Abstract
Objective
Septic arthritis is commonly caused by Staphylococcus aureus and is a medical emergency requiring antibiotics and joint irrigation. The bacteria produce α-toxin causing rapid cartilage cell (chondrocyte) death. Saline (0.9%NaCl) lavage is normally used to remove bacteria and toxins, however, its composition might be suboptimal to suppress the lethal effects of α-toxin. We utilized rabbit erythrocyte hemolysis as a sensitive, biologically relevant assay of α-toxin levels to determine if changes to osmolarity, temperature, pH, and divalent cation (Mg2+, Ca2+) concentration were protective.
Design
Erythrocytes were incubated in the various conditions and then exposed to α-toxin (“chronic” challenge) or incubated with α-toxin and then exposed to experimental conditions (“acute” challenge).
Results
Raising osmolarity from 300 mOsm (0.9%NaCl) to 400, 600, or 900 mOsm (sucrose addition) when applied chronically, significantly reduced hemolysis linearly. As an acute challenge, osmotic protection was significant and similar over 400 to 900 mOsm. Reducing temperature chronically from 37°C to 25°C and 4°C significantly reduced hemolysis, however, when applied as an acute challenge although significant, was less marked. Divalent cations (Mg2+, Ca2+ at 5mM) reduced hemolysis. Varying pH (6.5, 7.2, 8.0) applied chronically marginally reduced hemolysis. The optimized saline (0.9% NaCl; 900 mOsm with sucrose, 5 mM MgCl2 (37°C)) rapidly and significantly reduced hemolysis compared with saline and Hank’s buffered saline solution applied either chronically or acutely.
Conclusions
These results on the effect of S. aureus α-toxin on erythrocytes showed that optimizing saline could markedly reduce the potency of S. aureus α-toxin. Such modifications to saline could be of benefit during joint irrigation for septic arthritis.
Introduction
Septic arthritis, a destructive joint disease leading to permanent cartilage damage and disability, affects all ages with an incidence in Western Europe of 4 to 10 cases/100,000 persons/year.1,2 Staphylococcus aureus (S. aureus) accounts for 40% to 70% of all cases of septic arthritis1,3,4 and the incidence is rising due to factors including an aging population, increased use of immunosuppressive agents, musculoskeletal prosthetics, and surgical procedures. 5 Management of septic arthritis is to rapidly eliminate the bacteria and associated toxins through intravenous antibiotics and copious joint irrigation. 5 Despite eradication of the bacteria, the damage caused to articular cartilage persists in >50% of cases and may lead to osteoarthritis (OA).1,2,6
S. aureus produces an array of toxins, including exotoxins (including alpha [α], beta [β], gamma [γ], and delta [δ] hemolysin), 7 which are potent virulence factors.8-11 Recent work using in vitro bovine cartilage and an in vivo murine model of septic arthritis with isogenic mutants of S. aureus, has identified α-toxin as the primary agent causing the rapid death of cartilage cells (chondrocytes).12-14 Chondrocytes are the only cell type capable of maintaining the tissue’s resilience through the turnover of extracellular matrix proteins and their loss, through the action of α-toxin, will result in cartilage degradation. S. aureus α-toxin also has longer term damaging effects on chondrocytes, for example, it may increase expression of catabolic factors, including matrix metalloproteinases (MMPs) and inducible nitric oxide synthase (iNOS) leading to deleterious changes to cartilage metabolism.15-18 While there has been considerable attention given to the development of antibacterials for treating S. aureus infection, the protection of chondrocytes against the deleterious effects of α-toxin has not been as intensively investigated.
S. aureus α-toxin binds to the A Disintegrin And Metalloproteinase 10 (ADAM10) receptor present on animal and human articular chondrocytes and rabbit erythrocytes.19-21 This leads to the formation of a heptameric pore and rapid influx of Na+ and water, causing cell swelling and lysis, leading to the release of intracellular components resulting in inflammation. 22 Rabbit erythrocytes show only low sensitivity to other hemolysins 23 (in contrast to human erythrocytes 24 ) and are therefore an extremely flexible and sensitive model system for studying the interaction between this α-toxin and cell lysis. 24 Additionally, the release of hemoglobin can easily be measured spectrophotometrically, allowing the dynamic effects of biologically relevant activity of α-toxin on cell viability to be assessed. 24
The fluid used for joint irrigation is normally isotonic saline (0.9% NaCl; 300 mOsm), which is hypo-osmotic compared with normal synovial fluid (400 mOsm). 25 Previous work has shown that the sensitivity of chondrocytes to other forms of injury may be markedly reduced when the osmolarity of isotonic saline or culture medium (typically 300 mOsm) is increased.26,27 This raised the possibility that the saline currently used for irrigation might be suboptimal and that altering some of its properties might reduce the injurious effects of α-toxin and thus be chondroprotective against α-toxin. Accordingly, we have tested the effects of osmolarity (300, 400, 600, 900 mOsm), temperature (4°C, 25°C, 37°C), divalent cations (Ca2+ and Mg2+) and pH (6.5, 7.2, 8.0) on the potency of S. aureus α-toxin using the sensitive rabbit erythrocyte hemolysis assay. The aim of this study therefore was to determine if these relatively simple alterations to the properties of standard saline could reduce the damaging effect of S. aureus α-toxin.
