Abstract
Objective
To determine the characterization of chondrogenic properties of adeno-associated virus type 2 (AAV2)-delivered hFGF18, via analysis of effects on primary human chondrocyte proliferation, gene expression, and in vivo cartilage thickness changes in the tibia and meniscus.
Design
Chondrogenic properties of AAV2-FGF18 were compared with recombinant human FGF18 (rhFGF18) in vitro relative to phosphate-buffered saline (PBS) and AAV2-GFP negative controls. Transcriptome analysis was performed using RNA-seq on primary human chondrocytes treated with rhFGF18 and AAV2-FGF18, relative to PBS. Durability of gene expression was assessed using AAV2-nLuc and in vivo imaging. Chondrogenesis was evaluated by measuring weight-normalized thickness in the tibial plateau and the white zone of the anterior horn of the medial meniscus in Sprague-Dawley rats.
Results
AAV2-FGF18 elicits chondrogenesis by promoting proliferation and upregulation of hyaline cartilage–associated genes, including COL2A1 and HAS2, while downregulating fibrocartilage-associated COL1A1. This activity translates to statistically significant, dose-dependent increases in cartilage thickness in vivo within the area of the tibial plateau, following a single intra-articular injection of the AAV2-FGF18 or a regimen of 6 twice-weekly injections of rhFGF18 protein relative to AAV2-GFP. In addition, we observed AAV2-FGF18-induced and rhFGF18-induced increases in cartilage thickness of the anterior horn of the medial meniscus. Finally, the single-injection AAV2-delivered hFGF18 offers a potential safety advantage over the multi-injection protein treatment as evidenced by reduced joint swelling over the study period.
Conclusion
AAV2-delivered hFGF18 represents a promising strategy for the restoration of hyaline cartilage by promoting extracellular matrix production, chondrocyte proliferation, and increasing articular and meniscal cartilage thickness in vivo after a single intra-articular injection.
Introduction
Aging results in several characteristic changes in a number of tissues, the most pronounced of which are a reduction in cellularity and concentration of physiologically appropriate extracellular matrix (ECM).1 -3 Much like skin, tendon, and bones, articular cartilage thickness and cellularity decrease with age as a result of a homeostatic imbalance that contributes to an increased risk of osteoarthritis (OA).4,5 With age, degenerative processes exceed anabolic activity in cartilage, leading to a loss of hyaline cartilage ECM components and a decrease in the number of functional chondrocytes, eventually manifesting in symptomatic OA.4,6 Age is the leading risk factor for primary OA 7 and cartilage thickness is highly correlated with disease severity and progression to joint replacement surgery.8 -10 While inflammation is associated with OA, anti-inflammatory treatments have shown no clinical benefit in disease modification,11 -13 and as of today, there are no approved disease-modifying OA treatments in the United States or the European Union. Finally, loss of articular cartilage and the resulting joint space narrowing are strongly associated with long-term risk of total knee replacement surgery, which is a highly debilitating procedure with poor outcomes reported by as many as 1 in 5 patients. 14 All in all, OA and the associated cartilage pathology affect 30 million US adults amounting to $318.4 billion in incurred healthcare costs, or 22.5% of the noninstitutionalized adult healthcare system expenses. 14
Slowing the rate of cartilage loss or transiently reversing degenerative dynamics within cartilage may be critical to preventing or delaying progression to joint replacement surgery. Recent 5-year follow-up data from a placebo-controlled, multicenter, randomized clinical trial evaluating the safety and efficacy of a truncated, recombinant human FGF18 protein analog (rhFGF18) demonstrated dose-dependent, statistically significant increases in cartilage thickness relative to a placebo control. 15 Similar chondrogenic effects were also observed in rodents and horses,16,17 benefiting even the most severe, fully denudated phenotypes. 17 The protein injection approach, however, is limited by the requirement to administer up to 12 intra-articular doses per year in bi-lateral OA treatment. Moreover, protein injections may need to be continued indefinitely to prevent reversion of degenerative dynamics. 15
The anatomic advantages offered by the constrained environment of the joint capsule, which enables a high degree of vector localization and optimal transgene expression durability, 18 create a unique opportunity to treat cartilage loss and OA using gene therapy. Moreover, the ability of adeno-associated virus type 2 (AAV2) vectors to transduce cells of the joint capsule and stably express a broad range of proteins without eliciting an inflammatory response for long durations (12 months) can help overcome the dosing limitations of the rhFGF18 protein analog therapy.18,19 Such a prospective treatment must be safe and durable, while eliciting no pathological, uncontrollable growth in the areas of healthy cartilage. In this study, we directly compare AAV-delivered hFGF18 with the rhFGF18 protein in vitro and in vivo with the aim of characterizing the mechanisms of FGF18 activity and comparing treatment safety and chondrogenic potential.
Methods
Production of Protein and AAVs
Bacterially expressed, rhFGF18 (O76093) protein (AA27-199) was manufactured by Peprotech (Rocky Hill, NJ) (#100-28). Eukaryotically expressed rhFGF18 was produced by transient transfection of human embryonic kidney (HEK) cells with plasmids encoding the full-length cDNA of hFGF18 under the regulation of the cytomegalovirus (CMV) promoter (Arvys, Trumbull, CT). AAV2-FGF18 (AAC62240.1), AAV2-GFP (AAB02574.1), and AAV2-nLuc (QMV47821.1) were produced using triple-transfection and a HEK293 mammalian viral vector packaging system (VectorBuilder, Chicago, IL); all transgenes were expressed under the regulation of the CMV promoter and flanked downstream by the woodchuck hepatitis virus posttranscriptional regulatory element followed by the bovine growth hormone polyadenylation signal.
Cell Culture, Cytocompatibility, Transduction Efficiency
Chondrogenic properties of AAV2-hFGF18 were compared with HEK-expressed rhFGF18 protein and E. coli-expressed truncated rhFGF18 analog in vitro, relative to AAV2-GFP and PBS controls. Primary human articular chondrocytes (from healthy, non-damaged sections of joints) were cultured in Chondrocyte Differentiation Medium (CDM, Lonza #CC-3225, 5% fetal bovine serum [FBS]) supplemented with 70 µg/mL ascorbic acid and 1% antibiotic-antimycotic (anti-anti, Gibco, Waltham, MA, #15240062). Synoviocytes were cultured in Dulbecco’s Modified Eagle Medium (DMEM; Gibco #11965092) supplemented with 10% FBS (Gibco #16000-044) and 1% anti-anti. Primary human chondrocytes (isolated as described by Otero et al.) 20 and primary human fibroblast-like synoviocytes (Cell Applications, San Diego, CA, #408-05A) were cultured in 24-well plates with AAV2-FGF18 or AAV2-GFP (various multiplicities of infection [MOI]) for up to 168 hours, with 6 biological replicates per group. Cytocompatibility, proliferation, and transduction efficiency were measured by measuring cell counts (Hoechst nuclear stain) and presence of green fluorescent protein (GFP) over time (168 hours) after AAV2-GFP transduction by fluorescent microscopy.
Transwell Study Experiment
Synoviocytes were seeded at 7,500 cells per transwell insert (Sterlitech Corporation, Auburn, WA, #9320412) in DMEM supplemented with 10% FBS and 1% anti-anti and were allowed to settle overnight. The next day, the synoviocytes were transduced with AAV2-GFP (MOI 500,000) or AAV2-FGF18 (MOI 50,000 or 500,000) and incubated for 6 days to allow for expression. After 6 days, the transwell inserts were transferred to a 24-well plate containing 10,000 chondrocytes per well. Control wells of chondrocytes received transwells with either PBS or 1,000 ng/mL bacterially expressed rhFGF18 without synoviocytes. All conditions were tested in triplicate. After 24 and 48 hours (depending on the plate), EdU was added to the chondrocytes and the plate was incubated for an additional 24 hours (for a total of 48 or 72 hours). The click-chemistry reaction with EdU to assess proliferation was performed using a Click-iT™ Plus EdU AlexaFluor™ 488 Imaging Kit (Invitrogen, Waltham, MA, #C10637) according to the manufacturer’s instructions and imaged on an Olympus IX71 with DP80 camera and associated software.
