Abstract
Objective
The purpose of this study was to determine if there were variations in chondrocyte viability, matrix glycosaminoglycan (GAG), and water content between different areas of the articular surface of a bovine metatarsophalangeal joint, a common and reliable source of articular cartilage for experimental study, which may compromise the validity of using multiple samples from different sites within the joint.
Methods
Nine fresh cadaveric bovine metatarsophalangeal joints were obtained. From each joint, 16 osteochondral explants were taken from 4 facets, yielding a total of 144 cartilage specimens for evaluation of chondrocyte viability, matrix GAG, and water content. A less invasive method for harvesting osteochondral explants and for processing the biopsy for the assessment of chondrocyte viability was developed, which maintained maximal viability within each cartilage explant.
Results
There was no significant difference between the 16 biopsy sites from the different areas of the joint surface with respect to chondrocyte viability, matrix GAG and water content. Pooled data of all samples from each joint established the baseline values of chondrocyte viability to be 89.4% ± 3.8%, 94.4% ± 2.2%, and 77.9% ± 7.8%, in the superficial quarter, central half, and deep quarter (with regard to depth from the articular surface), respectively. The matrix GAG content of bovine articular cartilage was 6.06 ± 0.41 μg/mg cartilage, and the cartilage water content was 72.4% ± 1.5%. There were also no significant differences of these 3 variables between the different joints.
Conclusion
It is thus reasonable to compare biopsies obtained from different sites, as a biopsy from one site would be considered representative of the whole joint.
Introduction
Osteochondral explants are frequently used as an experimental model in cartilage research and a variety of approaches have been used depending on the study performed. Commonly, tissue samples take the form of full-depth osteochondral cylinders or tissue blocks with attached subchondral bone.1-3 Other investigators have prepared cartilage explants with the subchondral bone carefully removed or avoided,1,4-7 or have used more specific explants such as those reflecting osteoarthritic changes 8 or in which growth plate cartilage is present.8,9
In the majority of studies to date, cartilage explants have then been randomized with relatively little attention being paid to the origin of their location on the joint and therefore the results reflect the average response of the areas under investigation. However, the prevailing load has been shown to affect the cartilage thickness and matrix components of different sites within the same joint.10,11 This is reflected in the variation in chondrocyte morphology, collagen fiber orientation, and the type and amount of matrix proteoglycans which vary with cartilage depth.7,12-14 Thus, even though randomized cartilage explants are routinely used, it is still uncertain whether an explant from one site is representative of the whole joint.
This study was therefore designed to evaluate chondrocyte viability, glycosaminoglycan (GAG), and water content of the extracellular matrix (ECM) within carefully mapped areas of the bovine metatarsophalangeal joint surface, which is commonly used for a range of studies in the field of cartilage research.4,15,16 A standard method to harvest the cartilage explant from the joint and to examine these variables was also established. The hypothesis was that there were no differences across the joint with regard to chondrocyte viability, matrix GAG, and water content.
Methods
Materials
Chemicals were obtained from Sigma-Aldrich (Dorset, UK) unless otherwise stated. The cell viability probes 5-chloromethylfluorescein diacetate (CMFDA) and propidium iodide (PI) were purchased from Invitrogen (Paisley, UK) and were prepared in dimethyl sulfoxide (DMSO) as aqueous 7 μM stocks. Dulbecco’s modified Eagle medium (DMEM; glucose 4.5 g/L) was also obtained from Invitrogen.