Materials and Methods
Biological Materials, Tissue Culture, Reagents
Saline (0.9%; 300 mOsm) used clinically for irrigation was obtained from Baxter Healthcare Ltd., Norfolk, UK. Hank’s buffered saline solution (HBSS; 300 mOsm) was purchased from Invitrogen Ltd., Paisley, UK. HEPES (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid) buffer was obtained from Sigma-Aldrich Chemical Co., Gillingham, UK. TSA (tryptone soya agar), TSB (tryptone soya broth), and skimmed milk were obtained from Oxoid Ltd., Basingstoke, UK.
Rabbit Red Blood Cells
The rabbit red blood cell (RBC) hemolysis assay was used to semiquantitatively determine biologically relevant α-toxin activity. 24 Fresh, heparinized RBCs from Orygen Ltd., Penicuik, UK, were prepared at ~5% hematocrit in saline (0.9% NaCl) and kept at 5°C until required. Fresh blood was obtained weekly and was suitable for up to 5 days of experimentation after receipt.
Preparation of Bacterial Pellets and Supernatant Samples
S. aureus strain 8325-4 was kindly provided by Prof. T. J. Foster and stored at −80°C in 10% v/w skimmed milk. This strain is a well-characterized prophage-cured derivative of strain NCTC8325 that produces large amounts of α-toxin 28 and has comparable potency to clinical strains of S. aureus in terms of chondrocyte-damaging potential. 12 When required, bacteria were thawed and streaked onto TSA plates and cultured (24 hours; 37°C). TSB plates containing 2 µg/mL tetracycline were then prepared and inoculated with several single bacterial colonies from the TSA plates and incubated (24 hours; 37°C) with shaking. From this culture, serial dilutions were performed in saline to 10−6, spread on TSA plates and incubated (24 hours; 37°C). The number of colony forming units/mL (CFU/mL) in TSB was typically ~1 × 109. α-toxin-containing supernatants were obtained by centrifugation (800 × g; 10 minutes) of the TSB cultures, which were then filter-sterilized and stored (4°C) until required, which was within 1 week. To establish an appropriate time course at the beginning of a week’s experiments, the sensitivity of rabbit RBCs to α-toxin were assessed by adding a small volume of toxin to a 5% RBC suspension and incubating at 37°C for 60 minutes. Samples were taken every 10 minutes, centrifuged (10,000 × g; 10 seconds) and hemolysis determined by the absorbance of hemoglobin at 540 nm (Abs540) on a Nanodrop spectrophotometer (ThermoFisher Scientific, Waltham, MA, USA). Maximum (100%) hemolysis was determined by freeze/thawing a sample of the RBC suspension. Percent hemolysis (%H) was then calculated ((Abs540 of sample − Abs540 of negative control)/(Abs540 of 100% hemolysis − Abs540 of negative control) × 100%) to give a measure of α-toxin activity. The sensitivity of erythrocyte samples to α-toxin and the amount and potency of α-toxin produced from each S. aureus culture, were variable. Rabbit erythrocytes which were relatively insensitive to toxin requiring >1 hour of incubation before hemolysis was detectable, were not studied further. A rabbit blood sample that produced ~50% hemolysis after about 15 minutes was considered acceptable for experimentation.
Chronic or Acute Exposure of α-Toxin-Treated Erythrocytes to Various Solutions and Temperatures
Chronic Exposure
Suspensions of rabbit erythrocytes were initially exposed to the experimental conditions of osmolarity, temperature, pH, or divalent cations for 10 minutes before an aliquot of α-toxin was added, the cell suspension mixed quickly, and the time course of percent hemolysis (%H) measurements commenced.
Acute Exposure
The α-toxin-treated erythrocyte suspensions were initially incubated under control conditions and hemoglobin release measured until this reached 20% to 30% hemolysis. The cell suspension was then challenged with the various experimental conditions, and the extent of hemolysis determined until the end of the time course. The rate of change in %H/10 min for the chronic challenge was measured over 10 to 20 minutes and for the acute challenge, the time course was measured over 20 to 30 minutes after the start of the experiment. Data were shown as the change in %H/10 min. For the control condition in the “acute exposure” experiments for osmolarity and divalent cations, an identical volume of saline was added at the same time point to correct for α-toxin dilution. For the pH experiments, HEPES (10 mM) was present and pH adjusted using HCl or NaOH. For some experiments, erythrocytes were suspended in HBSS (pH 7.2).
