Abstract
Objective
Septic arthritis results from joint infection by Staphylococcus aureus, which produces potent α-toxin causing cell death, potentially leading to permanent cartilage damage. Treatment is by joint irrigation and antibiotics, although it is unclear if, following treatment with antibiotics which cause bacterial lysis, there is release of additional stored α-toxin.
Design
A rabbit erythrocyte hemolysis assay was optimised to assess biologically-active α-toxin from cultured S. aureus α-toxin strain DU5946. Hemoglobin release was measured spectrophotometrically following addition of a bacteriostatic antibiotic (linezolid) or a bacteriolytic antibiotic (penicillin). A bovine cartilage model of septic arthritis was used to test the protective effects of antibiotics against S. aureus infection.
Results
During S. aureus culture, α-toxin levels increased rapidly but the rate of rise was quickly (within 20 minutes) suppressed by linezolid (25 μg/mL). Penicillin also reduced the increase in α-toxin levels; however, the time course was relatively slow compared to linezolid even at high concentrations (50,000 U/mL). The efficacy of penicillin (250,000 U/mL) at reducing the rise in α-toxin was approximately 8% less than that of linezolid (P < 0.05) suggesting the presence of additional toxin. This could be due to a delayed action of penicillin, and/or release of a small pool of stored α-toxin from dying bacteria. In a bovine cartilage model, however, there was no difference between the protection of in situ chondrocytes against S. aureus by penicillin or linezolid (P > 0.05).
Conclusion
The results suggested that equally effective protection of chondrocytes against S. aureus septic arthritis may be obtained by the bacteriostatic or bacteriolytic antibiotics tested.
Introduction
Septic arthritis, resulting from joint inflammation secondary to infection, 1 can be highly destructive, leading to cartilage damage and joint failure.2-4 It affects all ages with an incidence in Western Europe of 4 to 10 cases/100,000 persons/year. 5 The incidence of septic arthritis is rising globally due to various factors, including an aging population, increased use of immunosuppressive agents, musculoskeletal prosthesis, and surgical procedures.5-7 Although numerous bacterial species may induce septic arthritis, Staphylococcus aureus accounts for 40% to 60% of cases2,3,8 with invasive health care–associated methicillin-resistant S. aureus (MRSA) infections accounting for 18 per 100,000 persons/year. 9 The bacteria enter the synovial joint mainly by hematogenous spread, 5 and the infection results in chondrocyte death, leading to cartilage degeneration and tissue erosion.2,3 Chondrocyte death is particularly important as these are the only cell type capable of maintaining cartilage resilience through their regulation of extracellular matrix metabolism. 10 Septic arthritis can lead to osteoarthritis (OA 11 ), which contributes to the high morbidity associated with the disease, resulting in fatal septicemia. 7
S. aureus produces an array of potential virulence factors (e.g., toxins, adhesins), which contribute to colonisation and subsequent tissue destruction. The toxins may damage host cells directly or facilitate evasion of the host immune response. 12 Depending on the strain, S. aureus may release toxins, including α-, β-, and γ-toxins in addition to damaging agents, including Panton-Valentine leucocidin (PVL) and super-antigens (e.g., toxic shock syndrome toxin). 13 A bovine cartilage explant model of S. aureus–induced septic arthritis, however, reported that the “pore forming” α-toxin is the key damaging agent causing rapid chondrocyte death whereas the β- and γ-toxins were relatively ineffectual.14,15 α-Toxin is active against a variety of mammalian cells, but has marked potency against rabbit red blood cells (RBCs) via the ADAM-10 (A Disintegrin And Metalloproteinase domain-containing protein-10) receptor, which is also present on human chondrocytes.16-18 Rabbit RBC hemolysis can be quantified spectrophotometrically and thus is a very useful and sensitive biological assay for S. aureus α-toxin.19,20
Treatment for septic arthritis includes intravenous antibiotics with joint lavage 1 ; however, retrospective studies indicate that some permanent cartilage damage develops in ~50% of cases,2-4 thus its rapid and effective treatment is essential. When patients present, the joint is aspirated to obtain a microbiological profile of the synovial fluid. As identification of the causative microorganism may take ~48 hours, patients are traditionally empirically commenced on intravenous antibiotics in combination with joint lavage. β-Lactam-type antibiotics such as benzyl penicillin (penicillin-G) and flucloxacillin are classed as bacteriolytic as they kill the bacteria through damage to the cell membrane, leading to cell swelling and lysis.21,22 This may release cellular contents, including S. aureus α-toxin, potentially causing additional chondrocyte death above that occurring when the bacteria were alive. In contrast, antibiotics such as linezolid and erythromycin are bacteriostatic at low concentrations by inhibiting bacterial growth and replication, which may then be followed by their removal by the immune system,23,24 whereas at higher levels they are bactericidal.24,25 Thus, these agents can be described as bacteriostatic/bactericidal. The choice of antibiotic for S. aureus septic arthritis treatment is important as it could influence the amount of α-toxin in the synovial space, chondrocyte death and subsequent cartilage damage.
Here, we have used 2 isogenic mutants (DU5946, DU5938) from the well-characterized prophage-cured derivative of NCTC8325 S. aureus 8325-4. 26 NCTC8325 was originally isolated from a patient with joint sepsis and its lineage remains a valuable resource for S. aureus research. 27 These mutants produce the range of toxins26,28,29 and the mutations only affect the synthesis of α-hemolysin (α)-toxin (Hla::Emr), β-hemolysin (Hlb::φ42E), and γ-hemolysin (Δhlg::Tcr). Thus, while both strains produce damaging agents, the mutant strain DU5946 only produces α-toxin and not β- or γ-hemolysin, whereas the DU5938 mutant does not produce α-, β-, or γ-toxins. Levels of biologically-active α-toxin during S. aureus culture, were assessed semiquantitatively using the sensitive rabbit hemolysis assay.19,20 Using this technique, we tested the hypothesis that α-toxin levels in the culture medium would be higher after the addition of high concentrations of a bacteriolytic antibiotic (penicillin G) compared with a bacteriostatic/bactericidal antibiotic (linezolid). The results demonstrated that there was only a small (<10%) further increase in α-toxin levels from S. aureus cultures following penicillin treatment compared with linezolid. We then tested these 2 antibiotics (in addition to 2 other antibiotics, flucloxacillin [bacteriolytic] and erythromycin [bacteriostatic/bactericidal]) for their ability to protect articular chondrocytes in a bovine cartilage model of septic arthritis. The results suggested that both penicillin and linezolid (as well as flucloxacillin and erythromycin) protected chondrocytes with equal efficacy suggesting that there was a negligible level of intracellular α-toxin released from S. aureus following antibiotic treatment.
Materials and Methods
Bacterial Strains and Reagents
Two isogenic mutants of S. aureus 8325-4 (DU5946, DU5938) containing mutations affecting the synthesis of α-hemolysin (α)-toxin (Hla::Emr), β-hemolysin (Hlb::φ42E), and γ-hemolysin (Δhlg::Tcr; Table 1 ) were used. The DU5946 strain produced α-toxin whereas this was not produced by the DU5938 strain, otherwise the strains were identical. Antibiotics were from Sigma-Aldrich, Irvine, UK. Fresh, filter-sterilized stock solutions of linezolid (PZ0014), Na-flucloxacillin (F0150000), and penicillin G (P3032;1477U/mg) were prepared in dH2O whereas erythromycin (E6376) was dissolved in ethanol-water (6 mg/mL).