Harvest of the Bovine Osteochondral Explants
Nine metatarsophalangeal joints of 3-year-old cows were obtained from a local abattoir, washed, skinned, and opened under sterile conditions within 6 hours of slaughter. Only healthy joints without macroscopic evidence of cartilage damage/degeneration were used. There were 8 facets in the joint (
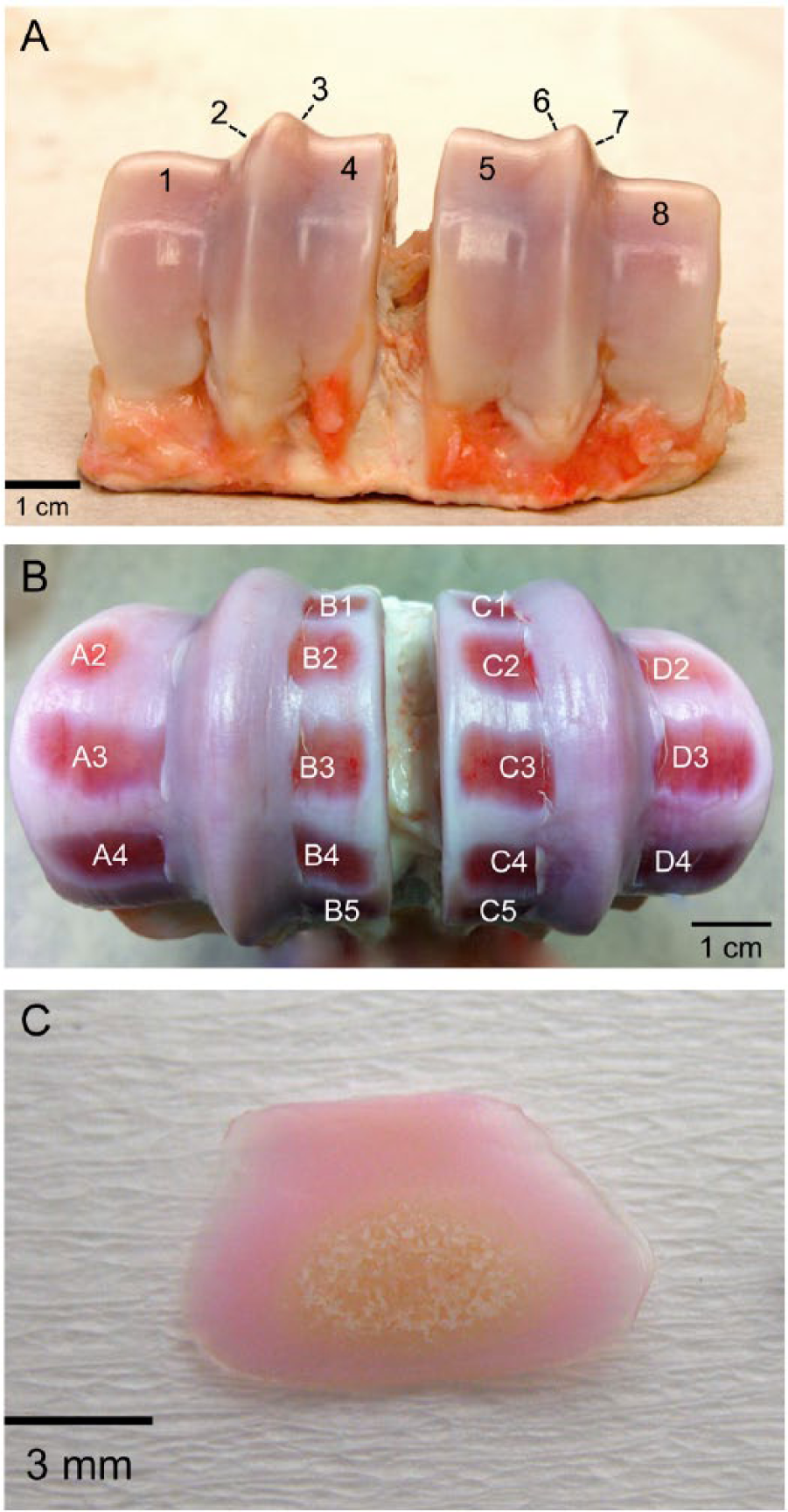
Cartilage sampling from the surface of a bovine metatarsophalangeal joint. (
Chondrocyte Viability Assessment
Explants were trimmed by using a rocking motion with a custom-made double-bladed cutting tool, to create 2 parallel straight edges on the cartilage explant (
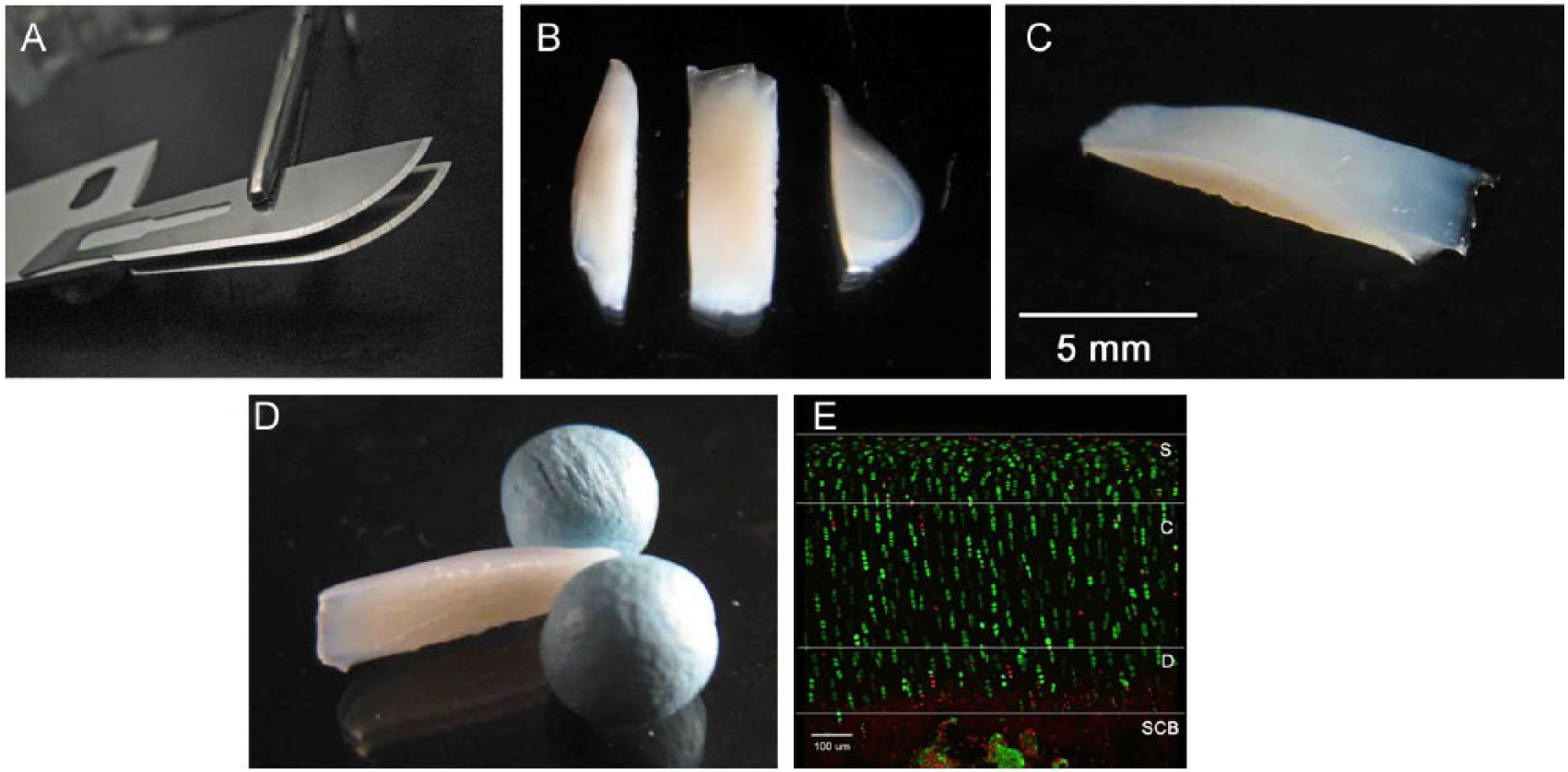
Preparation of cartilage samples for imaging, and visualization of fluorescently labeled in situ chondrocytes. (
An upright confocal laser scanning microscope (Zeiss LSM510 Axioskop, Carl Zeiss, Welwyn Garden City, UK) with a ×10 objective was used to acquire optical sections of CMFDA- and PI-labeled chondrocytes in the coronal plane, that is, through the cut-edge. The scanned images were reconstructed and analyzed using ImageJ software (Version 1.47, NIH, Bethesda, MD, USA). Articular cartilage was characterized into 3 regions on the basis of depth from the articular surface to the subchondral bone: the first quartile of cartilage was defined as the superficial quarter, followed by the central half as the middle 50% of the thickness, and the deep quarter as the last quartile (
Matrix Glycosaminoglycan Assessment
The content of sulfated glycosaminoglycan (GAG) in the extracellular matrix of cartilage was measured using a spectrophotometric microassay method.17,18 The 1,9-dimethylmethylene blue (DMMB) solution was maintained at pH 3.0. The dilution solution was Tris/HCl (50 mM) with a pH of 8.0. The standard solution was made from shark chondroitin sulfate with a concentration of 0.1 mg/mL.