Data Analysis and Statistics
Data are shown as means ± standard error of the mean (SEM) for N independent experiments and n replicates for each experiment (N(n)) and analyzed using GraphPad Prism Version 7.0b (GraphPad, San Diego, CA, USA). Nonparametric t tests and analyses of variance were performed as indicated and significant differences accepted when P < 0.05.
Results
Suppression of α-Toxin Damage by Raising Saline Osmolarity
To assess the effects of raising saline osmolarity (300 mOsm) above that of synovial fluid (i.e., ⩾400 mOsm) on the damaging effect of α-toxin on rabbit erythrocytes, 2 types of experiments were performed: (1) chronic challenge - where erythrocytes were exposed to the various osmotic conditions, including α-toxin throughout (
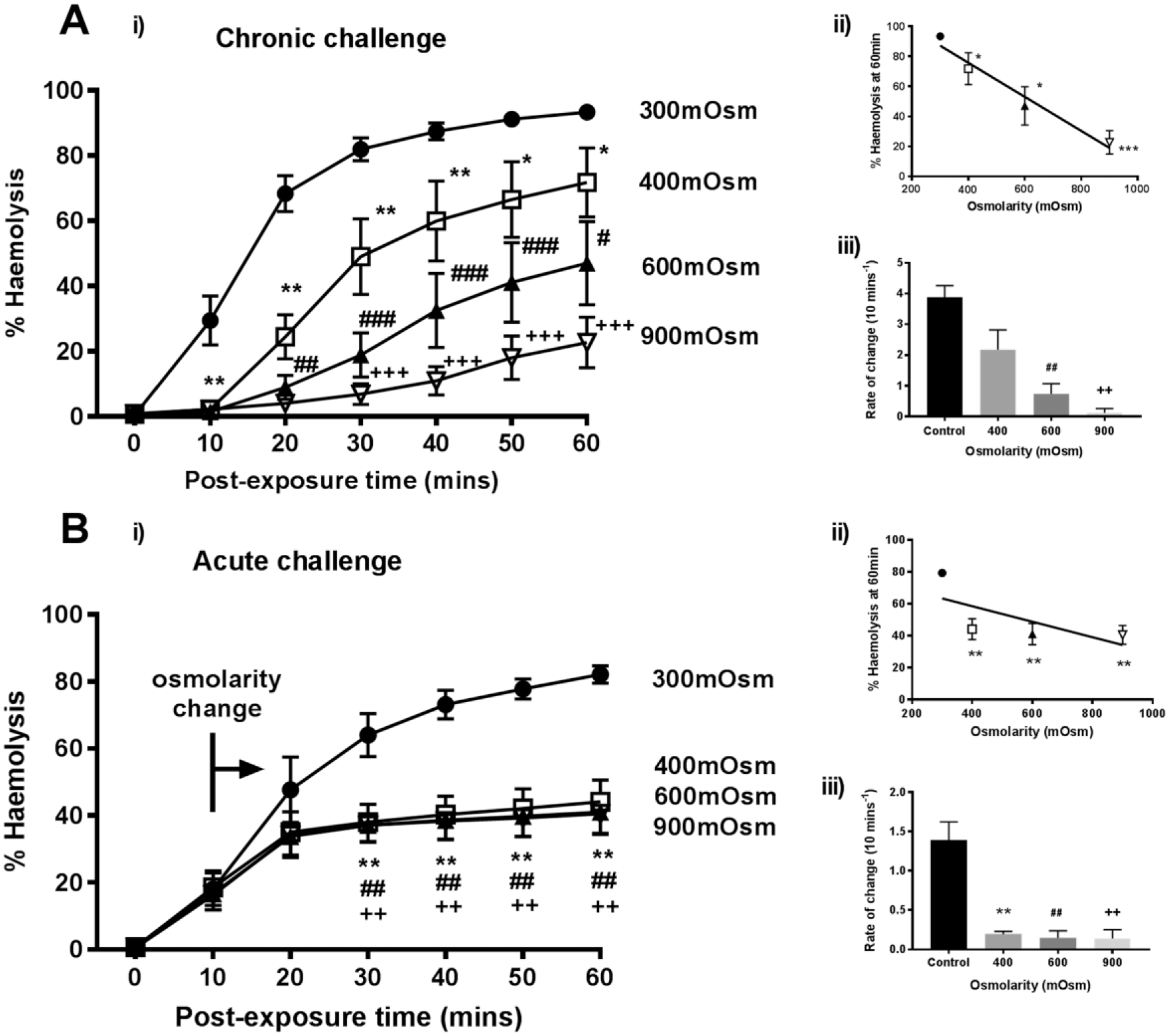
Raised osmolarity reduced rabbit erythrocyte hemolysis induced by Staphylococcus aureus α-toxin. The percent hemolysis (%H) was measured over 60 minutes under either (
When osmolarity was raised >300 mOsm, approximately 10 minutes after α-toxin addition (“acute challenge”), there was rapid and almost complete protection of erythrocytes (
The osmotic protection conferred by sucrose was compared with that of a different osmolyte (NaCl) to the same osmotic pressure. At 40 minutes, NaCl (600 mOsm) reduced %H from the control (300 mOsm) value of 66% ± 6% to 56% ± 8%, whereas with sucrose this was decreased further to 20% ± 3%. In additional experiments at the same time point, when the osmolarity was raised to 900 mOsm, the %H for the control was 68% ± 8% and reduced to 21% ± 7% in the presence of NaCl but was only 4% ± 2% with sucrose (data are means ± SD, N = 2). Although NaCl protected erythrocytes against α-toxin, it appeared less effective when present at the same osmolarity as sucrose.