The Bacterial Strains Used in the Study. a
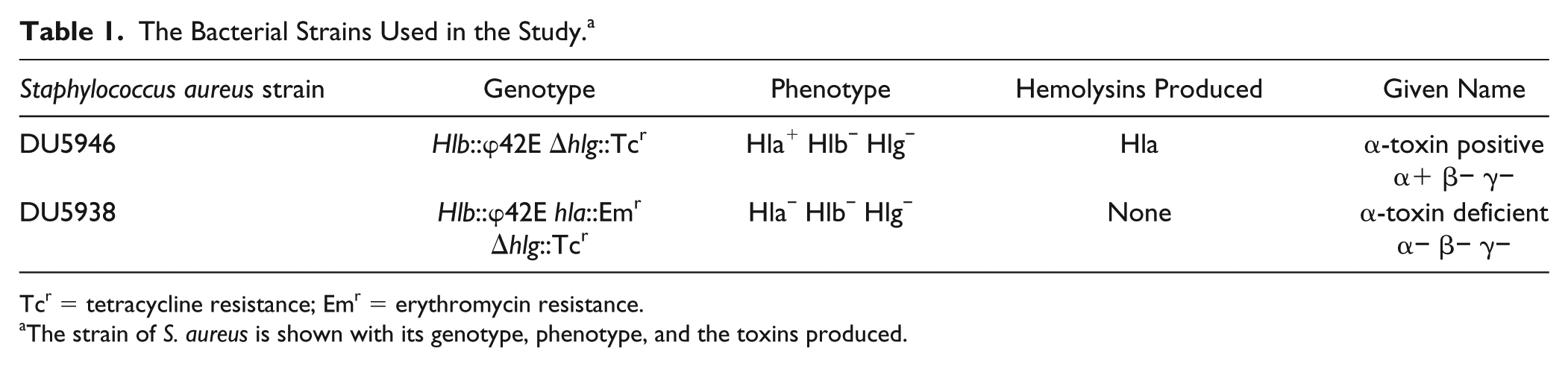
Tcr = tetracycline resistance; Emr = erythromycin resistance.
The strain of S. aureus is shown with its genotype, phenotype, and the toxins produced.
Preparation of Bacterial Pellets, Supernatant Samples, and Antibiotics
Bacteria were stored at −80°C in 10% v/w skimmed milk (Oxoid, Basingstoke, UK). When required, they were thawed and streaked onto tryptone soya agar (TSA; Oxoid) plates containing 2 µg/mL tetracycline (Cat. 87128; Sigma-Aldrich). Following incubation (24 hours; 37°C), tryptone soya broth (TSB; Oxoid), containing 2 µg/mL tetracycline, was inoculated with several single bacterial colonies from the TSA plate and incubated (24 hours; 37°C) with shaking. From this TSB culture, serial dilutions were performed in saline to 10−6, in order to calculate the number of colony-forming units (CFU). A CFU is defined as a unit used to estimate the number of viable bacteria capable of reproducing to form a colony of the same bacterial species—the number of CFUs is therefore a measure of the number of active bacteria in 1 mL of TSB cultured for 24 hours. Thereafter, 100 µL of 10−4, 10−5, and 10−6 dilutions were spread evenly onto TSA plates and incubated (37°C; 24 hours). Colonies were then counted using a colony counter (Stuart, Bibby Scientific, Stone, UK). Bacterial counts were performed on several cultures for each strain and a count of ~1 × 109 CFU/mL (range 0.8-1.25 × 109 CFU/mL) was routinely obtained. Cultures were then centrifuged (2,000 × g; 15 minutes) and the pellets resuspended by vortexing, washing in HBSS (Hank’s balanced salt solution) and centrifugation (×3). The supernatant was then removed and the pellets air-dried before being stored (−20oC) and used within 1 week. Based on the colony counts, a bacterial pellet contained ~1 × 105 CFU and the bacteria were used in the experiments described here at ~0.2 × 105 CFU in HBSS. TSA plates were prepared to assess effective doses of antibiotics against S. aureus whose viability was assessed by counting colonies plated on TSA in the presence/absence of antibiotics following incubation overnight at 37°C. For penicillin and linezolid, doses of 1000 U/mL and 25 µg/mL, respectively, were sufficient. For flucloxacillin and erythromycin, doses of 2 mg/mL and 10 µg/mL, respectively, were adequate and no viable bacteria were detected. For some experiments where α-toxin levels were assessed by the hemolysis method, higher doses of penicillin (to 250,000 U/mL) were used. Table 2 summarizes the antibiotics used with doses, together with sites of action and references. All doses used were substantially greater than the minimum inhibitory concentration (MIC) in the literature ( Table 2 ).
Antibiotics and Concentrations Used in Study.
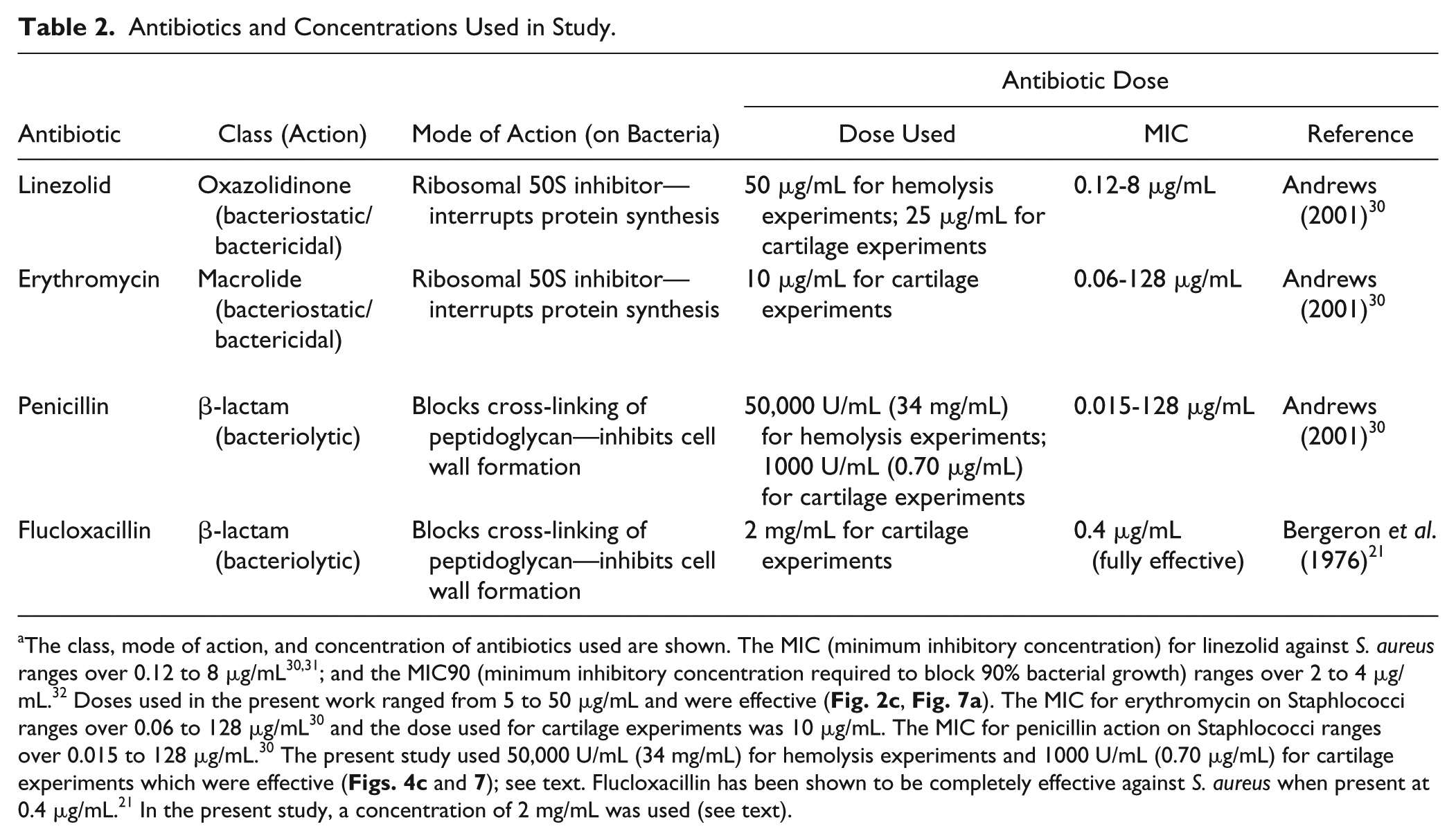
The class, mode of action, and concentration of antibiotics used are shown. The MIC (minimum inhibitory concentration) for linezolid against S. aureus ranges over 0.12 to 8 μg/mL30,31; and the MIC90 (minimum inhibitory concentration required to block 90% bacterial growth) ranges over 2 to 4 μg/mL. 32 Doses used in the present work ranged from 5 to 50 μg/mL and were effective ( Fig. 2c , Fig. 7a ). The MIC for erythromycin on Staphlococci ranges over 0.06 to 128 μg/mL 30 and the dose used for cartilage experiments was 10 μg/mL. The MIC for penicillin action on Staphlococci ranges over 0.015 to 128 μg/mL. 30 The present study used 50,000 U/mL (34 mg/mL) for hemolysis experiments and 1000 U/mL (0.70 μg/mL) for cartilage experiments which were effective ( Figs. 4c and 7 ); see text. Flucloxacillin has been shown to be completely effective against S. aureus when present at 0.4 μg/mL. 21 In the present study, a concentration of 2 mg/mL was used (see text).