The biopsied cartilage explants were trimmed with a skin biopsy punch of 2.5 mm diameter (Kai Industries, Seki, Japan) to obtain the central full-thickness area of cartilage tissue. This sample was weighed to obtain its “before-digested” wet weight, which included the weight of cartilage and subchondral bone. Papain (300 μg) was then added to digest the cartilage sample at 60°C for approximately 4 hours. After digestion, 10 μL of 1 M iodoacetic acid solution was added to stop the effect of papain, and the solution diluted with 4 mL Tris/HCl buffer. The undigested subchondral bone was weighed in order to calculate the true cartilage weight, which was the “before-digested” wet weight of the sample subtracted from the wet weight of the subchondral bone. The absorbance of the solution was measured immediately after adding 1 mL DMMB solution (within 10 seconds after mixture). The absorbance of the sample was compared with the standard solution to obtain the equivalent GAG weight of the cartilage matrix. This result (in micrograms) was then normalized to the total cartilage mass (in milligrams) to allow for any variation in the size of the cartilage specimen. Thus, GAG content was presented as the GAG mass (in μg) per cartilage mass (in mg), that is, “GAG (μg/mg cartilage)” in the Results section.
Cartilage Water Content
Excess moisture on cartilage explants was removed by placing them briefly and gently between folded filter paper prior to weighing (to obtain wet weight). The samples were then lyophilized at −55°C and 0.1 atm over 12 hours and then weighed to obtain the dry weight with the difference in weights representing the cartilage water weight. Water content was calculated using the formula: (cartilage water weight/cartilage wet weight) ×100%.
Statistical Analysis