RNA-Seq Analysis
Chondrocytes were seeded into 24-well plates at a density of 1 × 104 cells/cm2 in CDM supplemented with 70 µg/mL ascorbic acid and 1% anti-anti, and allowed to settle overnight. The next day, the chondrocytes were switched to serum-free CDM, supplemented with ascorbic acid and anti-anti as above, and serum-starved overnight. After serum starvation, the cells were switched to CDM supplemented with 0.1% FBS, 70 µg/mL ascorbic acid, and 1% anti-anti, and cultured with PBS, 1,000 ng/mL bacterially expressed rhFGF18, AAV2-FGF18 (MOI10, a dose previously confirmed as not capable of enhancing chondrocyte proliferation in culture), or AAV2-GFP (MOI10) for 4 days, using 5 biological replicates per group. RNA was isolated using the PureLink RNA Mini Kit (Invitrogen #12183025) and RNA-seq was performed by Tufts Genomics Core. Genome was aligned to hg38 using STAR (v2.7.0a) and analyzed with DESeq2 with a gene expression change Fold Cutoff Multiple of 3x or greater. Generic gene ontology (GO) analysis was performed by the GO Term Mapper provided by Princeton University, Lewis-Sigler Institute for Integrative Genomics using the Homo sapiens GOA @EBI + Ensembl databases analyzing biological process–related GO terms.
Animals
All animal care and experimental procedures used in this study were approved by the Institutional Animal Care and Use Committee at Tufts University (Protocol #B2021-106). Male Sprague-Dawley rats (300-375 g) were housed under standard conditions (14-hour light/10-hour dark cycle, standard chow diet ad libitum). The hind limbs of the rats were shaved, and each knee joint received a 50-μL intra-articular injection of 5 µg bacterially expressed rhFGF18 (twice weekly for 3 weeks), or a single 50-μL injection of 2 × 108 – 1 × 1011 viral genomes of AAV2 (AAV2-FGF18, AAV2-GFP, or AAV2-nLuc) using a 30-gauge needle. The knee was flexed and extended several times to distribute the fluid. A single rat received the same treatment in both knees.
Immunohistochemistry, Histology, and Thickness Analysis
Sagittal paraffin sections were taken at 5 µm thickness. For Saf O/fast green staining, deparaffinized slides were stained with 0.2% Gill’s hematoxylin (Ricca Chemical Company #3535-32) and blued in 3 changes of tap water. A solution of 0.02% fast green in 0.2% acetic acid was applied to the slides. The slides were then rinsed briefly with 1% acetic acid and incubated with a solution of 1% safranin O in water. Following mounting of coverslips, the slides were imaged on an ImageXpress Pico (Molecular Devices, San Jose, CA). The area of the meniscal tip was measured by super-positioning a 0.73″-height rectangular marker in the interior portion of the medial meniscus (anterior horn) and extending the rectangle to form a tangent with the meniscal tip and separation between the white (Safranin O high-density area) and red zones (fast-green high-density area). The tibial plateau was measured at 6 positions along the apex of the tibial cartilage by first positioning 6 equidistant rectangular markers (1.54″ × 1.54″) 1.49″ apart, shifting the markers into the hyaline cartilage zone, rotating the markers about their central axis to align with the cartilage surface, and extending the marker boundaries to span the hyaline cartilage zone from the subchondral bone to the synovial cavity. A total of n = 12 joints (6 animals, 2 joints) were injected with each treatment group. A total of 2 sections were collected per joint for staining and cartilage thickness measurement; all measurements were performed by blinded operators, who were not involved in animal handling and injection administration. Histological analysis of synovial inflammation was performed on Saf O–stained sections according to the OARSI Histopathology Initiative guideline for synovial inflammation scoring in rats. 21 Briefly, the anterior synovia and posterior synovia of each section were given a score 0-4 based on the thickness of the synovial lining, proliferation of synovial cells, and presence of inflammatory cells. For immunohistochemistry, deparaffinized slides were subjected to heat-induced antigen retrieval in 10 mM citrate, 0.05% Tween-20 (pH 6.0) in a vegetable steamer. Endogenous peroxidase activity was blocked with 3% hydrogen peroxide and the slides were blocked with 10% goat serum in PBS. The slides were incubated with rabbit anti-FGF18 (Invitrogen #PA5-106562) diluted 1:100 in 10% goat serum in PBS overnight, then washed thoroughly and incubated with goat anti-rabbit Immunoglobulin G (Vector Laboratories, Burlingame, CA, #BA-1000) diluted 1:100 in PBS. Detection was carried out using an ABC Kit (Vector Laboratories, #PK-4000) followed by a DAB Kit (Vector Laboratories, #SK-4100) according to the manufacturer’s instructions. Quantification of anti-FGF18 positive staining was performed using ImageJ 1.53t. Specifically, 4 regions of interest were selected in the tibial and femoral cartilage, such that at least 50% of the total visible cartilage were covered. Images were converted to 8-bit grayscale and a common threshold was applied. Following, over/under thresholding, using default settings, the Analyze Particles function was applied covering the full size and circularity range to obtain the total area covered by anti-FGF18 positive staining.
Bioluminescence Imaging
Durability of gene expression in the joint following intra-articular injection was confirmed in vivo by bioluminescence imaging of rats injected with AAV2-nLuc. Fifty microliters of a 1:50 dilution of fluorofurimazine solution (Nano-Glo® In Vivo Substrate, Promega, Madison, WI) were administered intra-articularly. Imaging was performed using the PerkinElmer IVIS Spectrum CT Biophotonic Imager 7 minutes following injection.
Joint Diameter Measurements
Joint diameter measurements were performed in triplicate per joint using mechanical Vernier calipers. Measurements were performed across all treatment groups (n = 6 animals, across both joints). Additional measurements were performed on uninjected animals to provide a population background average joint diameter, as well as at the 1- and 2-month timepoints following AAV2-FGF18 injection, AAV2-GFP control injection, and the bi-weekly injections of rhFGF18 protein.
Statistics
Blinding and randomization best practices were applied to all experiments to the extent possible. Raw data were analyzed for normality using the Anderson-Darling normality test. Normally distributed data were analyzed using analysis of variance (ANOVA), followed by Tukey’s pairwise comparison post hoc testing for multifactorial experiments and Student’s t-test for one factor, multilevel or two-factor one-level studies. Data are presented as mean ± standard deviation. A significant difference was considered at a P-critical value (Pcrit) of <0.05, except for RNA-seq analysis where a Pcrit of <0.01 was used to align with RNA-seq best practices. Confidence intervals are reported at 99% of the test statistic. All statistical analyses were performed using Minitab version 21.1 (State College, PA).
Results
To evaluate the cytocompatibility of the AAV2 vector, primary human chondrocytes and primary human synoviocytes were treated with increasing doses of AAV2 encoding a GFP reporter transgene ranging from an MOI of 1,000 to an MOI of 500,000 ( Fig. 1 ). Despite the high MOI used in the study, no dose-dependent cytotoxicity was observed in vitro for either chondrocytes or synoviocytes ( Fig. 1A and B , respectively). The number of GFP+ synoviocytes and chondrocytes rapidly increased between 24 and 168 hours following AAV2-mediated delivery of the GFP reporter transgene. An MOI of 50,000 resulted in 97 ± 3% GFP+ chondrocytes, while synoviocyte transduction at an MOI of 50,000 resulted in only 58 ± 16% GFP+ cells, suggesting that chondrocytes are more conducive to AAV2 transduction than synoviocytes in vitro.

