Abstract
Objective
Our study was performed to investigate whether micro-223 promotes diabetic Osteoarthritis (OA) progression by regulating cartilage degeneration and subchondral bone remodeling.
Methods
The expression of miR-223 in human normal cartilage, OA cartilage, and subchondral bone tissue with or without DM was detected by real-time quantitative reverse transcription polymerase chain reaction (qRT-PCR). miR-223 mimic or inhibitor was transfected into chondrocytes. Cell viability and apoptosis were assessed by 3-(4,5)-dimethylthiahiazo(-2)-3,5-diphenyltetrazolium bromide (MTT) and Terminal Deoxynucleotidyl Transferase(TdT)-mediated dUTP nick end labeling (TUNEL) assay, respectively.
Results
miR-223 was significantly higher in human diabetic OA cartilage and subchondral bone compared with normal OA and healthy control. Overexpression of miR-223 accelerated cartilage degeneration and subchondral bone sclerosis in diabetic OA mice, whereas miR-223 inhibition had the opposite effect. In vitro upregulation of miR-223 decreased proliferation and enhanced apoptosis of chondrocytes. Meanwhile, downregulation of miR-223 promoted glycosaminoglycan (GAG) production in chondrocytes.
Conclusion
miR-223 promotes diabetic OA progression by regulating cartilage degeneration and subchondral bone remodeling both in vitro and in vivo.
Introduction
Osteoarthritis (OA) is usually defined as a cartilage degenerative disease, which leads to progressive chondrocyte loss and cartilage failure. 1 Meanwhile, subchondral bone deterioration is commonly associated with articular cartilage defects and subchondral bone sclerosis, together with progressive cartilage degradation, is widely considered a hallmark of OA. 1 OA and type 2 diabetes mellitus (T2DM) are the two most common diseases that affect global population. 2 T2DM and OA often coexist partially because of their high prevalence and shared risk factors. For example, the correlation of OA with obesity is well known, 3 and a majority of people with T2DM suffer from obesity problems. 4 In addition, aging serves as a definite risk factor for both T2DM and OA. The estimated prevalence of T2DM in the United States is 4.6 million among individuals aged 18-44 years, and rises to 14.3 million people aged 45-64 years and 12.0 million people older than 65 years. 5 Similarly, occurrence of knee OA increases significantly along with age, affecting 14% of adults older than 25 years and 37% of those above the age of 60 years. 6 The concept of “diabetic OA” or “diabetes-induced OA phenotype” was first proposed by Berenbaum. 7 It is believed that hyperglycemia is one of the most important triggers of joint degradation in OA. Local increased concentration of glucose alters the cartilage matrix by increasing the formation of advanced glycation end products (AGEs) that can, in turn, activate chondrocytes and synoviocytes to produce prodegradative and pro-inflammatory mediators and modify the quality of the subchondral bone. 7 However, the potential mechanism that how hyperglycemia promotes OA progression remains partially unclear.
MicroRNAs (miRNAs) are a class of noncoding RNAs that regulate gene expression by binding the 3′ untranslated region (UTR) of their target messenger RNAs (mRNAs) and perform translational repression or degradation of mRNA. 8 miRNAs have been implicated not only in developmental and homeostatic events of vertebrates and invertebrates 9 but also are important mediators of cell-to-cell communication and coordinate biological functions including angiogenesis, cell proliferation, and immune response. 10 Moreover, miRNAs have been implicated as important players in the pathogenesis of various human diseases, including cancer, cardiovascular diseases, neural disease, viral infection, and immune diseases.11-15
In recent years, the miRNAs are found to be implicated in T2DM progression and OA pathophysiology. In terms of T2DM, modifications in the miRNA expression profile could lead to compensatory pancreatic β-cell mass expansion occurring under insulin resistance conditions in T2DM. 16 In addition, a growing number of studies suggested that a series of miRNAs are involved in diabetic complications. 17 For example, miR-92a has been found to protect pancreatic β-cell function by targeting Kruppel-like factor 2 (KLF2) in DM. 18 miR-200a has been shown to facilitate diabetic endothelial dysfunction via targeting Kelch-like ECH-associated protein 1/nuclear factor erythroid 2-related factor 2 (KEAP1/NRF2). 19 miR-26a plays an important role in regulating diabetic wound healing. 20 Meanwhile, miRNAs could perform both aggressive and protective roles during OA progression. For example, miR-103 could result in OA development by targeting Sox6. 21 Endisha also found that miR-34a-5p is found to promote joint destruction during OA progression. 22 Liebling found miR-181a inhibition could attenuate OA in facet and knee joints. 23 On the other hand, Ren found miR-140-3p ameliorates the progression of OA by targeting C-X-C motif chemokine receptor 4 (CXCR4). 24 Also, miR-320c inhibits development of OA through downregulation of Wnt signaling. 25 In addition, miRNA-335 relieves chondrocyte inflammation by activating autophagy in OA. 26
miR-223 is located on the X chromosome governed by an independent promoter not related to other gene products.27,28 Global miRNA expression analyses revealed that miR-223 is upregulated in many inflammation-related disorders, including rheumatoid arthritis (RA), 29 OA, 30 and inflammatory bowel disease (IBD). 31 Meanwhile, miR-223 is also upregulated in T2DM. 32 Lu et al. 33 showed an increased miR-223 levels in the diabetic heart and participated an adaptive/homeostatic response to restore Glut4 expression and glucose uptake. They showed that overexpression of miR-223 in cardiomyocytes increased Glut4 level and induced Glut4-mediated glucose uptake. 33 miR-223 has also been upregulated in many inflammatory states. For example, overexpression of miR-223 has been reported in the colonic mucosa of patients with ulcerative colitis (UC). 34 One study has shown that lentivirus-mediated silencing of miR-223 can reduce disease severity of experimental arthritis. 35 In addition, miRNA-223 could trigger inflammation in porcine aorta by activating Nod-like receptor family protein 3 (NLRP3) inflammasome. 36 These findings indicate that miR-223 is elevated in both DM and OA and acts as a pro-inflammatory factor in both situations.
Previous study demonstrated that peroxisomal dysfunction is associated with upregulation of apoptotic cell death via miR-223 induction in knee OA patients with T2DM. 37 Overexpression of miR-223 stimulates apoptotic cell death in human articular chondrocytes and induced severe cartilage destruction in diabetic mice. 37 All these studies indicated that miR-223 are important regulators for both T2DM and OA.
However, to the best of our knowledge, there were no studies available illustrating the potential role of miR-223 on diabetic OA and revealing its function on the interaction between DM and OA. Therefore, this study was carried out to explore the function of miR-223 on diabetic OA progression.