Statistical analyses were performed using Minitab 16 (Minitab Inc., State College, PA, USA). All data were tested for normality using the Kolmogorov-Smirnov test. Thereafter, parametric data were analyzed using paired or unpaired Student’s t tests if 2 sets of data were compared, or one-way analysis of variance (ANOVA) with post hoc Tukey’s tests if more than 2 sets of data were analyzed. For nonparametric data, the Mann-Whitney U test was used for comparison of 2 sets of independent results, while the Kruskal-Wallis test was used for 3 or more sets of nonparametric data. Data are presented as means ± standard deviation (SD) with the level of significance set at P < 0.05.
Results
A total of 9 different joints were used to test the 3 variables (3 joints for each variable), which were (a) chondrocyte viability, (b) GAG content, and (c) water content. For chondrocyte viability, the results from a total of 48 sites (3 joints, 16 sites per joint) showed that viability in the superficial quarter, central half, and deep quarter was 89.4% ± 3.8%, 94.4% ± 2.2%, and 77.9% ±7.8%, respectively (
Mapping of Chondrocyte Viability within Specified Regions across the Bovine Metatarsophalangeal Joint. a
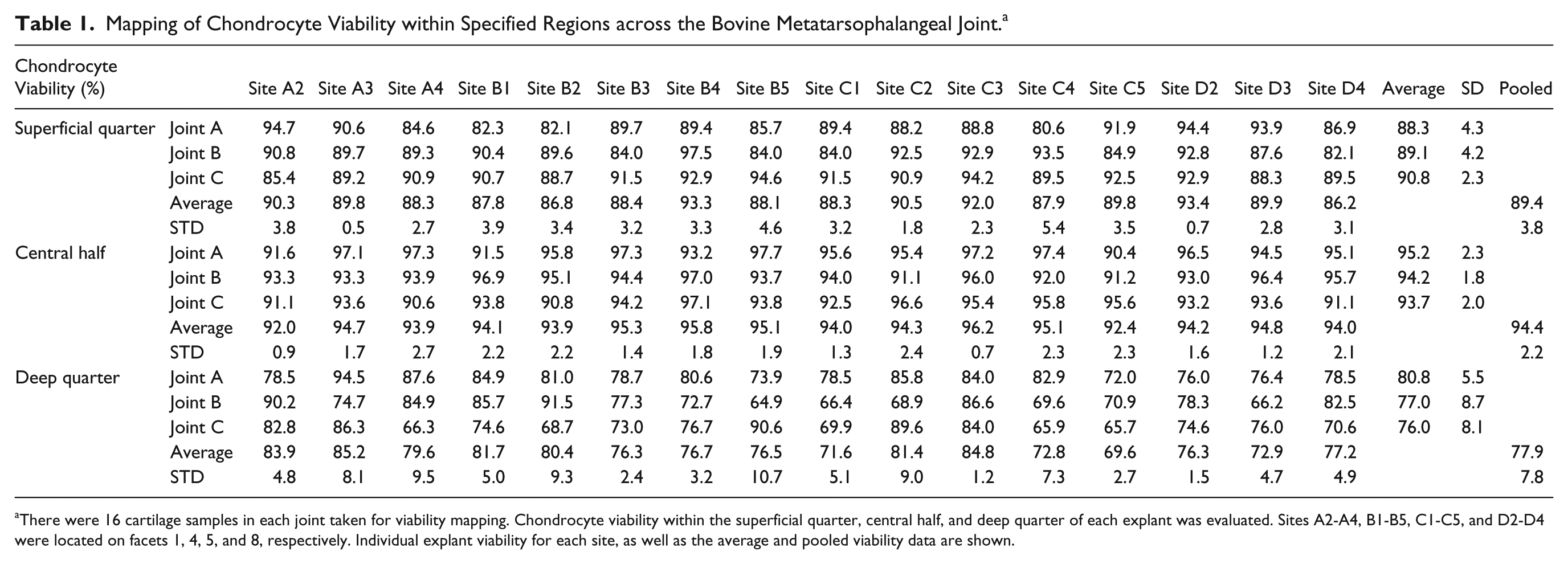
There were 16 cartilage samples in each joint taken for viability mapping. Chondrocyte viability within the superficial quarter, central half, and deep quarter of each explant was evaluated. Sites A2-A4, B1-B5, C1-C5, and D2-D4 were located on facets 1, 4, 5, and 8, respectively. Individual explant viability for each site, as well as the average and pooled viability data are shown.
The matrix GAG content, measured in 48 sites of 3 different joints, was 6.06 ± 0.41 μg/mg cartilage (
Mapping of Matrix Glycosaminoglycan (GAG) Content within Specified Regions across the Bovine Metatarsophalangeal Joint. a
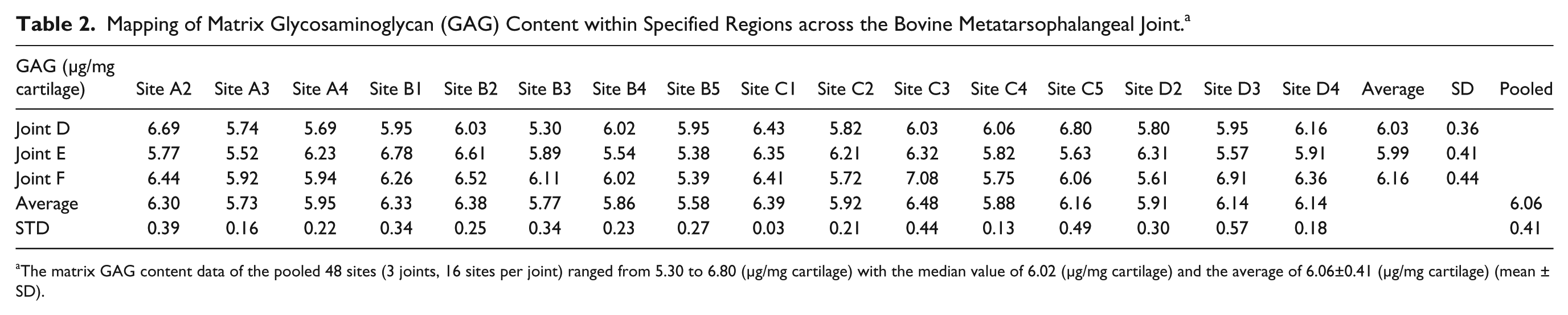
The matrix GAG content data of the pooled 48 sites (3 joints, 16 sites per joint) ranged from 5.30 to 6.80 (μg/mg cartilage) with the median value of 6.02 (μg/mg cartilage) and the average of 6.06±0.41 (μg/mg cartilage) (mean ± SD).
