of animals from treated groups was zero (0) on both occasions, on the day before surgery (D1) and on the day of treatment (D8), the saline control group however, exhibited a mean score of 0.11 ± 0.11 at D8. At the time of euthanasia, 7 weeks after treatment, the saline group score was 1.67 ± 0.41, the HA 1.6% was 0.25 ± 0.16 and the HA 2.4% was 0.33 ± 0.17. While there was no dose dependency observed in these experiments, both 1.6% and 2.4% concentrations of HA led to statistically significant reductions in the Kellgren and Lawrence score as compared with the control group (**
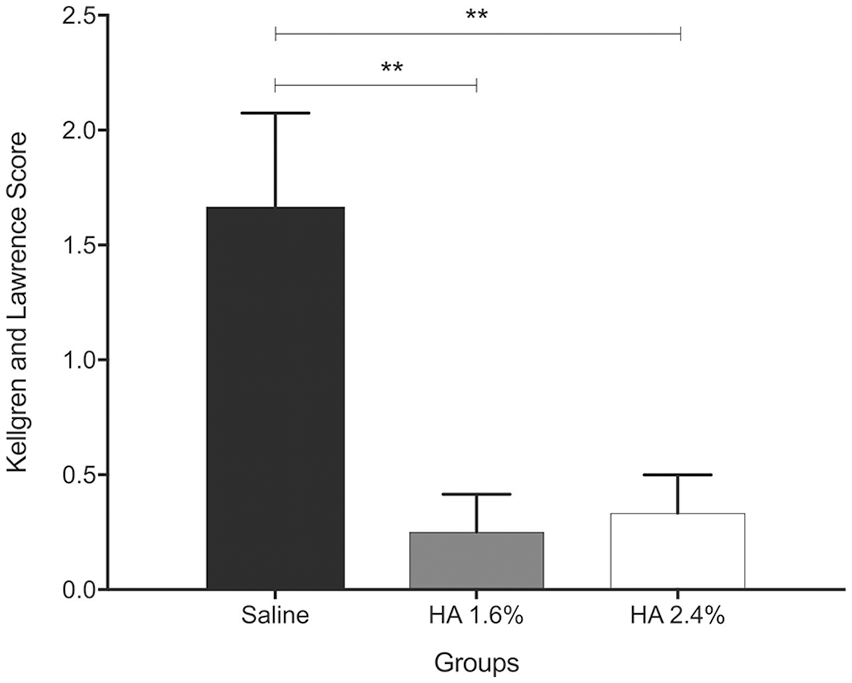
Radiographical grading of knee joints according to Kellgren and Lawrence score at euthanasia, presented as means ± SEM (**
Histopathology
At the time of euthanasia, the mean modified Mankin score obtained by evaluation of all 4 compartments was as follows: 7.19 ± 0.85 in the control group, while the mean scores for HA groups (1.6% and 2.4%) were 4.62 ± 0.67 and 3.53 ± 0.59, respectively. Both HA groups thereby achieved a statistically significant reduction when compared with the control (HA 1.6%:
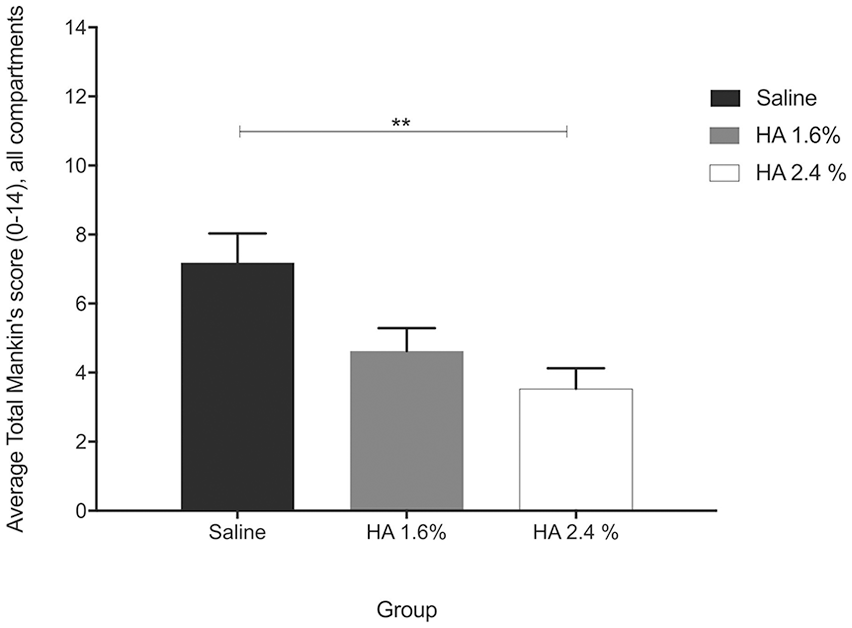
Histological grading according to the modified Mankin score. Overall score, all knee compartments presented as means ± SEM (**
In addition to the total Mankin score for all compartments, the scores for each individual compartment were also assessed (
Fig. 6
). There was a dose-dependent reduction in the Mankin score for the HA groups in comparison to the control group. The 2.4% HA treatment exhibited a statistically significant reduction in the trochlea (
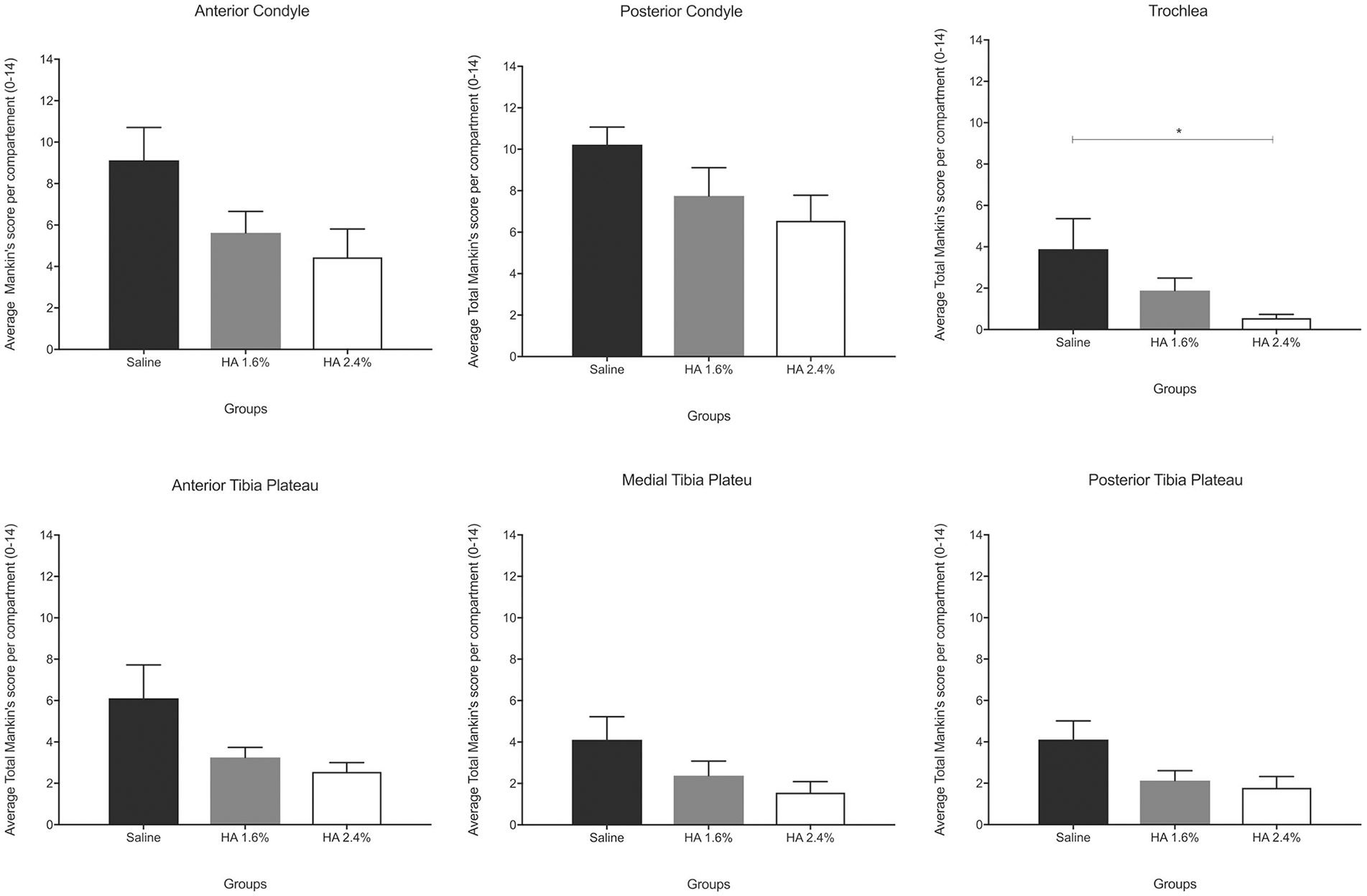
Histological grading according to the modified Mankin score. Overall score, individual knee compartments presented as means ± SEM. (*