Materials and Methods
Cartilage and Subchondral Bone Specimen Collection and Ethics Statement
Articular cartilage and subchondral bone tissue were obtained from primary OA patients receiving total knee replacement surgery (n = 45, 19 men and 26 women, aged 55-75 years), including 23 patients with diabetes and 22 patients without diabetes. Normal cartilage samples and subchondral bone tissues were obtained from 20 donors (10 men and 10 women, aged 54-72 years) following trauma or death. All samples were collected from knees and stored at −80 °C after promptly frozen by liquid nitrogen. There were no significant differences with regard to age and sex distribution among the diabetic OA, normal OA, and control groups. Informed consents were obtained from all patients before specimen collection. All protocols were carried out based on the Declaration of Helsinki, and the experiments were approved by the ethics committee of our hospital. The homeostasis model assessment of insulin resistance (HOMA-IR) was calculated in diabetic OA patients.
qRT-PCR Analysis
Total RNA was extracted from the chondrocytes by using TRIzol reagent (Invitrogen). The cDNA Synthesis Kit (Takara, Japan) was used for the synthesis of cDNA according to the manufacturer’s instructions. miRNA isolation was performed using the mirVana miRNA Isolation Kit (Life Technologies) followed by reverse transcription using the TaqMan miRNA Reverse Transcription Kit (Life Technologies). miR-223 quantitative polymerase chain reaction (qPCR) was performed using TaqMan miRNA assays (Life Technologies). RNA was then reverse transcribed into cDNAs using the Reverse Transcription System Kit (Takara, China). The cDNA templates were amplified by quantitative reverse transcription polymerase chain reaction (qRT-PCR) using SYBR Green PCR Mix (Takara). The miRNA housekeeping gene U6 was used as a control and other genes were normalized to β-actin. miR-223-F,5′-CGCUAUCUUUCUAUUAACUGACCAUAA-3′, miR-223-R,5′-CGCUAUCUUUCUAUUAUGACUCCAUAA-3′; COL2A1-F, 5′-atgacaatctggctcccaacactgc-3′, R-5′-gaccggccctatgtccacaccgaat-3′; MMP-13-F-5′-CGCCAGAAGAATCTG TCTTTAAA-3′; R-5′-CCA AAT TAT GGAGGAGATGC-3′. MMP-3 F-5′-TGCTGCTCATGAAATTGGCC-3′, R-5′-TCATCTTGAGACAGGCGGAA-3′; Aggrecan-F-5′-ACTCTGGGTTTTCGTGACTCT-3′, R 5′-ACACTCAGCGAGTTGTCATGG-3′; U6-F, 5′-CTCGCTTCGGCAGCACA-3′, R-5′-AACGCTTCACGAATTTGCG-3′; β-actin-F-5′-TCGTGCGTGACATTA AGGAG-3′, and R-5′-GTCAGGCAGCTCGTAGCTCT-3′.
Isolation and Culture of Primary Chondrocytes
To prepare the primary chondrocytes, the collected normal and OA cartilage was minced and pretreated with trypsin for 10 minutes. Then, the tissue slices were digested overnight (from 18:00, >15 h) with collagenase II in Dulbecco’s Modified Eagle Medium (DMEM) containing 10% fetal calf serum (FCS). The isolated cells were then passed through a filter to remove the residual cartilage matrix fragments, followed by centrifugation at 2,000 × g for 5 minutes. Afterward, cells were resuspended in DMEM supplemented with 10% FCS and antibiotics consisted of 100-U/ml penicillin and 100-μg/ml streptomycin. During experiments, the cells were subsequently cultured in the medium supplemented with
Fluorescence In Situ Hybridization for miR-223 in Chondrocytes
Fluorescence in situ hybridization (FISH) was performed as previously described. 40 Briefly, chondrocyte cells grown on the slides were washed with phosphate-buffered saline (PBS) and fixed in 4% paraformaldehyde. After protease reagent treatment, the slides were incubated with prehybridization buffer at 40 °C for 4 hours, and then hybridized with digoxin-labeled probe at 40 °C overnight. After washing and blocking, the slides were incubated with biotin-conjugated anti-digoxin antibody. The slides were then incubated with Cy3-fluorescein isothiocyanate (FITC) at 37 °C for 30 minutes after washing. The images were captured using a confocal microscope.
MiR-223 Transfection
Chondrocytes treated were digested and cultured in 6-well plates at 2.5 × 105 cells per well, with each well containing 2-ml DMEM/F12 with 5% fetal bovine serum. When the chondrocytes covered approximately 80% of the well area, they were transfected with miR-223 mimic/inhibitor (RiboBio, Guangzhou, China) using Lipofectamine 3000 (Thermo Fisher Scientific, Waltham, MA). Forty-eight hours post-transfection, the cells were treated with 30 nM of glucose for an additional 24 hours. After transfection for 24 hours, the culture medium was changed, and the culture was continued for an additional 24 hours.
Viability of OA Chondrocytes by MTT Assay
Chondrocytes (untransfected or transfected with miR-223 mimic/inhibitor) were seeded in 96-well plates (3 × 103 cells/well, 10 replicates per group). After incubation for 24 hours, 20 μl of 3-(4,5)-dimethylthiahiazo(-2)-3,5-diphenyltetrazolium bromide (MTT) reagent (5 mg/ml) was added, followed by incubation at 37 °C for 4 hours. The supernatant was aspirated, and 200 μl of dimethyl sulfoxide (DMSO) was added followed by shaking for 15 minutes. The absorbance was measured with a spectrophotometer at a wavelength of 490 nm.
TUNEL Assay
A TdT-mediated dUTP nick end labeling (TUNEL) Kit (Roche, Switzerland) was used for the detection and quantification of apoptosis. In brief, cells were fixed with 4% paraformaldehyde for at least 10 minutes after transfection and washed 3 times with PBS. Cells were then treated with 0.5% Triton X-100 for 20 minutes to permeabilize the cell membrane. Then, the cells were treated with the TUNEL reaction mixture in the dark for 30 minutes. Images were taken under a fluorescence microscope.
Glycosaminoglycan Measurement
OA chondrocytes treated with high concentration of glucose (untransfected or transfected with miR-223 mimic/inhibitor) were seeded in 6-well plates (5 × 105 cells/well) and cultured at 37 °C with 5% CO2 for 24 hours. A 1,9-dimethylmethylene blue assay (Sigma) was used to determine the GAG content according to the manufacturer’s instructions. Absorbance was measured at a wavelength of 525 nm using a fluorescence microplate reader.
Experimental Diabetic OA Mice Model
The diabetic OA model was established according to our previously published study. 41 60 C57BL/6 mice were treated humanely and with regard for alleviation of suffering. This animal study was approved and conducted by local institutional animal care and use committee. Mice were housed five per cage under standard laboratory conditions with 12-hour light/dark cycles.
The normal knee OA model was induced following transection of the anterior cruciate ligament (ACL) in the right knee, while the remaining 10 naive mice were referred as the control. Forty mice were then intraperitoneally injected with 100-mg/kg streptozotocin (STZ; Sigma) to mimic the model of diabetic condition and the remaining mice were regarded as nondiabetic OA. In brief, STZ was dissolved in sodium citrate buffer (pH: 4.5) and injected within 15 minutes of preparation. One week after STZ injection, the blood glucose levels that reached more than 400 mg/dl were defined as diabetic hyperglycemia.