Cytocompatibility ( synoviocytes and
synoviocytes and  chondrocytes (cytocompatibility: PTime > 0.05, PDose > 0.05, transduction efficiency: PTime < 0.05, PDose < 0.05, two-way ANOVA, n = 6). ANOVA = analysis of variance; MOI = multiplicities of infection.
chondrocytes (cytocompatibility: PTime > 0.05, PDose > 0.05, transduction efficiency: PTime < 0.05, PDose < 0.05, two-way ANOVA, n = 6). ANOVA = analysis of variance; MOI = multiplicities of infection.
Since the rhFGF18 protein analog evaluated in clinical trials is truncated (AA28-196), methionine initiated, and expressed in E. coli, 15 while the protein produced by an hFGF18 gene therapy would be the full-length glycosylated protein, we tested the ability of eukaryotically expressed rhFGF18 to promote dose-dependent proliferation, relative to the bacterial analog. The E. coli–expressed rhFGF18 protein analog (Bac) and HEK293-expressed rhFGF18 protein (HEK) both induced dose-dependent proliferation in primary human chondrocytes. This result was not statistically different between the two test groups but demonstrated statistical significance for the dose factor ( Fig. 2A and C ). A similar dose response was observed in chondrocytes treated at MOIs of 100 and 1,000 ( Fig. 2B and D ). Chondrocytes dosed at an MOI of 1,000 reached an 84% increase in cell count relative to baseline (t = 0), which was comparable with the 85% increase attained by the chondrocytes dosed with 10,000 ng/mL of the bacterially expressed rhFGF18 analog.
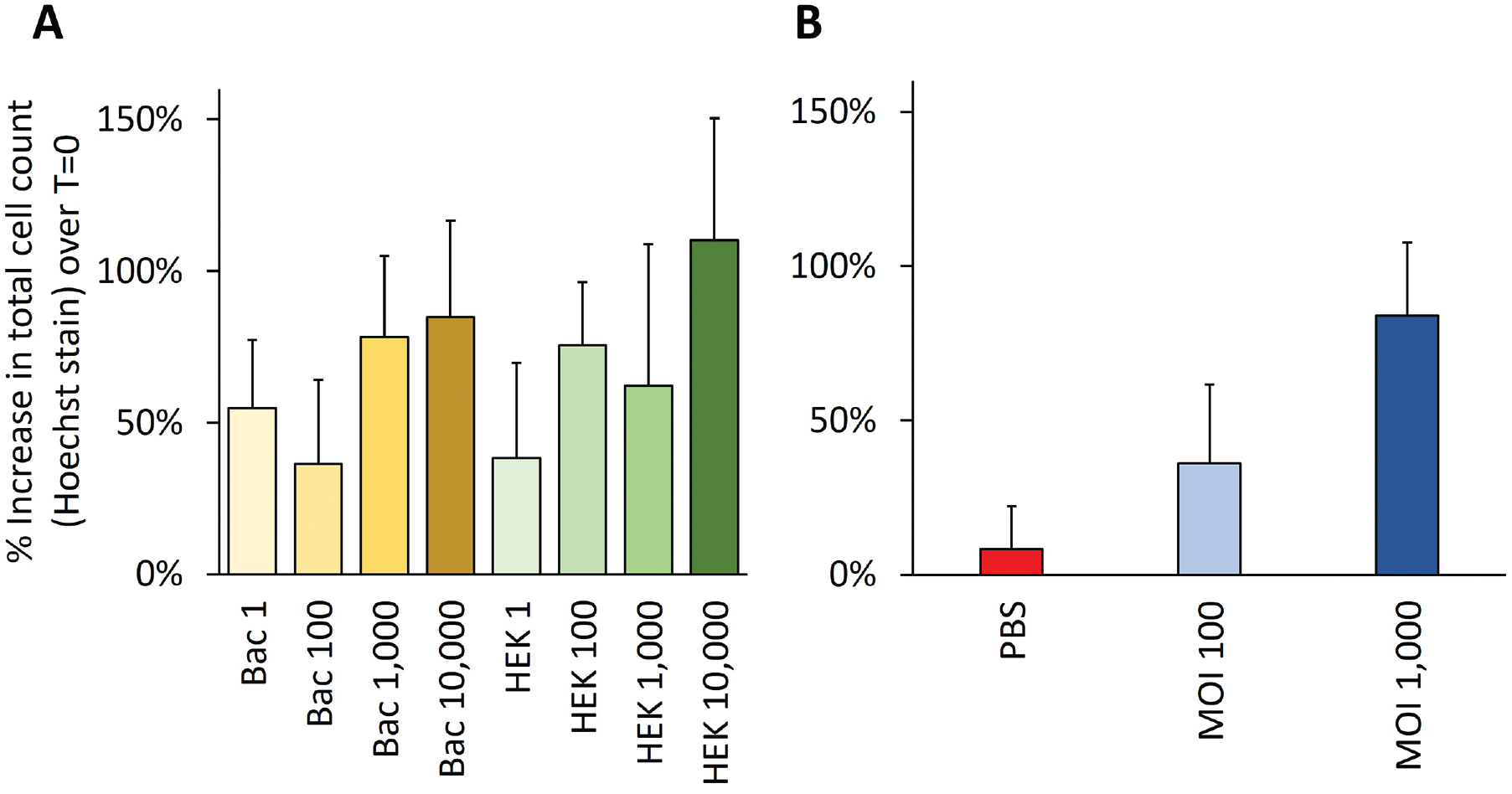
Proliferation of human chondrocytes upon stimulation by (
Since synoviocytes are oftentimes the target cell transduced by intra-articularly delivered AAV vectors,22 -24 we further evaluated the paracrine effect of hFGF18 expressed by synoviocytes in transwell culture with primary human chondrocytes ( Fig. 3 ). The number of proliferating chondrocytes increased by 134% between 48 and 72 hours following exposure to AAV2-FGF18 at MOI of 500,000; a 135% increase was observed for the rhFGF18 treatment group, while only a 6% increase was observed for the AAV2-GFP negative control dosed at MOI of 500,000.

Effect of  rhFGF18 protein and AAV2-FGF18 gene therapy at
rhFGF18 protein and AAV2-FGF18 gene therapy at  MOI 500k and
MOI 500k and  50k on chondrocytes in transwell culture with synoviocytes relative to
50k on chondrocytes in transwell culture with synoviocytes relative to  AAV2-GFP negative control (one-way ANOVA, P < 0.05; Tukey’s pairwise comparison grouping indicated by letter code, Pcrit = 0.05, n = 3). MOI = multiplicities of infection; GFP = green fluorescent protein; ANOVA = analysis of variance; AAV2 = adeno-associated virus type 2.
AAV2-GFP negative control (one-way ANOVA, P < 0.05; Tukey’s pairwise comparison grouping indicated by letter code, Pcrit = 0.05, n = 3). MOI = multiplicities of infection; GFP = green fluorescent protein; ANOVA = analysis of variance; AAV2 = adeno-associated virus type 2.
To determine whether FGF18 gene therapy would induce the same transcriptional profile as rhFGF18 protein in chondrocytes, we compared the gene expression profile of chondrocytes exposed to high doses of rhFGF18 protein (1,000 ng/mL) and sub-proliferative doses of AAV2-FGF18 (MOI 10). The experimental design allowed for the identification of a holistic set of genes upregulated by the rhFGF18 protein for comparison with the most significantly upregulated subset from the AAV2-FGF18 gene therapy–treated group. All but 2 genes (32/34 or 94%), TGM2 and SERPINA1, upregulated in the AAV2-FGF18-treated chondrocytes at the 3-fold cutoff (3x) were also upregulated following treatment with rhFGF18. Similarly, all genes (9/9 or 100%) downregulated by the AAV2-FGF18 gene therapy at the 3x cutoff were also downregulated in the rhFGF18-treated chondrocytes ( Fig. 4 ).