The factors that most influenced the Mankin score were cartilage structure, cellularity, and tidemark integrity. As with most other comparisons, there was a dose-dependent reduction in the Mankin score for the HA groups when compared with the control group (
Fig. 7
). Statistically significant improvements were observed in cartilage structure (2.4% HA,
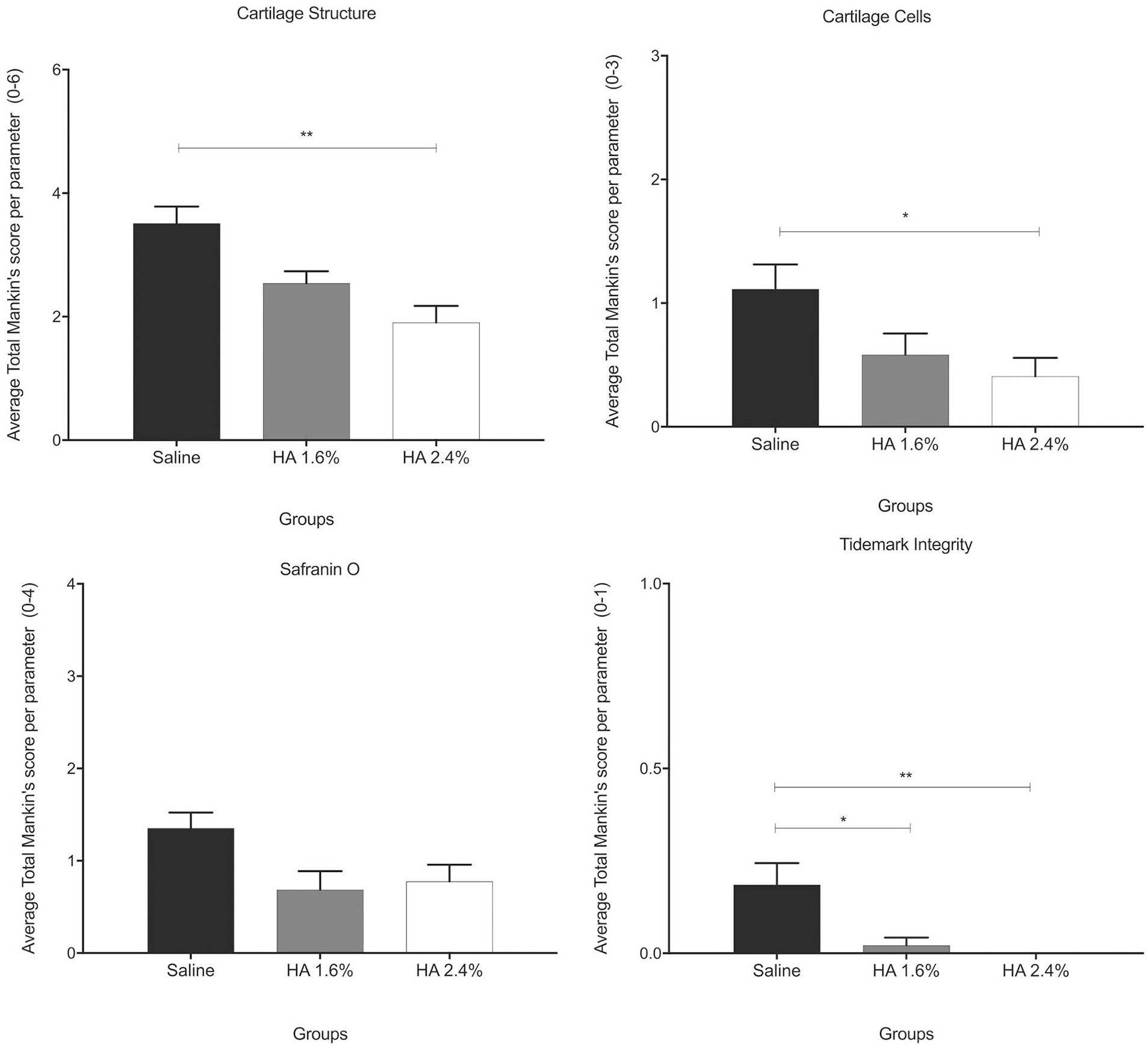
Graphic representation of the individual score for each category of the modified Mankin score presented as means ± SEM. (*
Safranin O staining was not significantly different between groups. Subchondral bone thickening was not observed in any group (data not shown). Representative histological pictures are shown in Figure 8 . Moderate surface fibrillation (arrow) and moderate upcoming bone front (+) are observed in saline group on both condyle compartments. None of those degenerative features were observed in HA-treated groups. Slight loss of safranin O (−) was observed in both condyle compartments of the saline group, and in posterior condyle of the 1.6% HA group. Small osteophytes were observed in 2.4% HA and saline groups (*).