Lentiviral miR-223 Precursors and Antisense Oligonucleotide
Expression vectors pMIF-cGFP-zeo (System Biosciences, Palo Alto, CA) encoding miR-223, and pmiR-ZIP-shRNA encoding miR-223 antisense oligonucleotides (miR-223-AS) (AM11746; Applied Biosystems Inc., Foster City, CA) were constructed, respectively. Expression vectors and pPACKF1 vectors were transferred into 293T cells. Lentiviral particle titers (infectious units/ml) were quantified using Lenti-X qRT-PCR Titration Kits (Clontech Laboratories Inc., San Francisco, CA) following enrichment at 1,000,000 × g for 60 minutes at 4 °C. Ten microliters of aliquots of lentiviral particle mixtures containing 1 × 109 infectious units/ml were then prepared for intra-articular injection.
Micro-Computed Tomography
Samples were analyzed under high-resolution mCT (SkyScan1176; Toyo Corporation, Tokyo, Japan), using the following parameters: source voltage, 40 kV; source current, 580 mA; pixel size, 12.47 mm; and spatial resolution, 9 mm. Images were reconstructed for analysis (CTanalyzer; Toyo Corporation). For human samples, cylindrical regions (0.5 mm in diameter and 0.5 mm in height) were obtained from the region of interest (ROI), defined as the area of greatest sclerotic change in the subchondral bone. In mice, ROI was the subchondral bone of the medial tibial plateau, with the bone volume/tissue volume (BV/TV, %) ratio.
TRAP Staining
Tartrate-resistant acid phosphatase (TRAP) staining was performed using a commercially available kit (Wako Pure Chemical Industries, Ltd., Osaka, Japan), according to the manufacturer’s protocol. TRAP-positive multinucleated cells, containing more than three nuclei, were recognized as osteoclasts and counted in the subchondral bone using Image J.