( up- and
up- and  down-regulated in AAV2-FGF18-treated chondrocytes in comparison with the full set of genes
down-regulated in AAV2-FGF18-treated chondrocytes in comparison with the full set of genes  up- and
up- and  down-regulated in rhFGF18 protein–treated chondrocytes. *ESM1 was also upregulated by AAV2-GFP control over PBS, however, by less than 3x. (
down-regulated in rhFGF18 protein–treated chondrocytes. *ESM1 was also upregulated by AAV2-GFP control over PBS, however, by less than 3x. ( up- and
up- and  down-regulated by AAV2-FGF18 and rhFGF18 protein analog, relative to PBS (n = 5). GFP = green fluorescent protein; GO = gene ontology; PBS = phosphate-buffered saline; AAV2 = adeno-associated virus type 2.
down-regulated by AAV2-FGF18 and rhFGF18 protein analog, relative to PBS (n = 5). GFP = green fluorescent protein; GO = gene ontology; PBS = phosphate-buffered saline; AAV2 = adeno-associated virus type 2.
Genes specifically associated with hyaline cartilage phenotype, such as HAS2, 25 ACAN (Aggrecan), 26 COL2A1, 27 and PRG4 (Proteoglycan 4), 28 were all upregulated in AAV2-FGF18-treated chondrocytes (with only ACAN not achieving statistical significance at a Pcrit = 0.01). In contrast, rhFGF18 protein–treated chondrocytes upregulated HAS2, COL2A1, and PRG4 (with PRG4 not achieving statistical significance at a Pcrit = 0.01); however, a slight non-statistically significant reduction in ACAN expression was observed ( Fig. 5 ). The fibrocartilage ECM–associated genes COL1A1, 29 ADAMTS15 (disintegrin and metalloproteinase with thrombospondin motifs), 30 and LOX (protein-lysine 6-oxidase, a lysyl oxidase family cuproenzyme) 31 were downregulated in the AAV2-FGF18-treated group and the rhFGF18 protein–treated group (with ADAMTS15 not achieving statistical significance in the protein-treated arm at a Pcrit = 0.01). Interestingly, AAV2-FGF18-treated chondrocytes also upregulated SOX9, a chondrocyte differentiation marker, 32 which was not observed in the rhFGF18 protein–treated group.
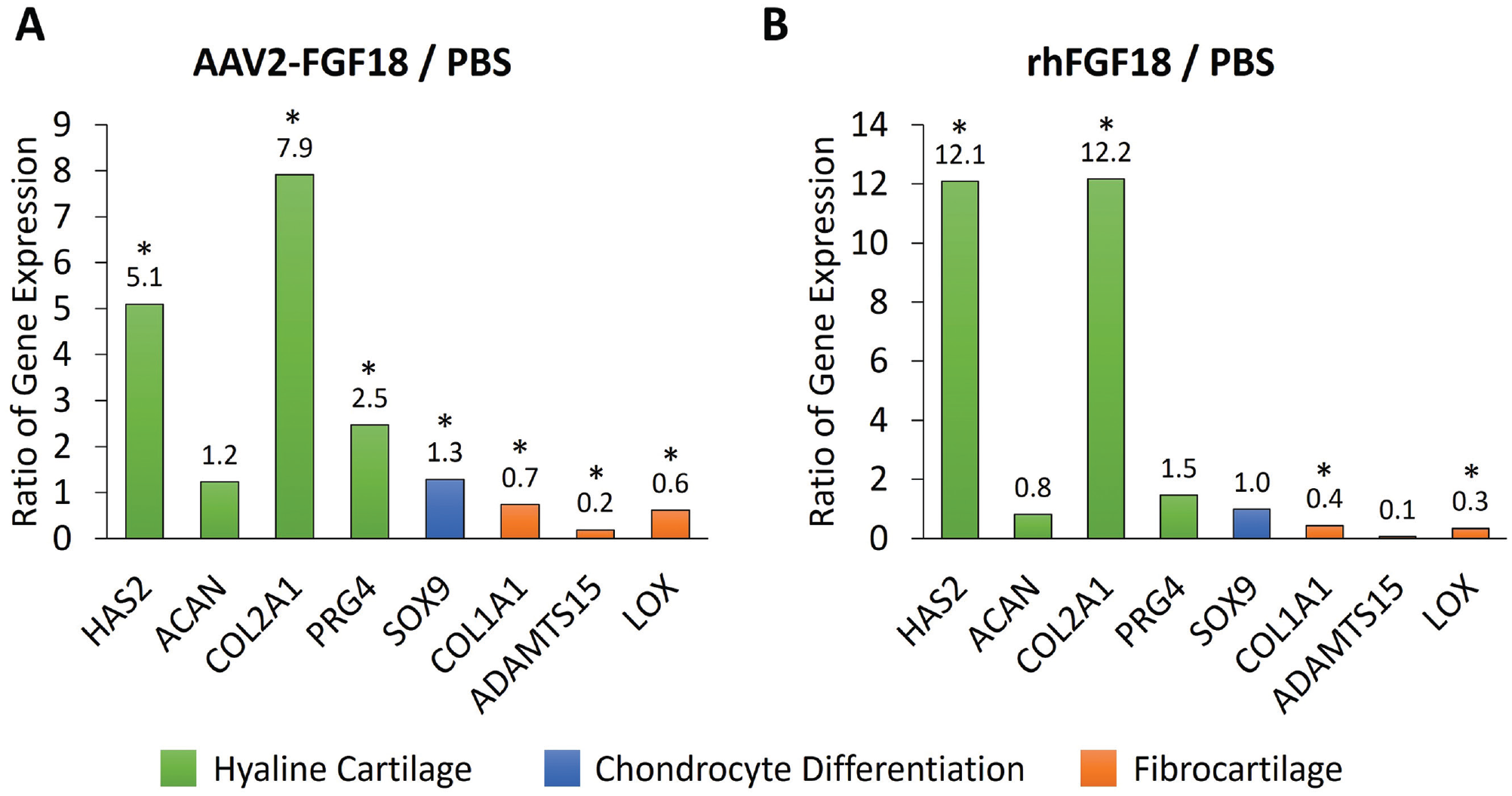
Effect of AAV2-FGF18 gene therapy (
To determine the durability of gene expression, we confirmed the presence of hFGF18 at the 2-month, terminal study timepoint using immunohistochemistry in the AAV2-FGF18-active group relative to AAV2-GFP control and AAV2-GFP with no primary antibody (Fig. 6A-C). In line with these findings, we observed an increase in FGF18 staining by 73% in AAV2-FGF18-treated knees on a relative basis over the natural expression levels of FGF18 in the AAV2-GFP-treated knees. The level of FGF18 staining observed in the “No Primary Antibody” negative control is effectively zero (Suppl. Fig. S1). In addition, we injected an AAV2 vector carrying nano-luciferase (nLuc) intra-articularly and observed the signal using bioluminescence imaging 4 months following dosing ( Fig. 6D ). A strong bioluminescent signal was detected locally in the joint following intra-articular administration of the furimazine substrate, indicating that the cells transduced by the vector expressed the reporter protein several months after a single injection of AAV2-nLuc.