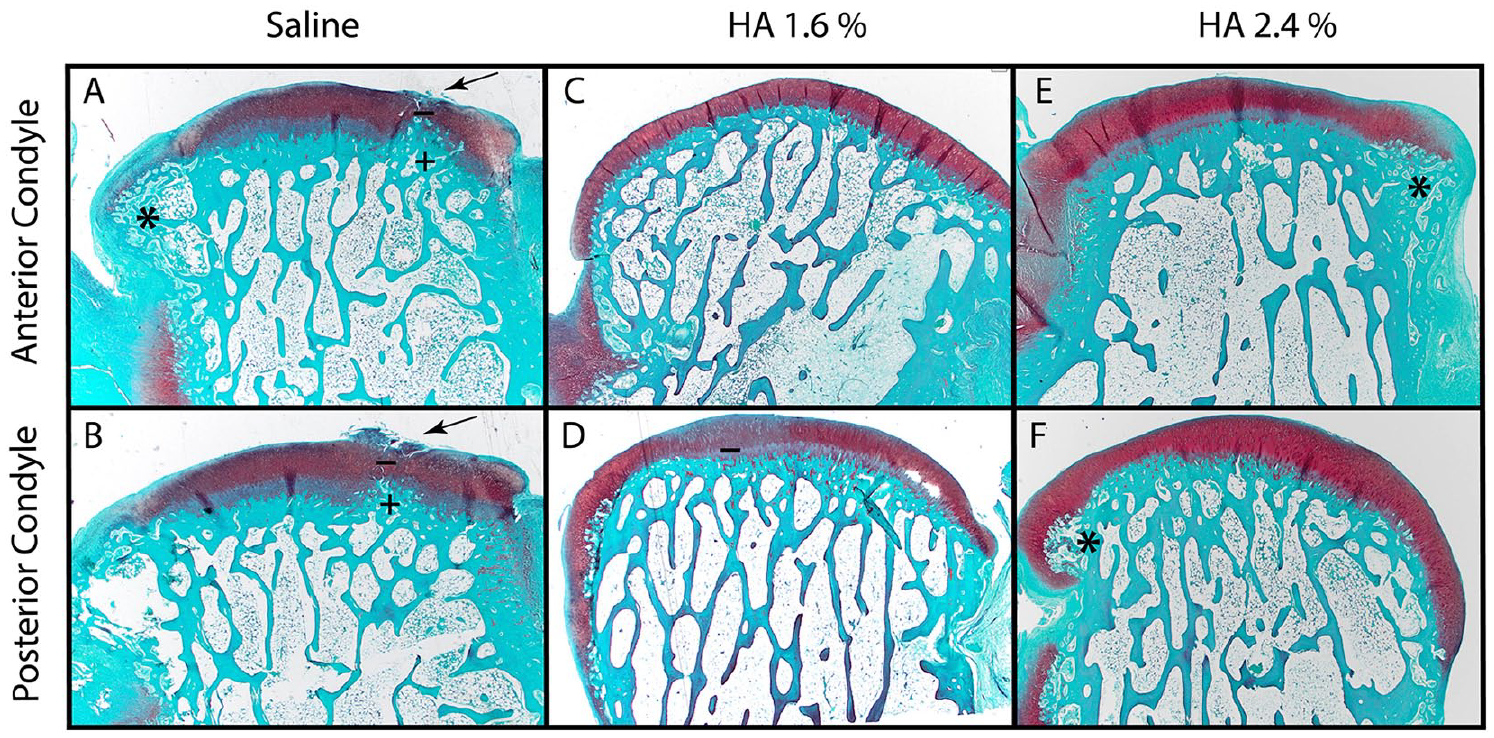
Representative histological pictures of each group. (*) Osteophyte, (arrows) surface fibrillation, (+) upcoming bone front, (–) partial loss of safranin O staining. In the control group (
No inflammation of the synovial membrane was observed in any groups (n = 8-9) ( Table 3 ). Only very little intimal hyperplasia up to 3 or 4 layers could be seen in all groups. One focus of lymphocytic infiltration could be seen in both HA groups.
Individual Score of Each Category of the Synovial Scoring System (Mean ± SEM).

SEM = standard error of the mean.
Animal died during study.
The synovial fluid was of normal appearance (normal viscosity, transparent without blood or pus) on aspiration.
Discussion
Tolerability of 2 concentrations of intra-articular HA compound (Sinovial) was investigated in the present study, along with its efficacy to delay disease progression in an ACLT rabbit model of OA.
Numerous studies have proposed the correlation between molecular weight of HA and its therapeutic effect.17,18 They have compared different compounds of similar molecular weight, 34 but to the best of our knowledge, this is the first preclinical study investigating the effect of single injection of Sinovial HA concentrations on OA progression.
In particular, it was demonstrated that animals treated with 2.4% HA, and to a lesser extent with 1.6% HA, exhibited a statistically significant reduction in cartilage erosion, joint narrowing, and cellularity as compared with a control group left untreated.
The ACLT rabbit model was selected for the present study, as it is a proven suitable model for the evaluation of the structural effects of a therapy on cartilage.
31
In addition, it has been conventionally used to study the effects of different types of OA treatments.