Rabbit Erythrocyte Hemolysis Assay for α-Toxin
The rabbit red blood cell (RBC) hemolysis assay was used to determine biologically-relevant levels of α-toxin released during S. aureus culture.19,20 Fresh, heparinized RBCs (Orygen, Penicuik, UK), were prepared at ~5% hematocrit in HBSS (pH 7.4; Invitrogen, Paisley, UK). At time zero and at specific points throughout the culture of S. aureus (see figures), aliquots were taken and centrifuged (8,000 × g; 30 seconds) and the supernatant aliquotted into microcentrifuge tubes, taking care not to disturb the pellet. The supernatants, which contained α-toxin, were then added to microcentrifuge tubes containing 5% RBC suspensions, mixed gently, and incubated (37°C; 60 minutes). The samples were then centrifuged (8,000 × g; 10 seconds) and the supernatant assessed for hemoglobin at 540 nm (Abs540) on a Nanodrop spectrophotometer (ThermoFisher Scientific, Waltham, MA, USA). Percentage hemolysis (%H) was then calculated ([Abs540 of sample – Abs540 of negative control]/[Abs540 of 100% hemolysis – Abs540 of negative control] × 100%). The sensitivity of RBC samples to α-toxin and the amount of α-toxin produced from each culture were variable. It was important to minimize this at the beginning of an experimental week, by initially testing erythrocytes from several rabbits on a freshly prepared batch of α-toxin. Erythrocytes that were relatively insensitive to α-toxin requiring >1 hour of incubation before hemolysis was detectable were not studied further and this reduced the variation in results obtained.
Assessment of In Situ Chondrocyte Viability by Confocal Laser Scanning Microscopy
Fresh, healthy (non-degenerate) metacarpophalangeal joints of 3-year-old cows from a local abattoir, were washed, skinned, dehoofed, and opened under aseptic conditions. Cartilage explants were cultured in Dulbecco’s modified Eagle’s medium (DMEM; pH 7.4; 37°C; Invitrogen) in the presence or absence of S. aureus and after 15 hours, the bacteria had caused measurable (~20%) chondrocyte death. Explants were then exposed to antibiotics (or control medium) and the incubation continued for up to 23 hours. At the time points indicated, explants were removed, rinsed in DMEM, and chondrocyte viability assessed as described. 33 Explants were incubated with 5-chloromethylfluorescein diacetate (CMFDA) and propidium iodide (PI) (both at 10 µM from Invitrogen, Paisley, UK) in DMEM (1 hour; 21°C). These probes labeled living or dead cells green or red, respectively. Explants were then fixed (4% formaldehyde; 4 hours; Fisher Scientific, Loughborough, UK), rinsed and stored in phosphate buffered saline (PBS), until imaged within 24 hours. Imaging of fluorescently-labeled in situ chondrocytes was performed as described.14,33 An upright Zeiss LSM510 Axioskop (Carl Zeiss, Welwyn Garden City, UK) confocal laser scanning microscope with a ×10 (0.3NA) dry objective was used to acquire axial images. The percentage cell death ([number of dead cells/total number of living and dead cells] × 100%) was calculated within a standard region of interest (ROI) using Volocity 4 software (Improvision, Coventry, UK). Within each ROI, individual cells, in both green (living) and red (dead) channels, were quantified based on percentage voxel (volumetric pixel) intensity. 14
Data Analysis and Statistics
Data are shown as means ± standard error of the mean (SEM) from at least 3 independent experiments, and were analyzed using GraphPad Prism Ver.6 (GraphPad, San Diego, CA, USA). Nonparametric t tests (Kruskal-Wallis) and analyses of variance were performed and significant differences accepted when P < 0.05.
Results
S. aureus α-Toxin Mediated Rabbit RBC Hemolysis
The strain of S. aureus (DU5946) that harbored the α-toxin (hla) gene was hemolytic on rabbit blood agar whereas the strain deficient in this gene (DU5938) was non-hemolytic ( Fig. 1a ). Supernatants were prepared from cultures of these strains and added to suspensions of rabbit erythrocytes in HBSS. There was a rapid increase in %H for the α-toxin-positive strain; however, the α-toxin-deficient strain had no detectable effect ( Fig. 1b ). Thus, by using rabbit erythrocytes in suspension as a model and determining hemoglobin release spectrophotometrically, biologically-active α-toxin could be quickly assessed semiquantitatively.
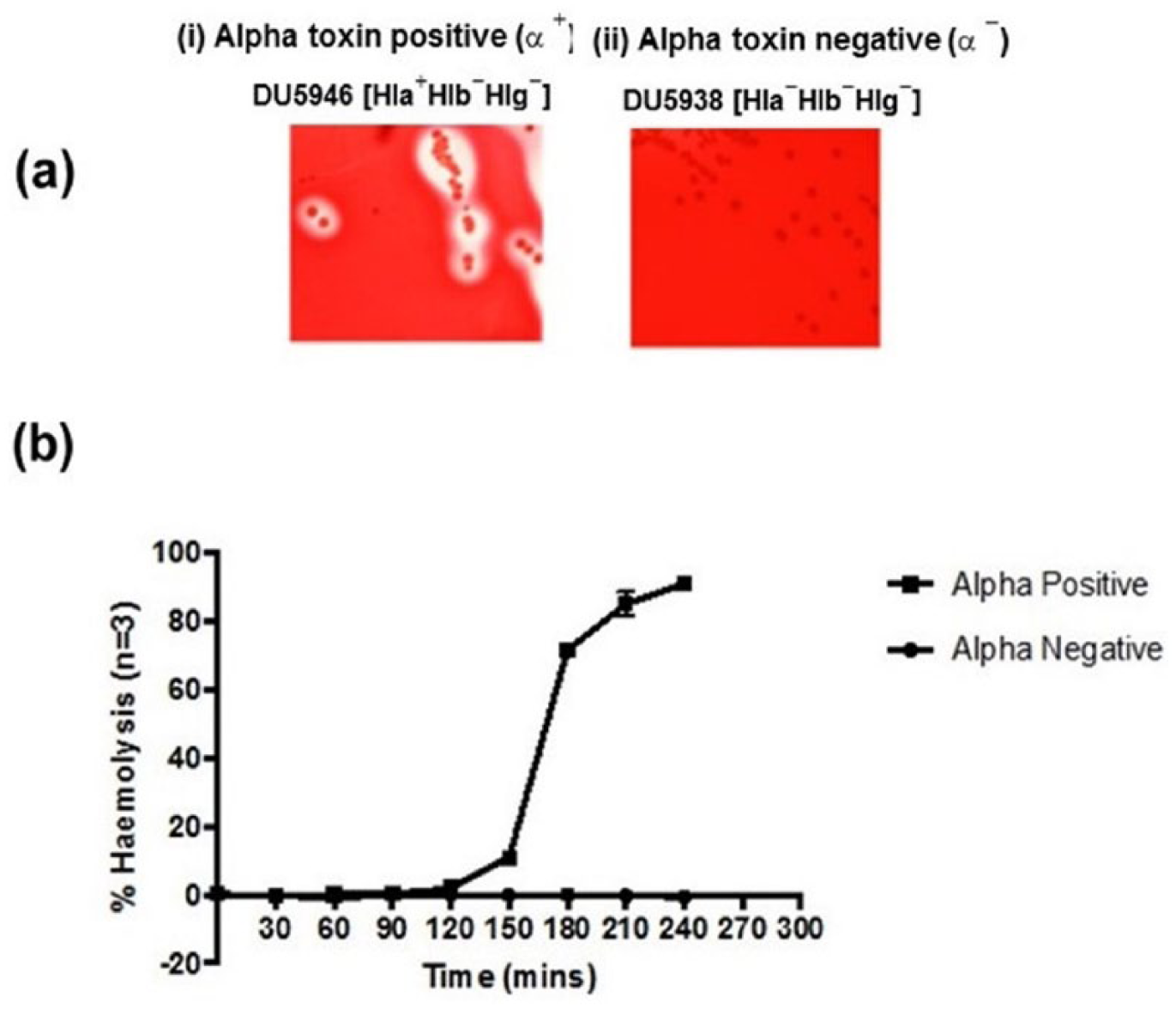
The hemolysis of rabbit erythrocytes following incubation with α-toxin positive or α-toxin negative strains of Staphylococcus aureus. (
Time Course of Linezolid Action
Single doses of linezolid (to concentrations of 5-50 μg/mL) were added to the α-positive S. aureus cultures (DU5946) at a point corresponding to ~50% hemolysis (after 80 minutes) and samples analysed for α-toxin-induced hemolysis ( Fig. 2a ). For all concentrations, there was a rapid reduction in the rate of rise of α-toxin-induced hemolysis, and at 25 and 50 μg/mL, there was no significant change in %H ( Fig. 2b ). Addition of linezolid to 50 μg/mL at 80-, 100-, 130-minute time points, rapidly suppressed any further increase in α-toxin-induced hemolysis ( Fig. 2c ). Linezolid (50 μg/mL) addition 60 minutes after the commencement of the S. aureus culture, abolished subsequent α-toxin-induced hemolysis compared with the untreated (infected) control ( Fig. 3a ). The same concentration of linezolid added after 100 minutes when there was ~40% hemolysis, also completely inhibited subsequent α-toxin-induced hemolysis to the same level after 40 and 60 minutes as that present at the start of the experiment (P < 0.001; Fig. 3b ). These results indicated that linezolid addition to S. aureus cultures rapidly (within ~20 minutes) inhibited α-toxin-induced hemolysis and that 25 and 50 μg/mL prevented any further rise.