Mapping of Cartilage Water Content within Specified Regions across the Bovine Metatarsophalangeal Joint.
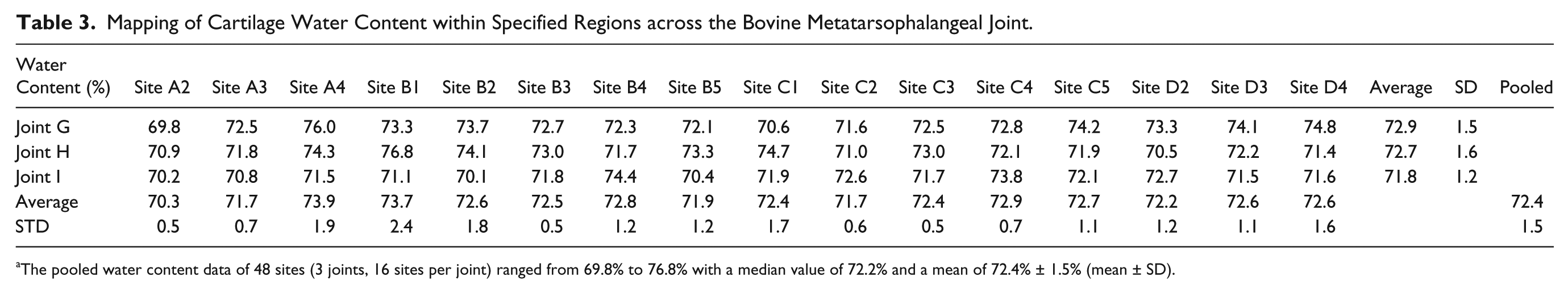
The pooled water content data of 48 sites (3 joints, 16 sites per joint) ranged from 69.8% to 76.8% with a median value of 72.2% and a mean of 72.4% ± 1.5% (mean ± SD).
Further comparisons between joints were performed demonstrating no significant difference between individual metatarsophalangeal joints of bovines in terms of their chondrocyte viability, matrix GAG, or water content (
Chondrocyte Viability, Glycosaminoglycan (GAG), and Water Content from the Pooled Data of 3 Joints. a
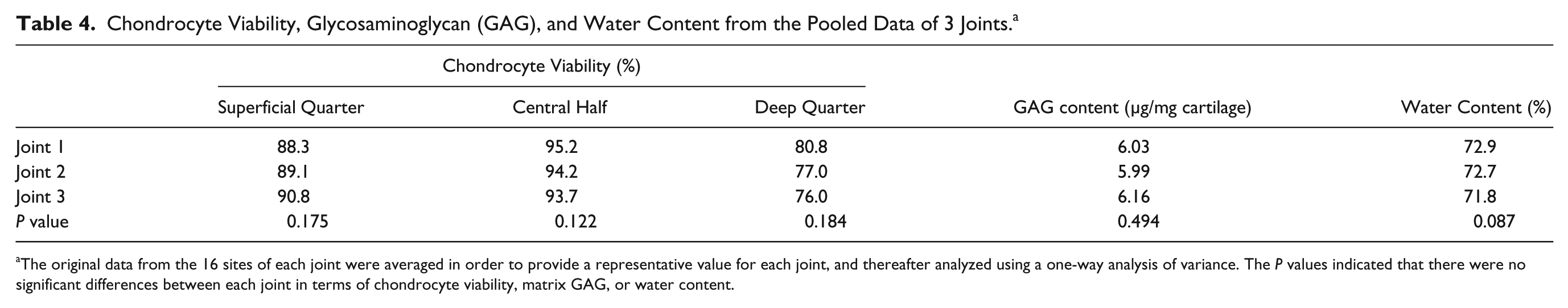
The original data from the 16 sites of each joint were averaged in order to provide a representative value for each joint, and thereafter analyzed using a one-way analysis of variance. The P values indicated that there were no significant differences between each joint in terms of chondrocyte viability, matrix GAG, or water content.
Discussion
The results from these 48 biopsy sites supported the hypothesis that there was no difference between the sites with respect to chondrocyte viability, matrix GAG or water content. The findings indicated that for these characteristics, a cartilage sample from any of these sites on the joint surface was representative of the whole joint. The results also confirmed that there was no statistical difference between these variables in joints from different individuals of the same species. However, the variability of the data was least for sites B3 and C3, both of which were located in the middle of the articular surface, which may indicate that these sites could be more suitable than others if only 1 or 2 cartilage samples are required from each bovine joint.