27
Our results are very similar to effects previously described for HA, HA-derivatives, and alginate-chitosan in comparable
There were several cases of periarticular nodules observed in treated animals. The present post-surgery complications, while a similar experimental design was previously safely used,26-28 are probably due to the combination of injection schedule, volume, viscosity, and injection being performed only 1 week following surgery. This time was probably too short to allow an eventless injection of a large volume of highly viscous HA solution. High volume and viscosity of the product increased pressure into the joint and let the polymer escape into adjacent soft tissues, thereby predisposing the host to infection and nodule formation. One-week interval between surgery and IA injection is common practice in rodents. Even when a high volume (900 µL) of a suspension of micro-beads was applied into the joint, 27 1-week interval was enough time for the tissues operated upon to self-repair and provide a leak-proof environment to the compound injected. However, in this particular case, injected compound was in the form of micro-beads that might have been efficiently retained into the joint despite incipient leakage. In the present settings, a high volume of a highly viscous compound and at high concentrations was injected, leading to a much higher pressure inside the joint as compared with saline that has not been associated with any postsurgery complication.
Therefore, in our view, complications are rather model-related than dose-related. Furthermore, no signs of inflammation were histologically observed in the synovial membrane or joint compartment in treated animals.
The lack of real untreated control could be considered a limitation of this preclinical study. Although numerous authors used phosphate-buffered saline (PBS) as control,9-12 it has been reported that PBS is effective in the treatment of OA, especially for pain, stiffness, and self-reported function.
34
However, Yoshioka
Sinovial, at different concentrations and volumes, has demonstrated clinically relevant pain relief, improved joint mobility and reduced use of rescue medication in knee OA as well as in other clinical conditions (i.e., hip OA, painful shoulder and hand OA). However, so far, evidence for disease modifying/structural activity of the product has never been demonstrated.22,24,34,36 The exact mechanism of action of HA is still to be defined; however, it is believed that several aspects contribute to the therapeutic effect achieved with HA in the treatment of OA. A mechanical effect due the viscosity of the product and biological actions including (but not limited to) anti-inflammatory, analgesic, anabolic, and chondroprotective effects.
One single injection of 2.4% HA solution exhibited the highest effect in the employed ACLT rabbit model for osteoarthritis as compared with a 1.6% HA solution or the control saline group. Treatment groups 1.6% and 2.4% had a lower Kellgren and Lawrence score, lower macroscopic score and lower modified Mankin score than the control group. There was a statistically significant dose-dependent effect in the macroscopic appearance of cartilage, globally correlating to less severe joint damage.
HA preparations are examples of locally administered treatments that have been considered largely free of systemic harms, albeit with some controversy about their efficacy. Nevertheless, a recent systemic review has raised concerns about the safety profile of HA. 37
The present preclinical study demonstrates that while increasing the HA concentration from 1.6% to 2.4%, no severe adverse events can be seen. On the other hand, an increase in the concentration improves the efficacy of the treatment, as demonstrated by radiographs and histological analysis (
In conclusion, the present study opens up the path to use Sinovial at a high concentration as a single injection to prevent cartilage degradation. Further studies and more evidences are deemed necessary to demonstrate this protective activity on human cartilage.
Footnotes
Authors Contributions
All authors were involved in drafting the article and revising it critically for important intellectual content and all authors approved the final version to be submitted for publication.
Acknowledgments and Funding
The authors are indebted to Miss Carol Benett for reviewing the English of the manuscript. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by IBSA Institut Biochimique SA, Pambo Noranco, Switzerland.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Aginko Research (Pomme Boissier and Pierre Mainil-Varlet) received an educational grant from IBSA Institut Biochimique SA for this preclinical study. Gisueppe Mautone is an employee of IBSA.
Ethical Approval
Ethical approval for this study was obtained from the IACUC and the Committee for Animal Protection of the Ministry of Health of the Czech Republic (Project Number 27, 2014).
Animal Welfare
The present study followed international, national, and/or institutional guidelines for human/animal treatment and complied with relevant legislations.