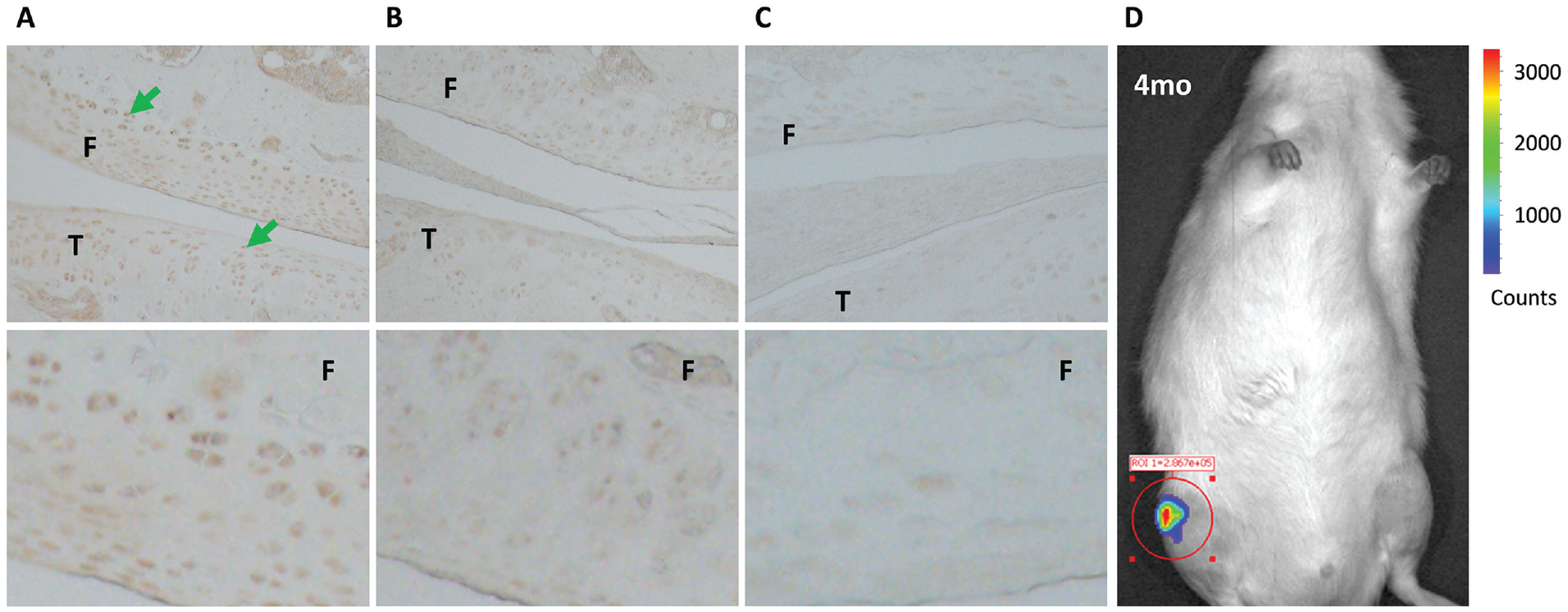
Histological sections stained for FGF18: (
We measured the thickness of articular cartilage in the tibial plateau at 6 positions along the sagittal plane and at the meniscal tip (white zone of the anterior horn of the medial meniscus) in Safranin O/fast green-stained sections to determine the effect of AAV2-FGF18 on cartilage thickness in vivo. We observed a qualitative increase in cartilage thickness in the AAV2-FGF18 and rhFGF18 treatment groups relative to the AAV2-GFP negative control. No observable effects to the subchondral bone, marrow content or morphology, as well as the pars intermedia section of the meniscus were noted in any of the histological sections ( Fig. 7 ). Finally, no signs of inflammation, abnormal growths, or degenerative processes within cartilage or underlying bone were observed within the areas examined, indicating optimal in vivo biocompatibility of the AAV2 vector and AAV2-FGF18 gene therapy.
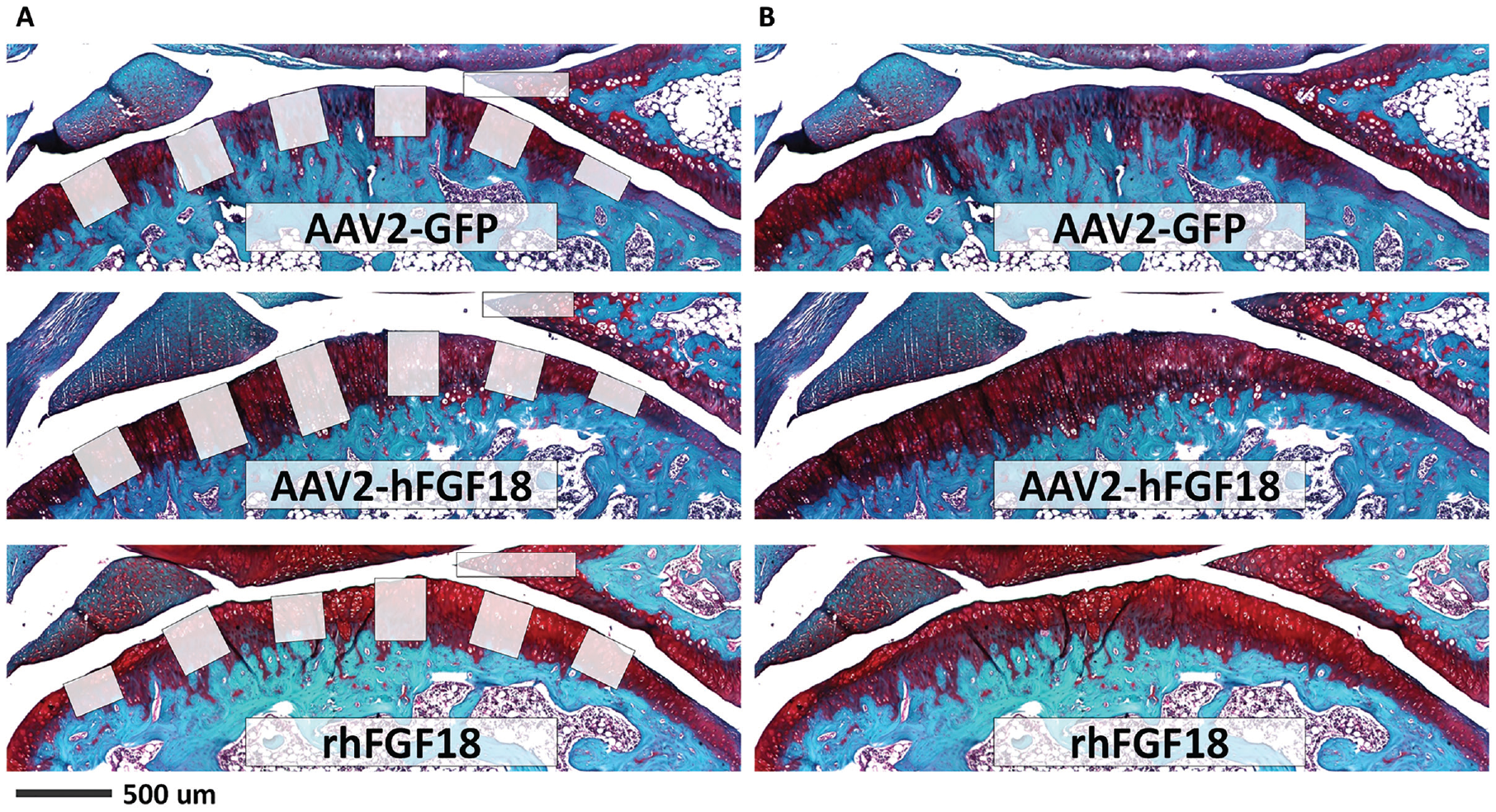
Safranin O-stained sagittal histology sections of rat knee joints with (
Quantitative, statistically significant increases in cartilage thickness were observed in the tibia and meniscal tip following normalization by animal weight ( Fig. 8 ). The normalized tibial cartilage thickness for the AAV2-FGF18 gene therapy ranged between 0.72 ± 0.15 µm/g (average ± standard deviation) and 0.67 ± 0.18 µm/g, while the normalized tibial cartilage thickness for the rhFGF18 protein treatment arm was 0.70 ± 0.15 µm/g, respectively. The control AAV2-GFP-dosed joints demonstrated an average cartilage thickness of 0.64 ± 0.17 µg/g at the 2-month timepoint. A statistically significant difference in cartilage thickness was achieved for the protein treatment arm and the highest gene therapy dose in the tibia relative to AAV2-GFP, which is known for its lack of chondrogenic or chondrodegenerative properties.19,33,34 Within the meniscus, the two top gene therapy doses attained statistical significance over the negative control, grouping together with the protein treatment; however, the protein did not achieve a statistically significant difference relative to the negative control using the Tukey’s pairwise comparison post hoc test. A dose response curve was observed in the tibial cartilage, while dose 2 AAV2-FGF18 appeared to perform best in the meniscal tip.