Statistical Analysis
Continuous data are expressed as the mean ± standard deviation (SD), and statistical analysis was performed by SPSS 20.0 (IBM Corp., Armonk, NY). The means of multiple groups were compared by one-way analysis of variance (ANOVA). Values of P < 0.05 or 0.01 were considered statistically significant.
Results
MiR-223 Was Increased in Human and Mice Diabetic OA Cartilage and Subchondral Bone
The expression of miR-223 in human and mice diabetic OA cartilage tissues, subchondral bone, and isolated chondrocytes was examined by qRT-PCR. qRT-PCR analysis showed that miR-223 expression was significantly higher in human and mice diabetic OA cartilage in comparison with nondiabetic OA cartilage (*P < 0.05 vs. nondiabetic OA) and normal cartilage (#P < 0.05 vs. normal control) (
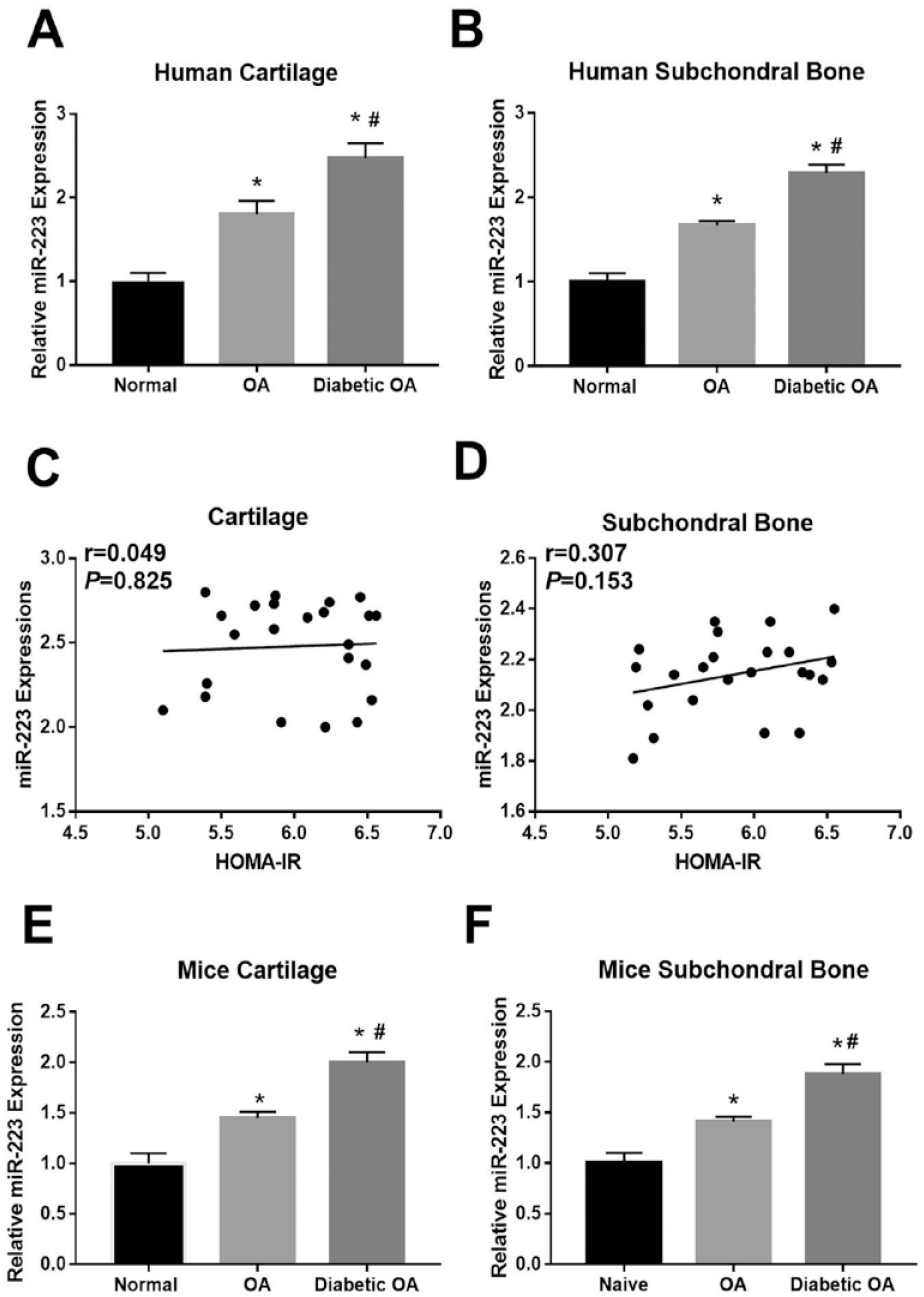
(
We further isolated chondrocytes from normal cartilage and OA cartilage and compared miR-223 expression using FISH (
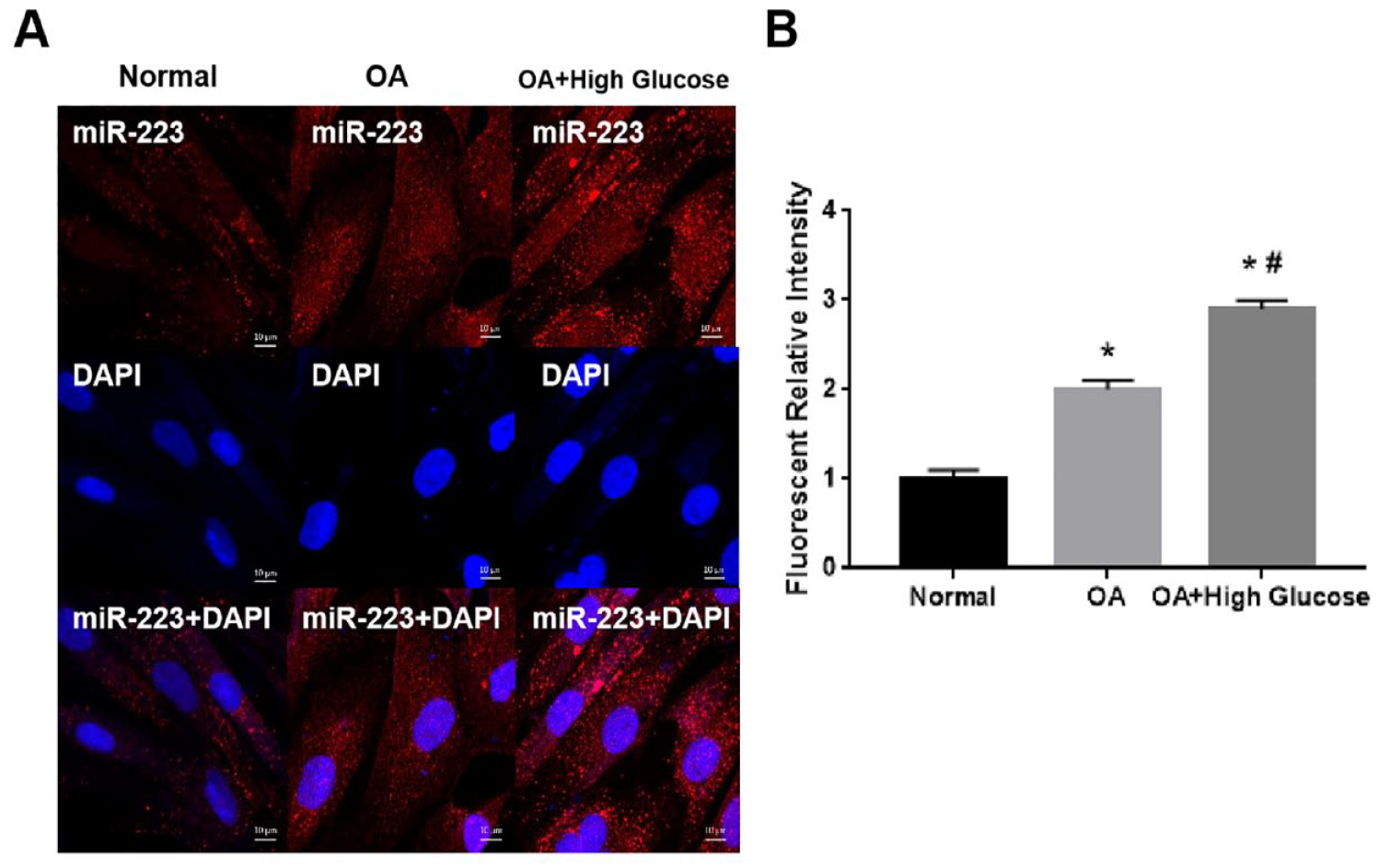
(
Effect of miR-223 on OA Chondrocyte Viability and Apoptosis
The effect miR-223 on OA chondrocyte viability and apoptosis treated or untreated with high glucose was further investigated. We found the absorbance values of human OA chondrocytes were significantly lower than those of normal chondrocytes (P < 0.05), indicating reduced viability of OA chondrocytes. Furthermore, when treated with 30-nM glucose, absorbance values were further markedly reduced. However, following transfection of the miRNA-223 inhibitor into OA chondrocytes with high glucose, the absorbance values of chondrocytes significantly increased compared with those of OA chondrocytes with the high-glucose group (P<0.05), indicating that inhibition of miR-223 could enhance the viability of OA chondrocytes. Overexpression of miR-223 may have exhibited the opposite effect (