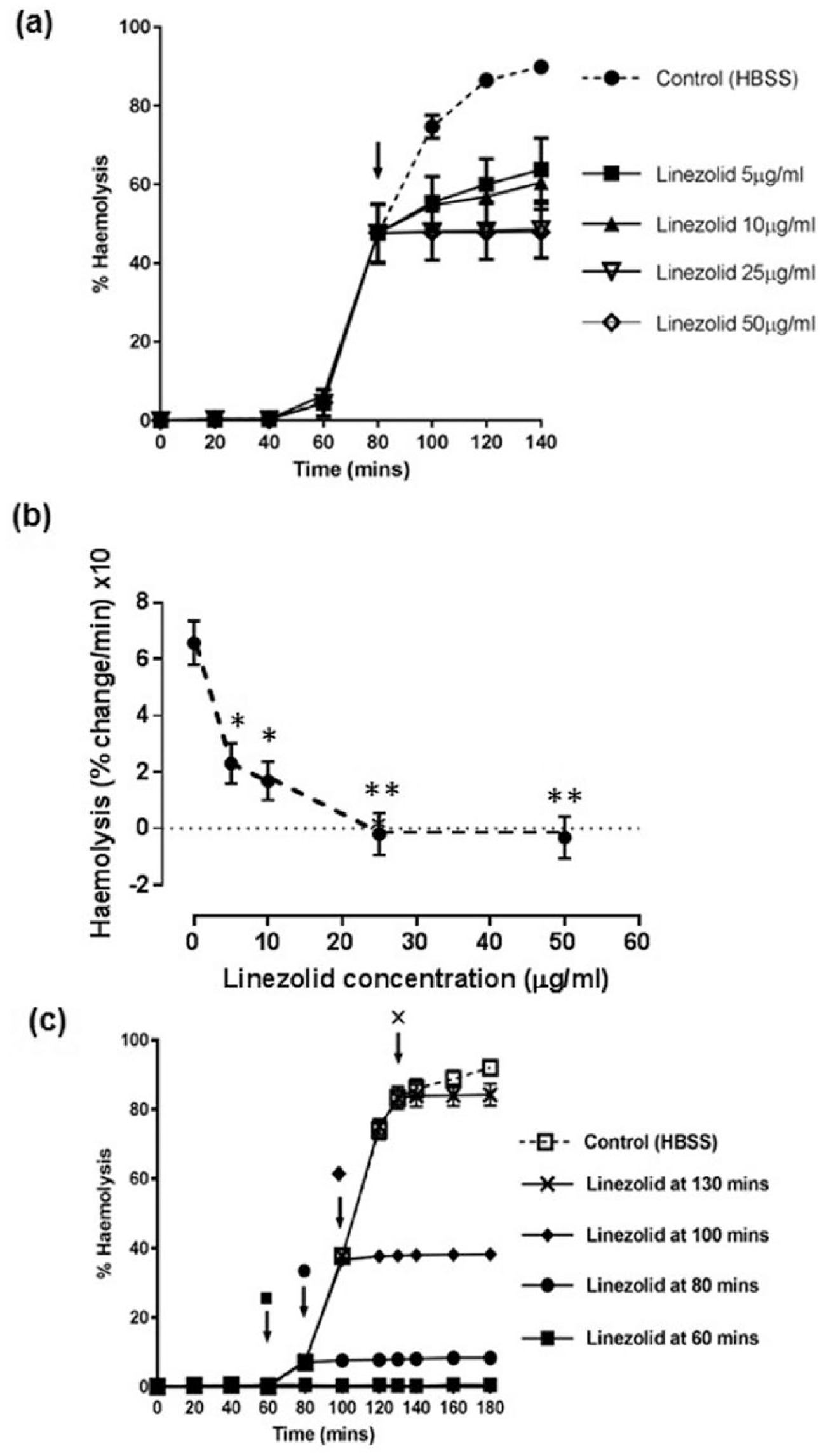
Effects of linezolid on α-toxin-induced hemolysis of rabbit erythrocytes. Panel (
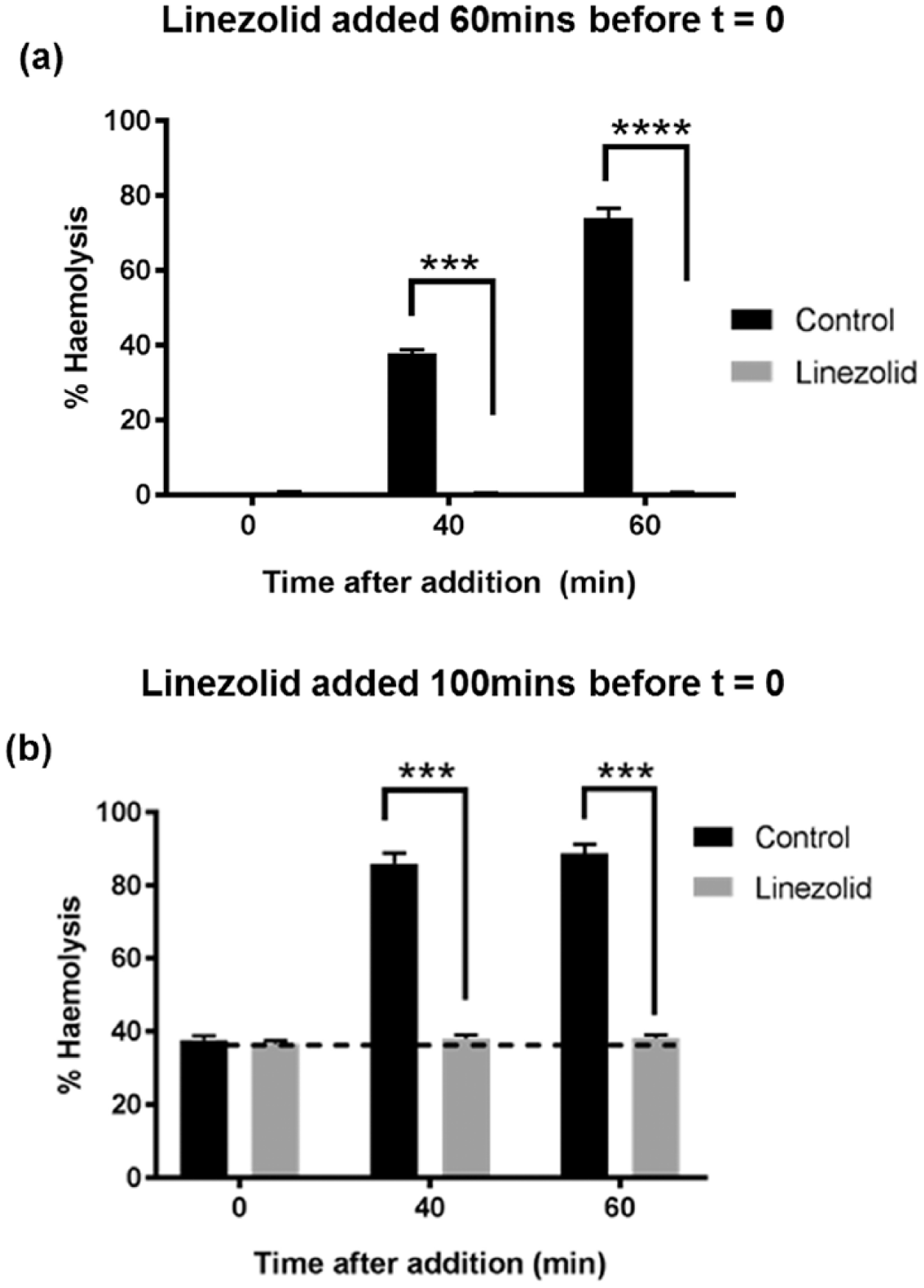
Inhibition of α-toxin-induced hemolysis of rabbit erythrocytes at different time points by prior treatment with linezolid. Linezolid (50 μg/mL) was added (
Time Course of Penicillin Action
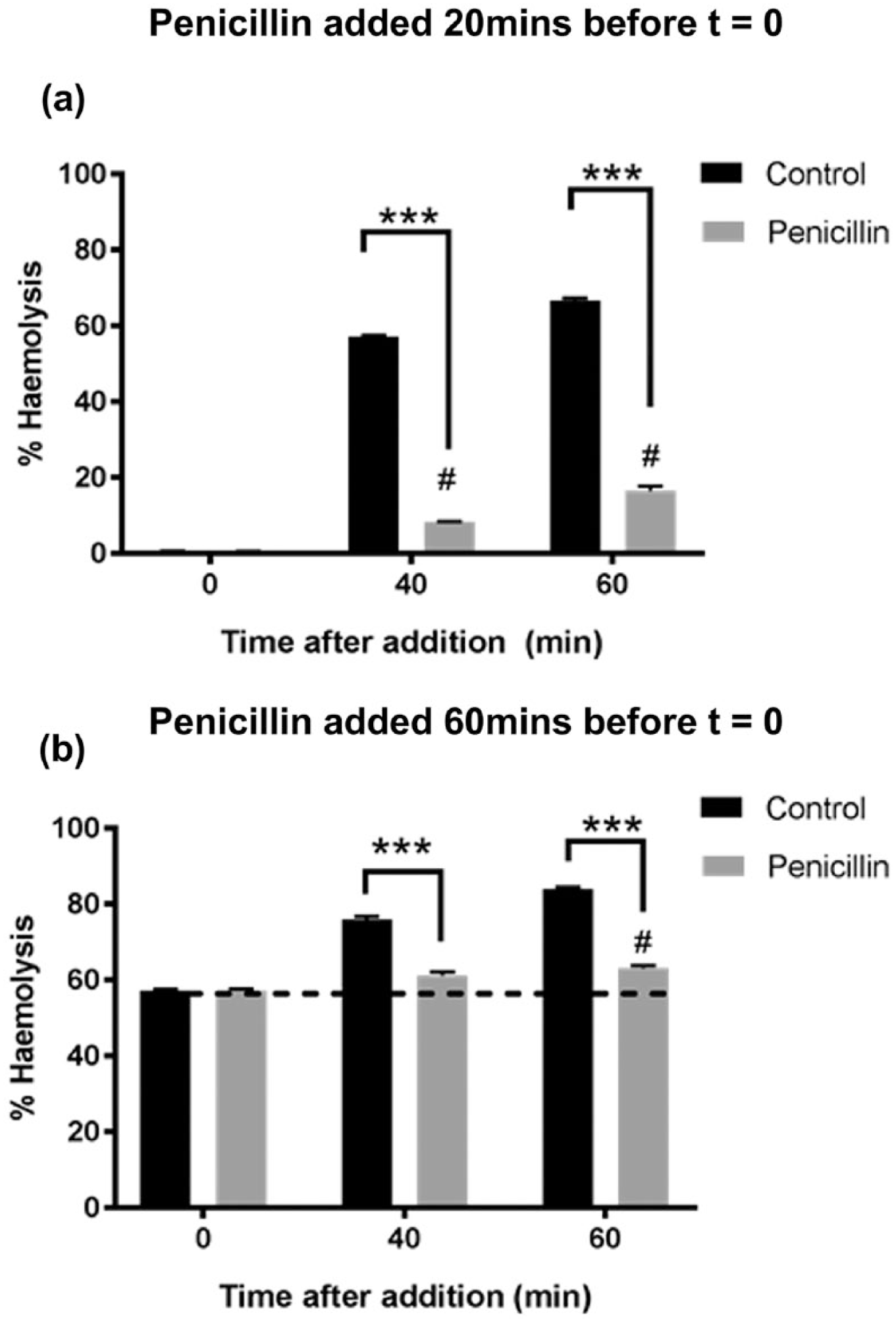
Single doses of penicillin G (to 5,000, 10,000, 25,000 or 50,000 U/mL) were added to the α-toxin positive S. aureus cultures at a time point corresponding to ~50% hemolysis (after 60 minutes) and samples analyzed for α-toxin-induced hemolysis ( Fig. 4a ). While at penicillin concentrations of 50,000 and 250,000 U/mL at 140 minutes there was a significant reduction in the rate of rise of α-toxin-induced hemolysis, the effect of lower concentrations was not significant ( Fig. 4b ). Addition of penicillin to 50,000 U/mL at different time points suppressed the increase in α-toxin-induced hemolysis ( Fig. 4c ); however, the time course of action was slower compared with linezolid ( Fig. 2c ). Penicillin addition 20 minutes after the start of the culture significantly inhibited hemolysis compared with the untreated (infected) control after 40 and 60 minutes ( Fig. 5a ). While in the presence of penicillin, there were significant increases in hemolysis at 40 and 60 minutes compared with the values at the time of drug addition (P < 0.05), these were not different (P = 0.12), indicating a plateau in α-toxin levels. When the same concentration of penicillin was added after 60 minutes, there was ~60% hemolysis at time zero and this significantly (P < 0.001) inhibited subsequent α-toxin-induced hemolysis after 40 and 60 mins ( Fig. 5b ). The extent of α-toxin-induced hemolysis at 60 minutes was significantly (P = 0.05) higher than the time zero point but not significantly different compared with the 40-minute time point ( Fig. 5b ). These results suggested that penicillin addition to S. aureus cultures progressively reduced the rate of increase of α-toxin-induced hemolysis. However, the time course appeared relatively slow compared with linezolid, and at the highest concentrations studied (50,000 U/mL), there was still a slight rise in α-toxin-induced hemolysis until the inhibition appeared complete.