Reducing Temperature of Saline Conferred Protection against α-Toxin-Induced Hemolysis
RBC suspensions were incubated for 10 minutes at the various temperatures, treated with α-toxin and then hemolysis determined. The chronic exposure to reduced temperature protected erythrocytes treated with α-toxin (
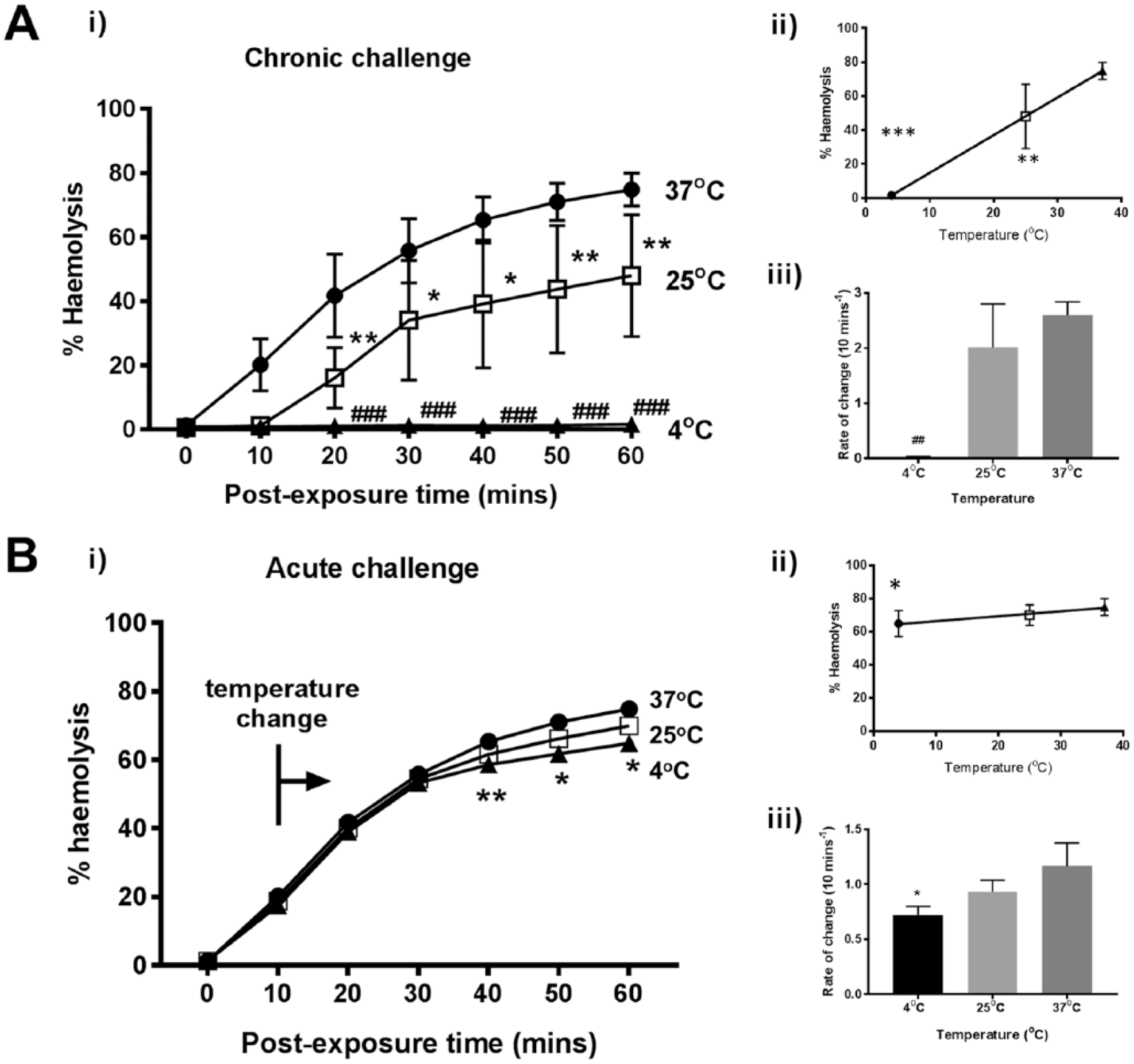
Decreasing temperature suppressed rabbit erythrocyte hemolysis induced by Staphylococcus aureus α-toxin. The percent hemolysis (%H) was measured over 60 minutes under either (
Protective Effect of Divalent Cations against α-Toxin-Induced Erythrocyte Hemolysis
For the chronic exposure experiments, divalent cations (Ca2+ or Mg2+ at 5 mM) produced a modest decrease in hemolysis with significant protection observed as early as 10 minutes with CaCl2 (P = 0.0149) and 20 mins with MgCl2 (P < 0.01;