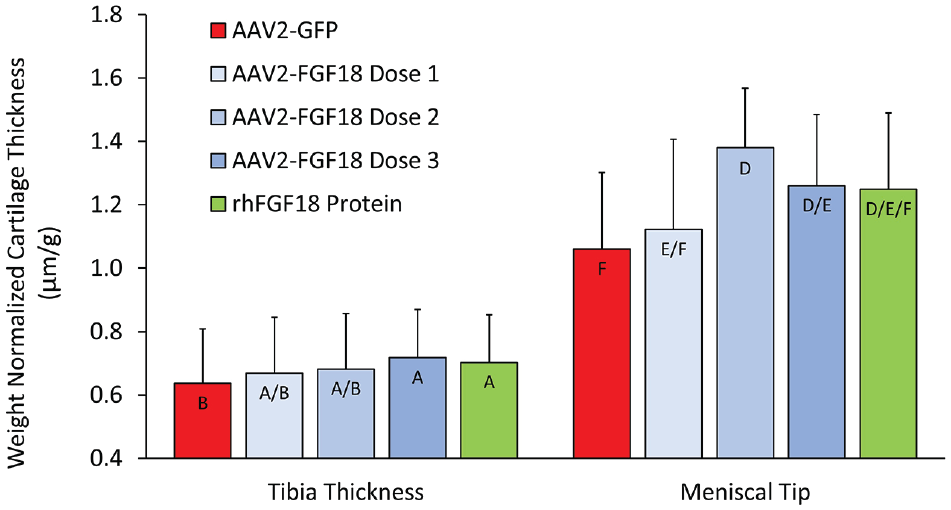
Weight-normalized cartilage thickness average by anatomical location following administration of rhFGF18 protein positive control, 3 doses of AAV2-FGF18 (dose 1 = 2 × 108 vg/joint, dose 2 = 1 × 1010 vg/joint, dose 3 = 1 × 1011 vg/joint) and AAV2-GFP negative control. All groups within anatomical location reach statistical significance against the AAV2-GFP negative control (one-way ANOVA, P < 0.05; Tukey’s pairwise comparison grouping indicated by letter code, Pcrit = 0.05, n = 12 joints, across 6 animals, 2 sections per joint). GFP = green fluorescent protein; ANOVA = analysis of variance; AAV2 = adeno-associated virus type 2.
Finally, we observed increased joint swelling following repeat administration of FGF18 over the 2-month study duration. The swelling was observed at both the 1- and 2-month timepoints, with partial resolution toward the end of the study ( Fig. 9 ). At the 1-month timepoint the multi-injection rhFGF18 protein-treated joints increased in diameter by 22.8% relative to no-injection baseline controls. Both the 1- and 2-month timepoints of the protein treatment arm were statistically different from all other test groups except AAV2-GFP at 2 months (two-way ANOVA, Tukey’s pairwise comparison, Pcrit = 0.05).

Joint diameters at the 1- and 2-month study timepoints relative to the no-injection baseline;  —AAV2-FGF18 single injection,
—AAV2-FGF18 single injection,  —AAV2-GFP control single injection,
—AAV2-GFP control single injection,  —rhFGF18 protein bi-weekly injections (two-way ANOVA, P < 0.05; Tukey’s pairwise comparison grouping indicated by letter code, Pcrit = 0.05, dashed red lines indicate 99% confidence interval of the mean of unoperated control, ⊕ indicates group mean, n = 12 joints across 6 animals per group). GFP = green fluorescent protein; ANOVA = analysis of variance; AAV2 = adeno-associated virus type 2.
—rhFGF18 protein bi-weekly injections (two-way ANOVA, P < 0.05; Tukey’s pairwise comparison grouping indicated by letter code, Pcrit = 0.05, dashed red lines indicate 99% confidence interval of the mean of unoperated control, ⊕ indicates group mean, n = 12 joints across 6 animals per group). GFP = green fluorescent protein; ANOVA = analysis of variance; AAV2 = adeno-associated virus type 2.
Discussion
OA bears many of the hallmarks of a classical disease of aging, where decreases in tissue cellularity as well as structurally important ECM concentration result in a progressively degenerative phenotype that eventually requires surgical intervention.4,35 While inflammatory cytokines have been hypothesized to possess both leading and contributory roles in OA,36,37 none of the anti-inflammatory therapies investigated to date have been able to demonstrate disease modification in controlled, randomized clinical trials. Only repeated administration of a chondrogenic rhFGF18 protein analog has demonstrated the ability to increase cartilage thickness in a predictable, dose-dependent manner, against a placebo control. 15 The protein injection approach, however, is a multi-dose therapy requiring up to 12 injections per year in bi-lateral OA treatment and may need to be sustained indefinitely to prevent reversal of cartilage gains. 15 To overcome the delivery challenges associated with rhFGF18 protein therapy, we have developed a novel hFGF18 gene therapy, using an AAV2 delivery vector, which has demonstrated a high degree of safety, durability, and localization following intra-articular administration.18,38
Our results demonstrate excellent transduction efficiency and cytocompatibility of the AAV2 vector with primary human synoviocytes and chondrocytes, the cell types that have been suggested as the primary targets for stable transgene expression in the joint. 39 Chondrocytes appeared to be more conducive to AAV2-mediated transduction as evidenced by higher percentage of GFP positive cells at significantly lower doses. After 168 hours in culture, >90% of chondrocytes were assessed as GFP+ by quantitative fluorescent microscopy at an MOI of 1,000, while only ~80% GFP+ synoviocytes were observed over the same timeframe at an MOI of 500,000 AAV2-GFP per cell ( Fig. 1 ). Despite these differences in transduction efficiency, both cells expressed significant amounts of GFP protein and did not demonstrate a statistically significant decline in cell count for any of the evaluated doses, confirming no dose-dependent cytotoxicity of the delivery vector.
In parallel, significantly lower doses of AAV2-FGF18 (MOI 100 and MOI 1,000) were able to induce chondroproliferative effects of a similar magnitude to that observed for protein doses previously evaluated in vitro, and used to inform in vivo and subsequent clinical dose selection 40 ( Fig. 2 ). Since this effect could be attributed to the previously suggested autocrine activity of the FGF18 protein 41 and transduction of synoviocytes may predominate in vivo, due to the lacunar isolation of chondrocytes, we further evaluated paracrine rhFGF18 activity using a transwell co-culture assay. Synoviocytes transduced with AAV2-FGF18 cultured in transwell plates with primary human chondrocytes demonstrated the ability of AAV2-FGF18 to mediate chondrocyte proliferation in a paracrine manner at a magnitude comparable with a high ambient dose of the rhFGF18 protein (1,000 ng/mL). These results support the potential of AAV2-FGF18 gene therapy to promote chondroproliferative effects following intra-articular delivery, regardless of the precise biodistribution within the joint, so long as at least some of the resident cells proximal to the synovial fluid are transduced.