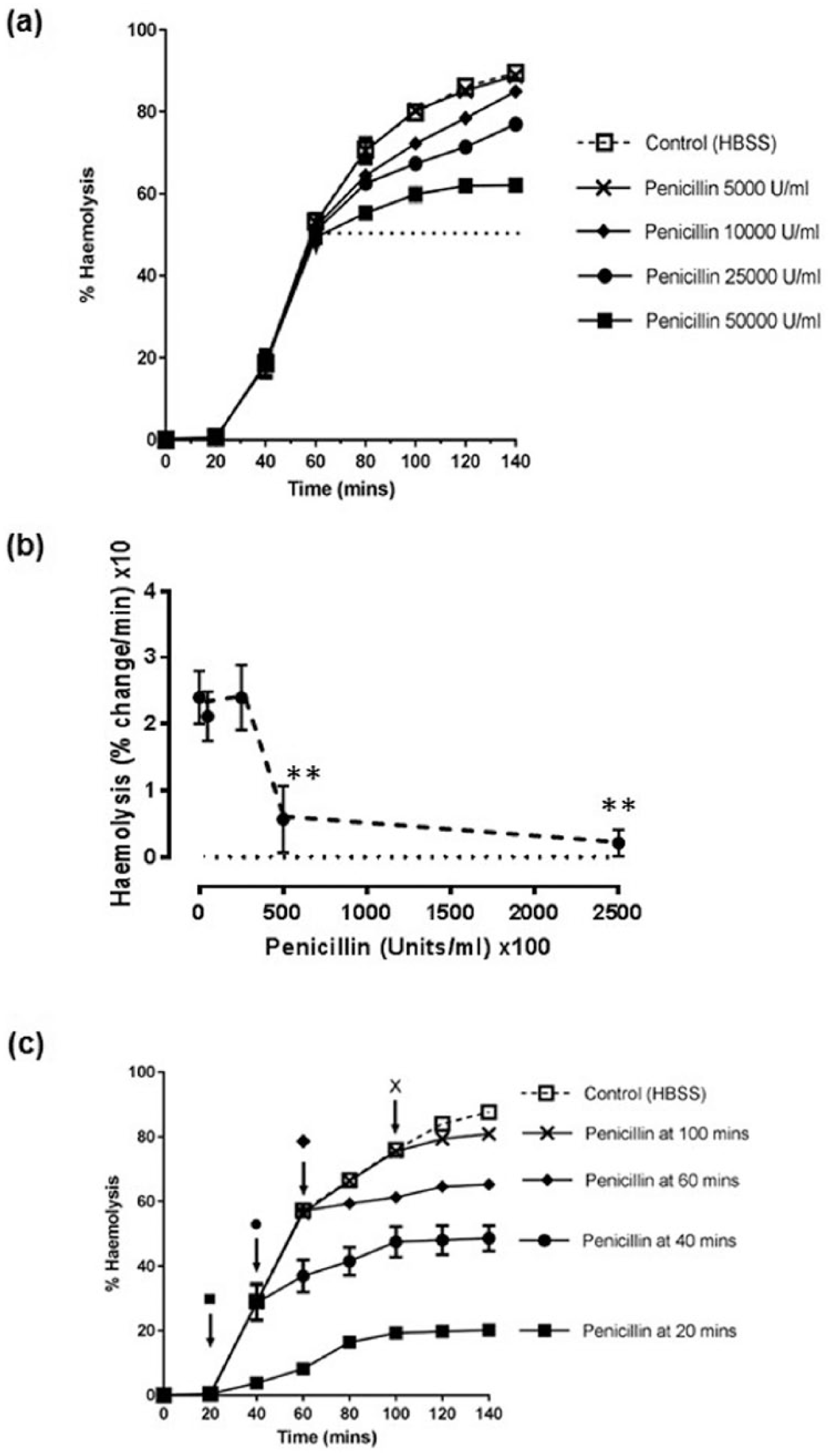
Effects of penicillin on α-toxin-induced hemolysis of rabbit erythrocytes. Panel (
Inhibition of α-toxin-induced hemolysis of rabbit erythrocytes at different time points by prior treatment with penicillin. Penicillin (50,000 U/mL) was added (
To directly compare the inhibitory effects of linezolid and penicillin using the same blood samples and S. aureus cultures, DU5946 cultures were incubated for 60 minutes, and then untreated (control) or dosed with penicillin (250,000 U/mL) or linezolid (25 μg/mL), and the medium analyzed for α-toxin for up to 80 minutes ( Fig. 6 ). After 20 minutes, both antibiotics significantly (P < 0.05) inhibited hemolysis; however, at 40, 60, and 80 minutes after antibiotic addition, the inhibition was significantly (by ~8%) greater (P < 0.05) in the presence of linezolid ( Fig. 6 ). The inhibition by penicillin did not change significantly compared with linezolid over the 40- to 80-minute period after antibiotic addition, suggesting that the inhibitory effect of penicillin was maximal.
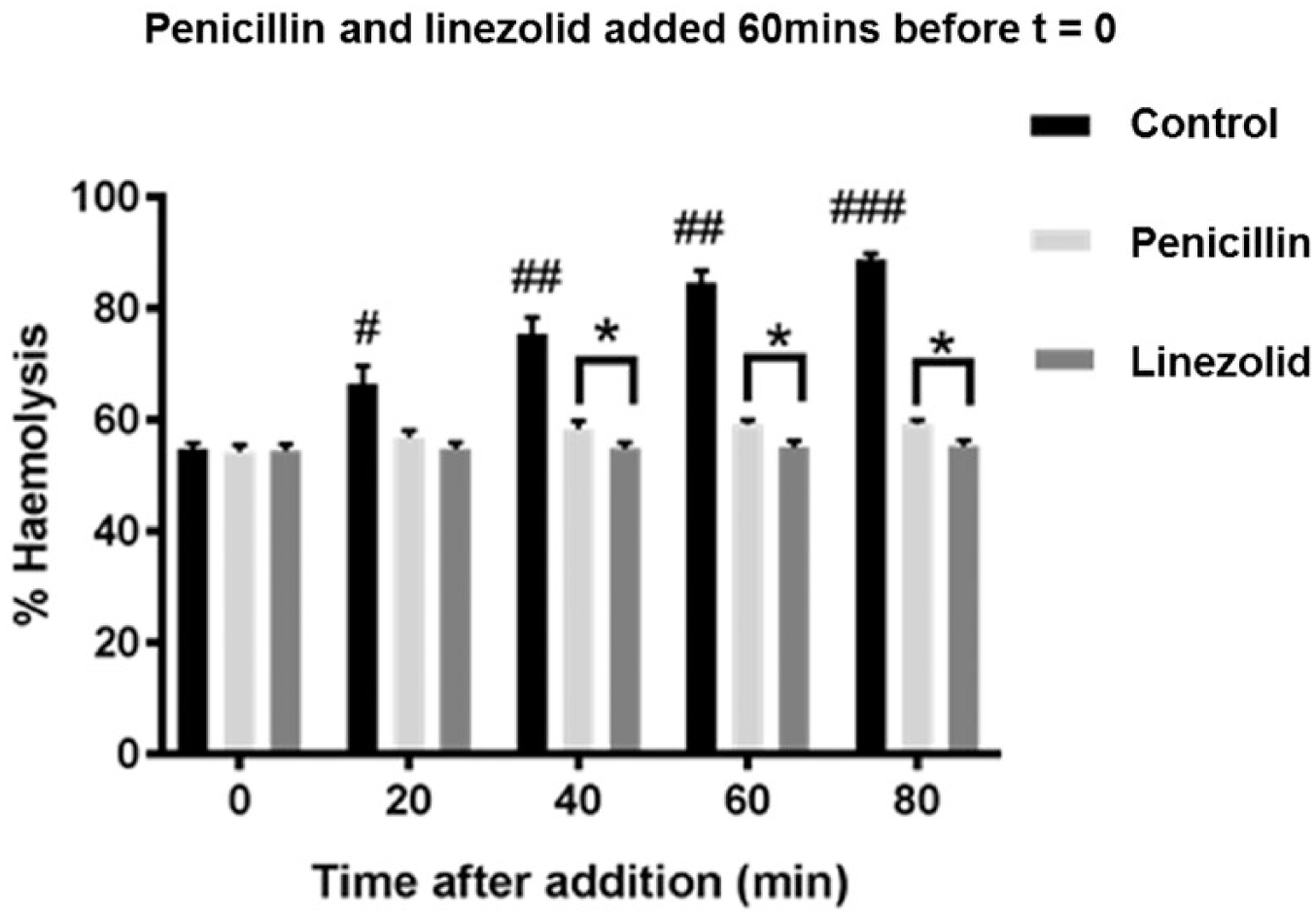
Comparison between the inhibitory effects of penicillin and linezolid on the extent of α-toxin-induced hemolysis. A single dose of penicillin (250,000 U/mL) or linezolid (25 μg/mL; final concentrations) was added to α-toxin positive strain (DU5946) Staphylococcus aureus cultures after 60 minutes (corresponding to approximately 55% hemolysis) and α-toxin-induced hemolysis determined. There was a significant reduction in levels of α-toxin-induced hemolysis for both drugs at 20, 40, 60, and 80 minutes compared with the untreated control at the same time points. However, the inhibition was significantly greater for the linezolid-treated cultures compared with those following penicillin addition at 40, 60, and 80 minutes (P < 0.01). Data shown are means ± SEM for at least 3 independent experiments.