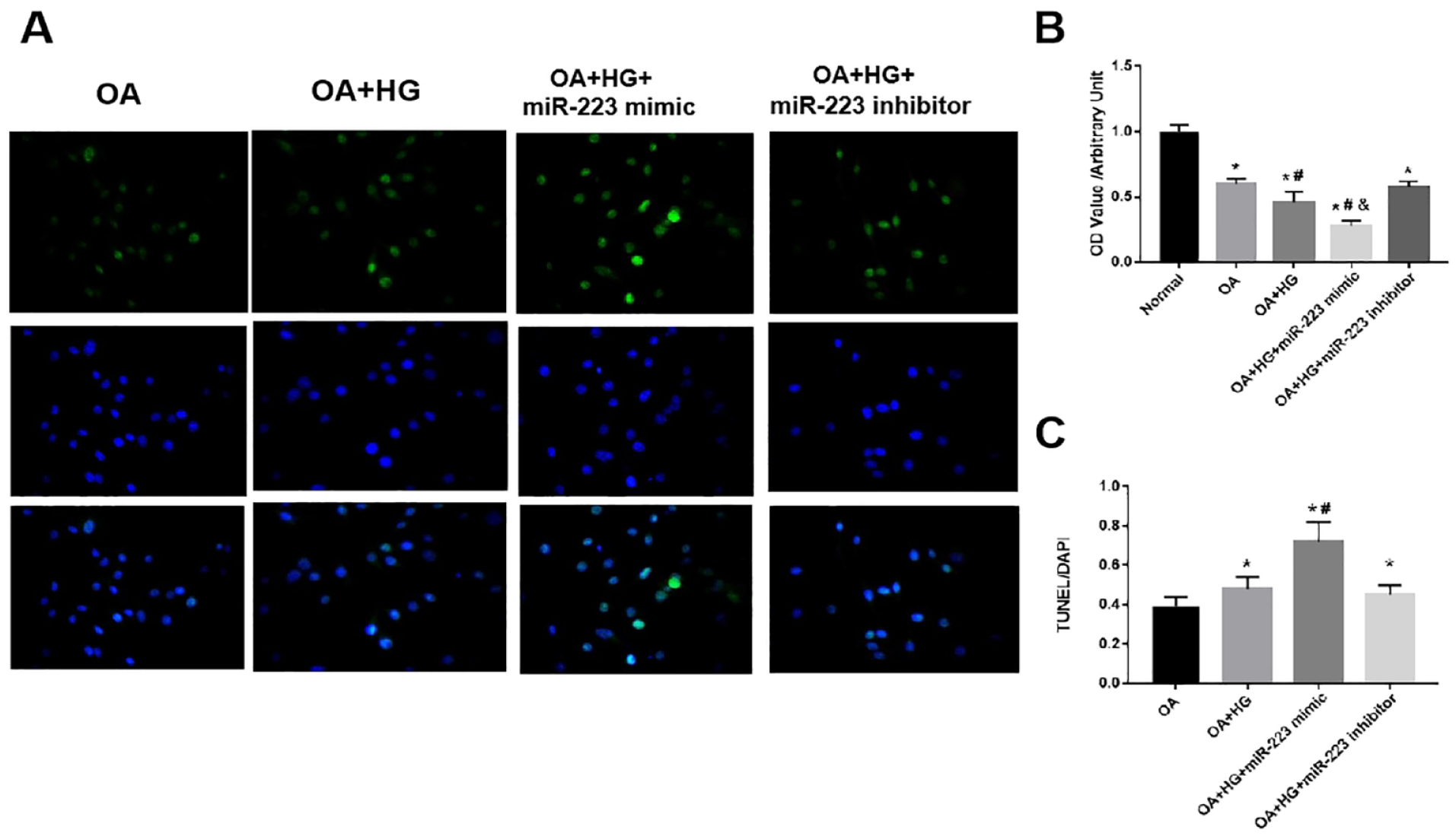
Effect of miR-223 on OA chondrocyte viability and apoptosis treated or untreated with high glucose. (
We performed TUNEL assay to further explore the effect and the potential role of miR-223 in apoptosis in high-glucose-treated OA chondrocytes. We found the OA + HG + miR-223 mimic group showed significantly increased number of apoptotic cells than the OA + HG group, whereas the OA + HG + miR-223 inhibitor group demonstrated significantly decreased number of apoptotic cells (
Effect of miRNA-223 on the Glycosaminoglycan Content of OA Chondrocytes
The effect of miRNA-223 mimic transfection on the glycosaminoglycan (GAG) content of human OA chondrocytes was examined using a GAG kit. As can be seen in Figure 3, the GAG content in human OA chondrocytes was significantly lower than that in normal chondrocytes, whereas high glucose treatment for OA chondrocytes further markedly decreased the GAG content. Moreover, miRNA-223 mimic transfection further drastically reduced the GAG content. However, on the other hand, transfection with the miRNA-223 inhibitor into high-glucose-treated chondrocytes could significantly increase the GAG content (
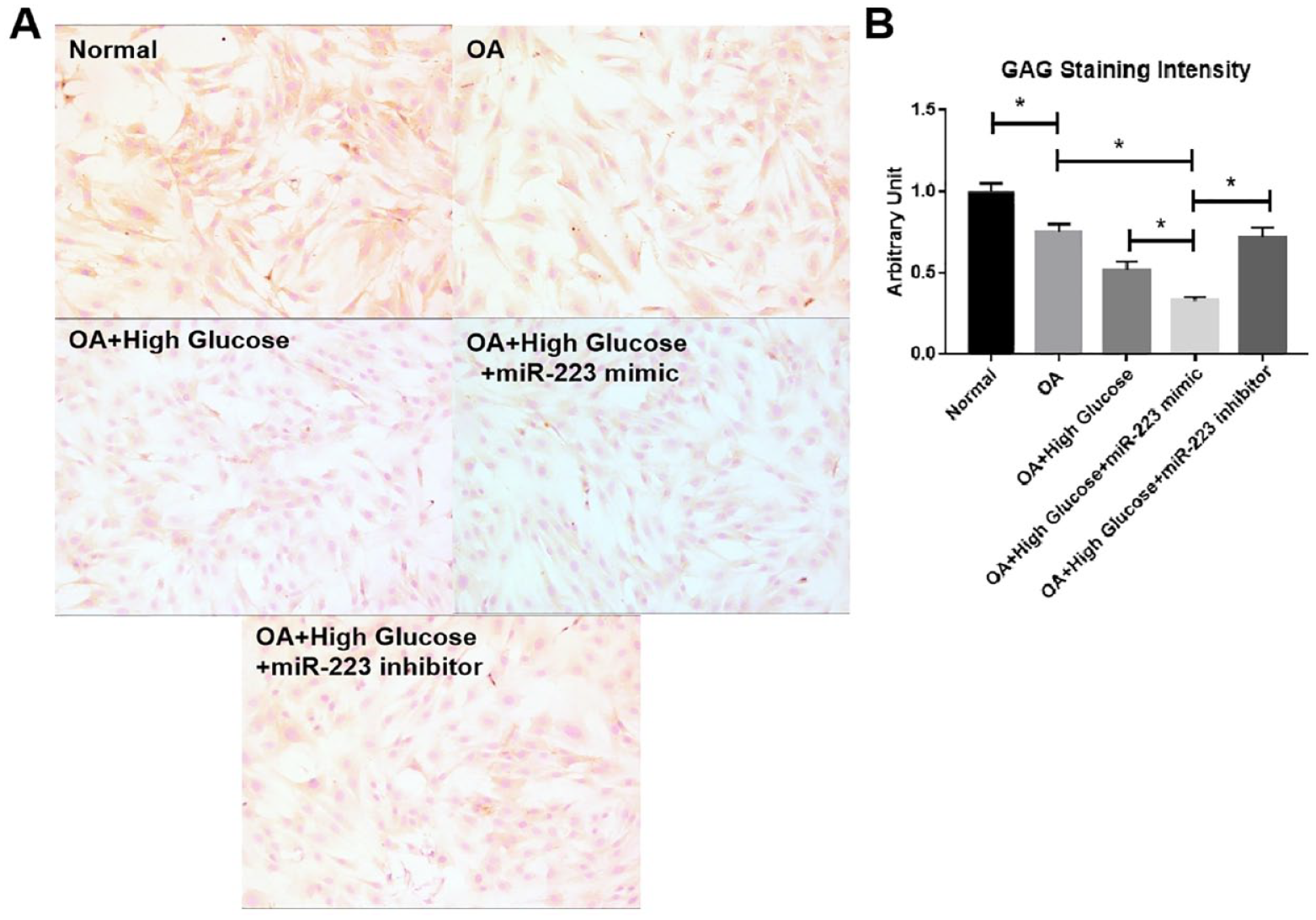
The effect of miRNA-223 on intracellular GAG production in human chondrocytes was measured using a GAG assay. (
Effects of miR-223 on the mRNA Levels of Cartilage Matrix Components and Matrix-Degrading Enzymes in Human Chondrocytes