Characterization of chondrocyte viability, matrix proteoglycan, and water content in fresh (day 0) joints was important because the data would provide baseline values for comparison with the results obtained under different experimental conditions. The data demonstrated that there would not be a sampling bias when the specimens were obtained from different locations of the joint surface. A knowledge of the variability of these values is also useful in the experimental design phase for power calculations. We are not aware of any data on this in the current literature even though cartilage explants from bovine joints have been used extensively for many years. The data from this joint mapping study helps rectify this deficiency.
From these results, it could be established that the baseline values of chondrocyte viability were 89.4% ± 3.8%, 94.4% ± 2.2%, and 77.9% ± 7.8%, in the superficial quarter, central half, and deep quarter, respectively. The matrix GAG content of bovine articular cartilage was 6.06 ± 0.41 μg/mg cartilage, and the cartilage water content was 72.4% ± 1.5%. However, when comparing the results from different studies, it is important to take into account the differences in the materials and methods between the studies. For example, when studying human surgical specimens, Amin et al. (2008) reported that chondrocyte viabilities within cartilage explants from human knee joints were 86.4%, 91.9%, and 82.2% in the superficial quarter, central half, and deep quarter, respectively. 19 Pun et al. 20 also studied cartilage explants of human knee joints and demonstrated that chondrocyte viability on day 0 was 80.5%, 80.0%, and 83.0% for the superficial quarter, central half, and deep quarter, respectively. Chondrocyte viability in these surgical specimens may have been reduced because of (a) cartilage degeneration itself or (b) as a result of the surgical manipulation of the cartilage explant or (c) due to the cutting action—as uncut cartilage would be expected to show less chondrocyte death.
The measured water content of cartilage in the current study was 72.4% ± 1.5%. This was the average value across all the areas in the present study, and is in agreement with the randomized samples that have been used in previous reports. 21 For the GAG content of the extracellular matrix, Hoemann et al. 22 reported that fresh cartilage explants from the bovine shoulder joint contained 4.9 to 5.8 μg/mg cartilage. Their values were slightly lower than the results presented here, which may have been due to the samples originating from a different joint with a different loading pattern, however it was perhaps more likely due to the different harvesting technique. The explants in their study were harvested from the joint directly with 6 mm biopsy punches. Because of the hardness of the subchondral bone, this biopsy technique may have caused more stress to the cartilage explants than the technique used in the current study. This might have resulted in more matrix GAG loss from the cutting margin of the biopsied explants.
When chondrocyte viability is assessed, the method of harvesting and cutting the cartilage explants plays an important role because the blade applies pressure on the tissue which adversely affects the cell viability.23-25 It is to be expected that some of the cells at the surface of a cut-edge would be dead, and the affected region has been found to be approximately within a 10 μm depth from the cut-edge.15,19,26 However, there is no direct way to detect cell viability without affecting the natural status of the chondrocytes to some extent. Therefore, as a result of the processing of the tissue, the “examined viability” of a fresh bovine joint in the present study was likely to be lower than the “real viability” present in vivo, which would be expected to be close to 100%. This small drop in viability should be taken into account when considering the results.
The cutting effect was unavoidable. However, if this was consistent for every sample, the viability results should be comparable. Previous authors15,27 have reported that the effect of cutting is reproducible and that more living cells are preserved if new scalpel blades are used for each cut. The present study has provided additional evidence for this. In addition, Amin et al. 15 demonstrated that cell viability during cutting could be markedly improved if it was performed in the presence of a hyperosmotic solution. Furthermore, the similarity of the results between the cartilage biopsies, suggested that the methodology used in this study, that is, using new scalpel blades for each cartilage biopsy and the parallel cutting of explants by 2 blades with a “rocking motion,” produced a similar cutting effect in every sample and thus the effect of the cut on cartilage was reproducible.