Chondrocyte Protection in a Bovine Cartilage Septic Arthritis Model by Antibiotics
S. aureus DU5946 was cultured with cartilage explants and in control (no antibiotics) samples, there was almost complete (>90%) chondrocyte death after ~21 hours ( Fig. 7a and b ). In contrast, when infected cartilage was treated with penicillin or linezolid after 15 hours, there was no subsequent chondrocyte death and no difference between the potency of the 2 drugs ( Fig. 7a and b ). Similar experiments were performed with flucloxacillin and erythromycin (data not shown). In the untreated infected samples, there was complete (~100%) chondrocyte death after ~24 hours. Treatment of infected cartilage samples with flucloxacillin (2 mg/mL) or erythromycin (10 μg/mL) after 18 hours, prevented any subsequent chondrocyte death and there was no significant difference (P > 0.05) between the 2 drugs at any time point studied up to 26 hours after antibiotic addition. Levels of chondrocyte death in the control (i.e., uninfected and untreated) cartilage samples were <2%. These results suggested that both bacteriolytic (penicillin, flucloxacillin) and bacteriostatic/bactericidal (linezolid, erythromycin) antibiotics were equally effective at protecting chondrocytes in the bovine cartilage model of S. aureus septic arthritis.
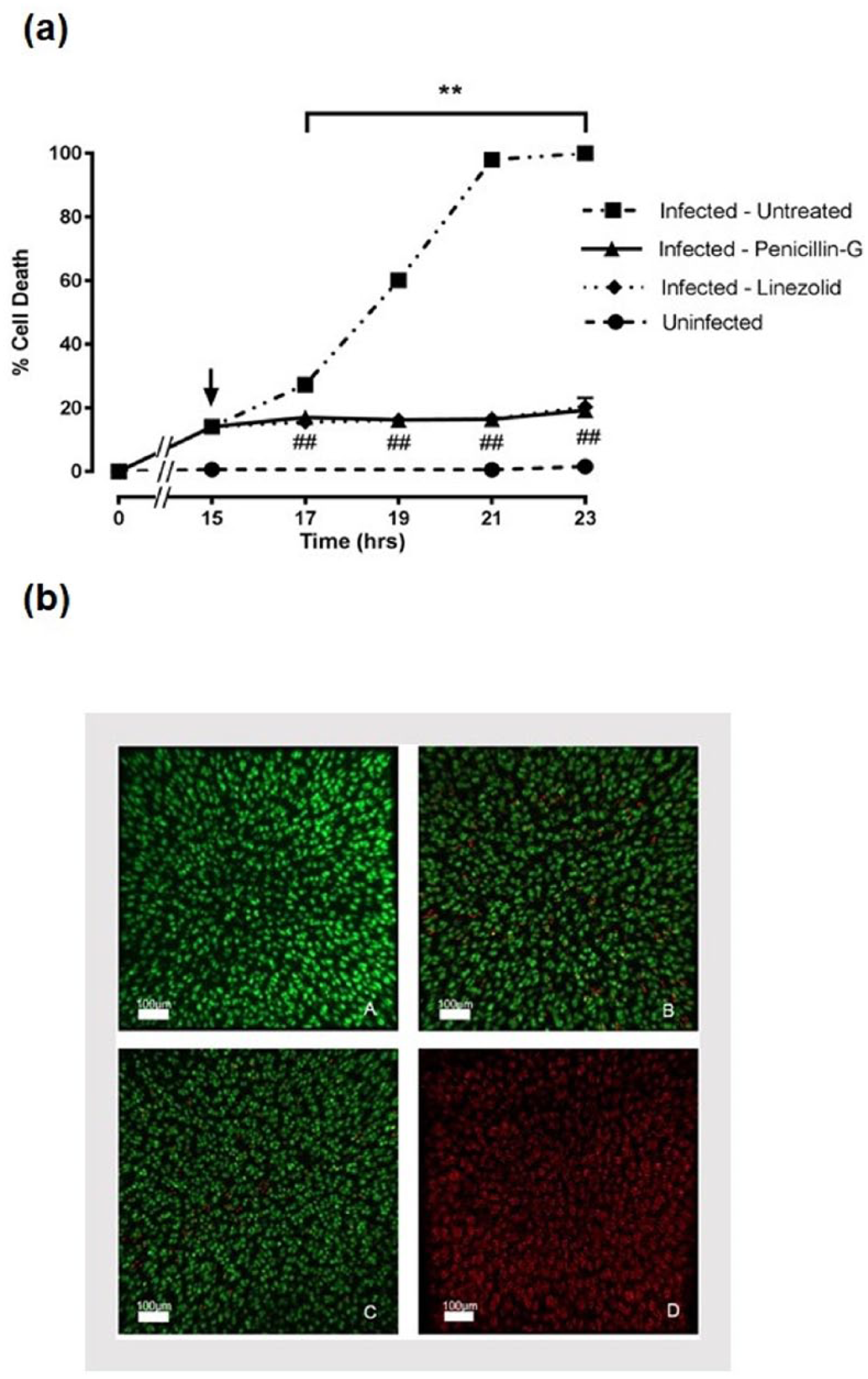
Protection of in situ chondrocytes by penicillin or linezolid against Staphylococcus aureus infection in a bovine model of septic arthritis. Bovine cartilage was incubated with α-toxin positive strain (DU5946) S. aureus cultures for 15 hours, which produced approximately 20% chondrocyte death (infected), or in the absence of bacteria (uninfected). Cultures were then continued untreated, or following the addition (at the arrow) of penicillin (1,000 U/mL) or linezolid (25 μg/mL). Data shown are means ± SEM for 6 independent experiments. Examples of confocal images of chondrocytes in bovine cartilage taken after 23 hours are shown for the following conditions (
Discussion
The rabbit erythrocyte hemolysis assay determined the time course of biologically-relevant α-toxin produced during culture of S. aureus in the presence or absence of bacteriostatic/bactericidal and bacteriolytic antibiotics. The antibiotics tested could limit or prevent a rise in α-toxin levels; however, there were differences in their time courses of action. The bacteriostatic/bactericidal agent, linezolid, appeared more rapid and potent than the bacteriolytic antibiotic, penicillin. There was, however, no evidence of a detectable “pulse” of α-toxin-induced hemolysis following addition of any of the antibiotics studied, suggesting there was no substantial intracellular “pool” of stored α-toxin in the S. aureus strain DU5946. In the bovine cartilage model of septic arthritis, S. aureus cultures rapidly caused chondrocyte death. However, chondrocytes were completely protected against S. aureus by the antibiotics tested (linezolid, flucloxacillin, penicillin, erythromycin). These results were important for elucidating whether or not there was a significant intracellular store of α-toxin as this may be relevant for antibiotic selection during S. aureus–induced septic arthritis.