To further investigate the mechanism of the effect of miRNA-223 on cartilage matrix components and matrix-degrading enzymes in OA chondrocytes, human OA chondrocytes treated with high glucose transfected with miR-223 mimic or inhibitor were used, followed by assessment of the expression of type 2 collagen A1 (COL2A1), aggrecan, matrix metalloproteinase-3 (MMP-3), and matrix metalloproteinase-13 (MMP-13) by qRT-PCR. As demonstrated in
Figure 5
, the mRNA expressions of COL2A1 and aggrecan were drastically lower, whereas MMP-3 and MMP-13 were markedly increased in the OA + HG group compared with the normal and OA groups (*P < 0.05 vs. normal, #P < 0.05 vs. OA) (
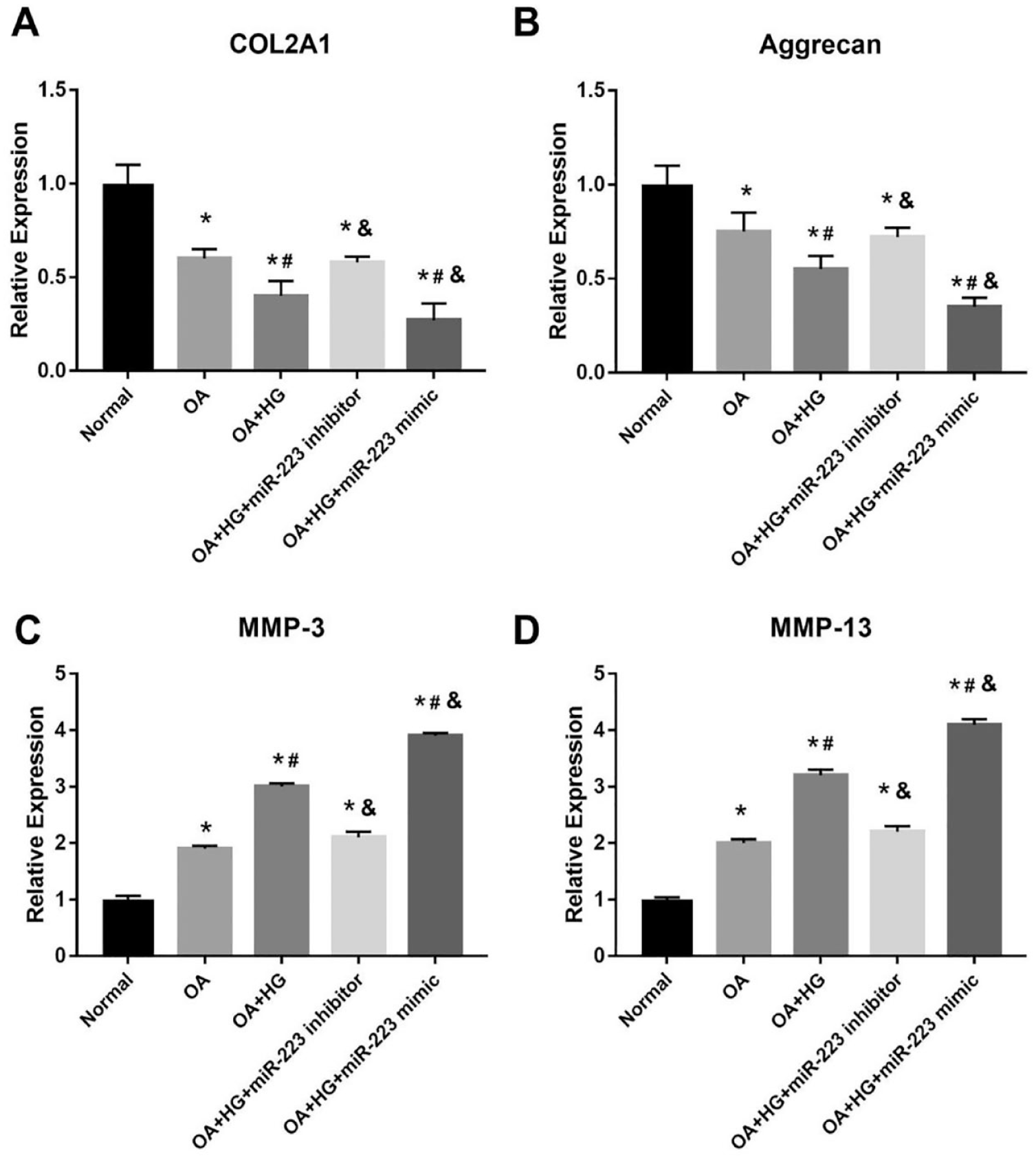
The effects of miR-223 on the mRNA levels of cartilage matrix components and matrix-degrading enzymes in human articular chondrocytes. (
Effect of miRNA-223 on the Cartilage Changes in Diabetic OA Mice Model
All mice survived the experimental procedures, without experiencing any adverse events. The pure OA group showed progressive degenerative change compared with the normal group. Furthermore, the diabetic OA mice group exhibited much more cartilage degeneration than pure OA mice and adding miR-223 intrainjection further worsened cartilage damage, whereas this condition could be attenuated by adding miR-223 antisense intra-injection (Fig. 6). Accordingly, The OARSI score in the diabetic OA group was significantly higher than that in the normal control group and OA group at 8 weeks (P < 0.05; Fig. 6). The diabetic OA + miR-223 OE group showed higher Osteoarthritis Research Society International (OARSI) scores, whereas the diabetic OA + miR-223 AS group showed lower OARSI scores in comparison with the diabetic OA group (P < 0.05; Fig. 6).