Subsequently, we assessed the ability of AAV2-FGF18 to promote the upregulation of hyaline cartilage–associated genes, while downregulating fibrocartilage-related genes in culture. Hyaline is the natural cartilage form in articular joints, while the presence of fibrocartilage following surgical focal defect repair procedures (such as microfracture) was previously suggested to result in decreased durability of repair. 42 Treatment of primary human chondrocytes with AAV2-FGF18 and rhFGF18 protein upregulated HAS2 and COL2A1 relative to the PBS baseline; however, only AAV2-FGF18 was able to achieve a statistically significant increase in PRG4. HAS2 is an essential hyaluronan (HA) synthesis component, specifically responsible for the production of high molecular mass HA, which is abundant in hyaline cartilage. 25 Similarly, COL2A1 and PRG4 are secreted proteins deemed essential for articular cartilage structural integrity.27,28,43 As such, the upregulation of HAS2, COL2A1, and PRG4 is further supportive of the chondroanabolic activity of hFGF18.
Conversely, COL1A1 and LOX were downregulated in the protein and AAV2-FGF18 treatment groups, while only the AAV2-FGF18 treatment group was able to achieve a statistically significant reduction in ADAMTS15. Previous studies have associated high levels of COL1A1, a fibrocartilage component, with decreased durability following abnormal articular cartilage repair caused by surgical resurfacing procedures such as microfracture. 44 Similarly, lysyl oxidase family cuproenzymes were suggested to play a role in undesirable collagen crosslinking and LOX downregulation has been observed to promote articular cartilage regeneration. 31 Finally, ADAMTS15, an important metalloproteinase responsible for catabolic activity, 45 has been associated with pathologic OA, and tempering of ADAMTS15 was suggested as a potentially viable approach to prevent progressive cartilage degeneration. 46
Interestingly, Sox9, a chondrocyte differentiation–associated transcription factor, was only upregulated in the AAV2-FGF18 and not the rhFGF18 protein treatment group; however, the degree of upregulation was relatively small compared with baseline, only achieving a 1.3-fold increase relative to PBS. Unbiased RNA-seq analysis further demonstrated that the most impacted gene expression GO terms mapped to anatomic structure development (22 of 43 or 51%), signaling (19 of 43 or 44%), and cell differentiation (10 of 43 or 23%), which is expected for the regenerative phenotype of the intervention ( Fig. 4 ). Moreover, we observed a number of other similarly upregulated and downregulated genes including ESM1, NTSR1, MMP1 and ANGPTL7, COPG2IT1, pentraxin-related protein 3 (PTX3), respectively (Suppl. Fig. S2), all which have previously been previously associated with skeletal development and maintenance.47-52 These findings further suggest similarity in activity between the rhFGF18 protein and AAV2-FGF18 gene therapy, regardless of the differences in delivery modality. Conversely, TGM2, a calcium-dependent acyltransferase with observed involvement in cartilage homeostasis, 53 and SERPINA1, which has previously been associated with chondrogenesis and chondrocyte differentiation, 54 require further investigation. Pathway analysis and mechanistic studies may help elucidate whether the differences can be attributed to the degree of hFGF18 accumulation in the cytoplasm, delivery, or pathways activated by the AAV2 capsid.
All in all, the results of gene expression analysis suggest that the mechanism of hFGF18 activity is comprised of 2 complementary anabolic components driving chondrocyte proliferation and hyaline cartilage ECM production ( Fig. 10 ) with multiple direct (lubricin/PRG4, collagen 2/COL2A1) and indirect pathways (ADAMTS1, 5, 15, and MMP2).55-57 In parallel, FGF18 appears to suppress expression of fibrocartilage-associated genes via direct downregulation of COL1A1 and LOX, as well as indirect downregulation of genes that promote fibrosis including PTX3 and insulin-like growth factor I.58,59
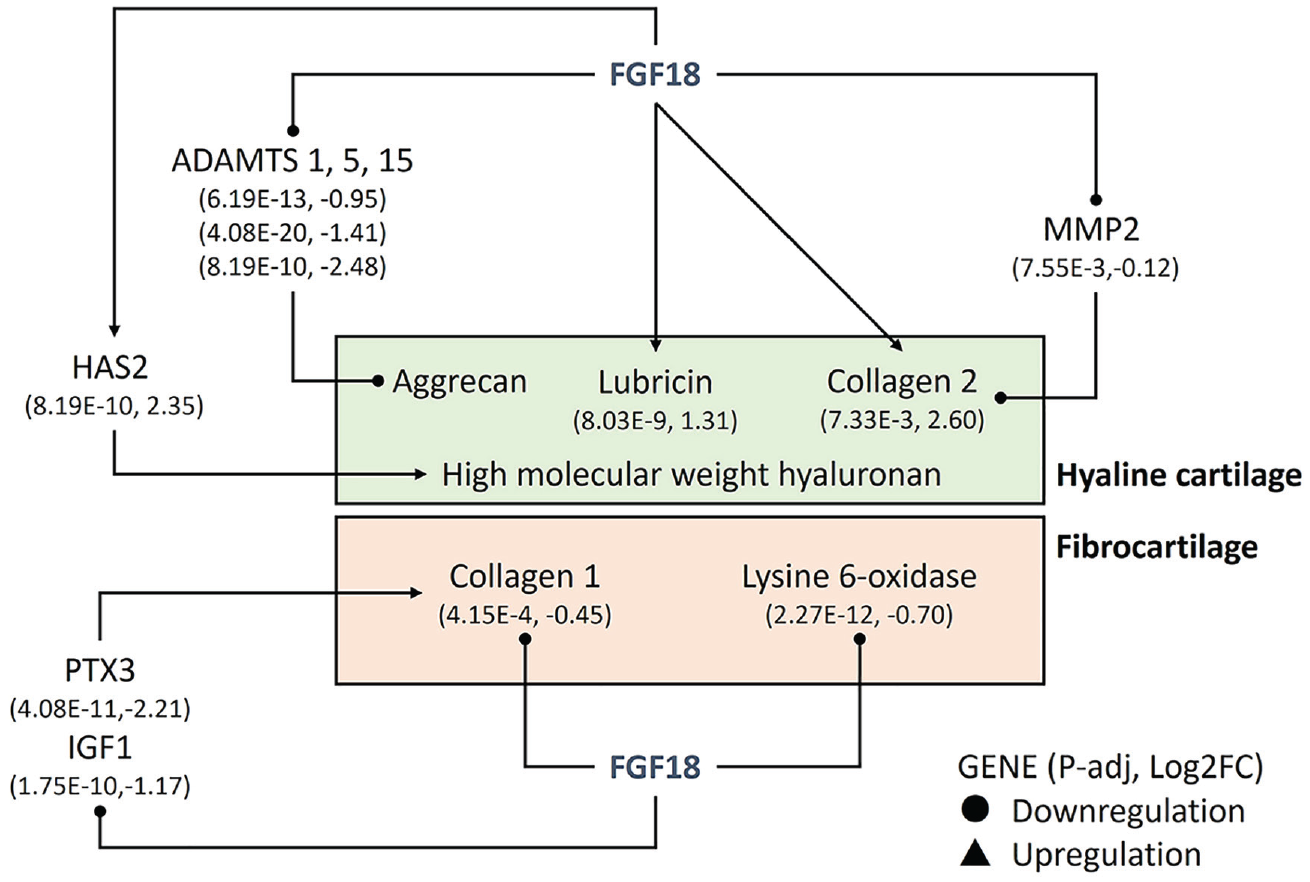
Hyaline cartilage– and fibrocartilage-associated pathways affected by AAV2-FGF18 administration, as determined by RNA-seq analysis (n = 5). Direct hyaline cartilage promoting effects observed in RNA-seq analysis: lubricin (PRG4) and collagen 2 (COL2A1). Indirect hyaline cartilage promoting effects: ADAMTS1, 5, 15, and matrix metalloprotease-2. Direct fibrocartilage suppressing effects: Collage 1 (COL1A1) and lysine 6-oxidase. Indirect fibrocartilage suppressing effects: PTX3 and IGF1. PTX3 = pentraxin-related protein 3; IGF1 = insulin-like growth factor 1; AAV2 = adeno-associated virus type 2.