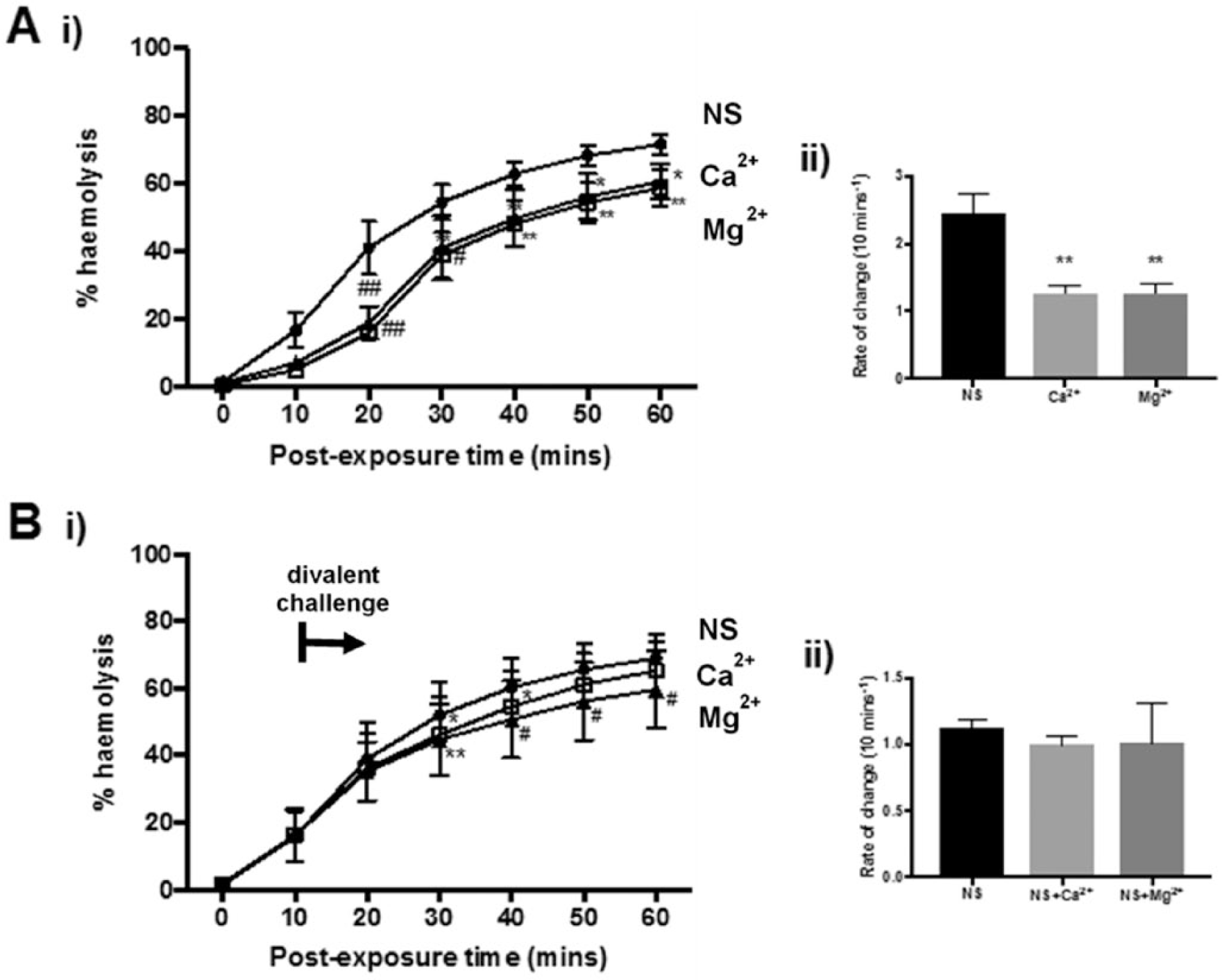
Divalent cations inhibited rabbit erythrocyte hemolysis induced by Staphylococcus aureus α-toxin. The percent hemolysis (%H) was measured over 60 minutes under either (
Influence of pH on α-Toxin-Induced Erythrocyte Hemolysis
Altering saline pH might provide some protection against the damaging effects of α-toxin. A significant decrease in %H occurred between pH 6.5 and 8.0 (2-way analysis of variance; P = 0.0015) at 60 minutes (
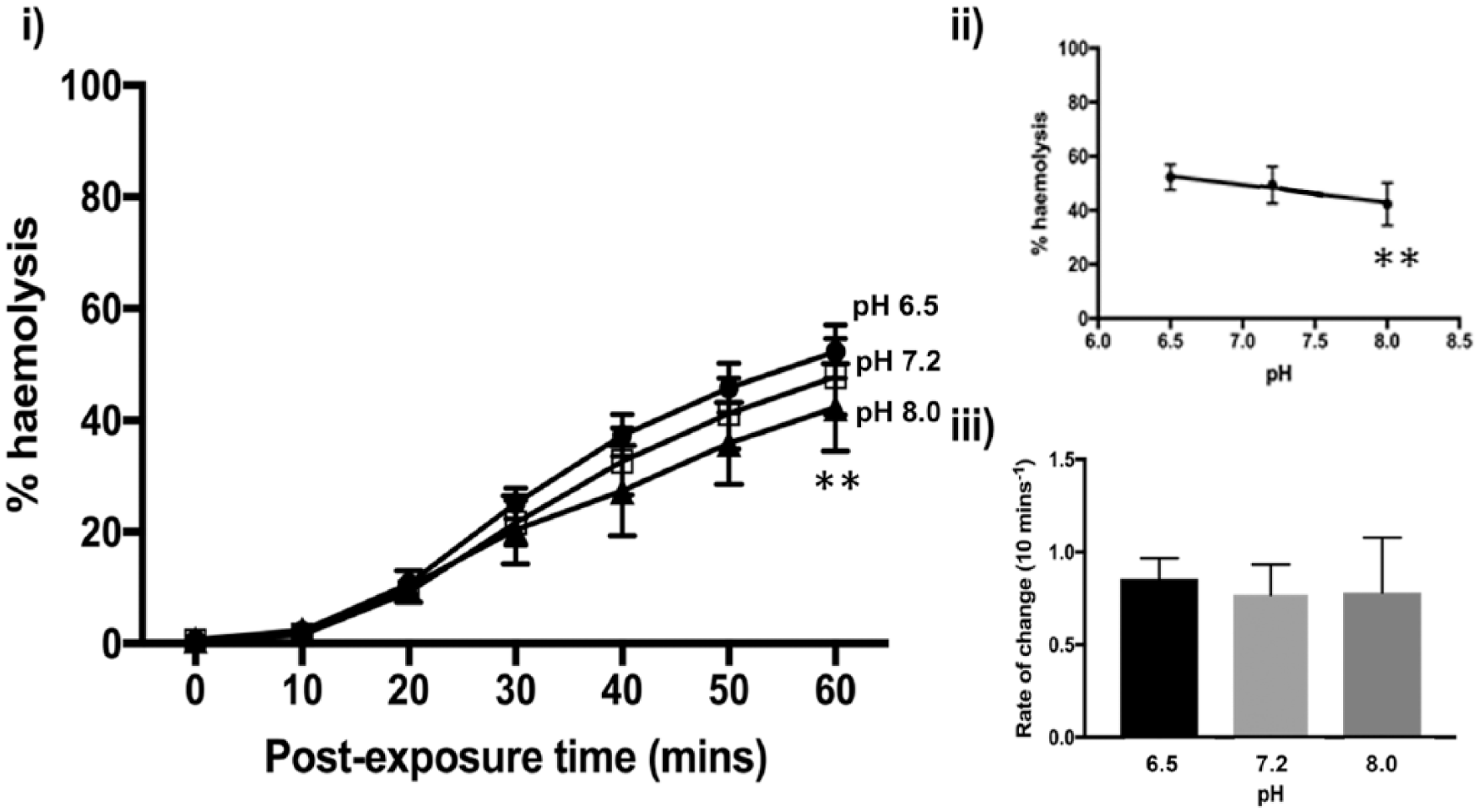
Effect of varying pH on rabbit erythrocyte hemolysis induced by Staphylococcus aureus α-toxin. The percent hemolysis (%H) was measured over 60 minutes under chronic challenge at pH values of 7.2 (control) (□), 6.5 (•), and 8.0 (▲) in Hank’s buffered saline solution (HBSS) containing the buffer HEPES (10 mM) with pH altered using HCl or NaOH (all at 300 mOsm). The panel labeled (