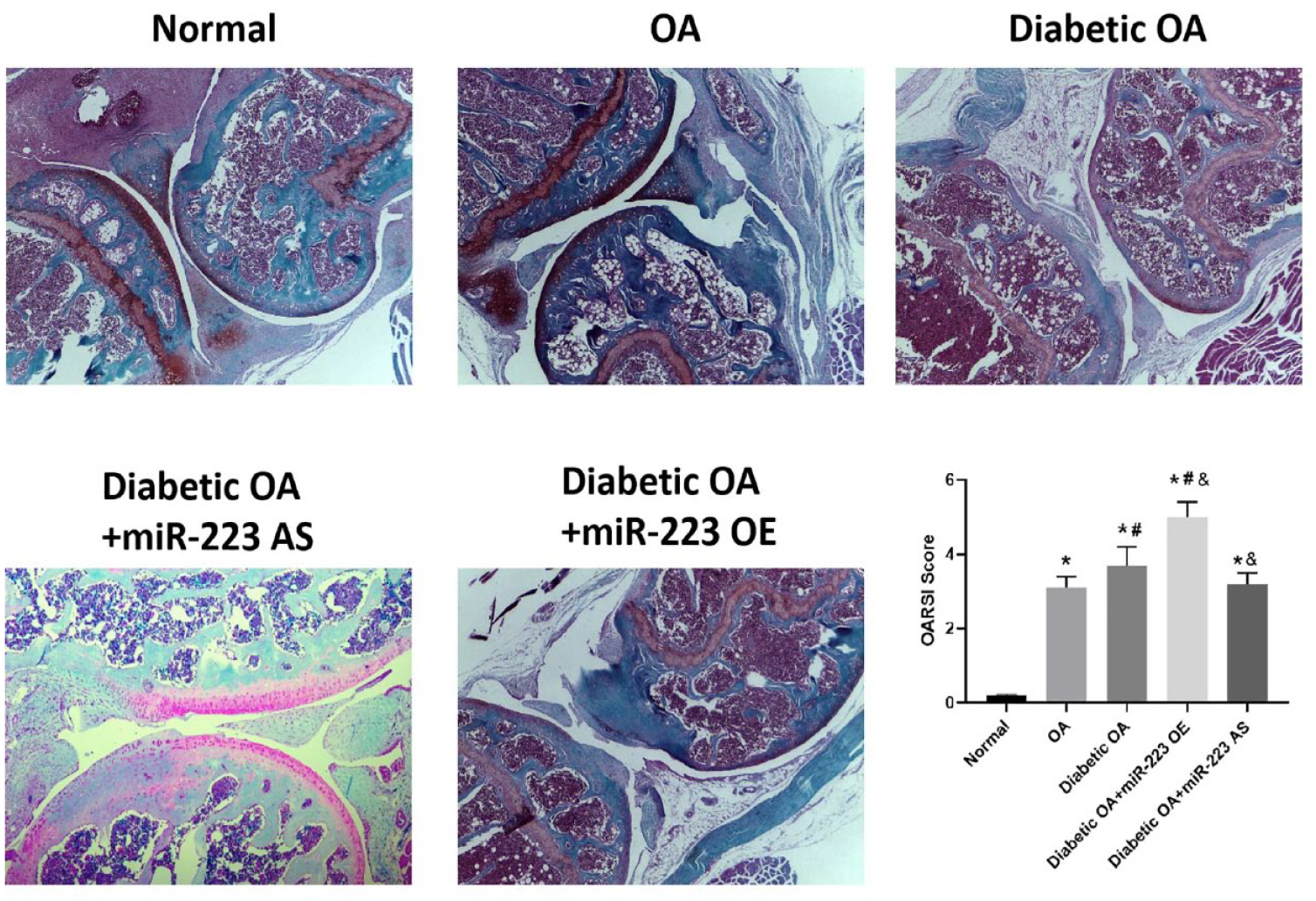
Representative figures of Safranin O (SO) staining of the knee joint in each group and the OARSI score was used to quantify the staining results. Data were expressed as mean ± SD. *P < 0.05 versus normal, #P < 0.05 versus OA, &P < 0.05 versus diabetic OA. OARSI = Osteoarthritis Research Society International; OA = osteoarthritis; OE = overexpression.
Effect of miRNA-223 on the BV/TV Ratio and Osteoclastic Activity in Subchondral Bone
Analysis of mCT images showed that the BV/TV ratio of the medial plateau’s subchondral bone in the miR-223 overexpression group was significantly higher than that in the miR-223-OE NC group at 8 weeks (P < 0.05;
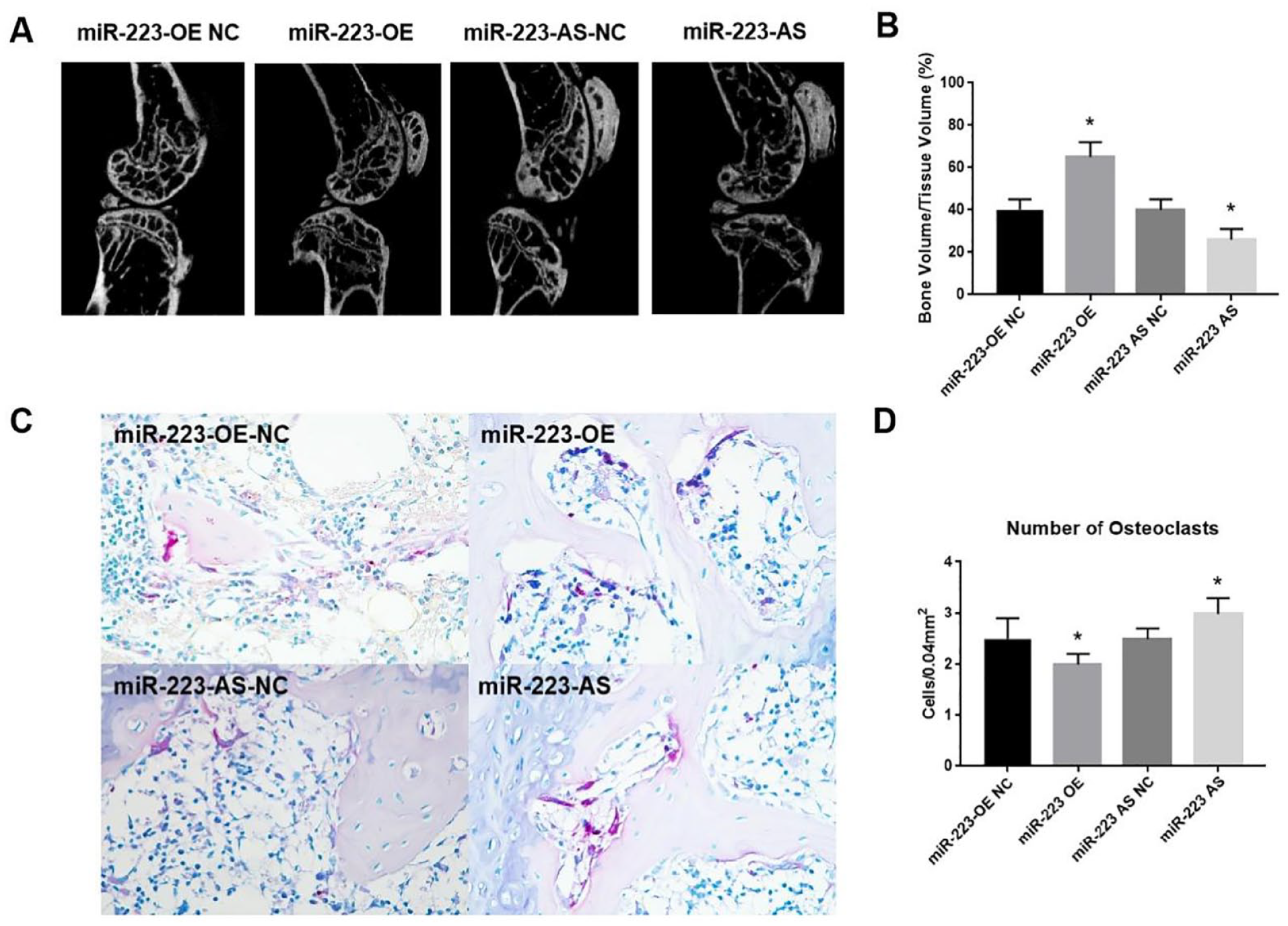
Effect of miRNA-223 on the BV/TV ratio and osteoclastic activity in subchondral bone. (
Accordingly, the ratio of TRAP-positive cells to bone marrow cells in mice in the miR-223-OE group was significantly lower than that in mice in the miR-223-OE NC group (P < 0.05; Fig. 7C and
Discussion
In the past decades, the detailed factors that linked T2DM with OA have been widely explored, although they remain incompletely known. Generally, T2DM exerts a pathogenic effect on OA via oxidative stress and low-grade chronic inflammation resulting from chronic hyperglycemia and insulin resistance. In addition, T2DM acts potentially as a risk factor for OA progression and has a negative impact on arthroplasty outcomes. 42 However, molecular mechanisms underlying this link between the two diseases were not fully investigated yet.