We further confirmed the safety of the AAV2 delivery vector by administering doses up to 1 × 1011 vg per joint via intra-articular injection. We observed no abnormal growths or tumors in cartilage, meniscus, subchondral bone, or the proximal bone marrow over the study duration. Furthermore, neither the AAV2-GFP control nor the AAV2-FGF18 gene therapy treatment arm produced any clearly observable chondro- or osteo-degenerative processes, inflammatory infiltrates, or other qualitative attributes of tissue degradation or cellular inflammation. In contrast to the multiple injection rhFGF18 protein treatment, the single injection AAV2-hFGF18 approach did not elicit joint swelling or inflammation. The synovitis we observed with rhFGF18 treatment was therefore attributed to the consistent disruption of the joint capsule due to repeated injections, which likely drove the pathology. These findings are in line with the previously published studies of viral vector–delivered rhFGF18 in the pinna and intravertebral disk,41,60 which also did not report any treatment-related side effects.
To our knowledge, increases in tibial and meniscal cartilage thickness in healthy knee joints have not been previously reported for either rhFGF18 protein treatment or viral vector–delivered FGF18. In the present study, both rhFGF18 protein injections and the AAV-hFGF18 treatment arm demonstrated statistically significant increases in tibial cartilage thickness over the AAV2-GFP negative control reaching increases of 9.4% and 12.5%, respectively. The highest dose of the AAV2-FGF18 group and the protein treatment group achieved statistical significance over the negative control in the tibia. While previous studies using smaller numbers of animals did not observe statistically significant increases in tibial cartilage thickness of healthy rodent joints dosed with rhFGF18 protein, those studies were limited by their sample size and lack weight normalization, which was previously demonstrated to be associated with cartilage thickness. 16 Moreover, early studies using adenoviral vector–delivered hFGF18 to the mouse ear pinna observed chondrocyte proliferation, upregulation of ECM production, and locally increased tissue thickness attributed to increased cartilage accumulation. 41 The observed increase in cartilage thickness of the tibial plateau (28-37 µm relative to a 298-µm control baseline) may be sufficient to delay age-related cartilage erosion and symptom onset based on previously observed rates of cartilage erosion in OA and aging.8-10,15 Interestingly, the increase in cartilage thickness was also observed at the meniscal tip, where administration of an intermediate dose of AAV2-FGF18 resulted in a 30% increase in the white cartilaginous zone, relative to the AAV2-GFP control. The protein treatment group increased the thickness of the meniscal tip by 18%; however, statistical significance was not achieved, likely due to the sample size and magnitude of effect. This increase in cartilage was observed in the white zone segment of the meniscus, which is known to be comprised of a more hyaline cartilage phenotype with an increased concentration of type II collagen.61,62 Unlike in the case of the tibia, the most pronounced increase in the meniscal cartilage did not occur at the highest dose, and while this effect may be attributed to random variation, it is likely that the dose-chondrogenesis relationship includes a global maximum, and a peak-dose effect may be different for various anatomical structures. While chondroregenerative treatments focused on articular cartilage are currently under late-stage clinical evaluation, 15 the application of said treatments to repair of meniscal tissues has not been previously reported. At the same time, we did not observe any joint swelling following single injections of AAV2-GFP or AAV2-FGF18, whereas the protein treatment arm demonstrated significantly increased joint diameters relative to the uninjected population background controls, peaking at the 1-month timepoint and partially resolving by the second month. The rhFGF18 treatment group–associated swelling was attributed to inflammation of the synovial membrane, likely resulting from the repeated disruption of the joint capsule following the series of twice-weekly intra-articular injections (Suppl. Fig. S3). While it is unclear whether swelling is a phenomenon that will translate to the significantly larger human joint, and the rhFGF18 protein injection treatment appears safe in placebo-controlled randomized human trials, 15 further evaluation of the multi-dose-associated swelling effect with a focus on joint size to dosing needle aspect ratio and injection frequency is warranted.
In conclusion, intra-articular administration of AAV2-FGF18 appeared safe and effective at increasing cartilage thickness in a manner comparable with rhFGF18 protein treatment. The single-injection AAV2-FGF18 appears mechanistically similar to rhFGF18 treatment, while offering potential safety and pharmacology advantages related to the frequency of required injections. The ability to increase chondrocyte proliferation, upregulate several hyaline cartilage ECM–related genes, and promote the increase of cartilage thickness in rat knee joints supports the potential advancement of the hFGF18 gene therapy into disease model efficacy testing. Both age-related chondrodegenerative pathologies such as OA and focal cartilage lesions may benefit from general increases in cartilage thickness following AAV2-mediated delivery of FGF18. Finally, the observed effects of hFGF18 gene therapy on the meniscal tip warrant further evaluation of this approach in regenerative repair of meniscal tears and extrusions, which have been previously associated with increased OA progression due to reduced meniscal coverage of the tibial plateau.63,64
Supplemental Material
sj-jpg-1-car-10.1177_19476035231158774 – Supplemental material for Adeno-Associated Virus-Delivered Fibroblast Growth Factor 18 Gene Therapy Promotes Cartilage Anabolism
Supplemental material, sj-jpg-1-car-10.1177_19476035231158774 for Adeno-Associated Virus-Delivered Fibroblast Growth Factor 18 Gene Therapy Promotes Cartilage Anabolism by Judith M. Hollander, Alex Goraltchouk, Miraj Rawal, Jingshu Liu, Francesco Luppino, Li Zeng and Alexey Seregin in CARTILAGE
Supplemental Material
sj-jpg-2-car-10.1177_19476035231158774 – Supplemental material for Adeno-Associated Virus-Delivered Fibroblast Growth Factor 18 Gene Therapy Promotes Cartilage Anabolism
Supplemental material, sj-jpg-2-car-10.1177_19476035231158774 for Adeno-Associated Virus-Delivered Fibroblast Growth Factor 18 Gene Therapy Promotes Cartilage Anabolism by Judith M. Hollander, Alex Goraltchouk, Miraj Rawal, Jingshu Liu, Francesco Luppino, Li Zeng and Alexey Seregin in CARTILAGE
Supplemental Material
sj-jpg-3-car-10.1177_19476035231158774 – Supplemental material for Adeno-Associated Virus-Delivered Fibroblast Growth Factor 18 Gene Therapy Promotes Cartilage Anabolism
Supplemental material, sj-jpg-3-car-10.1177_19476035231158774 for Adeno-Associated Virus-Delivered Fibroblast Growth Factor 18 Gene Therapy Promotes Cartilage Anabolism by Judith M. Hollander, Alex Goraltchouk, Miraj Rawal, Jingshu Liu, Francesco Luppino, Li Zeng and Alexey Seregin in CARTILAGE
Footnotes
Author Contributions
J.M.H., A.G., L.Z., and A.S. have given substantial contributions to the conception and the design of the manuscript, and J.M.H., A.G., M.R., J.L., and A.S. contributed to acquisition, analysis, and interpretation of the data. All authors have participated to drafting the manuscript, and F.L. and L.Z. revised it critically. All authors read and approved the final version of the manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The funding for this work was provided by Remedium Bio, Inc.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: L.Z. serves as an advisor to Remedium Bio, Inc.
Ethical Approval
All methods involving animals in this study were approved by the Tufts University Institutional Animal Care and Use Committee (Protocol #B2021-106).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