Articular cartilage is traditionally divided into 4 zones, that is, superficial, middle, deep, and calcified zones. However, the thickness of each zone is highly dependent on species, the joint studied and the stage of the animal’s development.28-30 The border of each zone can be difficult to identify reliably and reproducibly, especially in the confocal images. We found that the change in chondrocyte viability occurred predominantly in the first and deepest quartiles. Therefore, we used the terms “superficial quarter”, “central half”, and “deep quarter” to indicate the first 25%, the next 50%, and the last 25% of cartilage, respectively, for getting more reliable and repeatable boundaries by quartile percentage rather than chondrocyte shape or topographical arrangement.
Different zonal arrangements have been used by others, for example, Lipshitz et al. 12 who examined a different metric, that is, the hexosamine content in different layers of cartilage taken from the bovine medial femoral condyle. This cartilage was approximately 1200 μm thick. They stated that they took successive sections of 250 μm thickness (although earlier in the methods they stated 50-100 μm thick slices were cut). The first cut was stated to be 200 to 250 μm below the surface. This is similar to our first optical section as our cartilage thickness was approximately 800 to 1000 μm. They found that the hexosamine content and swelling ratio of adult bovine articular cartilage varied with depth from the articular surface. Nevertheless, they did not define the borders of each zone by the hexosamine content or the swelling ratio. It is important to note that if different zonal definitions are used chondrocyte viability would be expected to change accordingly. However, the comparisons of chondrocyte viability in the same region, but from different biopsy sites will be largely unaltered. In addition, in the present study, measurements of GAG and water content were performed on full depth cartilage explants instead of dividing explants into the different regions. It is known that these have spatial distribution patterns in cartilage. 14 However, for the simplification of the tests, these two variables were measured in full depth.
For matrix GAG measurement, it would be difficult to excise the subchondral bone accurately from the explant without losing any cartilage tissue by leaving it on the bone. This might increase the inaccuracy of the GAG measurement since a substantial proportion of the matrix GAG is located in the deep quarter of cartilage.12,14 Thus full-depth explants, which included a small amount of subchondral bone, were taken in the present study to circumvent this problem. The inclusion of the subchondral bone has been taken into account when determining other cartilage properties. Furthermore, the GAG assessment using the DMMB assay involved a comparison with the standard chondroitin sulfate of shark cartilage. Consequently, some nuances should be taken into account such as the impurity of the standard shark cartilage.31,32 The molecular weight difference between the standard chondroitin sulfate and the tested cartilage sample containing chondroitin sulfate, keratan sulfate, and other small proteoglycans should also be considered if the DMMB assay was used. 31 However, although this is a limitation of the method, the influence of this was reduced by using the same standard solution throughout all the experiments in the current study.
To conclude, the present study demonstrated that the outcome measures (specifically, chondrocyte viability as measured by CLSM and GAG by the DMMB assay) had good reliability and repeatability and therefore, the number of repeat experiments could be kept within a reasonably low range. For the bovine metatarsophalangeal joint, there were no significant differences between chondrocyte viability, matrix GAG and water content of full-depth cartilage samples taken across the joint as described. Therefore, a cartilage biopsy taken from one of these sites accurately represented these properties of all the other sites that were studied.
Footnotes
Acknowledgments and Funding
The authors thank Dr Trudi Gillespie, IMPACT facility, The University of Edinburgh, for CLSM guidance, Mrs Anne Pryde, Department of Hepatology, The University of Edinburgh, for spectrophotometry guidance and Scotbeef Ltd., Bridge of Allan, UK, for providing bovine feet. The support of an Overseas Research Grant from Taipei Medical University – Shuang Ho Hospital is gratefully acknowledged. Neither funding source had any influence on the study design, data collection, data analysis and interpretation, and manuscript preparation.
Author Contributions
Conception and design: Y-C. Lin, A.C. Hall, A.H.R.W. Simpson
Collection and assembly of data: Y-C. Lin, A.H.R.W. Simpson
Analysis and interpretation of data: Y-C. Lin, A.C. Hall, I.D.M. Smith, D.M. Salter, A.H.R.W. Simpson
Drafting of the manuscript: Y-C. Lin, A.C. Hall, A.H.R.W. Simpson
Critical revision: Y-C. Lin, A.C. Hall, I.D.M. Smith, A.H.R.W. Simpson
Final approval of the article: Y-C. Lin, A.C. Hall, I.D.M. Smith, D.M. Salter, A.H.R.W. Simpson
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was not sought for the present study because cadaveric animal joints from an abattoir were used.