This study investigated the potential role of miR-223 in the development of diabetic OA. Along with the previous studies, our findings revealed that miR-223 not only influenced the metabolism of cartilage but also participated in the process of bone remodeling of subchondral bone, based on the clinical sample examinations, in vitro cell study, and in vivo animal model. This work implicated miR-223 may serve as important in leading to diabetic OA progression. The potential mechanism was illstrated as figure 8 (Figure 8).
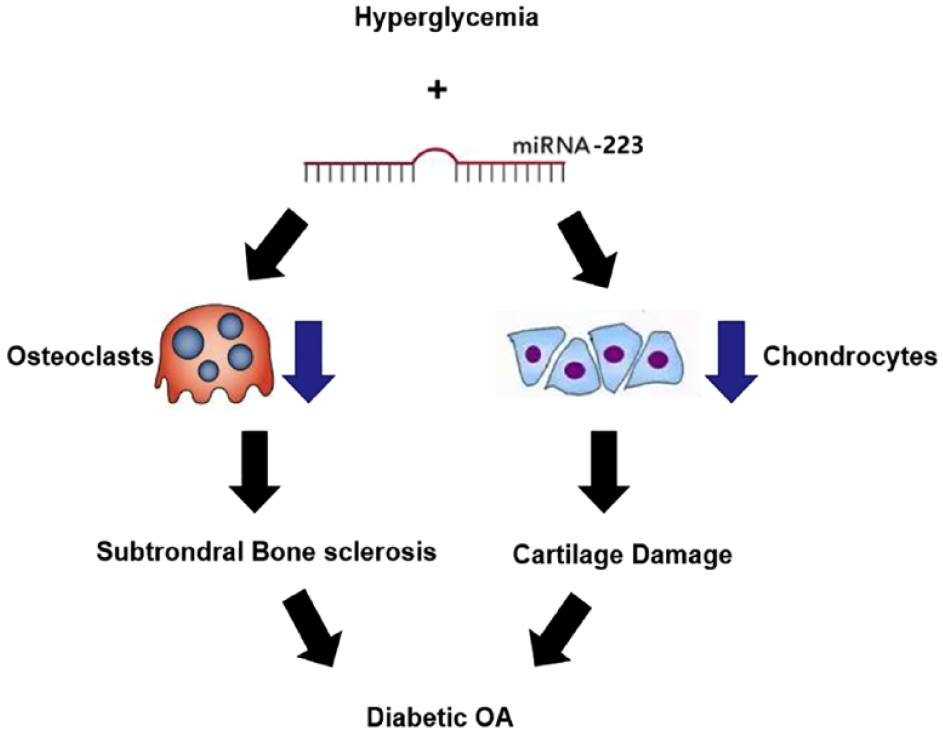
Schema of diabetic OA progression through miR-223 to cartilage and subchondral bone metabolism. OA = osteoarthritis.
We first found that miR-223 expression was significantly upregulated in the diabetic OA group in comparison with OA and normal cartilage and subchondral bone, indicating miR-223 is both involved in the process of cartilage damage and subchondral bone remodeling. Previous studies showed that miR-223 expression was higher in the OA group than in normal controls, 43 and lentivirus-mediated silencing miR-223 could ameliorate collagen-induced arthritis in mice. 43 However, there are no significant differences with regard to the cartilage change defined by OARSI scores during intrainjection with miR-223 OE or miR-223 AS into the normal control mice, indicating miR-223 may perform its function under inflammation states.
miR-223 has also been implicated in many DM complications, and inhibition of miR-223 could also relieve these complications. For example, inhibition of miR-223 attenuates the NLRP3 inflammasome activation, fibrosis, and apoptosis in diabetic cardiomyopathy. 44 miR-223 was also been proved to participate in the pathogenesis of diabetic retinopathy. 45 qRT-PCR assay showed that the level of miR-223 was overexpressed in DM samples and human retinal endothelial cells (hRECs) in hyperglycemia. 45
We next transfected miR-223 mimic and inhibitor into chondrocytes treated with high glucose, and we found miR-223 overexpression caused decreased chondrocyte viability and GAG production along with increased chondrocyte apoptosis, whereas miR-223 inhibition had opposite effect. In consistent with another study, Kim found peroxisomal dysfunction is associated with upregulation of apoptotic cell death via miR-223 induction in knee OA patients with T2DM, and overexpression of miR-223 caused apoptotic cell death in chondrocytes and contribute to severe cartilage destruction in diabetic mice. 37 Besides, we found miR-223 could also affect cartilage matrix components COL2A1 and aggrecan as well as matrix-degrading enzymes MMP-3 and MMP-13.
In addition to cartilage damage, subchondral bone remodeling also plays important roles in diabetic OA progression. Chen et al. 46 investigated alterations in subchondral bone remodeling, microstructure, and strength in knees from T2D patients and their association with cartilage degradation and found abnormal bone remodeling may contribute to the early pathogenesis of T2D-associated knee OA. We found overexpression of miR-223 accelerates subchondral bone sclerosis in diabetic OA mice, whereas miR-223 inhibition had opposite effect. One previous study explored the effect of upregulation of miR-223 on osteoclastogenesis induced in vitro by RANKL. The findings demonstrated that a number of TRAP-positive osteoclasts were drastically reduced, indicating that signaling pathways downstream of RANKL are the targets of miR-223. 29
There were some limitations that should be noted. First, the detailed mechanism that miR-223 causing cartilage damage and subchondral bone was not investigated and should be explored in the future study. Next, only miR-223 was detected in this study, and other noncoding RNAs that involved both DM and OA situations also need examination in the future.
Conclusion
Collectively, on the basis of our clinical findings that miR-223 is upregulated in both diabetic OA cartilage and subchondral bone, in further study, we also observed that in both in vitro hyperglycemia condition and diabetic OA model, overexpression of miR-223 facilitates collagen degradation and subchondral bone remodeling. Further exploration of the intensified mechanisms responsible for miR-223 involved in diabetes-induced OA needs warrant, thus developing novel and effective therapeutic strategies for this chronic condition.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Science and Technology Program of Guangzhou, China (grant number: 2021324353) and the Fundamental Research Funds for the Central Universities (grant number: 21620452).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This study was approved by the First Affiliated Hospital, Jinan University. All the patients signed the written informed consent.
Availability of Data and Materials
The data sets used and/or analyzed during this study are available from the corresponding author on reasonable request.