Effect of Optimized Saline on α-Toxin-Induced Erythrocyte Hemolysis
On the basis of the previous results, an optimized saline solution (0.9% NaCl, 900 mOsm, 5 mM MgCl2, 37°C) was prepared. This was compared with normal saline (0.9% NaCl) and HBSS (which contains Ca2+) in its ability to protect erythrocytes against α-toxin (
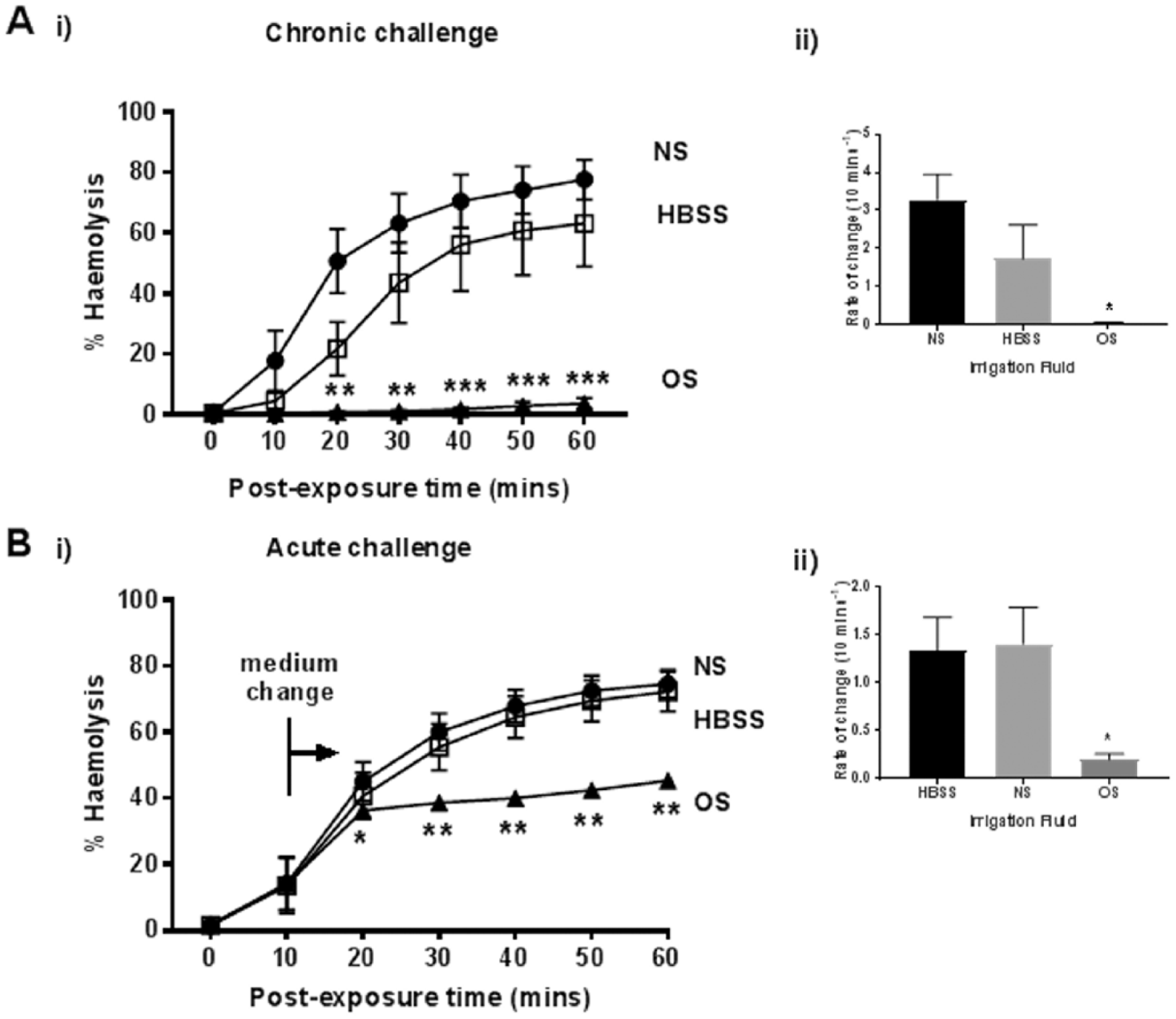
The protective effect of optimized saline on rabbit erythrocyte hemolysis induced by Staphylococcus aureus α-toxin. The percent hemolysis (%H) was measured over 60 minutes under either (
Discussion
During treatment for S. aureus septic arthritis, it is important that the synovial fluid and infected tissues of the joint are rinsed quickly with a benign solution to remove bacteria and associated toxins. Traditionally, saline (0.9% NaCl) is used; however, this might be suboptimal for suppressing α-toxin activity and there may be opportunities for its composition and other properties to be modified to protect chondrocytes. To assess the protective effects of these modified solutions, the release of hemoglobin from rabbit erythrocytes was used as a sensitive and biologically relevant measure of S. aureus α-toxin activity. The results suggested that increased osmolarity, reduced temperature, divalent cations, and to a lesser extent alkaline pH, could significantly reduce the damaging effect of α-toxin suggesting that relatively simple modifications to saline could be of benefit during joint irrigation for septic arthritis.