The rabbit RBC hemolysis bioassay was used to determine the time course of α-toxin release during S. aureus culture. The measurement of hemoglobin released following the interaction between α-toxin and erythrocytes provided a dynamic and sensitive assay for the lethal effects of α-toxin, and was highly flexible experimentally and reproducible, allowing several conditions (e.g., drug concentrations) to be tested at the same time points. The other toxins/damaging agents produced by S. aureus had a negligible effect on rabbit erythrocytes emphasizing the specific nature of this assay for α-toxin ( Fig. 1 ). It was noted that some blood samples were very sensitive to α-toxin whereas others were relatively unresponsive and so it was necessary to initially screen each blood sample for sensitivity against a S. aureus culture. Once a blood sample was deemed acceptable, the time course of hemolysis to α-toxin was highly reproducible. In addition, the ability of S. aureus cultures to produce sufficient quantities of potent α-toxin was variable and probably depended on prevailing culture conditions. The difference in potency of batches of α-toxin was observed, as for example, in one series of experiments, 60 minutes elapsed before the S. aureus culture started to produce measurable α-toxin levels ( Fig. 2a ). In another set of experiments performed with a different α-toxin preparation, after only 40 minutes, hemolysis had already increased to 20% ( Fig. 4a ). The rabbit erythrocyte model for determining the effects of antibacterials on α-toxin levels might be considered a limitation. However, rabbit erythrocytes contain the S. aureus α-toxin receptor ADAM-10 also present on chondrocytes of animals and normal and degenerate human cartilage.16-18 Our previous work has demonstrated that in a cartilage model of septic arthritis, α-toxin is the key damaging agent to chondrocytes and thus the rabbit erythrocyte hemolysis assay is appropriate to determine the effects of antibacterials on α-toxin release from S. aureus cultures.14,15
Linezolid at 25 or 50 µg/mL rapidly abolished the rise in α-toxin-induced hemolysis ( Fig. 2c ) probably because of its potent action on bacterial protein synthesis and replication. 34 However, linezolid may have both bacteriostatic and/or bactericidal effects on S. aureus depending on concentration and experimental conditions. 22 Therefore, the reduced rate of hemolysis in linezolid-treated cultures may be the result of bacteriostatic, bactericidal, or combined bacteriostatic/bactericidal action. Whatever the mechanism, at 50 µg/mL linezolid ( Fig. 3a and b ), there was no progressive change in hemolysis ( Figs. 2c and 3b ) indicating no additional release of α-toxin from dying/dead bacteria. However, future work would be required to investigate the effect of varying concentrations of linezolid on the bacteriostatic and/or bactericidal modes of actions on S. aureus α-toxin. It should be noted that to test our hypothesis of whether there was a detectable intracellular store of α-toxin, we used concentrations considerably above the MIC ( Table 2 ) and have not atttempted to study the effects of clinically-relevant doses.
The action of penicillin was slower compared with linezolid as evidenced by the relatively tardy inhibition of α-toxin-induced hemolysis even at a high concentration (50,000 U/mL; Figs. 4c and 5a and b ). There are 2 possible explanations for this finding. The most likely interpretation of these results is because of the slower time course of the antibiotic inhibiting bacterial cell wall synthesis.21,22 The level of hemolysis after penicillin addition was higher than that with linezolid, and did not change with time ( Fig. 6 ). This suggested that the inhibition by both drugs was maximal with no further bacterial death or inhibition of division. However, it is also possible that the slightly elevated level of hemolysis (~8% of the total) with a high dose of penicillin ( Fig. 6 ) could have been because of the release of the small additional amount of intracellular α-toxin from the dying/dead bacteria.
The presence of a small amount of intracellular S. aureus α-toxin is implied by others using different methods to cause bacterial damage. For example, Duncan and Cho 35 suggested that mechanical disruption of S. aureus released 1% to 2% of total toxin present in cultures. However, this might have been influenced by the simultaneous release of degradative enzymes leading to an underestimation in the amount of active toxin. Monecke et al. 36 using alkali treatment, noted that there was only a “small” (but unquantified) level of stored α-toxin following bacterial lysis. For S. aureus therefore, there might only be a small intracellular store with the α-toxin released rapidly after synthesis. On the other hand, pneumolysin, the toxin produced by Streptococcus pneumoniae, is almost exclusively stored intracellularly and may be released during bacterial lysis.37,38 In a S. pneumoniae rabbit meningitis model, addition of the bacteriolytic agent ceftriaxone, released additional quantities of pneumolysin compared with the nonbacteriolytic agent rifampin, with the additional toxin causing further cellular damage and inflammation. 39
In the S. aureus septic arthritis cartilage model, linezolid and penicillin ( Fig. 7a and b ) and flucloxacillin and erythromycin (see Results) were equally effective. This might appear to conflict with the suggestion above that there was a small intracellular “pool” of α-toxin. It is possible that the methods for determining chondrocyte death were not sufficiently sensitive to detect the small increase from the release of stored α-toxin by penicillin. However, there was no significant difference in percentage chondrocyte viability between linezolid- and penicillin-treated explants so this does not seem likely. Alternatively, α-toxin levels in the medium and thus the cartilage matrix may already be high and any small increase following penicillin treatment could be without further detectable effect. The α-toxin released from S. aureus will have a rapid effect on the rabbit erythrocytes as pore formation and hemolysis proceeds promptly. 19 However, in cartilage, toxin permeability may be hindered by the cartilage extracellular matrix. 40 It is also possible that the dynamics of toxin binding and pore formation to bovine chondrocytes proceeds with a different time course compared to rabbit erythrocytes. ADAM-10 has been identified as an important receptor for α-toxin binding 41 mediating changes to intracellular Ca2+ signaling 42 and is present on human chondrocytes and upregulated in osteoarthritis.16-18 The binding site density on chondrocytes may also be different compared to rabbit erythrocytes which could further influence the time course of the α-toxin effect.
The rapid inhibition by linezolid on the rise in S. aureus α-toxin levels compared with penicillin is likely due to a bacteriostatic action on S. aureus replication, and in vivo this would be followed by bacteria removal by the immune system. Septic arthritis may be regarded as a medical emergency 3 and thus clinically there would be benefit in administering this antibacterial agent or others in this class for the most rapid action possible to protect joint tissues from further damage. It is possible that bacteriolytic antibiotics (e.g., penicillin, flucloxacillin) may lead to the release of additional inflammatory components (e.g., teichoic acids, peptidoglycans) from S. aureus potentially causing further chondrocyte damage. However, all the drugs tested (penicillin, linezolid [ Fig. 7 ]; flucloxacillin, erythromycin [see Results]), were equally effective at protecting chondrocytes against S. aureus. This suggests that these agents, if released from dead/dying bacteria, do not have a significant effect on chondrocyte viability and support the view that the α-toxin alone was the primary cause of chondrocyte death. 15 It is also worth noting that bacteriostatic/bactericidal agents, for example, linezolid, reduce the expression of S. aureus α-toxin.29,34 This is in marked contrast to subinhibitory doses of β-lactams, which increase S. aureus hla (α-toxin) mRNA expression43,44 and therefore might cause concern in the treatment of osteoarticular infection by these bacteria. Furthermore, linezolid reduces expression of other staphylococcal toxins such as PVL in PVL-associated staphylococcal pneumonia. 45 Since PVL-producing strains of S. aureus may also be present and can cause complications in septic arthritis, 46 linezolid may offer a further advantage over bacteriolytic antibiotics. Although our results demonstrated that these antibacterials are protective against chondrocytes in vitro, the response in vivo is likely to be far more complex as our model of S. aureus-induced septic arthritis does not include the host’s immune response.
While it can be particularly challenging to extrapolate the results from the in vitro situation to the clinical setting, there are 3 main findings from this study that are relevant. (a) The time course of the bacteriostatic/bactericidal antibiotic linezolid on limiting the rise α-toxin was clearly more rapid than for the bacteriolytic antibiotic penicillin (
Fig. 2
vs.
Fig. 4
). (
This present study used a rabbit RBC assay to determine α-toxin levels produced by S. aureus in culture following treatment with antibiotics. Linezolid rapidly limited any further increase in α-toxin levels by S. aureus while penicillin had a slower time course of action. There was a small additional release of α-toxin following penicillin addition suggesting a delayed action and/or a small intracellular store of α-toxin. Both drugs were effective at protecting chondrocytes in our S. aureus septic arthritis model, however linezolid may be the preferential choice due to its primarily bacteriostatic action and rapid effect.
Footnotes
Acknowledgments and Funding
We thank Prof. T. J. Foster, Department of Microbiology, Trinity College, Dublin, for supplying S. aureus strains, Dr. A. Kubasik-Thayil, IMPACT facility, University of Edinburgh, for confocal laser scanning microscopy guidance, and K. Wόjcik of Scotbeef Ltd, Bridge of Allan, Stirlingshire, for providing bovine feet. We also thank A. Boyle and Dr. C. J. Doherty for helpful comments. The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by an independent peer-reviewed grant from Pfizer Grant No. WI177684.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