It could be considered that the rabbit hemolysis assay for determining methods for protecting cells against the damaging effects of α-toxin would not be an appropriate model. However, rabbit erythrocytes contain the S. aureus α-toxin receptor ADAM10, which is also present on chondrocytes of animals and normal and degenerate human cartilage.19-21 The measurement of hemoglobin release following the interaction between α-toxin and erythrocytes provided a dynamic and sensitive assay for the lethal effects of α-toxin and was highly flexible experimentally and reproducible. While it would be possible to test these conditions on cartilage explants, interpreting the results obtained under these various conditions might not be as straightforward, as for example access to the receptor in cartilage zones could be delayed and/or restricted. While studies on isolated chondrocytes could be of benefit, the receptor may be damaged or its sensitivity altered as a result of the enzymic treatment of cartilage, which is required for the release of chondrocytes. Our previous work has demonstrated that S. aureus α-toxin is the key damaging agent to chondrocytes in a cartilage model of septic arthritis.12,14 Thus, although the rabbit erythrocyte model could be considered a limitation in this study, it nevertheless yielded valuable information about whether protection against α-toxin was possible and identified alterations to the irrigation fluid which could potentially be extended to detailed in vivo and clinical studies on S. aureus septic arthritis.
Two protocols were used, which would broadly correspond to different stages of α-toxin action on the cell membrane, which is time-dependent with the binding and pore formation occurring within 2 to 3 minutes.
24
For the chronic challenge, erythrocytes were equilibrated with the various conditions (osmolarity, temperature, pH, divalent cations), before toxin was added and the hemolysis time course commenced. For the acute challenge, the time course was started by α-toxin addition to the RBC suspension, and when there was 20% to 30% hemolysis, the erythrocytes were exposed to the experimental conditions. Thus, the chronic exposure would mainly represent the effect of experimental conditions on early steps of toxin action but for the acute exposure, pore formation would be complete and the pathological changes (i.e., increased ion permeability, cell swelling) would be underway with hemolysis following. The acute exposure would be closer to the clinical situation where the majority of cells in the joint would already have been exposed to prevailing levels of α-toxin, and cell injury/death would be proceeding. While the overall effects of chronic and acute challenges of osmolarity, reduced temperature and divalent cation concentration (
Raising osmolarity prior to toxin addition (chronic osmotic challenge) reduced the rate of hemolysis in a dose-dependent manner (
Preincubating erythrocytes at 25°C delayed the damaging action following α-toxin addition. However, once established, the rate of increase in hemolysis was the same as for cells equilibrated at a physiological temperature (
There was a mild but significant protection of erythrocytes when pH was increased from 6.5 to 8.0 (
Ca2+ and Mg2+ produced small but significant protection against α-toxin (
On the basis of these experiments, we tested an optimized saline applied as either a chronic or an acute challenge and observed substantial protection of erythrocytes against α-toxin (
While the present results were obtained using the rabbit RBC model, some caution should be exercised when extrapolating these results to the protection of chondrocytes within the cartilage matrix. Previous studies using a bovine osteochondral explant model have shown that S. aureus α-toxin can rapidly penetrate the matrix and cause chondrocyte death. 12 We have also shown that chondrocyte volume changes very quickly (within minutes) following alterations to extracellular osmolarity. 39 Thus there is the expectation that by raising osmolarity, protection of in situ chondrocytes against α-toxin should be achieved in the same way this has been demonstrated with rabbit erythrocytes. With these observations in mind and taking the results from the present study together with previous observations, the beneficial effects of modifying the irrigation saline used during joint lavage should be considered in further in vivo animal and/or clinical research. The use of a relatively benign, inexpensive, drug-free and rapidly effective modified saline as part of the normal lavage process is potentially an attractive novel method for limiting the damaging action of S. aureus α-toxin during septic arthritis.
S. aureus infections have been treated with β-lactams (e.g., penicillin) for decades, but the appearance and rapid spread of methicillin-resistant S. aureus (MRSA) have all but eliminated these antibacterials for treatment. 40 Non-antibiotic treatment is therefore an area of important research interest since suppressing activity of bacterial toxins either by influencing toxin production, or blocking their action would not only make the bacteria less pathogenic, but may also increase their susceptibility to host immune defence. 41 For example, inhibition of S. aureus pathogenesis by interfering with the signal transduction pathways for virulence using the RNA III inhibiting peptide has been described. 42 This peptide reduced the pathology and delayed the onset of disease symptoms in models of S. aureus infection, including septic arthritis. 42 Other methods include an α-toxin antibody, 43 cyclodextrin-lipid complexes to suppress the damaging effect of S. aureus α-toxin 44 and nanoparticle-based α-toxin entrapment to deliver the nondisrupted pore-forming toxin for immune processing. 45 These methods could be particularly important for cells with high levels of the ADAM10 receptor, 21 which would render them particularly sensitive to S. aureus α-toxin. In summary, the development of the optimized irrigation saline described here potentially offers a cheap, very rapid (within minutes) and relatively benign method to suppress the damaging effects of α-toxin and may be of benefit during joint irrigation for septic arthritis caused by S. aureus.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank Prof. T. J. Foster, Trinity College, Dublin for providing Staphylococcus aureus strain 8325-4.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
